Abstract
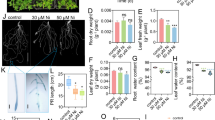
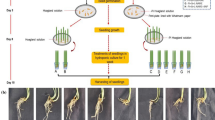
Copper nanoparticles have enhanced the germination and wheat development. To explain the effects of copper nanoparticles on shoot of Pakistan-13 and NARC-11, proteomic technique was used. The physiological responses such as weights/lengths of seedling, shoot, and root of wheat varieties were increased on 10-ppm copper nanoparticle exposure. The number of proteins related to protein metabolism was increased in Pakistan-13 while protein metabolism and photosynthesis-related proteins were increased in NARC-11, treated with copper nanoparticles compared to untreated plants. Abundance of proteins related to glycolysis and tricarboxylic acid cycle was increased on copper nanoparticle exposure in Pakistan-13 and NARC-11. However, the abundance of proteins related to photosynthesis and tetrapyrole synthesis was decreased on copper nanoparticle exposure in Pakistan-13 and NARC-11. Chlorophyll content such as chlorophyll a, chlorophyll b, and total chlorophyll was decreased on copper nanoparticle exposure in Pakistan-13 and NARC-11. The rate of photosynthesis and carbon assimilation decreased on copper nanoparticle exposure. These results suggest that copper nanoparticles mend the seedling growth of wheat, which might be concomitant with the enhancement of protein abundance related to glycolysis and tricarboxylic acid cycle in wheat varieties.









Similar content being viewed by others
References
Abbas SH, Sohail M, Hussain I, Saleem M, Qamar M, Aslam M, Imran M (2014) Grain yield of newly developed wheat cultivar (narc 2011) as enhanced by foliar application of humic acid under rainfed conditions. Sarhad J Agric 30:173–178
Ahmed A (2014) Pakistan develops Ug99-resistant wheat varieties. Published Feb 23, 2014 08:16AM. https://www.dawn.com/news/1307208
Alam MM, Siwar C, Molla RI, Toriman ME, Talib B (2011) Climate change and vulnerability of paddy cultivation in north-West Selangor, Malaysia: a survey of farmers’ assessment. Voice of Academia 6:45–56
Arnon DI (1949) Copper enzymes in isolated chloroplasts, polyphenoxidase in beta vulgaris. Plant Physiol 24:1–15
Auguar-Fernandez MA, Hullmann A (2014) A boost for safer nanotechnology. Nano Today 2:56
Balsemao-Pires E, Jaillais Y, Olson BJ, Andrade LR, Umen JG, Chory J, Sachetto-Martins G (2011) The Arabidopsis translocator protein (AtTSPO) is regulated at multiple levels in response to salt stress and perturbations in tetrapyrrole metabolism. BMC Plant Biol 11:880
Barrena R, Casals E, Colon J, Font X, Sanchez A, Puntes V (2009) Evaluation of the ecotoxicity of model nanoparticles. Chemosphere 75:850–857
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brosch M, Yu L, Hubbard T, Choudhary J (2009) Accurate and sensitive peptide identification with mascot percolator. J Proteome Res 8:3176–3181
Cao Z, Rossi L, Stowers C, Zhang W, Lombardini L, Ma X (2018) The impact of cerium oxide nanoparticles on the physiology of soybean (Glycine max (L.) Merr.) under different soil moisture conditions. Environ Sci Pollut Res Int 25:930–939
Challinor AJ, Ewert F, Arnold S, Simelton E, Fraser E (2009) Crops and climate change: progress, trends, and challenges in simulating impacts and informing adaptation. J Exp Bot 60:2775–2789
Chang WWP, Huang L, Shen M, Webster C, Burlingame AL, Roberts JKM (2000) Patterns of protein synthesis and tolerance of anoxia in root tips of maize seedlings acclimated to a low-oxygen environment, and identification of proteins by mass spectrometry. Plant Physiol 122:295–318
Chaves MM (1991) Effects of water deficits on carbon assimilation. J Exp Bot 42:1–16
Chen R, Ratnikova TA, Stone MB, Lin S, Lard M, Huang G, Hudson JS, Ke PC (2010) Differential uptake of carbon nanoparticles by plant and mammalian cells. Small 6:612–617
Collins JF, Prohaska JR, Knutson MD (2010) Metabolic crossroads of iron and copper. Nutr Rev 68:133–147
Czarnecki O, Grimm B (2012) Post-translational control of tetrapyrrole biosynthesis in plants, algae, and cyanobacteria. J Exp Bot 63:1675–1687
Delfani M, Firouzabadi MB, Farrokhi N, Makarian H (2014) Some physiological responses of black-eyed pea to iron and magnesium nanofertilizers. Commun Soil Sci Plant Anal 45:530–540
Dhawan A, Bajpayee M, Parmar D (2009) Comet assay: a reliable tool for the assessment of DNA damage in different models. Cell Biol Toxicol 25:5–32
Dietz KJ, Herth S (2011) Plant nanotoxicology. Trends in plant sci 16:582–589
Dimkpa CO, McLean JE, Latta DE, Manangon E, Britt DW, Johnson WP, Boyanov MI, Anderson AJ (2013) CuO and ZnO nanoparticles: phtotoxicity, metal speciation, and induction of oxidative stress in sand-grown wheat. J Nanopart Res 14:1125–1129
Dreher K, Callis J (2007) Ubiquitin, hormones and biotic stress in plants. Annu Bot 99:787–822
Du W, Tan W, Yin Y, Ji R, Peralta-Videa JR, Guo H, Gardea-Torresdey JL (2018) Differential effects of copper nanoparticles/microparticles in agronomic and physiological parameters of oregano (Origanum vulgare). Sci Total Environ 618:306–312
Du W, Gardea-Torresdey JL, Ji R, Yin Y, Zhu J, Peralta-Videa JR, Guo H (2015) Physiological and biochemical changes imposed by CeO2 nanoparticles on wheat: a life cycle field study. Environ Sci Technol 49:884–893
Du W, Sun Y, Ji R, Zhu J, Wu J, Guo H (2011) TiO2 and ZnO nanoparticles negatively affect wheat growth and soil enzyme activities in agricultural soil. J Environ Monit 13:822–828
Frazier TP, Burklew CE, Zhang B (2014) Titanium dioxide nanoparticles affect the growth and micro RNA expression of tobacco(Nicotiana tabacum). Funct Integr Genomics 14:75–83
Ghafariyan MH, Malakouti MJ, Dadpour MR, Stroeve P, Mahmoudi M (2013) Effects of magnetite nanoparticles on soybean chlorophyll. Environ. Sci. Technol. 47:10645–10652
Guicherd P, Peltier JP, Gout E, Bligny R, Marigo G (1997) Osmotic adjustment in Fraxinus excelsior L. malate and mannitol accumulation in leaves under drought conditions. Trees 11:155–161
Gutierrez L, Wuytswinkel OV, Castelain M, Bellini C (2007) Combined networks regulating seed maturation. Trends Plant Sci 12:294–300
Hochstrasser M (1996) Ubiquitin-dependent protein degradation. Annu Rev Genet 30:405–439
Hodges M, Flesch V, Gálvez S, Bismuth E (2003) Higher plant NADP dependent isocitrate dehydrogenases, ammonium assimilation and NADPH production. Plant Physiol Biochem 41:577–585
Jacob DL, Barchardt JD, Navaratnam L, Otte ML, Bezbaruah A (2013) Uptake and translocation of Ti from nanoparticles in crops and wetland plants. Int J Phytoremediat 15:142–153
Jiang Y, Huang B (2002) Protein alterations in tall fescue in response to drought stress and abscisic acid. Crop Sci 42:202–207
Johnson AC, Bowesa MJ, Crossley A, Jarvie HP, Jurkschat K, Jürgensa MD, Lawlor AJ, Park B, Rowland P, Spurgeon D, Svendsen C, Thompson IP, Barnes RJ, Williams RJ, Xua N (2011) An assessment of the fate, behaviour and environmental risk associated with sunscreen TiO2 nanoparticles in UK field scenarios. Sci Total Environ 409:2503–2510
Kang G, Li G, Xu W, Peng X, Han Q, Zhu Y, Guo T (2012) Proteomics reveals the effects of salicylic acid on growth and tolerance to subsequent drought stress in wheat. J Proteome Res 11:6066–6079
Kausar R, Arshad M, Shahzad A, Komatsu S (2013) Proteomics analysis of sensitive and tolerant barley genotypes under drought stress. Amino Acids 44:345–359
Khatoon A, Rehman S, Hiraga S, Makino T, Komatsu S (2012) Organ-specific proteomics analysis for identification of response mechanism in soybean seedlings under flooding stress. J Proteome 75:5706–5723
Khot LR, Sankaran S, Maja JM, Ehsani R, Schuster EW (2012) Applications of nanomaterials in agricultural production and crop protection: a review. Crop Prot 35:64–70
Komatsu S, Han C, Nanjo Y, Altaf-Un-Nahar M, Wang K, He D, Yang P (2013) A label-free quantitative proteomic analysis of abscisic acid effect in early-stage soybean under flooding. J Proteome Res 12:4769–4784
Larue C, Laurette J, Herlin-Boime N, Khodja H, Fayard B, Flank A, Frisset F, Brisset M (2012) Accumulation, translocation, and impact of TiO2 nanoparticles in wheat (Triticum aestivum spp.): influence of diameter and crystal phase. Sci Total Environ 43:197–208
Lee WM, An YJ, Yoon H, Kweon HS (2008) Toxicity and bioavailability of copper nanoparticles to the terrestrial plants mung bean (Phaseolus radiatus) and wheat (Triticum aestivum): plant agar test for water-insoluble nanoparticles. Nanomater Environ 27:1915–1921
Li J, Hu J, Xiao L, Wang Y, Wang X (2018a) Interaction mechanisms between α-Fe2O3, γ-Fe2O3 and Fe3O4 nanoparticles and Citrus maxima seedlings. Sci Total Environ 625:677–685
Li M, Chen D, Liu Y, Chuang CY, Kong F, Harrison PJ, Zhu X, Jiang Y (2018b) Exposure of engineered nanoparticles to Alexandrium tamarense (Dinophyceae): healthy impacts of nanoparticles via toxin-producing dinoflagellate. Sci Total Environ 610-611:356–366
Lin D, Xing B (2007) Phytotoxicity of nanoparticles: inhibition of seed germination and root growth. Environ Pollut 150:243–250
Ma H, Williams PL, Diamond SA (2013) Ecotoxicity of manufactured ZnO nanoparticles -a review. Environ Pollut 172:76–85
Ma Y, Kuang L, He X, Bai W, Ding Y, Zhang Z (2010) Effects of rare earth oxide nanoparticles on root elongation of plants. Chemosphere 78:273–279
Mechin V, Thevenot C, Guilloux ML, Prioul JL, Damerval C (2007) Developmental analysis of maize endosperm proteome suggests a pivotal role for pyruvate orthophosphate dikinase. Plant Physiol 143:1203–1219
Millar AH, Sweetlove LJ, Giege P, Leaver CJ (2001) Analysis of the Arabidopsis mitochondrial proteome. Plant Physiol 127:1711–1727
Miller GW, Huang IJ, Welkie GW, Pushnik JC (1995) Function of iron in plants with special emphasis on chloroplasts and photosynthetic activity. Iron Nutrition in Soils and Plants 59:19–28
Mohammadi P, Moieni A, Hiraga S, Komatsu S (2012) Organ-specific proteomic analysis of drought-stressed soybean seedlings. J Proteome 75:1906–1923
Monica RC, Cremonini R (2009) Nanoparticles and higher plants. Caryologia 62:161–165
Moon J, Parry G, Estelle M (2004) The ubiquitin-proteasome pathway and plant development. Plant Cell 16:3181–3195
Muckhopadhyay D, Riezman H (2007) Proteasome-independent functions of ubiquitin in endocytosis and signaling. Sci 315:201–205
Munns R, James RA, Läuchli A (2006) Approaches to increasing the salt tolerance of wheat and other cereals. J Exp Bot 57:1025–1043
Musante C, White JC (2012) Toxicity of silver and copper to Cucurbita pepo: differential effects of nano and bulk-size particles. Environ Toxicol 27:510–517
Musrati RA, Kollarova M, Mernik N, Mikulasova D (1998) Malate dehydrogenase: distribution, function and properties. Gen Physiol Biophys 17:193–210
Mustafa G, Sakata K, Komatsu S (2015) Proteomic analysis of flooded soybean roots exposed to aluminum oxide nanoparticles. J Proteome 128:280–297
Mustafa G, Sakata K, Komatsu S (2016) Proteomic analysis of soybean root exposed to varying sizes of silver nanoparticles under flooding stress. J Proteome 148:113–125
Nair PM, Chung IM (2014) A mechanistic study on the toxic effect of copper oxide nanoparticles in soybean (Glycine max L.) root development and lignification of root cells. Biol Trace Elem Res 162:342–352
Nair R, Varghese SH, Nair BG, Maekawa T, Yoshida Y, Kumar DS (2010) Nanoparticulate material delivery to plants. Plant Sci 179:154–163
Nanjo Y, Skultety L, Uvackova L, Klubicova K, Hajduch M, Komatsu S (2012) Mass spectrometry-based analysis of proteomic changes in the root tips of flooded soybean seedlings. J Proteome Res 11:372–385
Nekrasova OE, Mendez MG, Chernoivanenko IS, Tyurin-Kuzmin PA, Kuczmarski ER, Gelfand VI, Goldman RD, Minin AA (2011) Vimentin intermediate filaments modulate the motility of mitochondria. Mol Biol Cell 22:2282–2289
Ngo QB, Dao TH, Nguyen HC, Tran XT, Nguyen TV, Khuu TD, Huynh TH (2014) Effects of nanocrystalline powders (Fe, co and cu) on the germination, growth, crop yield and product quality of soybean (Vietnamese species DT-51). Adv Nat Sci Nanosci Nanotechnol 5:1–7
Nowack B, Bucheli TD (2007) Occurrence, behavior and effects of nanoparticles in the environment. Environ Pollut 150:5–22
Oh WM, Roy SK, Kamal AH, Cho K, Cho SW, Park CS, Choi JS, Komatsu S, Woo SH (2014) Proteome analysis of roots of wheat seedlings under aluminum stress. Mol Biol Rep 41:671–681
Olsen JV, de Godoy LMF, Li G, Macek B, Mortensen P, Pesch R, Makarov A, Lange O, Horning S, Mann M (2005) Parts per million mass accuracy on an orbitrap mass spectrometer via lock mass injection into a C-trap. Mol Cell Proteomics 4:2010–2021
Oukarroum A, Barhoumi L, Pirastru L, Dewez D (2013) Silver nanoparticle toxicity effect on growth and cellular viability of the aquatic plant Lemna gibba. Environ Toxicol Chem 32:902–907
Pakistan Agriculture Research Council (PARC) (2017) Wheat and barley research program. http://www.parc.gov.pk/index.php/en/csi/137-narc/crop-sciences-institue/710-wheat-and-barley-research-program
Perreault F, Popovic R, Dewez D (2014) Different toxicity mechanisms between bare and polymer-coated copper oxide nanoparticles in Lemna gibba. Environ Pollut 185:219–227
Pokhrel LR, Dubey B (2013) Evaluation of developmental responses of two crop plants exposed to silver and zinc oxide nanoparticles. Sci Total Environ 45:321–332
Quarrie SA, Quarrie SP, Radosevic D, Rancic D, Kaminska A, Barnes JD, Leverington M, Ceoloni C, Dodig D (2006) Dissecting a wheat QTL for yield present in a range of environments: from the QTL to candidate genes. J Exp Bot 57:2627–2637
Rafique G, Arshad N, Khakhar MF, Qazi IA, Hamza A, Vivic N (2014) Growth response of wheat to titania nanoparticles application. NJES 7:42–46
Rastogi A, Zivcak M, Sytar O, Kalaji HM, He X, Mbarki S, Brestic M (2017) Impact of metal and metal oxide nanoparticles on plant: a critical review. Front Chem 5:78
Riahi-Madvar A, Rezaee F, Alili V (2012) Effects of alumina nanoparticles on morphological properties and antioxidant system of Triticum aestivum. Iranian J Plant Pathol 3:595–603
Rico CM, Peralta-Videa JR, Gardea-Torresdey JL (2015) Differential effects of cerium oxide nanoparticles on rice, wheat, and barley roots: a Fourier transform infrared (FT-IR) microspectroscopy study. Appl Spectrosc 69:287–295
Roco MC (2001) International strategy for nanotechnology research and development. J Nanopart Res 3:353–360
Rogalski M, Schottler MA, Thiele W, Schuleze WX, Bock R (2008) Rpl33, a nonessential plastid-encoded ribosomal protein in tobacco, is required under cold stress conditions. Plant Cell 20:2221–2237
Rukes, Kan L, Mulkey TJ (1994) Measurement on the effects of light quality and other factors on the rate of photosynthesis. Biosci 20:7–11
Sairam PK, Dharmar K, Chinnusamv V, Meena RI (2009) Waterlogging-induced increase in sugar mobilization, fermentation, and related gene expression in the roots of mung bean (Vigna radiata). J Plant Physiol 166:602–616
Sang T, Shan X, Li B, Shu S, Sun J, Guo S (2016) Comparative proteomic analysis reveals the positive effect of exogenous spermidine on photosynthesis and salinity tolerance in cucumber seedlings. Plant Cell Rep 35:1769–1782
Schlicke H, Richter A, Rothbart M, Brzezowski P, Hedtke B, Grimm B (2015) Function of tetrapyrroles, regulation of tetrapyrrole metabolism and methods for analyses of tetrapyrroles. Procedia Chem 14:171–175
Shah V, Belozerova I (2009) Influence of metal nanoparticles on the soil microbial community and germination of lettuce seeds. Water Air Soil Pollut 197:143–148
Sharma P, Bhatt D, Zaidi MGH, Pardha Saradhi P, Khanna PK, Arora S (2012) Silver nanoparticle-mediated enhancement in growth and antioxidant status of Brassica juncea. Appl Biochem Biotechnol 167:2225–2233
Shewry PR (2008) Wheat. J Exp Bot 60:1537–1553
Shi J, Abid AD, Kennedy IM, Hristova KR, Silk WK (2011) Toduck weeds (Landoltia punctata), nanoparticulate copper oxide is more inhibitory than the soluble copper in the bulk solution. Environ Pollut 159:1277–1282
Shinoda K, Tomita M, Ishihama Y (2010) emPAI calc for the estimation of protein abundance from large scale identification data by liquid chromatography-tendem mass spectrometry. Bioinformatics 26:576–577
Singh D, Kumar A (2015) Effects of nano silver oxide and silver ions on growth of Vigna radiate. Bull Environ Contam Toxicol 95:379–384
Song U, Shin M, Lee G, Roh J, Kim Y, Lee EJ (2013) Functional analysis of TiO2 nanoparticle toxicity in three plant species. Biol Trace Elem Res 155:93–103
Stone SL (2014) The role of ubiquitin and the 26S proteasome in plant abiotic stress signaling. Front Plant Sci. https://doi.org/10.3389/fpls(2014)00135
Sun D, Hussain HI, Yi Z, Rookes JE, Kong L, Cahill DMM (2016) Mesoporous silica nanoparticles enhance seedling growth and photosynthesis in wheat and lupin. Chemosphere 152:81–91
Tanaka R, Tanaka A (2007) Tetrapyrrole biosynthesis in higher plants. Annu Rev Plant Biol 58:321–346
Thornton PK, Ericksen PJ, Herrero M, Challinor AJ (2010) Climate variability and vulnerability to climate change: a review. Glob Change Biol 20:3313–3328
Usadel B, Nagel A, Thimm O, Redestig H, Blaesing OE, Palacios-Rofas N, Selbig J, Hannemann J, Piques MC, Steinhauser D, Scheible WR, Gibon Y, Morcuende R, Weicht D, Meyer S, Stitt M (2005) Extension of the visualization tool MapMan to allow statistical analysis of arrays, display of corresponding genes, and comparison with known responses. Plant Physiol 138:1195–1204
Usadel B, Poree F, Nagel N, Lohse M, Czedik-Eysenberg A, Stitt M (2009) A guide to using MapMan to visualize and compare omics in plants: a case study in the crop species, maize. Plant Cell Environ 32:1211–1229
Vannini C, Domingo G, Onelli O, Mattia FD, Bruni I, Marsoni M, Bracale M (2014) Phytotoxic and genotoxic effects of silver nanoparticles exposure on germinating wheat seedlings. J Plant Physiol 171:1142–1148
Vierstra PD (2009) The ubiquitin/26S proteasome system at the nexus of plant biology. Nat Rev Mol Cell Biol 10:385–397
Wang H, Kou X, Pei Z, Xiao JQ, Shan X, Xing B (2011) Physiological effects of magnetite (Fe3O4) nanoparticles on perennial ryegrass (Lolium perenne L.) and pumpkin (Cucurbita mixta) plants. Nanotoxicology 5:30–42
Wang X, Han H, Liu X, Gu X, Chen K, Lu D (2012) Multi-walled carbon nanotubes can enhance root elongation of wheat (Triticum aestivum) plants. J Nanopart Res 14:841
Wiesner M, Lowry RGV, Alvarez P, Dionysiou D, Biswas P (2006) Assessing the risks of manufactured nanomaterials. Environ. Sci. Technol. 40:4336–4345
Wu J, Lu H, Zhu G, Chen L, Chang Y, Yu R (2017) Regulation of membrane fixation and energy production/conversion for adaptation and recovery of ZnO nanoparticle impacted Nitrosomonas europaea. Appl Microbiol Biotechnol 101:2953–2965
Yang L, Watts DJ (2005) Particles surface characteristics may play an important role in phtotoxicity of alumina nanoparticles. Toxicol Lett 158:122–132
Yang T, Xu Z, Wen J, Yang L (2009) Factors influencing bioleaching copper from waste printed circuit boards by Acidithiobacillus ferrooxidans. Hydrometallurgy 97:29–32
Yasmeen F, Raja NI, Mustafa G, Sakata K, Komatsu S (2016a) Quantitative proteomic analysis of post-flooding recovery in soybean root exposed to aluminum oxide nanoparticles. J Proteome 143:136–150
Yasmeen F, Raja NI, Razzaq A, Komatsu S (2016b) Gel-free/label-free proteomic analysis of wheat shoot in stress tolerant varieties under iron nanoparticles exposure. Biochim Biophys Acta 1864:1586–1598
Yasmeen F, Raja NI, Razzaq A, Komatsu S (2017) Proteomic and physiological analyses of wheat seeds exposed to copper and iron nanoparticles. Biochim Biophys Acta 1865:28–42
Yasmeen F, Razzaq A, Iqbal MN, Jhanzab HM (2015) Effect of silver, copper and iron nanoparticles on wheat germination. Int J Biosci 6:112–117
Yuan L, Richardson CJ, Ho M, Willis CW, Colman BP, Wiesner MR (2018) Stress responses of aquatic plants to silver nanoparticles. Environ. Sci. Technol. 52:2558–2565
Zheng L, Hong F, Lu S, Liu C (2005) Effect of nano-TiO2 on strength of naturally aged seeds and growth of spinach. Biol Trace Elem Res 105:83–91
Zhou DJS, Li L, Wang Y, Weng N (2011) Quantifying the adsorption and uptake of CuO nanoparticles by wheat root based on chemical extractions. J Environ Sci 23:1852–1857
Zhu H, Han J, Xiao JQ, Jin Y (2008) Uptake, translocation and accumulation of manufactured iron oxide by pumpkin plants. J Environ Monit 10:713–717
Acknowledgements
The authors acknowledge the financial support provided by the Higher Education Commission Pakistan through International Research Support Initiative Program.
Author information
Authors and Affiliations
Contributions
FY and SK conceived and designed research. FY conducted experiments. SK, NI, and NIR contributed new reagents or analytical tools. FY and SK analyzed data. FY wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Key Message
This article has highlighted the effects of Cu NPs on morphology of wheat varieties and change in proteomics enabled us to understand the mechanism targeted by Cu NPs.
Rights and permissions
About this article
Cite this article
Yasmeen, F., Raja, N.I., Ilyas, N. et al. Quantitative Proteomic Analysis of Shoot in Stress Tolerant Wheat Varieties on Copper Nanoparticle Exposure. Plant Mol Biol Rep 36, 326–340 (2018). https://doi.org/10.1007/s11105-018-1082-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11105-018-1082-2