Abstract
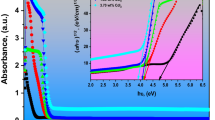
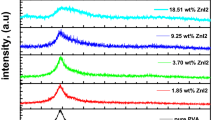
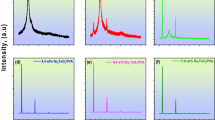
Polyvinyl alcohol (PVA) incorporated with various amounts of ammonium iodide salt (NH4I) composite polymer electrolyte films were prepared via the casting process. The XRD analysis, as a tool for structural investigation, confirms the polymer electrolytes formation. The transmittances of the studied samples are decreased with increasing NH4I fillers. The UV–Vis absorption spectra are shifted to the higher wavelengths, which indicate their importance for light shielding devices. The bandgaps decrease with the increase in NH4I content, attributed to the increase in the crystallite size. The calculated Urbach energy was found to increase with increasing NH4I salt fillers. The samples' optical limiting is carefully investigated using a He–Ne laser beam of wavelength = 632.8 nm. This result enhances light absorption behavior and makes the material suitable for optical UV-protection devices. The correlation between dielectric properties and conductivity is also understood and discussed. The dielectric permittivity ε' of the samples is associated with the dipole, and polarization increased with the addition of NH4I salt content as the particle size increased according to XRD analysis. AC impedance spectroscopy was carried out to disclose the PVA doped with NH4I polymer electrolyte films as a function of various NH4I salt fillers. The analysis via complex electric modulus gives abnormal behavior by adding NH4I salt content, where the minimization of the electrode polarization can be achieved. Our results indicate that the newly designed composite polymeric electrolyte films are commonly appropriate for electronic and optoelectronic devices.













Similar content being viewed by others
References
V. Parameswaran, N. Nallamuthu, P. Devendran, E.R. Nagarajan, A. Manikandan, Electrical conductivity studies on ammonium bromide incorporated with zwitterionic polymer blend electrolyte for battery application. Phys. B 515, 89–98 (2017). https://doi.org/10.1016/j.physb.2017.03.043
R.A. Senthil, J. Theerthagiri, J. Madhavan, A.K.M. Arof, High performance dye-sensitized solar cell based on 2-mercaptobenzimidazole doped poly(vinylidinefluoride-co-hexafluoropropylene) based polymer electrolyte. J. Macromol. Sci. A 53(4), 245–251 (2016). https://doi.org/10.1080/10601325.2016.1143322
K. Omri, I. Najeh, L. El Mir, Influence of annealing temperature on the microstructure and dielectric properties of ZnO nanoparticles. Ceram. Int. 42, 8940–8948 (2016). https://doi.org/10.1016/j.ceramint.2016.02.151
K. Omri, A. Bettaibi, K. Khirouni, L. El Mir, The optoelectronic properties and role of Cu concentration on the structural and electrical properties of Cu doped ZnO nanoparticles. Phys. B 537, 167–175 (2018). https://doi.org/10.1016/j.physb.2018.02.025
K. Omri, F. Alharbi, Synthesis and effect of temperature on morphological and photoluminescence properties of TiO2 nanoparticles. Appl. Phys. A 125, 696 (2019). https://doi.org/10.1007/s00339-019-2993-y
S.B. Aziz, M.A. Rasheed, A.M. Hussein, H.M. Ahmed, Fabrication of polymer blend composites based on [PVA-PVP] (1–x):(Ag2S) x (0.01 ≤ x ≤ 0.03) with small optical band gaps: structural and optical properties. Mater. Sci. Semicond. Process. 71, 197–203 (2017). https://doi.org/10.1016/j.mssp.2017.05.035
S.B. Aziz, R.B. Marif, M.A. Brza, A.N. Hassan, H.A. Ahmad, Y.A. Faidhalla, M.F.Z. Kadir, Structural, thermal, morphological and optical properties of PEO filled with biosynthesized Ag nanoparticles: new insights to band gap study. Results Phys. 13, 102220 (2019). https://doi.org/10.1016/j.rinp.2019.102220
S.B. Aziz, H.M. Ahmed, A.M. Hussein, A.B. Fathulla, R.M. Wsw, R.T. Hussein, Tuning the absorption of ultraviolet spectra and optical parameters of aluminium doped PVA based solid polymer composites. J. Mater. Sci. 26, 8022–8028 (2015). https://doi.org/10.1007/s10854-015-3457-6
R.M. Abdullah, S.B. Aziz, S.M. Mamand, A.Q. Hassan, S.A. Hussein, M.F.Z. Kadir, Reducing the crystallite size of spherulites in PEO-based polymer nanocomposites mediated by carbon nanodots and Ag nanoparticles. Nanomaterials 9, 874 (2019). https://doi.org/10.3390/nano9060874
H.T. Ahmeda, O.G. Abdullah, Structural and ionic conductivity characterization of PEO:MC-NH4I proton conducting polymer blend electrolytes-based films. Results Phys. 16, 102861 (2020). https://doi.org/10.1016/j.rinp.2019.102861
C.S. Ramya, S. Selvasekarapandian, G. Hirankumar, T. Savitha, P.C. Angelo, Investigation on dielectric relaxations of PVP– NH4SCN polymer electrolyte. J. Non-Cryst. Solids 354, 1494–1502 (2008). https://doi.org/10.1016/j.jnoncrysol.2007.08.038
S.L. Agrawal, S. Markandey, T. Mridula, M.D. Mrigank, P. Kamalesh, Dielectric relaxation studies on [PEO–SiO2]: NH4SCN nanocomposite polymer electrolyte films. J. Mater. Sci. 44, 6060–6068 (2009). https://doi.org/10.1007/s10853-009-3833-9
S. Ramesh, K.Y. Ng, Characterization of polymer electrolytes based on high molecular weight PVC and Li2SO4. Curr. Appl. Phys. 9, 329–332 (2009). https://doi.org/10.1016/j.cap.2008.03.002
K.P. Radha, S. Selvasekarapandian, S. Karthikeyan, M. Hema, C. Sanjeeviraja, Synthesis and impedance analysis of proton-conducting polymer electrolyte PVA:NH4F. Ionics 19, 1437–1447 (2013). https://doi.org/10.1007/s11581-013-0866-5
R. Hemalatha, M. Alagar, S. Selvasekarapandian, B. Sundaresan, V. Moniha, G. Boopathi, P.C. Selvin, Preparation and characterization of proton-conducting polymer electrolyte based on PVA, amino acid proline, and NH4Cl and its applications to electrochemical devices. Ionics 25, 141–154 (2019). https://doi.org/10.1007/s11581-018-2564-9
S. Sivadevi, S. Selvasekarapandian, S. Karthikeyan, N. Vijaya, M.G.F. Kingslin, C. Sanjeeviraja, Structural and AC impedance analysis of blend polymer electrolyte based on PVA and PAN. Int. J. Sci. Res. 2(10), 1–3 (2013). https://doi.org/10.15373/22778179/OCT2013/121
S. Selvasekarapandian, M. Hema, J. Kawamura, O. Kamishima, Characterization of PVA-NH4NO3 polymer electrolyte and its application in rechargeable proton battery. J. Phys. Soc. Jpn. 79, 163–168 (2010). https://doi.org/10.1143/JPSJS.79SA.163
K.P. Radha, S. Selvasekarapandian, Characterization of PVA: NH4F: ZrO2 composite polymer electrolytes. Int. J. Sci. Res. 1, 118–119 (2012). https://doi.org/10.15373/22778179/OCT2012/43
G. Hirankumar, S. Selvasekarapandian, N. Kuwata, J. Kawamura, T. Hattori, Thermal, electrical and optical studies on the poly (vinyl alcohol) based polymer electrolytes. J. Power Sources 144, 262–267 (2005). https://doi.org/10.1016/j.jpowsour.2004.12.019
M. Hema, S. Selvasekarapandian, D. Arunkumar, A. Sakunthala, H. Nithya, FTIR XRD and AC impedance spectroscopic study on PVA based polymer electrolyte doped with NH4X (X=Cl, Br, I). J. Non-Cryst. Solids 355, 84–90 (2009). https://doi.org/10.1016/j.jnoncrysol.2008.10.009
C.S. Ramya, S. Selvasekarapandian, T. Savitha, G. Hirankumar, Transport mechanism of Cu-ion conducting PVA based solid polymer electrolyte. Ionics 11, 436–441 (2005). https://doi.org/10.1007/BF02430262
G. Hirankumar, S. Selvasekarapandian, M.S. Bhuvaneswari, R. Baskaran, M. Vijayakumar, Ag+ ion transport studies in poly vinyl alcolhol based polymer electrolyte system. J Solid State Electrochem. 10, 193–197 (2006). https://doi.org/10.1007/s10008-004-0612-z
P.D. Vinoth, S. Selvasekarapandian, R. Bhuvaneswari, M. Premalatha, S. Monisha, D. Arunkumar, K. Junichi, Development and characterization of proton conducting polymer electrolyte based on PVA, amino acid glycine and NH4SCN. Solid State Ionics 298, 15–22 (2016). https://doi.org/10.1016/j.ssi.2016.10.016
S. Sikkanthar, S. Karthikeyan, S. Selvasekarapandian, D. Arunkumar, H. Nithya, J. Kawamura, Structural, electrical conductivity and transport analysis of PAN–NH4Cl polymer electrolyte system. Ionics 22, 1085–1094 (2016). https://doi.org/10.1007/s11581-016-1645-x
G. Boopathi, S. Pugalendhi, S. Selvasekarapandian, M. Premalatha, S. Monisha, G. Aristatil, Development of proton conducting biopolymer membrane based on agar-agar for fuel cell. Ionics 23, 2781–2790 (2016). https://doi.org/10.1007/s11581-016-1876-x
M. Hema, S. Selvasekarapandian, D. Arunkumar, A. Sakunthala, H. Nithya, FTIR, XRD and ac impedance spectroscopic study on PVA based polymer electrolyte doped with NH4X (X=Cl, Br, I). J. Non Cryst. Solids 355, 84–90 (2009). https://doi.org/10.1016/j.jnoncrysol.2008.10.009
M.H. Buraidah, A.K. Arof, Characterization of chitosan/PVA blended electrolyte doped with NH4I. J. Non-Cryst. Solids 357, 3261–3266 (2011). https://doi.org/10.1016/j.jnoncrysol.2011.05.021
R. Khalil, Impedance and modulus spectroscopy of poly(vinyl alcohol)-Mg[ClO4]2 salt hybrid films. Appl. Phys. A 12, 1234 (2017). https://doi.org/10.1007/s00339-017-1026-y
I.S. Yahia, M.I. Mohammed, A.M. Nawar, Multifunction applications of TiO2/poly(vinyl alcohol) nanocomposites for laser attenuation applications. Phys. B 556, 48–60 (2019). https://doi.org/10.1016/j.physb.2018.12.031
I.S. Yahia, M.I. Mohammed, Facile synthesis of graphene oxide/PVA nanocomposites for laser optical limiting: band gap analysis and dielectric constants. J. Mater. Sci. 29, 8555–8563 (2018). https://doi.org/10.1007/s10854-018-8869-7
S. Divakara, S. Madhu, R. Somashekar, Stacking faults and microstructural parameters in non-mulberry silk fibres. Pramana J. Phys. 73, 927–938 (2009). https://doi.org/10.1007/s12043-009-0159-8
I.H. Hall, R. Somashekar, Determination of crystal size and disorder from the X-ray diffraction photograph of polymer fibres. 2. Modelling intensity profiles. J. Appl. Cryst. 24, 1051–1059 (1991). https://doi.org/10.1107/S0021889891007707
R. Somashekar, I.H. Hall, P.D. Carr, The determination of crystal size and disorder from X-ray diffraction photographs of polymer fibres. 1. The accuracy of determination of Fourier coefficients of the intensity profile of a reflection. J. Appl. Cryst. 22, 363–371 (1989). https://doi.org/10.1107/S0021889889004085
R. Somashekar, H. Somashekarappa, X-ray diffraction-line broadening analysis: paracrystalline method. J. Appl. Cryst. 30, 147–152 (1997). https://doi.org/10.1107/S0021889896010023
S.-D. Cho, J.-Y. Lee, K.-W. Paik, Effects of particle size on dielectric constant and leakage current of epoxy/barium titanate (BaTiO3) composite films for embedded capacitors. Adv. Electron. Mater. Packag. (Cat. No.01EX506) 63–68 (2001). https://doi.org/10.1109/emap.2001.983959
A. Bouzidi, W. Jilani, I.S. Yahia, H.Y. Zahran, M.A. Assiri, Optical analysis and UV-blocking filter of cadmium iodide-doped polyvinyl alcohol polymeric composite films: synthesis and dielectric properties. J. Inorg. Organomet. Polym Mater. (2020). https://doi.org/10.1007/s10904-020-01534-5
D. Coskun, B. Gunduz, M.F. Coskun, Synthesis, characterization and significant optoelectronic parameters of 1-(7-methoxy-1-benzofuran-2-yl) substituted chalcone derivatives. J. Mol. Struct. 1178, 261–267 (2019). https://doi.org/10.1016/j.molstruc.2018.10.043
B. Gündüz, Optical properties of poly[2-methoxy-5-(3′,7′-dimethyloctyloxy)-1,4- phenylenevinylene] light-emitting polymer solutions: effects of molarities and solvents. Polym. Bull. 72(12), 3241–3267 (2015). https://doi.org/10.1007/s00289-015-1464-7
S.B. Aziz, Morphological and optical characteristics of chitosan(1–x):Cux (4 ≤ x ≤ 12) based polymer nano-composites: optical dielectric loss as an alternative method for tauc’s model. Nanomaterials 7, 444 (2017). https://doi.org/10.1016/j.mssp.2017.05.035
S.B. Aziz, A.Q. Hassan, S.J. Mohammed, W.O. Karim, M.F.Z. Kadir, H.A. Tajuddin, N.N.M.Y. Chan, structural and optical characteristics of PVA:C-dot composites: tuning the absorption of ultra violet (UV) region. Nanomaterials 9(2), 216 (2019). https://doi.org/10.3390/nano9020216
S.B. Aziz, Modifying poly(Vinyl Alcohol) (PVA) from insulator to small-bandgap polymer: a novel approach for organic solar cells and optoelectronic devices. J. Electron. Mater. 45, 736–745 (2016). https://doi.org/10.1007/s11664-015-4191-9
S.B. Aziz, M.A. Brza, M.M. Nofal, R.T. Abdulwahid, S.A. Hussen, A.M. Hussein, W.O. Karim, A comprehensive review on optical properties of polymer electrolytes and composites. Materials 13(17), 3675 (2020). https://doi.org/10.3390/ma13173675
K.S. Hemalatha, K. Rukmani, Concentration dependent dielectric, AC conductivity and sensing study of ZnO-polyvinyl alcohol nanocomposite films. Int. J. Nanotechnol. 14, 961–973 (2017). https://doi.org/10.1504/IJNT.2017.086778
M. Abdelaziz, M.M. Ghannam, Influence of titanium chloride addition on the optical and dielectric properties of PVA films. Phys. B 405(3), 958–964 (2010). https://doi.org/10.1016/j.physb.2009.10.030
S.B. Aziz, M. Rasheed, H. Ahmed, Synthesis of polymer nanocomposites based on [Methyl Cellulose](1–x):(CuS)x (0.02 M ≤ x ≤ 0.08 M) with desired optical band gaps. Polymers 9(12), 194 (2017). https://doi.org/10.3390/polym9060194
Y. Khairy, M.M. Abdel-Aziz, H. Algarni, A.M. Alshehri, I.S. Yahia, H.E. Ali, The optical characteristic of PVA composite films doped by ZrO2 for optoelectronic and block UV-visible applications. Mater. Res. Express. (2019). https://doi.org/10.1088/2053-1591/ab4e34
W. Al-Taa’y, M.A. Nabi, R.M. Yusop, E. Yousif, B.M. Abdullah, J. Salimon, N. Salih, S.I. Zubairi, Effect of nano ZnO on the optical properties of poly (vinyl chloride) films. Int. J. Polym. Sci. (2014). https://doi.org/10.1155/2014/697809
A. Bouzidi, W. Jilani, H. Guermazi, I.S. Yahia, H.Y. Zahran, G.B. Sakr, The effect of zinc iodide on the physicochemical properties of highly flexible transparent poly (vinyl alcohol) based polymeric composite films: opto-electrical performance. J. Mater. Sci. 30, 11799–11806 (2019). https://doi.org/10.1007/s10854-019-01552-1
N. Sabry, M.I. Mohammed, I.S. Yahia, Optical analysis, optical limiting and electrical properties of novel PbI2/PVA polymeric nanocomposite films for electronic optoelectronic applications. Mater. Res. Express 6, 115339 (2019). https://doi.org/10.1088/2053-1591/ab4c24
F.F. Muhammad, S.B. Aziz, S.A. Hussein, Effect of the dopant salt on the optical parameters of PVA:NaNO3 solid polymer electrolyte. J. Mater. Sci. 26, 521–529 (2015). https://doi.org/10.1007/s10854-014-2430-0
S.K. Tripathy, Refractive indices of semiconductors from energy gaps. Opt. Mater. 46, 240–246 (2015). https://doi.org/10.1016/j.optmat.2015.04.026
T.S. Moss, Relations between the refractive index and energy gap of semiconductors. Phys. Stat. Sol. (b) 131, 415 (1985). https://doi.org/10.1002/pssb.2221310202
R.R. Reddy, S. Anjaneyulu, Analysis of the moss and ravindra relations. Phys. Status Sol. (b) 174(2), K91–K93 (1992). https://doi.org/10.1002/pssb.2221740238
M. Anani, C. Mathieu, S. Lebid, Y. Amar, Z. Chama, H. Abid, Model for calculating the refractive index of a III-V semiconductor. Comput. Matter. Sci. 41, 570 (2008). https://doi.org/10.1016/j.commatsci.2007.05.023
V. Kumar, J. K. Singh Ind, Model for calculating the refractive index of different materials. J. Pure Appl. Phys. 48 (2010) 571–574. http://nopr.niscair.res.in/handle/123456789/9962.
F.M. Ali, Synthesis and characterization of a novel erbium doped poly(vinyl alcohol) films for multifunctional optical materials. J. Inorg. Organomet. Polym. 30, 2418–2429 (2020). https://doi.org/10.1007/s10904-019-01386-8
T. Abdel-Baset, M. Elzayat, S. Mahrous, Characterization and optical and dielectric properties of polyvinyl chloride/silica nanocomposites films. Int. J. Polym. Sci. 2016, 13 (2016). https://doi.org/10.1155/2016/1707018
H. Eliasson, I. Albinsson, B.E. Mellander, Conductivity and dielectric properties of AgCF3SO3-PPG. Mater. Res. Bull. 35, 1053–1065 (2000). https://doi.org/10.1016/S0025-5408(00)00309-3
S.M. Salem, Effect of iron on the electrical properties of lead–bismuth glasses. J. Mater. Sci. 44, 5760–5767 (2009). https://doi.org/10.1007/s10853-009-3807-y
D.K. Das-Gupta, Molecular processes in polymer electrets. J. Electrost. 515, 159–166 (2001). https://doi.org/10.1016/S0304-3886(01)00090-0
B.A. Shujahadeen, M.F.Z. Kadir, M.H. Hamsan, H.J. Woo, M.A. Brza, Development of polymer blends based on PVA: POZ with low dielectric constant for microelectronic applications. Sci. Rep. 9, 13163 (2019). https://doi.org/10.1038/s41598-019-49715-8
S.B. Aziz, R.M. Abdullah, Crystalline and amorphous phase identification from the tanδ relaxation peaks and impedance plots in polymer blend electrolytes based on [CS: AgNt] x: PEO(x–1)(10≤x≤50). Electrochim. Acta 285, 30–46 (2018). https://doi.org/10.1016/j.electacta.2018.07.233
N. Kulshrestha, B. Chatterjee, P. Gupta, Characterization and electrical properties of polyvinyl alcohol-based polymer electrolyte films doped with ammonium thiocyanate. Mater. Sci. Eng. B 184, 49–57 (2014). https://doi.org/10.1016/j.mseb.2014.01.012
R. Ramani, R. Ramachandran, G. Amarendra, S. Alam, Direct correlation between free volume and dielectric constant in a fuorine-containing polyimide blend. J. Phys. 618, 012025 (2015). https://doi.org/10.1088/1742-6596/618/1/012025
N. Mazuki, A.P.P. Abdul Majeed, A.S. Samsudin, Study on electrochemical properties of CMC-PVA doped NH4Br based solid polymer electrolytes system as application for EDLC. J. Polym. Res. 27, 135 (2020). https://doi.org/10.1007/s10965-020-02078-5
S. Ramesh, A.H. Yahaya, A.K. Arof, Dielectric behaviour of PVC-based polymer electrolytes. Solid State Ionics 152, 291–294 (2002). https://doi.org/10.1016/S0167-2738(02)00311-9
L.C. Hsu, M.C. Yang, T. Higashihara, W.C. Chen, M. Ueda, Synthesis and characterization of poly(2,6-dialkoxy-1,5- naphthylene)s with low dielectric constants. Polym. J. 50, 277–280 (2018). https://doi.org/10.1038/s41428-017-0011-9
H. Sun, Y. Lv, C. Zhang, X. Zuo, M. Li, X. Yue, Z. Jiang, Materials with low dielectric constant and loss and good thermal properties prepared by introducing perfuorononenyl pendant groups onto poly(ether ether ketone). RSC Adv. 8, 7753 (2018). https://doi.org/10.1039/C7RA13600E
O.G. Abdullah, S.B. Aziz, M.A. Rasheed, Incorporation of NH4NO3 into MC-PVA blend-based polymer to prepare proton-conducting polymer electrolyte films. Ionics 24, 777–785 (2018). https://doi.org/10.1007/s11581-017-2228-1
S.B. Aziz, Role of dielectric constant on Ion transport: reformulated arrhenius equation. Adv. Mater. Sci. Eng. 2016, 2527013 (2016). https://doi.org/10.1155/2016/2527013
A.S. Samsudin, M.I.N. Isa, Structural and ionic transport study on CMC doped NH4Br: a new types of biopolymer electrolytes. J. Appl. Sci. 12(2), 174–179 (2012). https://doi.org/10.3923/jas.2012.174.179
M. Marzantowicz, J.R. Dygas, F. Krok, A. Łasinska, Z. Florjanczyk, E. Zygadło-Monikowska, In situ microscope and impedance study of polymer electrolytes. Electrochim. Acta 51, 1713–1727 (2006). https://doi.org/10.1016/j.electacta.2005.02.098
A.K. Jonscher, Dielectric Relaxation in Solids (Chelsea Dielectrics, London, 1983).
A.S. Marf, R.M. Abdullah, B.A. Shujahadeen, Structural, morphological, electrical and electrochemical properties of PVA: CS-based proton-conducting polymer blend electrolytes. Membranes 10, 71 (2020). https://doi.org/10.3390/membranes10040071
W. Jilani, A. Bouzidi, I.S. Yahia, H. Guermazi, H.Y. Zahran, G. Saker, Effect of organic inorganics on structural properties, linear optics and impedance spectroscopy of methyl orange (C.I. acid orange 52) doped polyvinyl alcohol composite thin films. J. Mater. Sci. 29, 16446–16453 (2018). https://doi.org/10.1007/s10854-018-9736-2
B.A. Shujahadeen, M.A. Brza, M.H. Hamsan, M.F.Z. Kadir, S.K. Muzakir, R.T. Abdulwahid, Effect of ohmic-drop on electrochemical performance of EDLC fabricated from PVA: dextran: NH4I based polymer blend electrolytes. J. Mater. Res. Technol. 9(3), 3734–3745 (2020). https://doi.org/10.1016/j.jmrt.2020.01.110
N.M.J. Rasali, S.K. Muzakir, A.S. Samsudin, A study on dielectric properties of the cellulose derivative-NH4Br-glycerol based the solid polymer electrolyte system. Makara J. Technol. 21, 65–69 (2017). https://doi.org/10.7454/mst.v21i2.3082
T. Siddaiah, P. Ojha, N.O. Gopal, Ch. Ramu, H. Nagabhushana, Thermal, structural, optical and electrical properties of PVA/MAA: EA polymer blend filled with different concentrations of lithium perchlorate. J. Sci. Adv. Mater. Devices 3, 456–463 (2018). https://doi.org/10.1016/j.jsamd.2018.11.004
A.P. Fonseca, D.S. Rosa, F. Gaboardi, S. Neves, Development of a biodegradable polymer electrolyte for rechargeable batteries. J. Power Sources 155(2), 381–384 (2006). https://doi.org/10.1016/j.jpowsour.2005.05.004
S.B. Aziz, Occurrence of electrical percolation threshold and observation of phase transition in chitosan(1–x): AgIx (0.05_x_0.2)-based ion-conducting solid polymer composites. Appl. Phys. A 122, 706 (2016). https://doi.org/10.1007/s00339-016-0235-0
S.B. Aziz, Z.H.Z. Abidin, Electrical and morphological analysis of chitosan: AgTf solid electrolyte. Mater. Chem. Phys. 144, 2802–2886 (2014). https://doi.org/10.1016/j.matchemphys.2013.12.029
S.B. Aziz, R.M. Abdullah, M.A. Rasheed, H.M. Ahmed, Role of ion dissociation on DC conductivity and silver nanoparticle formation in PVA:AgNt based polymer electrolytes: deep insights to ion transport mechanism. Polymers 9, 338 (2017). https://doi.org/10.3390/polym9080338
H. Hassib, A.A. Razik, Dielectric properties and AC conduction mechanism for 5,7-dihydroxy- 6-formyl-2-methylbenzo-pyran-4-one bis-schi_base. Solid State Commun. 147, 345–349 (2008). https://doi.org/10.1016/j.ssc.2008.06.034
A. Manjunath, T. Deepa, N.K. Supreetha, M. Irfan, Studies on AC electrical conductivity and dielectric properties of PVA/NH4NO3 solid polymer electrolyte films. Adv. Mater. Phys. Chem. 5, 295–301 (2015). https://doi.org/10.4236/ampc.2015.58029
R. Macovez, M. Zachariah, M. Romanini, P. Zygouri, D. Gournis, J.L. Tamarit, Hopping conductivity and polarization effects in a fullerene derivative salt. J. Phys. Chem. C 118, 12170–12175 (2014). https://doi.org/10.1021/jp503298e
S.B. Aziz, Z.H.Z. Abidin, Electrical conduction mechanism in solid polymer electrolytes: new concepts to arrhenius equation. J. Soft Matter 2013, 8 (2013). https://doi.org/10.1155/2013/323868
H.T. Ahmed, O.G. Abdullah, Structural and ionic conductivity characterization of PEO:MC-NH4I proton conducting polymer blend electrolytes based films. Results Phys. 16, 102861 (2020). https://doi.org/10.1016/j.rinp.2019.102861
S.B. Aziz, T.J. Woo, M.F.Z. Kadir, H.M. Ahmed, A conceptual review on polymer electrolytes and ion transport models. J. Sci. 3, 1–17 (2018). https://doi.org/10.1016/j.jsamd.2018.01.002
S.B. Aziz, Study of electrical percolation phenomenon from the dielectric and electric modulus analysis. Bull. Mater. Sci. 38, 1597–1602 (2015). https://doi.org/10.1007/s12034-015-0978-9
S.B. Aziz, R.M. Abdullah, M.F.Z. Kadir, H.M. Ahmed, Non suitability of silver ion conducting polymer electrolytes based on chitosan mediated by barium titanate (BaTiO3) for electrochemical device applications. Electrochim. Acta (2018). https://doi.org/10.1016/j.electacta.2018.11.081
S.B. Aziz, S.M. Mamand, The study of dielectric properties and conductivity relaxation of ion conducting chitosan:NaTf based solid electrolyte. Int. J. Electrochem. Sci. 13, 10274–10288 (2018). https://doi.org/10.20964/2018.11.05
S.B. Aziz, R.M. Abdullah, Crystalline and amorphous phase identification from the tanδ relaxation peaks and impedance plots in polymer blend electrolytes based on [CS:AgNt]x:PEO(x–1) (10≤x≤50). Electrochim. Acta 285, 30–46 (2018). https://doi.org/10.1016/j.electacta.2018.07.233
Acknowledgements
The authors would like to acknowledge the support of the Ministry of Education, Kingdom of Saudi Arabia, for this research through a Grant (PCSED-019-18) under the Promising Centre for Sensors and Electronic Devices (PCSED) at Najran University, Kingdom of Saudi Arabia. Also, the authors express their appreciation to the Deanship of Scientific Research at King Khalid University for funding this work through research groups program under Grant Number (R.G.P.2/64/40).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mohammed, M.I., Bouzidi, A., Zahran, H.Y. et al. Ammonium iodide salt-doped polyvinyl alcohol polymeric electrolyte for UV-shielding filters: synthesis, optical and dielectric characteristics. J Mater Sci: Mater Electron 32, 4416–4436 (2021). https://doi.org/10.1007/s10854-020-05184-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-020-05184-8