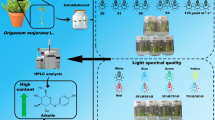
Abstract
Lilium candidum L. is a plant with rich cultural traditions and huge potential in floriculture and modern medicine. Our study analyzed the effects of light emitting diodes (LEDs) of variable quality on adventitious organogenesis induced in bulb scales placed on Murashige and Skoog medium. In addition to the spectral compositions of red (100%), blue (100%), and red and blue light (RB ratio 7:3), RB light was mixed in equal proportions (50%) with green (RBG), yellow (RBY), UV (RBUV), and far red (RBfR) light. We also tested white LED (Wled). Fluorescent lamp light (Fl) and darkness (D) served as controls. We assessed the effects of light on the morphometric features as well as the physiological and biochemical quality of the bulblets (adventitious bulbs). Bulb formation was observed under all light qualities, even in darkness, albeit to a limited extent. White LED light (Wled) treatment translated into the highest efficiency of bulblet formation and the greatest number of bulbs with developed leaves. The leaves, even though the shortest ones, also developed as a result of RBG treatment, and this light mixture enhanced the diameter of the forming bulblets. The bulbletsunder this light spectrum accumulated the most chlorophyll and carotenoids. The bulblets formed under B, RBfR and RBG LED had more than two times higher phenoliclevels than those formed under R LED and Fl. The bulbletsformed under B LED were the richest in soluble sugars, similarly to those developing in darkness (D).
Key message
Efficient micropropagation of Lilium candidum L. was achieved on media without growth regulators under different LED light quality which affected organogenesis, photosynthetic pigments, soluble phenolics and sugars composition in bulblets.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lilium candidum L. (also known as Madonna lily) is a plant of symbolic and ethnic significance. Initially, it was a Jewish symbol (Zaccai et al. 2009), and then it became a Christian personification of Maria associated with purity and virginity. It was planted in monastery gardens and used as art motif (Fulton 2004; Kabatliyska 2007; Husti and Cantor 2015). Madonna lily is also known in traditional folk medicine, has been used to heal wounds, burns and ulcers, and accelerated their healing. Modern medicine does not use the potential of L. candidum, but some of its pro-health properties have been proven. It shows anti-tumor, antiviral, and hepatoprotective potential, and the bulbs contain valuable metabolites, especially flavonoids and saponins (Mimaki et al. 1998; Vahálkova et al. 2000; Erdoğan et al. 2001; Yarmolinsky et al. 2009; Munafo and Gianfagna 2015; Patocka and Navratilova 2019).
L. candidum L. is a bulb geophyte of Liliaceae family, characterized by early flowering and pure white flowers with a strong and pleasant fragrance. It is native to the countries of the Mediterranean basin, and in Israel the species is recognized as endangered and under protection (Zaccai et al. 2009; Ӧzen et al. 2012). A low rate of generative reproduction negatively affects L. candidum population abundance. The apparent self-sterility of the species means that the plants do not set viable seeds, and even cross-pollination does not guarantee the formation of germinating seeds (Mynett 1992).
Research has been conducted on the micropropagation of many species and cultivars of lily (Bakhshaie et al. 2016), with just a few focusing on L. candidum. The studies mostly concern initiation of in vitro culture and elimination of numerous bacterial and fungal infections with antibiotics and fungicides toxic to the plant tissues (Altan et al. 2010; Burun and Sahin 2013). Bulbscales are the best explants for efficient formation of adventitious organs in in vitro cultures of Lilium (Bakhshaie et al. 2016). Leaves collected from plants grown in a greenhouse or in vitro (Khawar et al. 2005), or in vitro induced callus are used less frequently (Saadon and Zaccai 2013).
Steady advances in the development of the light emitting diodes (LED) technology make it applicable in plant micropropagation. LED lamps have a number of advantages over traditionally used light sources. They are characterized by low electricity consumption, high efficiency, low heat emission, longer durability, small size, and most importantly, they emit light of a specific wavelength (Gupta and Jatothu 2013; Bantis et al. 2018). Bornwaßer and Tantau (2012) reported a more even distribution of light on a shelf board under LED lighting than a traditional fluorescent lamp. LED lamps allow for manipulating the spectral quality of light and make it possible to gain a specific response of plant material, and even plants of higher quality. The light spectrum affects the direction of in vitro organogenesis and plant ability to acclimatize to ex vitro conditions (Pawłowska et al. 2018a). LED lighting was demonstrated to influence the content of starch, soluble sugars and photosynthetic pigments in plants propagated in vitro. Moreover, light wavelength affects the production of secondary metabolites (Pawłowska et al. 2018a; Cioć et al. 2018; Tomaszkiewicz et al. 2022; Klimek-Szczykutowicz et al. 2022). LEDs make it possible to conduct basic research on plant response under strictly controlled in vitro conditions by activating specific morphogenetic, biochemical, and physiological mechanisms (Gupta and Jatothu 2013; Bantis et al. 2018).
For many geophytes, light is necessary for the regeneration of storage organs in in vitro cultures, as confirmed in the studies on Fritillaria thunbergii (Paek and Murthy 2002), Nerine bowdenii (Jacobs et al. 1992), and Lilium oriental hybrid ‘Casablanca’ (Lian et al. 2003). Phytochrome is responsible for the mechanism of plant response to light, and the induction and direction of organogenesis is influenced by the quality of light (Ascough et al. 2008). The research conducted on Solanum tuberosum (Goeden and Tong 2003), which is a model system for tuberization, showed that blue light and its receptor cryptochrome affect the formation of storage organs in in vitro cultures. The process is poorly understood in ornamental plants (Ascough et al. 2008). The effects of light on biometrical parameters during in vitro organogenesis were demonstrated in various species and cultivars of lily. Light quality affected the frequency of plantlet (Simmonds and Cumming 1976), leaf (Takayama and Misawa 1979; Aguettaz et al. 1990), shoot (Han et al. 2005), and bulblet formation (Lian et al. 2003), as well as bulb dry weight (Varshney et al. 2000).
The first studies on the effects of light quality during adventitious organogenesis in bulbous plants were conducted under monochromatic fluorescent lamps. They showed that in in vitro cultures of Hyacinthus orientalis blue light stimulates the development of adventitious shoots and red light stimulates the formation of adventitious bulbs (Bach and Świderski 2000; Bach and Pawłowska 2006). LED technologies were used in research on a few species and cultivars of lily, and demonstrated the effect of light quality on plant growth and development (Bakhshaie et al. 2016). Lian et al. (2002) achieved the greatest incremental growth of Lilium oriental hybrid 'Pesaro' bulb under 1:1 mixture of red and blue LED.
The aim of this study was to determine the influence of light emitted by light-emitting diodes of different quality (eight combinations) on the formation of adventitious organs on L. candidum bulbscales in in vitro cultures. The spectral compositions most frequently used in research, that is red and blue, were supplemented with additional wave ranges of green, yellow, UV, far red, and white LED. We assessed the morphogenetic response of regenerating bulbscales, as well as the physiological and biochemical quality of the obtained bulbs based on their content of photosynthetic pigments, soluble sugars, and soluble phenolics.
Material and methods
Plant material
Adventitious bulbs of L. candidum grown in vitro (Gadzinowska et al. 2017) were used as the experimental material. They were formed on bulbscales in Murashige and Skoog (MS) medium (1962), without growth regulators, with the addition of 6% sucrose, and then cooled in the dark at 4 °C for 12 months. The starting bulbs of uniform size, 15 mm in diameter, and with 11–12 scales (Fig. 1a, b) were divided into individual scales (bulbscales) that were used as explants (Fig. 1c).
Culture condition and light treatment
The bulbscales were placed on MS medium containing 3% sucrose, pH 5.7, solidified with 0.5% BioAgar (BIOCORP, Poland). On each Petri dish (Ø 90 mm) with 30 ml of the medium, 8–9 scales from a single starting bulb were placed (Fig. 1c), with 3 the smallest inner scales being discarded.
The cultures were maintained in a phytotron at 23/21 °C (day/night), 80% relative humidity, and 16 h photoperiod (16 h day/8 h night).
Experimental LED panels (PXM, Poland) (Pawłowska et al. 2018a) allowed for setting and testing 8 different combinations of LED light quality (different wavelengths): 100% blue of 430 nm (B); 100% red of 670 nm (R); mix of 70% red and 30% blue (RB); 50% RB and 50% far red of 730 nm (RBfR); 50% RB and 50% yellow of 600 nm (RBY); 50% RB and 50% UV of 400 nm (RBUV); 50% RB and 50% green of 528 nm (RBG); 33.3% warm white (2700 K), 33.3% neutral white (4500 K), and 33.3% cool white (5700 K) (Wled) (Fig. 2a, b). Fluorescent lamp (Fl) (OSRAM LUMILUX Cool White L 36 W/840) (Fig. 2a) and darkness (D) were used as controls. For all light combinations, the photosynthetic photon flux density (PPFD) was 40 µmol m−2 s−1. LI-250A light meter with a Q 50604 sensor (Li-COR) and BTS256 spectrometer (Gigahertz-Optik) were used to determine the parameters of the LED panels.
Light spectra used in experiment. a—LEDs (%): 70 red + 30 blue (RB); 100 blue (B); 100 red (R); white: 33.3 warm + 33.3 neutral + 33.3 cool (Wled) and fluorescent lamp (Fl), b—LEDs (%): 35 red + 15 blue + 50 yellow (RBY); 35 red + 15 blue + 50 UV (RBUV); 35 red + 15 blue + 50 far red (RBfR) and 35 red + 15 blue + 50 green (RBG)
A total of 10 light combinations (including darkness) were tested, each of 10 replicates (Petri dishes) with 8–9 bulbscales.
After 8 weeks of the culture, biometric observations were carried out and plant material was collected for chemical analysis. Samples of the plant media were also taken to determine their content of soluble sugars.
The experimental method
Morphometrical observation
We assessed the biometric parameters of the organs formed on the bulbscales. To this end, we determined the ratio of the bulbscales regenerating bulblets, bulblets with leaves, and bulblets regenerating roots, i.e. the percent of scales regenerating the organ to the number of scales placed on the medium expressed as a ratio.
The numbers of formed bulblets, bulblets with leaves (minimum 5 mm in length), and adventitious roots per a starting bulb were determined. We also measured the diameter of the adventitious bulbs, and the length of leaves and adventitious roots.
Biochemical analyses
The bulblets formed under all light combinations were pulverized, frozen at − 80 °C, and kept until further analysis. Each biochemical analysis involved five 1 g samples of bulblet material from each light combination.
The analyzes were performed with a Freezone 4.5 freeze dryer (Labconco, USA), a QIAGEN TissueLyser II homogenizer (QIAGEN, Germany), a laboratory Eppendorf Centrifuge 5415 R (Bioridge Centrifuge, China), and a spectrophotometer Amersham Biosciences Ultrospec 2100 pro (Amersham Biosciences, United Kingdom).
Analysis of photosynthetic pigments content
The samples were lyophilized and then homogenized. The homogenates (3 mg) were poured over with 1 ml of 96% ethanol and macerated overnight in a refrigerator. The supernatants were analyzed in a spectrophotometer, and absorbance was measured at 470, 649, and 664 nm. Concentrations (µg/ml) of chlorophyll a (Ca), chlorophyll b (Cb), and carotenoids (Cc) were calculated from the following formulas: Ca = 13.36A664–5.19A649; Cb = 27.43A649–8.12A664; Cc = (1000A470–2.13Ca–97.63Cb)/209 (where A denoted absorbance) (Sumanta et al. 2014).
Content of soluble carbohydrates in adventitious bulbs
One gram of sample tissue was taken and homogenized for 2 min at a rate of 25 strokes per second with a steel ball only, and then at 20 strokes per second for 1 min with 1 ml of deionized water. Then the test tubes were centrifuged at 15 °C for 10 min (12,800 rpm, 15.2 g). The analysis was performed with the anthrone method (Dische 1962). The light absorption value of the samples were measured in a spectrophotometer at 620 nm. After preparing a series of glucose solutions with known concentrations, a standard curve was obtained and then used for the calculation of glucose content in the tested material.
Content of soluble phenolics in adventitious bulbs
The samples were lyophilized and then homogenized. The homogenates (3 mg) were poured over with 1 ml of 80% ethanol and macerated in a refrigerator for 48 h. The analysis was performed according to the methodology of Singleton and Rossi (1965). The absorbance was measured spectrophotometrically at 760 nm.
Content of soluble carbohydrates in the medium
The medium samples were taken from the spots where the explants were placed with a margin of approximately 1 mm. They were kept frozen at − 80 °C until further analysis. For the analysis, the medium was heated and 5 µl were taken and dissolved in 3 ml of distilled water. The analysis was performed using the anthrone method (Dische 1962), similarly as for the content of soluble carbohydrates in the bulblets.
Statistical analysis
All the study findings were analyzed statistically (ANOVA) using the Statistica 13.3 software (StatSoft, TIBCO Software Inc., Palo Alto, CA, USA). The post hoc multiple range Duncan test was used. Significantly different means were separated at p ≤ 0.05. The strength and direction of the correlation between the investigated parameters were determined with Statistica 13.3 package (StatSoft). A map of the correlations between the adventitious organogenesis and photosynthetic pigments was created.
Results and discussion
Morphological effects of different spectral combinations on bulblet formation
This study on micropropagation of L. candidum showed intense formation of adventitious bulbs on the bulbscales growing on the media without exogenous growth regulators. Earlier papers on micropropagation of L. candidum investigated the effects of medium PGR on organogenesis (Khawar et al. 2005; Burun and Sahin 2013; Saadon and Zaccai 2013; Akshay et al. 2021). Some researchers noted the formation of abnormal bulblets and disturbed development of L. candidum leaves on the PGR supplemented media (Lian et al. 2003; Saadon and Zaccai 2013). The bulblets we obtained on the media free of exogenous PGR showed a normal structure. We achieved a comparable or even higher organogenesis efficiency without PGRs than the other researchers (Khawar et al. 2005; Burun and Sahin 2013; Akshay et al. 2021). This indicates that it is possible to reduce the use of chemicals in in vitro bulb reproduction. Our research also confirmed the usefulness of bulbscales as explants for efficient adventitious organogenesis. The bulblets formed at the basal parts of the scales that contain more endogenous compounds necessary for the initiation of adventitious buds (Khawar et al. 2005).
Most experiments on in vitro cultures of Lilium genus involved traditional fluorescent lamps (Bakhshaie et al. 2016). Two papers describing the use of LED in in vitro cultures of lily have been published so far (Lian et al. 2002; Prokopiuk et al. 2018), and they investigated red, blue, and red–blue mix light spectra. These wavelengths are the most important for controlling the differentiation and growth of plants, with different forms of synergistic interactions of those light spectra (Silva et al. 2014). These spectra closely match that of chlorophyll absorption (Lian et al. 2002). In a study by Lian et al. (2002), Lilium oriental hybrid 'Pesaro' usually (over 80%) formed bulblets under a fluorescent lamp, but high efficiency per single scale (up to 3.9 bulblets) was also observed under blue LED and a mix of blue and red LED. The same LED spectra were reported as conducive to the most effective adventitious organogenesis on Lilium regale scales (Prokopiuk et al. 2018).
Additional wavelengths supplementing the basic red and blue mix, i.e. far red, yellow, green, UV, or white LED, has so far been rarely tested in in vitro research, and their impact on plant growth and development is unknown. The exclusion of exogenous PGRs allowed for monitoring the effects of light, and in such conditions we achieved a high efficiency of the adventitious organogenesis of L. candidum (on PGR free medium). In our study the bulbscales under all investigated light combinations and in darkness developed bulblets (adventitious bulbs) and adventitious roots, and some of them also formed leaves (Table 1). The scales kept in darkness only developed bulblets and roots but not leaves. The adventitious organs formed at the basal part of the bulb scale (Fig. 3a–g). The bulblets were made of flat and wide scales adjacent to each other. Their shade of green depended on the light spectrum (Fig. 3b–g), and those kept in the darkness were white (Fig. 3a). The tested light combinations affected the efficiency of the adventitious organ formation and their morphometric parameters (Table 1). Bulblets were observed on 79–100% of the explants. Our results were good under RB LED and fluorescence light, but when assessing a total organogenesis efficiency, we concluded that the addition of yellow and far red spectrum to this RB mix increased the formation of bulblets. They were most often observed under RBY light (and in the darkness), and the least often under blue light. Although an average of 16.3 bulblets per one starting bulb formed under the control fluorescent light, the efficiency of this process was at the same statistical level for the six LED light combinations: B, RB, RBfR, RBY, RBG, and Wled (from 12.7 to 14.4, respectively). Bulb formation was inhibited by red light (6.5 bulblets) and when the RB spectrum was in 50% replaced with UV (8.3 bulblets), but was the least efficient in the darkness (3 bulblets).The addition of yellow wavelength to the red and blue spectrum turned out conducive to vegetative growth of potato plantlets in vitro (Li et al. 2018).
Adventitious organogenesis on Lilium candidum bulb scales under different light quality in vitro: a darkness (D); b fluorescent lamp (Fl); under LED light (%): c 100 blue (B); d 100 red (R); e 70 red + 30 blue (RB); f 35 R + 15 B + 50 green (RBG) and g white: 33.3 warm, 33.3 neutral + 33.3 cool (Wled). Bar = 1 cm
The formation of L. candidum bulblets was the most intensively stimulated by white LED (Wled) that can be suggested as an economic alternative to traditional fluorescent lamps. Under this light, one starting bulb produced as many as 23 bulblets over an 8 week growth cycle. The formation of bulblets and bulblets with leaves, both of which are potential source of ex vitro regenerants, Wled light was the most efficient. The bulbs placed under control fluorescent light yielded about 21.5 regenerants but those under Wled more often developed leaves. The positive effect of white LED on the efficiency of micropropagation was confirmed in Vanilla planifolia, where white led induced twice as many shoots per explant as blue or red LEDs (Bello-Bello et al. 2016).
So far, there have been few studies on the effects of UV light emitted by LEDs on organogenesis in vitro. Tomaszewicz et al. (2022) demonstrated a negative effect of UV presence in the light spectrum on the growth of Cyathea delgadii gametophytes in vitro. UV light is tested mainly with light sources other than LED as an elicitor and a mutagenic agent (Ramakrishna and Ravishankar 2011; Tůmová and Tůma 2011; Espinosa-Leal et al. 2018; Xu et al. 2012). Abbasi et al. (2021) observed a decrease in cell proliferation and even death of Fagonia indica callus with an increasing dose of UV radiation in in vitro cultures. Victório et al. (2011) showed also tissue damage and decreased number of Phyllanthus tenellus shoots. This corresponds to the organogenesis results we witnessed, i.e., lowered regeneration coefficients and reduced morphometric features of the formed and assessed organs. The formation of L. candidum bulblets was also reduced in the darkness. According to Lian et al. (2002), this might be associated with photoperception and triggering regeneration capabilities by photomorphogenic pigments.
The diameter of the resulting bulblets can be a critical factor for their ex vitro performance upon transplantation (Lian et al. 2002). In our experiment the bulblets of the largest diameter (5.4 mm) developed under RB light supplemented witch green light (Fig. 3f). Their diameter was by almost 1 mm larger than under standard fluorescent lamp, although this was the same statistical level. A mix of red and blue LED increased the diameter of 'Pesaro' lily bulblets (Lian et al. 2002). White LED increased the diameter of pseudobulbs in Bletilla ochracea culture (Godo et al. 2011). RB LED supplemented with green light enhanced the diameter of Madonna lily bulblets and produced the largest bulblets of which 50% developed leaves. However, these leaves where the shortest (statistically as well as under RBUV and RBY lights) as compared with other light variants.
In our study leaves rarely developed on the bulblets formed under the tested light combinations, and they never developed in the darkness (Table 1). Most bulbs that grew leaves (43%) were kept under RB spectrum enriched with a green diode but the intensity of this process was very low (0.7 bulblets with leaves per one starting bulb). Contrary to that, 29% of bulblets formed under Wled developed leaves, and the yield was the highest among all light combinations (8.5 leaves per one starting bulb). In the remaining tested light combinations, leaves were observed on 3–15% of explants, with an efficiency of about 1–2 bulblets with leaves. An exception was fluorescent light, with the rate of over 5.3 bulblets that developed leaves.
Green light also shortened the leaves of Alternanthera brasiliana (Macedo et al. 2011), Vitis 'Manicure Finger' (Li et al. 2017), and Bletilla ochracea, whose leaves were also narrower (Godo et al. 2011). In Oncidium 'Grower Ramsey', green light inhibited shoot differentiation (Mengxi et al. 2011). According to Li et al. (2017), the limiting effect of green light on leaf growth may be caused by the "shade stress" effect. In natural conditions, this is related to the characteristics of the spectral composition of light reaching deep into the forest layers. In the shade, the light spectrum is rich in green wavelengths (Golovatskaya and Karnachuk 2015). In such conditions, plants invest, e.g., in shoot elongation at the expense of developing other organs, such as leaf blades. This is related to the shadow avoidance mechanism. By competing for light, plants adjust their structure to lift the leaves higher where there is more light (Casal 2012; Pierik and de Wit 2013; Li et al. 2017). This was experimentally confirmed, as the application of 100% green light increased shoot length in Achillea millefolium cultures (Alvarenga et al. 2015), and Vanilla planifolia (Bello-Bello et al. 2016) plants were taller and produced more leaves. However, the response of different plant species to light, including green and yellow light, can be different. Contrary to previous results, green light stimulated leaf area growth of Capsicum annuum plants (Casierra-Posada et al. 2014), and shoot growth in Lactuca sativa (Johkan et al. 2012). In Solanum tuberosum the addition of yellow light improved vegetative growth (Li et al. 2018).
Doughter and Bugbee (2001) pointed out similarities in the effects of green but also yellow light on some plant physiological mechanisms. Those wavelengths are equally and similarly transmitted through the canopy in nature. Therefore, yellow light can also limit plant growth, chlorophyll synthesis (Doughter and Bugbee 2001) or induction of protocorm-like bodies of Oncidium (Liu et al. 2011). However, the influence of yellow light is still not well understood and described in the literature. Researchers usually classify wavelengths of 500–600 nm as green light, which additionally limits the observations on the properties of yellow light (580–600 nm) (Li et al. 2018). In this research, green light increased root regeneration and elongation, similarly to darkness, where L. candidum bulbs produced the longest roots (28 mm). In these conditions the roots grew at the basal plates of all formed bulblets. Adventitious roots grew from the basal plates of the bulblets formed under all light combinations, with varying intensity, ranging from 23% (B) to 88% (Fl) (Table 1). The greatest number of roots was observed under the mixed light spectrum of RB and Wled (approx. 8, i.e. 0.6 per bulblet), as well as under Fl (13, i.e. 0.8 per bulblet). Under the remaining combinations the roots formed less frequently. Fluorescent light stimulated the formation of greater number of roots in 'Pesaro' lily (Lian et al. 2002) and PLB Oncidium (Mengxi et al. 2011).
Commonly used fluorescent lamps usually lack far red in their light spectrum. It is very important for plant development, for example stem elongation (Werbroucket al. 2012) or phytochrome activity. It can also significantly affect the propagation ratio by increasing it and improving multiplication (Miler et al. 2019). In our experiment, the longest leaves were observed during regeneration under RBfR light.
Effect of spectral combination on photosynthetic pigment content
Light is a key factor in the production of chlorophyll, which is an important component of photosynthesis. The quality of light used is also crucial for chlorophyll biosynthesis. Due to reduction of 5-aminolevulinic acid (ALA) precursor, red light creates unfavorable conditions for its synthesis, as opposed to blue light, which increases the accumulation of chlorophyll. A long-lasting application of blue light enhances the synthesis of ALA and eliminates the unfavorable effect of red light in this respect (Fan et al. 2013). The literature data confirm the inhibitory effect of red light on the production of chlorophyll that we also observed in our study (Li et al. 2010; Fan et al. 2013; Habiba et al. 2014; Bello-Bello et al. 2016; Coelho et al. 2021), and carotenoids (Liu et al. 2011; Fan et al. 2013; Pawłowska et al. 2018b; Coelho et al. 2021) in in vitro plant cultures. RBUV light also inhibited the synthesis of photosynthetic pigments, both chlorophylls and carotenoids, in L. candidum cultures. Our experiment confirmed that both chlorophyll a and b, lower levels were observed in the bulbs formed under RBUV (and a fluorescent lamp). UV radiation induces the uncoupling of the chlorophyll molecules in the light-harvesting system that results in reduced photosynthesis in many plant species (Jovanić et al. 2022). However, the response of chlorophyll to UV light may be different for different species and growing conditions (Begum et al. 2021). Chlorophyll a and b may also have different sensitivity to UV light, which is related to their dissimilar pathways (Ranjbarfordoei et al. 2009; Shaukat et al. 2011).
In the darkness, the content of photosynthetic pigments, especially carotenoids, is very low. In our studies the bulblets in these conditions hardly contain any pigments (Fig. 4a–d). The level of carotenoids was 6 times lower were seen in the bulblets formed under the darkness, and this value was comparable with R LED light (Fig. 4c). In their in vitro study Klimek-Szczykutowicz et al. (2022) demonstrated chlorophyll synthesis inhibition in darkness and resulting etiolation. Pigment deficiency alters the structure of the chloroplasts and inhibits their further differentiation into other organelles such as chromoplasts (Masuda et al. 1996). It is only during photomorphogenesis that the coordinated increase in the levels of chlorophylls and carotenoids in combination with other components results in the formation of a functional photosynthetic apparatus (von Lintig et al. 1997).
Photosyntetic pigments: a chlorophyll a, b chlorophyll b, c carotenoids, d chlorophyll a + b content in in vitro bulblets of Lilium candidum under different light quality: in the darkness (D); under fluorescence lamp (Fl); under LED light (%): 100 blue (B); 100 red (R); 70 red + 30 blue (RB); 35 R + 15 B + 50 far red (RBfR); 35 red + 15 blue + 50 yellow (RBY); 35 red + 15 blue + 50 UV (RBUV); 35 red + 15 blue + 50 green (RBG) and white: 33.3 warm, 33.3 neutral + 33.3 cool (Wled). Data are presented as means ± standard deviations. Different letters indicate significant differences between values according to Duncan’s multiple range test at p ≤ 0.05
We detected the highest amount of chlorophyll a (over 440 µg/g dw) in the bulblets under the RBG. It was almost 3 timed lower in the bulblets formed under R and RBUV LED, and those from under the control fluorescent lamp. Chlorophyll b content was also the highest (202 µg/g dw), however, in the bulblets under Wled it was at the same statistical level (147 µg/g dw). Green LED light (RBG) increased also the content of carotenoids (96 µg/g dw), which was the highest. The research literature contains examples of positive effects of adding green light to the spectrum on the content of photosynthetic pigments in cultured plant tissues. In plants, green light is mainly a signal and is not involved in photosynthesis. Its stimulating effect is explained by compensating for the insufficient amount of photosynthetically active light (Golovatskaya and Karnachuk 2015). For example, potato (Solanum tuberosum L.) plantlet leaves accumulated more chlorophyll a (Li et al. 2018), and Achillea millefolium L. revealed the highest content of photosynthetic pigments (chlorophyll a, chlorophyll b and carotenoids) when exposed to green LED light (Alvarenga et al. 2015). Protocorm-like bodies (PLBs) of Dendrobium kingianum contained similar amounts of chlorophyll as those grown under blue LED or a fluorescent lamp (Habiba et al. 2014). In Oncidium sp. green light produced effects comparable to fluorescence (Mengxi et al. 2011). In an experiment carried out in Brassica campestris L., a similar content of chlorophyll a and carotenoids was shown in plants exposed to green and yellow LED as compared to those exposed to blue light. For green light this relationship was also confirmed in terms of the content of chlorophyll b (Fan et al. 2013). Green and yellow light favored the production of photosynthetic pigments in Aeollanthus suaveolens (Araújo et al. 2021) and Lippia dulcis (Rocha et al. 2022). Our study demonstrated an increased production of photosynthetic pigments under green LED (RBG).
Effect of spectral combination on soluble phenol content
The bulblets formed under different light combinations had variable content of soluble phenolics (Fig. 5). We confirmed the inhibitory effect of red LED (2 times less than under control (Fl) on soluble phenolics content in the bulblets of L. candidum, while the other tested LED spectra significantly increased tissue accumulation of soluble phenolics as compared with the control light of the fluorescent lamp. Also the bulblets kept in darkness accumulated more phenols than those under the Fl light. High content of phenolics was detected in the plants grown under B, RBfR, and RBG light (8.8, 8.5, and 7.6 ng/g dw, respectively), and it was 3–3.5 times more than under R light (Fig. 5).The same effect of light spectra was determined as for the content of photosynthetically active pigments. Similar results were reported by Bach et al. (2015) in their research on Lachenalia, with the use of monochromatic lamps. Bulb content of phenols was enhanced under blue light and reduced under red light. Phenolics biosynthesis in plants is controlled by external factors, including light that may stimulate the process or even be its indispensable component (Ghasemzadeh et al. 2010). The main absorption spectrum of flavonoids occurs between 400 and 430 nm, that is the spectrum of blue light by which their production is stimulated (Ebisawa et al. 2008; Silva et al. 2017; Taulavuori et al. 2018; Coelho et al. 2021; Rocha et al. 2022). The main absorption spectrum for many phenolic compounds matches UVB wavelength and their synthesis is stimulated by UVB light. UV light is often used in research as an elicitor to intensify the production of secondary metabolites (Ramakrishna and Ravishankar 2011; Tůmová and Tůma 2011; Espinosa-Leal et al. 2018).
Soluble phenolics content in in vitro bulblets of Lilium candidumunder different light quality: in the darkness (D); under fluorescence lamp (Fl); under LED light (%): 100 blue (B); 100 red (R); 70 red + 30 blue (RB); 35 R + 15 B + 50 far red (RBfR); 35 red + 15 blue + 50 yellow (RBY); 35 red + 15 blue + 50 UV (RBUV); 35 red + 15 blue + 50 green (RBG) and white: 33.3 warm, 33.3 neutral + 33.3 cool (Wled). Data are presented as means ± standard deviations. Different letters indicate significant differences between values according to Duncan’s multiple range test at p ≤ 0.05
The results of other authors confirm the stimulating effect of other LED wavelengths on the accumulation of phenolic compounds that we noticed in our experiments. Hossen (2007) reported the highest rutin content in Fagopyrum esculentum sprouts under RBG light. The leaves of Betula pendula exposed to far red and UVB contained more simple phenolic compounds than the variant deprived of UVB light (Tegelberg et al. 2004). Literature data show high variability in the content of phenolic compounds depending on the species, organs, tissues, or cells (Chang et al. 2019; Hsie et al. 2019).
Effect of spectral combination on soluble sugar content
High content of soluble sugars in the bulblets and their lower concentrations in the postculture medium can be explained by the storage role of the bulbs that accumulate nutrients, including sugars (Ascough et al. 2008). This happens especially during the period of vegetative growth (Pouris et al. 2020). Carbohydrates make up a significant proportion of the dry weight of lily bulbs and are absorbed in large amounts from the culture medium (Langens-Gerrits et al. 2003). Bach et al. (1992) showed higher percentage content of soluble sugars in leaf explants of Hyacinthus orientalis than in the starting medium. Light significantly affects carbohydrate metabolism in higher plants (Kowalik 1982; Chen et al. 2018). In our study the tested light spectra affected the content of soluble sugars in the adventitious bulbs but with lower variability than in the case of photosynthetic pigments and soluble phenolics (from 369.7 to 564.7 mg/g dw) (Fig. 6a, b). The bulbs formed in the darkness (D) and under B light were the most abundant in soluble sugars (above 550 mg/g dw) (Fig. 6a). For Pancratium maritimum bulblets exposure to ambient light enhanced total carbohydrate content that exceeded 700 mg/g dw (Pouris et al. 2020). Blue LED enhanced soluble sugar content also in Brassica campestris (Fan et al. 2013) and promoted sugar in potato tuberizing in vitro (Li et al. 2020). Blue (or far-red) light qualities are more effective than conventional fluorescent lamps in enhancing carbohydrate accumulation (Heo et al. 2006). Sugar is required for hypocotyl elongation in response to darkness (Zhang et al. 2015; Stewart et al. 2011; Oh et al. 2012), which may explain its increased content under these conditions. Lachenalia plants grown in darkness also contained more soluble sugars than the illuminated ones (Bach et al. 2015). The effect of light on the accumulation of soluble sugars depends also on the investigated plant species (Liu et al. 2011; Li et al. 2018).
Soluble sugars contents: a—in in vitro bulblets of Lilium candidum. b—in culture media under different light quality: in the darkness (D); under fluorescence lamp (Fl); under LED light (%): 100 blue (B); 100 red (R); 70 red + 30 blue (RB); 35 R + 15 B + 50 far red (RBfR); 35 red + 15 blue + 50 yellow (RBY); 35 red + 15 blue + 50 UV (RBUV); 35 red + 15 blue + 50 green (RBG) and white: 33.3 warm, 33.3 neutral + 33.3 cool (Wled). Data are presented as means ± standard deviations. Different letters indicate significant differences between values according to Duncan’s multiple range test at p ≤ 0.05
The concentration of soluble sugars in the starting medium, determined before establishing the culture, was 103 mg/ml. The content of soluble sugars in the medium at the end of the culture ranged from 99.8 to 173.4 mg/ml of the medium. It was the highest in the media kept under fluorescent light and B, R, and RBG LEDs, and the lowest under RB and white LED (Fig. 6b). A joint presence of light, temperature, and high humidity causes agar degradation and breaks down chemical bonds (Freile-Pelegrín et al. 2007). This may also determine the content of soluble sugars in the postculture medium. Depending on the type of light used, this content may even be higher than in the starting medium, as reported in our research. Bach et al. (1992) confirmed this tendency and claimed it depended on the type and concentration of carbohydrates in the medium.
Correlation analysis between morphometrical and physiological traits
We found a positive correlation between the number of scales regenerating plants and the root regeneration ratio. Greater number of resulting bulblets stimulated also the formation of bulblets with leaves, length of leaves, and number of roots but inhibited root elongation. Enhanced bulblet diameter was associated with a greater number of bulblets with leaves. Both these parameters showed a positive correlation with the content of all examined photosynthetic pigments. The frequency of adventitious roots formation on the bulbscales was associated with an increase in their number. We also confirmed a strong and positive correlation with the content of photosynthetic pigments (Table 2).
Conclusion
Light quality has affected adventitious organogenesis, photosynthetic pigments, soluble phenolics and soluble carbohydrates concentrations in L. candidum bulblets. The white LED light (Wled) proved to be the best light condition for efficient bulblets production. The chlorophyll a, chlorophyll b and carotenoids content was the highest under the mix of red and blue LED light supplemented with green (RBG). The diameter of the forming bulblets was the greatest. Darkness and red light (R) led to the bulblet etiolation. Soluble phenolics content was highest in plants cultured under blue LED light (B). Darkness and blue LED light (B) stimulated the accumulation of soluble sugars in bulblets.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- B:
-
100% Blue LED light
- D:
-
Darkness
- Fl:
-
Fluorescent lamp
- g dw:
-
Gram of dry weight
- LED:
-
Light-emitting diode
- MS:
-
Murashige and Skoog culture medium
- R:
-
100% Red LED light
- RB:
-
70% Red + 30% blue LED light
- RBfR:
-
35% Red + 15% blue + 50% far red LED light
- RBG:
-
35% Red + 15% blue + 50% green LED light
- RBUV:
-
35% Red + 15% blue + 50% UV LED light
- RBY:
-
35% Red + 15% blue + 50% yellow LED light
- Wled:
-
33.3% Warm + 33.3% neutral + 33.3% cool white LED light
References
Abbasi BH, Khan T, Khurshid R, Nadeem M, Drouet S, Hano Ch (2021) UV-C mediated accumulation of pharmacologically signifcant phytochemicals under light regimes in in vitro culture of Fagonia indica (L.). Sci Rep. https://doi.org/10.1038/s41598-020-79896-6
Aguettaz P, Paffen A, Delvallee I, Van der Linde P, De Klerk GJ (1990) The development of dormancy in bulblets of Lilium speciosum generated in vitro: 1. The effects of culture conditions. Plant Cell Tissue Organ Cult 22:167–172. https://doi.org/10.1007/BF00033631
Akshay MP, Pooja PG, Sonali D (2021) In vitro micropropagation of Lilium candidum bulb by application of multiple hormone concentrations using plant tissue culture technique. Int J Res Appl Sci Biotechnol 8(2):244–253. https://doi.org/10.31033/ijrasb.8.2.32
Altan F, Bürün B, Şahin N (2010) Fungal contaminants observed during micropropagation of Lilium candidum L. and the effect of chemotherapeutic substances applied after sterilization. Afr J Biotechnol 9(7):991–995. https://doi.org/10.5897/AJB08.090
Alvarenga ICA, Pacheco FV, Silva ST, Bertolucci SKV, Pinto JEBP (2015) In vitro culture of Achillea millefolium L.: quality and intensity of light on growth and production of volatiles. Plant Cell Tiss Organ Cult 122:299–308. https://doi.org/10.1007/s11240-015-0766-7
Araújo DX, Rocha TT, Carvalho AA, Bertolucci SKV, Medeiros APR, Ribeiro FNS, Barbosa SM, Pinto JEBP (2021) Photon flux density and wavelength influence on growth, photosynthetic pigments and volatile organic compound accumulation in Aeollanthus suaveolens (Catinga-de-mulata) under in vitro conditions. Ind Crops Prod 168:113597. https://doi.org/10.1016/j.indcrop.2021.113597
Ascough GD, van Staden J, Erwin JE (2008) In vitro storage organ formation of ornamental geophytes. In: Janick J (ed) Horticultural reviews, vol 34. Wiley, Hoboken, pp 417–445. https://doi.org/10.1002/9780470380147.ch7
Bach A, Pawłowska B (2006) Effect of light qualities on cultured in vitro ornamental bulbous plants. In: da Teixeira Silva JA (ed) Floriculture, ornamental plant biotechnology. Global Science Books Ltd, Middlesex, pp 271–276
Bach A, Świderski A (2000) The effect of light quality on organogenesis of Hyacinthus orientalis L. in vitro. Acta Biol Cracov Series Bot 42:115–120
Bach A, Pawłowska B, Pułczyńska K (1992) Utilization of soluble carbohydrates in shoot and bulb regeneration of Hyacinthus orientalisL. in vitro. Acta Hort 325:487–492. https://doi.org/10.17660/ActaHortic.1992.325.67
Bach A, Kapczyńska A, Dziurka K, Dziurka M (2015) Phenolic compounds and carbohydrates in relation to bulb formation in Lachenalia ‘Ronina’ and ‘Rupert’ in vitro cultures under different lighting environments. Sci Hortic 188:23–29. https://doi.org/10.1016/j.scienta.2015.02.038
Bakhshaie M, Khosravi S, Azadi P, Bagheri H, Tuyl JM (2016) Biotechnological advances in Lilium. Plant Cell Rep 35:1799–1826. https://doi.org/10.1007/s00299-016-2017-8
Bantis F, Smirnakou S, Ouzounis T, Koukounaras A, Ntagkas N, Radoglou K (2018) Current status and recent achievements in the field of horticulture with the use of light-emitting diodes (LEDs). Sci Hortic 235:437–451. https://doi.org/10.1016/j.scienta.2018.02.058
Begum HA, Hamayun M, Shad N, Khan W, Ahmad J, Khan MEH, Jones DA, Ali K (2021) Effects of UV radiation on germination, growth, chlorophyll content, and fresh and dry weights of Brassica rapa L. and Eruca sativa L. Sarhad J Agricult 37(3):1016–1024. https://doi.org/10.17582/journal.sja/2021/37.3.1016.1024
Bello-Bello JJ, Martínez-Estrada E, Caamal-Velázquez JH, Morales-Ramos V (2016) Effect of LED light quality on in vitro shoot proliferation and growth of vanilla (Vanilla planifolia Andrews). Afr J Biotechnol 15(8):272–277. https://doi.org/10.5897/AJB2015.14662
Bornwaßer T, Tantau HJ (2012) Evaluation of LED lighting systems in in vitro cultures. Acta Hort 956:555–562. https://doi.org/10.17660/ActaHortic.2012.956.66
Burun B, Sahin O (2013) Micropropagation of Lilium candidum L.: a rare and native bulbous flower of Turkey. Bangladesh J Bot 42(2):185–187. https://doi.org/10.3329/bjb.v42i1.15913
Casal JJ (2012) Shade avoidance. Arabidopsis Book 10:e0157. https://doi.org/10.1199/tab.0157
Casierra-Posada F, Matallana-Díaz YA, Zapata-Cassiera E (2014) Growth of bell pepper plants (Capsicum annuum) affected by coloured covers. GesundePflanzen 66:149–155. https://doi.org/10.1007/s10343-014-0328-7
Chang HP, Kim NS, Park JS, Lee SY, Lee JW, Park SU (2019) Effects of light-emitting diodes on the accumulation of glucosinolates and phenolic compounds in sprouting canola (Brassica napus L.). Foods 8(2):76. https://doi.org/10.3390/foods8020076
Chen L, Xue X, Yang Y, Chen F, Zhao J, Wang X, Khan AT, Hu Y (2018) Effects of red and blue LEDs on in vitro growth and microtuberization of potato single-node cuttings. Front Agric Sci Eng 5(2):197–205. https://doi.org/10.15302/J-FASE-2018224
Cioć M, Szewczyk A, Żupnik M, Kalisz A, Pawłowska B (2018) LED lighting affects plant growth, morphogenesis and phytochemical contents of Myrtus communis L. in vitro. Plant Cell Tiss Organ Cult 132:433–447. https://doi.org/10.1007/s11240-017-1340-2
Coelho AD, Souza CK, Bertolucci SKV, Carvalho AA, Santos GC, Oliveira T, Marques EA, Salimena JP, Pinto JEBP (2021) Wavelength and light intensity enhance growth, phytochemical contents and antioxidant activity in micropropagated plantlets of Urtica dioica L. Plant Cell Tissue Organ Cult (PCTOC) 145:59–74. https://doi.org/10.1007/s11240-020-01992-2
Dische Z (1962) General color reactions. In: Whistler RL, Wolfram ML (eds) 187 Carbohydrate chemistry. Academic Press, New York, pp 477–512
Doughter TAO, Bugbee B (2001) Evidence for yellow light suppression of lettuce growth. Photochem Photobiol 73(2):208–212. https://doi.org/10.1562/0031-8655(2001)073%3c0208:EFYLSO%3e2.0.CO;2
Ebisawa M, Shoji K, Kato M, Shimomura K, Goto F, Yoshihara T (2008) Supplementary ultraviolet radiation B together with blue light at night increased quercetin content and flavonol synthase gene expression in leaf lettuce (Lactucasativa L.). Environ Control Biol 46:1–11. https://doi.org/10.2525/ecb.46.1
Erdoğan I, Sener B, Rahman A (2001) Etioline, a steroidal alkaloid from Lilium candidum L. Biochem Syst Ecol 29:535–536. https://doi.org/10.1016/S0305-1978(00)00075-2
Espinosa-Leal CA, Puente-Garza CA, García-Lara S (2018) In vitro plant tissue culture: means for production of biological active compounds. Planta 248:1–18. https://doi.org/10.1007/s00425-018-2910-1
Fan XX, Zang J, Xu ZG, Guo SR, Jiao XL, Liu XY, Gao Y (2013) Effects of different light quality on growth, chlorophyll concentration and chlorophyll biosynthesis precursors of non-heading Chinese cabbage (Brassica campestris L.). Acta Physiol Plant 35:2721–2726. https://doi.org/10.1007/s11738-013-1304-z
Freile-Pelegrín Y, Madera-Santana T, Robledo D, Veleva L, Quintana P, Azamar JA (2007) Degradation of agar films in a humid tropical climate: thermal, mechanical, morphological and structural changes. Polym Degrad Stab 92:244–252. https://doi.org/10.1016/j.polymdegradstab.2006.11.005
Fulton R (2004) The Virgin in the garden, or why flowers make better prayers. Spiritus: J Christian Spirit 4(1):1–23. https://doi.org/10.1353/scs.2004.0008
Gadzinowska J, Cioć M, Prokopiuk B (2017) Madonna lily (Lilium candidumL.) in vitro cultures establishment. 6th International Conference for Young Researchers. Multidirectional Research in Agriculture, Forestry and Technology, 24–25 April 2017, Kraków, 158
Ghasemzadeh A, Jaafar HZE, Rahmat A, Wahab PEM, Halim MRA (2010) Effect of different light intensities on total phenolics and flavonoids synthesis and anti-oxidant activities in young ginger varieties (Zingiber officinale Roscoe). Int J Mol Sci 11:3885–3897. https://doi.org/10.3390/ijms11103885
Godo T, Fujiwara K, Guan K, Miyoshi K (2011) Effect of wavelength of LED-light on in vitroasymbiotic germination and seedling growth of Bletilla ochraceaSchltr. (Orchidaceae). Plant Biotechnol 28:397–400. https://doi.org/10.5511/plantbiotechnology.11.0524a
Goeden KR, Tong CB (2003) The effects of blue light on potato tuberisation. Abstr, 543. Poster presented for the annual meeting of the American Society Plant Biologists.
Golovatskaya IF, Karnachuk RA (2015) Role of green light in physiological activity of plants. Russ J Plant Physiol 62(6):727–740. https://doi.org/10.1134/S1021443715060084
Gupta SD, Jatothu B (2013) Fundamentals and applications of light-emitting diodes (LEDs) in in vitro plant growth and morphogenesis. Plant Biotechnol Rep 7:211–220. https://doi.org/10.1007/s11816-013-0277-0
Habiba AU, Kazuhiko S, Ahasan M, Alam M (2014) Effects of different light quality on growth and development of protocorm-like bodies (PLBs) in Dendrobium kingianumcultured in vitro. Bangladesh Res Publ J 10(2):223–227
Han BH, Yae BW, Yu HJ, Peak KY (2005) Improvement of in vitro micropropagation of Lilium Oriental hybrid ‘Casablanca’ by the formation of shoots with abnormally swollen basal plates. Sci Hortic 103:351–359. https://doi.org/10.1016/j.scienta.2004.07.003
Heo JW, Shin KS, Kim SK, Peak KY (2006) Light quality affects in vitro growth of grape “Teleki 5BB.” J Plant Biol 49(4):276–280. https://doi.org/10.1007/BF03031155
Hossen Z (2007) Light emitting diodes increase phenolics of buckwheat (Fagopyrum esculentum) sprouts. J Plant Interact 2(1):71–78. https://doi.org/10.1080/17429140701288228
Hsie BS, Bueno AIS, Bertolucci SKV, Carvalho AA, Cunha SHB, Martins ER, Pinto JEBP (2019) Study of the influence of wavelengths and intensities of LEDs on the growth, photosynthetic pigment, and volatile compounds production of Lippia rotundifolia Cham in vitro. J Photochem Photobiol, B: Biol. https://doi.org/10.1016/j.jphotobiol.2019.111577
Husti A, Cantor M (2015) Sacred connection of ornamental flowers with religious symbols. ProEnvironment 8:73–79
Jacobs G, Richard G, Allderman LA, Theron KI (1992) Direct and indirect organogenesis in tissue cultures of Nerine bowdenii Watts. Acta Hort 325:475–479. https://doi.org/10.17660/ActaHortic.1992.325.65
Johkan M, Shoji K, Goto F, Hahida S, Yoshihara T (2012) Effect of green light wavelength and intensity on photomorphogenesis and photosynthesis in Lactuca sativa. Environ Exp Bot 75:128–133. https://doi.org/10.1016/j.envexpbot.2011.08.010
Jovanić BR, Radenković B, Despotović-Zrakić M, Bogdanović Z, Barać D (2022) Effect of UV-B radiation on chlorophyll fluorescence, photosynthetic activity and relative chlorophyll content of five different corn hybrids. J Photochem Photobiol 10:1115. https://doi.org/10.1016/j.jpap.2022.100115
Kabatliyska Z (2007) Flowers in the Bulgarian monasteries – interior and exterior, traditions and future. Academia Danubiana 5:10–13
Khawar KM, Cocu S, Parmaksiz I, Sarihan EO, Özcan S (2005) Mass proliferation of Madonna Lily (Lilium candidum L.) under in vitro conditions. Pak J Bot 37(2):243–248
Klimek-Szczykutowicz M, Prokopiuk B, Dziurka K, Pawłowska B, Ekiert H, Szopa A (2022) The infuence of diferent wavelengths of LED light on the production of glucosinolates and phenolic compounds and the antioxidant potential in in vitro cultures of Nasturtium ofcinale (watercress). Plant Cell Tissue Organ Cult (PCTOC) 149:113–122. https://doi.org/10.1007/s11240-021-02148-6
Kowalik W (1982) Blue light effects on respiration. Plant Physiol 33(1):51–72. https://doi.org/10.1146/ANNUREV.PP.33.060182.000411
Langens-Gerrits M, Kuijpers A-M, De Klerk G-J, Croes A (2003) Contribution of explant carbohydrate reserves and sucrose in the medium to bulb growth of lily regenerated on scale segments in vitro. Physiol Plant 117:245–255. https://doi.org/10.1034/j.1399-3054.2003.1170212.x
Li H, Xu Z, Tang C (2010) Effect of light-emitting diodes on growth and morphogenesis of upland cotton (Gossypium hirsutum L.) plantlets in vitro. Plant Cell Tissue Organ Cult 103:155–163. https://doi.org/10.1007/s11240-010-9763-z
Li Ch, Xu Z, Dong R, Chang S, Wang L, Khalil-Ur-Rehman M, Tao J (2017) An RNA-Seq analysis of grape plantlets grown in vitro reveals different responses to blue, green, red led light, and white fluorescent light. Front Plant Sci 8:78. https://doi.org/10.3389/fpls.2017.00078
Li R, Huang W, Wang X, Liu X, Xu Z (2018) Effects of yellow, green, and different blue spectra on growth of potato plantlets in vitro. HortScience 53(4):541–546. https://doi.org/10.21273/HORTSCI12848-18
Li R, Long J, Yan Y, Luo J, Xu Z, Liu X (2020) Addition of white light to monochromatic red and blue lights alters the formation, growth, and dormancy of in vitro-grown Solanum tuberosum L. microtubers. HortScience 55(1):71–77. https://doi.org/10.2173/HORTSCI14548-19
Lian ML, Murthy HN, Paek KY (2002) Effects of light emitting diodes (LED) on the in vitro induction and growth of bulblets of Lilium oriental hybrid ‘Pesaro.’ Sci Hortic 94:365–370. https://doi.org/10.1016/S0304-4238(01)00385-5
Lian ML, Chakrabarty D, Paek KY (2003) Bulblet formation from bulbscale segments of Lilium using bioreactor system. Biol Plant 46:199–202. https://doi.org/10.1023/A:1022890208500
Liu M, Xu Z, Yang Y, Yijie F (2011) Effects of different spectral lights on Oncidium PLBs induction, proliferation, and plant regeneration. Plant Cell, Tissue Organ Cult 106(1):1–10. https://doi.org/10.1007/s11240-010-9887-1
Macedo AF, Leal-Costa MV, Tavares ES, Lage CLS, Esquibel MA (2011) The effect of light quality on leaf production and development of in vitro -cultured plants ofAlternanthera brasiliana Kuntze. Environ Exp Bot 70:43–50. https://doi.org/10.1016/j.envexpbot.2010.05.012
Masuda T, Takabe K, Ohta H, Shioi Y, Takamiya K (1996) Enzymatic activities for the synthesis of chlorophyll in pigment-deficient variegated leaves of Euonymus japonicus. Plant Cell Physiol 37(4):481–487. https://doi.org/10.1093/oxfordjournals.pcp.a028970
Mengxi L, Zhigang X, Yang Y, Yijie F (2011) Effects of different spectral lights on Oncidium PLBs induction, proliferation, and plant regeneration. Plant Cell Tiss Organ Cult 106:1–10. https://doi.org/10.1007/s11240-010-9887-1
Miler N, Kulus D, Woźny A, Rymarz D, Hajzer M, Wierzbowski K, Nelke R, Szeffs L (2019) Application of wide-spectrum light-emitting diodes in micropropagation of popular ornamental plant species: a study on plant quality and cost reduction. In Vitro Cell Dev Biol Plant 55:99–108. https://doi.org/10.1007/s11627-018-9939-5
Mimaki Y, Tadaaki S, Minpei K, Yutaka S, Yoshio H (1998) New steroidal constituents from the bulbs of Lilium candidum. Chem Pharm Bull 46(11):1829–1832. https://doi.org/10.1248/cpb.46.1829
Munafo JP Jr, Gianfagna T (2015) Chemistry and biological activity of steroidal glycosides from the Lilium genus. Nat Prod Rep 32(3):454–477. https://doi.org/10.1039/C4NP00063C
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Mynett K (1992) Lilie. Powszechne Wydawnictwo Rolnicze i Leśne, Warsaw Poland, pp 7–45
Oh E, Zhu JY, Wang ZY (2012) Interaction between BZR1 and PIF4 integrates brassinosteroid and environmental responses. Nat Cell Biol 14:802–809. https://doi.org/10.1038/ncb2545
Paek KY, Murthy HN (2002) High frequency of bulblet regeneration from bulb scale sections of Fritillaria thunbergii. Plant Cell Tissue Org Cult 68:247–252. https://doi.org/10.1023/A:1013952803887
Patocka J, Navratilova Z (2019) Bioactivity of Lilium candidum L : a mini review. Biomed J Sci Technol Res. https://doi.org/10.26717/BJSTR.2019.18.003204
Pawłowska B, Cioć M, Prokopiuk B (2018a) How LED light rooting in vitro affected Gerbera acclimatization efficiency. Acta Hortic 1201:583–590. https://doi.org/10.17660/ActaHortic.2018.1201.78
Pawłowska B, Żupnik M, Szewczyk-Taranek B, Cioć M (2018b) Impact of LED light sources on morphogenesis and levels of photosynthetic pigments in Gerbera jamesonii grown in vitro. Hortic Environ Biotechnol 59:115–123. https://doi.org/10.1007/s13580-018-0012-4
Pierik R, de Wit M (2013) Shade avoidance: phytochrome signalling and other aboveground neighbour detection cues. J Exp Bot 65(11):2815–2824. https://doi.org/10.1093/jxb/ert389
Pouris J, Meletiou-Christou M-S, Chimona C, Rhizopoulou S (2020) Seasonal functional partitioning of carbohydrates and proline among plant parts of the sand daffodil. Agronomy 10(4):539. https://doi.org/10.3390/agronomy10040539
Prokopiuk B, Cioć M, Maślanka M, Pawłowska B (2018) Effects of light spectra and benzyl adenine on in vitro adventitious bulb and shoot formation of Lilium regale E. H. Wilson. Propag Ornam Plants 18(1):12–18
Ramakrishna A, Ravishankar GA (2011) Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal Behav 6(11):1720–1731. https://doi.org/10.4161/psb.6.11.17613
Ranjbarfordoei A, van Damme P, Samson R (2009) Elevated ultraviolet-B radiation influences photosynthetic pigments and soluble carbohydrates of sweet almond [Prunus dulcis (Miller) D. webb]. Electron J Environ Agric Food Chem 8(11):1077–1084
Rocha TT, Araújo DX, Carvalho AA, Germano CM, Santos MF, Lameira OA, Bertolucci SKV, Pinto JEBP (2022) In vitro culture of Lippia dulcis (Trev.): light intensity and wavelength effects on growth, antioxidant defense, and volatile compound production. In Vitro Cell Dev Biol Plant 58:636–652. https://doi.org/10.1007/s11627-022-10270-z
Saadon S, Zaccai M (2013) Lilium candidum bulblet and meristem development. In Vitro Cell Dev Biol Plant 49:313–319. https://doi.org/10.1007/s11627-013-9496-x
Shaukat SS, Zaidi S, Khan MA (2011) Effect of supplemental UV-B radiation on germination, seedling growth, and biochemical responses of sunflower (Helianthus annuus L.). Fuuast J Biol 1(1):27–33
Silva MMA, Oliveira ALB, Oliveira-Filho RA, Gouveia-Neto AS, Camara TJR, Willadino LG (2014) Effect of blue/red LED light combination on growth and morphogenesis of Saccharum officinarum plantlets in vitro. Proc. SPIE 8947, Imaging, Manipulation, and Analysis of Biomolecules, Cells, and Tissues XII, 89471X.https://doi.org/10.1117/12.2036200
Silva ST, Bertolucci SKV, Cunha SHB, Lazzarini LES, Tavares MC, Pinto JEBP (2017) Effect of light and natural ventilation systems on the growth parameters and carvacrol content in the in vitro cultures of Plectranthusamboinicus (Lour.) Spreng. Plant Cell Tissue Organ Cult 129:501–510. https://doi.org/10.1007/s11240-017-1195-6
Simmonds JA, Cumming BG (1976) Propagation of Lilium hybrids. II. Production of plantlets from bulb-scale callus cultures for increased propagation rates. Sci Hortic 5:161–170. https://doi.org/10.1016/0304-4238(76)90078-9
Singleton VS, Rossi JA Jr (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagent. Amer J Enol Viticult 16:144–157.http://www.ajevonline.org/content/16/3/144.full.pdf+html
Stewart JL, Maloof JN, Nemhauser JL (2011) PIF genes mediate the effect of sucrose on seedling growth dynamics. PLoS ONE 6:e19894. https://doi.org/10.1371/journal.pone.0019894
Sumanta N, Haque CI, Nishika J, Suprakash R (2014) Spectrophotometric analysis of chlorophylls and carotenoids from commonly grown fern species by using various extracting solvents. Res J Chem Sci 4(9):63–69. https://doi.org/10.1055/s-0033-1340072
Takayama S, Misawa M (1979) Differentiation in Liliumbulbscales grown in vitro. Effect of various cultural conditions. Physiol Plant 46:184–190. https://doi.org/10.1111/j.1399-3054.1979.tb06555.x
Taulavuori K, Pyysalo A, Taulavuori E, Julkunen-Tiitto R (2018) Responses of phenolic acid and flavonoid synthesis to blue and blue-violet light depends on plant species. Environ Exp Bot 150:183–187. https://doi.org/10.1016/j.envexpbot.2018.03.016
Tegelberg R, Julkunen-Tiitto R, Aphalo PJ (2004) Red:far-red light ratio and UV-B radiation: their effects on leaf phenolics and growth of silver birch seedlings. Plant, Cell Environ 27:1005–1013. https://doi.org/10.1111/j.1365-3040.2004.01205.x
Tomaszkiewicz W, Cioć M, Dos Santos SK, Grzyb M, Pietrzak W, Pawłowska B, Mikuła A (2022) Enhancing in vitro production of the tree fern Cyathea delgadiiand modifying secondary metabolite profiles by LED lighting. Cells 11(3):486. https://doi.org/10.3390/cells11030486
Tůmová L, Tůma J (2011) The effect of UV light on isoflavonoid production in Genista tinctoria culture in vitro. Acta Physiol Plant 33:635–640. https://doi.org/10.1007/s11738-010-0566-y
Vachálkova A, Eisenreichowá E, Haladová M, Mučaji P, Jóźová B, Novotny L (2000) Potential carcinogenic and inhibitory activity of compounds isolated from Lilium candidum L. Neoplasma 47(5):313–318
Varshney A, Dhawan V, Srivastava PS (2000) A protocol for in vitro mass propagation of Asiatic hybrids of lily through liquid stationary culture. In Vitro Cell Dev Biol Plant 36:383–391. https://doi.org/10.1007/s11627-000-0069-4
Victório CP, Leal-Costa MV, Tavares ES, Kuster RM, Lage CLS (2011) Effects of supplemental UV-A on the development, anatomy and metabolite production of Phyllanthus tenellus cultured in vitro. Photochem Photobiol 87:685–689. https://doi.org/10.1111/j.1751-1097.2011.00905.x
von Lintig J, Welsh R, Bonk M, Giuliano G, Batschauer A, Kleining H (1997) Light-dependent regulation of carotenoid biosynthesis occurs at the level of phytoene synthase expression and is mediated by phytochrome in Sinapis alba and Arabidopsis thaliana seedlings. Plant J 12(3):625–34. https://doi.org/10.1046/j.1365-313x.1997.00625.x
Werbrouck S, Buyle H, Geelen D, Van Labeke MC (2012) Effect of red-, far-red- and blue-light-emitting diodes on in vitro growth of Ficus benjamina. Acta Hort 961(961):533–538. https://doi.org/10.17660/ActaHortic.2012.961.70
Xu L, Naeem U, Najeeb MS, Wan GL, Jin ZL, Khan F, Zhou WJ (2012) In vitro mutagenesis and genetic improvement. In: Gupta SK (ed) Technological innovations in major world oil crops, vol 2. Springer, New York, pp 151–173. https://doi.org/10.1007/978-1-4614-0827-7_6
Yarmolinsky L, Zaccai M, Ben-Shabat S, Mills D, Huleihel M (2009) Antiviral activity of ethanol extracts of Ficus benjamina and Lilium candidum in vitro. New Biotechnol 26:307–313. https://doi.org/10.1016/j.nbt.2009.08.005
Zaccai M, Ram A, Mazor I (2009) Lilium candidum: flowering characterization of wild Israeli ecotypes. Israel J Plant Sci 57:297–302. https://doi.org/10.1560/IJPS.57.4.297
Zhang Y, Liu Z, Wang J, Chen Y, Bi Y, He J (2015) Brassinosteroid is required for sugar promotion of hypocotyl elongation in Arabidopsis in darkness. Planta 242(4):881–893. https://doi.org/10.1007/s00425-015-2328-y
Ӧzen F, Temeltaş H, Aksoy Ӧ (2012) The anatomy and morphology of the medicinal plant, Lilium candidum L. (Liliaceae) distributed in Marmara region of Turkey. Pak J Bot 44(4):1185–1192
Acknowledgements
The project was supported by the Polish Ministry of Science and Higher Education.
Funding
The project was supported by the Polish Ministry of Science and Higher Education.
Author information
Authors and Affiliations
Contributions
PP: writing of original draft, project administration, validation, formal analysis, investigation, data curation and visualization. MC: validation, review and editing. KH: methodology, software, validation and resources. BST: review and editing. BP: supervision, conceptualization, methodology, validation, resources, review and editing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Informed consent
Informed consent was obtained from all individual participants included in the study. Additional informed consent was obtained from all individual participants for whom identifying information is included in this article.
Human and animal rights
This research did not involve experiments with human or animal participants.
Additional information
Communicated by Christophe Hano.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pałka, P., Cioć, M., Hura, K. et al. Adventitious organogenesis and phytochemical composition of Madonna lily (Lilium candidum L.) in vitro modeled by different light quality. Plant Cell Tiss Organ Cult 152, 99–114 (2023). https://doi.org/10.1007/s11240-022-02391-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-022-02391-5