Abstract
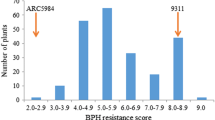
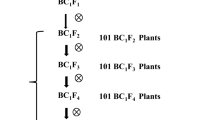
The brown planthopper (Nilaparvata lugens Stål; BPH) is one of the most serious rice pests worldwide. Growing resistant varieties is the most effective way to manage this insect, and wild rice species are a valuable source of resistance genes for developing resistant cultivars. BPH27 derived from an accession of Guangxi wild rice, Oryza rufipogon Griff. (Accession no. 2183, hereafter named GX2183), was primarily mapped to a 17-cM region on the long arm of the chromosome four. In this study, fine mapping of BPH27 was conducted using two BC1F2 populations derived from introgression lines of GX2183. Insect resistance was evaluated in the BC1F2 populations with 6,010 individual offsprings, and 346 resistance extremes were obtained and employed for fine mapping of BPH27. High-resolution linkage analysis defined the BPH27 locus to an 86.3-kb region in Nipponbare. Regarding the sequence information of rice cultivars, Nipponbare and 93-11, all predicted open reading frames (ORFs) in the fine-mapping region have been annotated as 11 types of proteins, and three ORFs encode disease-related proteins. Moreover, the average BPH numbers showed significant differences in 96–120 h after release in comparisons between the preliminary near-isogenic lines (pre-NILs, lines harboring resistance genes) and BaiR54. BPH growth and development were inhibited and survival rates were lower in the pre-NIL plants compared with the recurrent parent BaiR54. The pre-NIL exhibited 50.7 % reductions in population growth rates (PGR) compared to BaiR54. The new development in fine mapping of BPH27 will facilitate the efforts to clone this important resistant gene and to use it in BPH-resistance rice breeding.






Similar content being viewed by others
References
Alam SN, Cohen MB (1998a) Detection and analysis of QTLs for resistance to the brown planthopper, Nilaparvata lugens, in a doubled-haploid rice population. Theor Appl Genet 97:1370–1379
Alam SN, Cohen MB (1998b) Durability of brown planthopper, Nilaparvata lugens, resistance in rice variety IR64 in greenhouse selection studies. Entomol Exp Appl 89:71–78
Athwal DS, Pathak MD, Bacalangco EH, Pura CD (1971) Genetics of resistance to brown planthoppers and green leafhoppers in Oryza sativa L. Crop Sci 11:747–750
Cha YS, Ji H, Yun DW, Ahn BO, Lee MC, Suh SC, Lee CS, Ahn EK, Jeon YH, Jin ID, Sohn JK, Koh HJ, Eun MY (2008) Fine mapping of the rice Bph1 gene, which confers resistance to the brown planthopper (Nilaparvata lugens Stål), and development of STS markers for marker-assisted selection. Mol Cells 26(2):146–151
Chen JW, Wang L, Pang XF, Pan QH (2006) Genetic analysis and fine mapping of a rice brown planthopper (Nilaparbata lugens Stål) resistance gene bph19(t). Mol Genet Genomics 275:321–329
Cohen MB, Alam SN, Medina EB, Bernal CC (1997) Brown planthopper, Nilaparvata lugens, resistance in rice cultivar IR64: mechanism and role in successful N. lugens management in Central Luzon, Philippines. Entomol Exp Appl 85:221–229
Daniel X, Lacomme C, Morel JB, Roby D (1999) A novel myb oncogene homolog in Arabidopsis thaliana related to the hypersensitive cell death. Plant J 20:57–66
Du B, Zhang WL, Liu BF, Hu J, Wei Z, Shi ZY, He RF, Zhu LL, Chen RZ, Han B, He GC (2009) Identification and characterization of Bph14, a gene conferring resistance to brown planthopper in rice. Proc Natl Acad Sci USA 106(52):22163–22168
Edwards OR (2001) Interspecific and intraspecific variation in the performance of three pest aphid species on five grain legume hosts. Entomol Exp Appl 100:21–30
Guzzo F, Portaluppi P, Grisi R, Barone S, Zampieri S, Franssen H, Levi M (2005) Reduction of cell size induced by enod40 in Arabidopsis thaliana. J Exp Bot 56(412):507–513
Hirabayashi H, Ogawa T (1995) RFLP mapping of Bph-1 (brown planthopper resistance gene) in rice. Breed Sci 45:369–371
Hirabayashi H, Angeles ER, Kaji R, Ogawa T, Brar DS, Khush GS (1998) Identification of brown planthopper resistance gene derived from O. officinalis using molecular markers in rice. Breed Sci 48(Suppl):82 (in Japanese)
Hospital F, Chevalet C, Mulsant P (1992) Using markers in gene introgression breeding programs. Genetics 132:199–210
Hu XB, Song FM, Zheng Z (2006) Molecular cloning and expression analysis of rice OsTVLP1, encoding a protein with similarity to TGF-receptor interacting proteins and vacuolar assembly Vam6p/Vps39p proteins. DNA Seq 17:152–158
Huang Z, He GC, Shu LH, Li XH, Zhang QF (2001) Identification and mapping of two brown planthopper resistance genes in rice. Theor Appl Genet 102:929–934
IRRI (1988) Standard evaluation system for rice. International Rice Research Institute, Manila
Ishii T, Brar DS, Multani DS, Khush GS (1994) Molecular tagging of genes for brown planthopper resistance and earliness introgressed from Oryza australiensis into cultivated rice, O. sativa. Genome 37:217–221
Jairin J, Phengrat K, Teangdeerith S, Vanavichit A, Toojinda T (2007) Mapping of a broad-spectrum brown planthopper resistance gene, Bph3, on rice chromosome 6. Mol Breed 19:35–44
Jena KK, Jeung JU, Lee JH, Choi HC, Brar DS (2006) High-resolution mapping of a new brown planthopper (BPH) resistance gene, Bph18(t), and marker-assisted selection for BPH resistance in rice (Oryza sativa L.). Theor Appl Genet 112:288–297
Kabis MA, Khush GS (1988) Genetic analysis of resistance to brown planthopper in rice, Oryza sativa L. Plant Breed 100:54–58
Kawaguchi M, Murata K, Ishii T, Takumi S, Mori N, Nakamura C (2001) Assignment of a brown planthopper (Nilaparvata lugens Stål) resistance gene bph4 to rice chromosome 6. Breed Sci 51:13–18
Khush GS, Karim AR, Angeles EG (1985) Genetics of resistance of rice cultivar ARC10550 to Bangladesh brown planthopper biotype. J Genet 64(2):121–125
Klingler J, Creasy R, Gao LL, Nair RM, Calix AS, Jacob HS, Edwards OR, Singh KB (2005) Aphid resistance in Medicago truncatula involves antixenosis and phloem-specific, inducible antibiosis, and maps to a single locus flanked by NBS-LRR resistance gene analogs. Plant Physiol 137:1445–1455
Lakshminarayana A, Khush GS (1977) New genes for resistance to the brown planthopper in rice. Crop Sci 17:96–100
Li RB, Li LS, Wei SM, Wei YP, Chen YZ, Bai DL, Yang L, Huang FK, Lv WL, Zhang XJ, Li XY, Yang XQ, Wei YW (2006) The evaluation and utilization of new genes for brown planthopper resistance in common wild rice (Oryza rufipogon Griff.). Mol Plant Breed 4(3):365–371
Li RB, Chen YZ, Wei YP, Liu C, Ma ZF, Huang DH, Zhang YX, Lv WL (2009) Mapping of a wide compatibility locus in indica rice using SSR markers. Mol Breed 24(2):135–140
Ling KC, Tiongco ER, Aguiero VM (1977) Transmission of rice ragged stunt disease. Int Rice Res Newsl 2(6):11–12
Liu GQ, Yan H, Fu Q, Qian Q, Zhang ZT, Zhai WX, Zhu LH (2001) Mapping of a new gene for brown planthopper resistance in cultivated rice introgressed from Oryza eichingeri. Chin Sci Bull 46:738–742
Luo JJ, Hao W, Jin J, Gao JP, Lin HX (2008) Fine mapping of Spr3, a locus for spreading panicle from African cultivated rice (Oryza glaberrima Steud.). Mol Plant 1:830–838
Massague J (1998) TGF-beta signal transduction. Annu Rev Biochem 67:753–791
McCouch SR, CGSNL (2008) Gene nomenclature system for rice. Rice 1:72–84
Murai H, Hashimoto Z, Sharma PN, Shimizu T, Murata K, Takumi S, Mori N, Kawasaki S, Nakamura C (2001) Construction of a high-resolution linkage map of a rice brown planthopper (Nilaparvata lugens Stål) resistance gene bph2. Theor Appl Genet 103:526–532
Murata K, Fujiwara M, Kaneda C, Takumi S, Mori N, Nakamura C (1998) RFLP mapping of a brown planthopper (Nilaparvata lugens Stål) resistance gene bph2 of indica rice introgressed into a japonica breeding line ‘Norin-PL4’. Genes Genet Syst 73:359–364
Murata K, Fujiwara M, Murai H, Takumi S, Mori N, Nakamura C (2001) Mapping of a brown planthopper (Nilaparvata lugens Stål) resistance gene Bph9 on the long arm of rice chromosome 12. Cereal Res Commun 29:245–250
Murray MG, Thompson WK (1980) Rapid isolation of high molecular-weight plant DNA. Nucleic Acids Res 8:4321–4325
Myint KKM, Fujita T, Matsumura M, Sonoda T, Yoshimura A, Yasui H (2012) Mapping and pyramiding of two major genes for resistance to the brown planthopper (Nilaparvata lugens [Stål]) in the rice cultivar ADR52. Theor Appl Genet 124:495–504
Nemoto H, Ikeda R, Kaneda C (1989) New genes for resistance to brown planthopper, Nilaparvata lugens Stål, in rice. Jpn J Breed 39:23–28
Normile D (2008) Reinventing rice to feed the world. Science 321:330–333
Painter RH (1951) Insect resistance in crop plants. The Macmillan Company, New York, p 520
Qiu YF, Guo JP, Jing SL, Zhu LL, He GC (2010) High-resolution mapping of the brown planthopper resistance gene Bph6 in rice and characterizing its resistance in the 9311 and Nipponbare near isogenic backgrounds. Theor Appl Genet 121:1601–1611
Qiu YF, Guo JP, Jing SL, Tang M, Zhu LL, He GC (2011) Identification of antibiosis and tolerance in rice varieties carrying brown planthopper resistance genes. Entomol Exp Appl 141:224–231
Qiu YF, Guo JP, Jing SL, Zhu LL, He GC (2012) Development and characterization of japonica rice lines carrying the brown planthopper resistance genes BPH12 and BPH6. Theor Appl Genet 124(3):485–494
Raffaele S, Rivas S, Roby D (2006) An essential role for salicylic acid in AtMYB30-mediated control of the hypersensitive cell death program in Arabidopsis. FEBS Lett 580:3498–3504
Rahman ML, Jiang WZ, Chu SH, Qiao YL, Ham TH, Woo MO, Lee JH, Khanam MS, Chin JH, Jeung JU, Brar DS, Jena KK, Koh HJ (2009) High-resolution mapping of two rice brown planthopper resistance genes, Bph20(t) and Bph21(t), originating from Oryza minuta. Theor Appl Genet 119(7):1237–1246
Renganayaki K, Feitz AK, Sadasivam S, Pammi S, Harrington SE, McCouch SR, Kumar SM, Reddy AS (2002) Mapping and progress toward map-based cloning of brown planthopper biotype-4 resistance gene introgressed from Oryza officinalis into cultivated rice, O. sativa. Crop Sci 42:2112–2117
Rivera CT, Ou SH, Lida TT (1966) Grassy stunt disease of rice and its transmission by Nilaparvata lugens (Stål). Plant Dis Rep 50:453–456
Rivers RL, Dean RM, Chandy G, Hall JE, Roberts DM, Zeidel ML (1997) Functional analysis of nodulin 26, an aquaporin in soybean root nodule symbiosomes. J Biol Chem 272:16256–16261
Seo PJ, Park CM (2010) MYB96-mediated abscisic acid signals induce pathogen resistance response by promoting salicylic acid biosynthesis in Arabidopsis. New Phytol 186(2):471–483
Sogawa K (1982) The rice brown planthopper: feeding physiology and host plant interactions. Annu Rev Entomol 27:49–73
Sun LH, Su CC, Wang CM, Zhai HQ, Wan JM (2005) Mapping of a major resistance gene to the brown planthopper in the rice cultivar Rathu Heenati. Breed Sci 55:391–396
Vailleau F, Daniel X, Tronchet M, Montillet JL, Triantaphylides C, Roby D (2002) A R2R3-MYB gene, AtMYB30, acts as a positive regulator of the hypersensitive cell death program in plants in response to pathogen attack. Proc Natl Acad Sci USA 99:10179–10184
Wang YY, Wang XL, Yuan HY, Chen RZ, Zhu LL, He RF, He GC (2008) Responses of two contrasting genotypes of rice to brown planthopper. Mol Plant Microbe Interact 21:122–132
Wilson RC, Long FX, Maruoka EM, Cooper JB (1994) A new proline-rich early nodulin from Medicago truncatula is highly expressed in nodule meristematic cells. Plant Cell 6:1256–1275
Yamazaki Y, Tsuchiya R, Takahashi Y, Asanuma T, Shidahara Y, Sakaniwa S (2010) Rice genes in Oryza base. Nat Preced. doi:10.1038/npre.2010.5069.1
Yang HY, Ren X, Weng QM, Zhu LL, He GC (2002) Molecular mapping and genetic analysis of a rice brown planthopper (Nilaparvata lugens Stål) resistance gene. Hereditas 136:39–43
Yang HY, You AQ, Yang ZF, Zhang FT, He RF, Zhu LL, He GC (2004) High resolution genetic mapping at the Bph15 locus for brown planthopper resistance in rice (Oryza sativa L.). Theor Appl Genet 110:182–191
Acknowledgments
The authors thank C. Liu, Z. F. Ma, Y. P. Wei, L. S. Li and Y. Z. Chen for gene mapping and field work. This research was supported by the Major Program of National Science and Technology Project for Breeding of Transgenic Biological Varieties (2009ZX08009-047B), National High Technology Research and Development Program (863) (2007AA021405, 2007AA10Z191), Guangxi Natural Science Foundation (0833025) and Guangxi Science and Technology Projects (0815011-6-1, 09-007-08).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by T. Sasaki.
D. Huang and Y. Qiu contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
122_2012_1975_MOESM2_ESM.tif
Supplementary Figure S1 BPH-resistance genes on rice chromosome 4. Mapping region of the resistance genes: BPH12 (formerly Bph12(t)) from O. latifolia, mapped between RM16459 and RM1305 (Qiu et al. 2012); Bph15 from O. officinalis, mapped between RG1 and RG2 (Yang et al. 2004); Bph17 from cv. Rathu Heenati, mapped between RM8213 and RM5953 (Sun et al, 2005); Bph20(t) from O. minuta, mapped between B42 and B44 (Rahman et al. 2009); bph16 from O. officinalis, mapped between G271 and R93 (Hirabayashi et al. 1998); BPH6 (formerly Bph6) from cv. Swarnalata, mapped between RM6997 and RM5742 (Qiu et al. 2010). BPH27 from O. rufipogon, mapped between RM16846 and RM16853 (TIFF 1728 kb)
Rights and permissions
About this article
Cite this article
Huang, D., Qiu, Y., Zhang, Y. et al. Fine mapping and characterization of BPH27, a brown planthopper resistance gene from wild rice (Oryza rufipogon Griff.). Theor Appl Genet 126, 219–229 (2013). https://doi.org/10.1007/s00122-012-1975-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-012-1975-7