Abstract
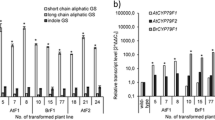
Catharanthus roseus produces many pharmaceutically important terpenoid indole alkaloids (TIAs) such as vinblastine, vincristine, ajmalicine, and serpentine. Past metabolic engineering efforts have pointed to the tight regulation of the TIA pathway and to multiple rate-limiting reactions. Transcriptional regulator ORCA3 (octadecanoid responsive Catharanthus AP2-domain protein), activated by jasmonic acid, plays a central role in regulating the TIA pathway. In this study, overexpressing ORCA3 under the control of a glucocorticoid-inducible promoter in C. roseus hairy roots resulted in no change in the total amount of TIAs measured. RT-qPCR results showed that ORCA3 overexpression triggered the upregulation of transcripts of most of the known TIA pathway genes. One notable exception was the decrease in strictosidine glucosidase (SGD) transcripts. These results corresponded to previously published results. In this study, ORCA3 and SGD were both engineered in hairy roots under the control of a glucocorticoid-inducible promoter. Co-overexpression of ORCA3 and SGD resulted in a significant (p < 0.05) increase in serpentine by 44 %, ajmalicine by 32 %, catharanthine by 38 %, tabersonine by 40 %, lochnericine by 60 % and hörhammericine by 56 % . The total alkaloid pool was increased significantly by 47 %. Thus, combining overexpression of a positive regulator and a pathway gene which is not controlled by this regulator provided a way to enhance alkaloid production.






Similar content being viewed by others
Abbreviations
- 7-DLGT:
-
7-deoxyloganetic acid-O-glucosyl transferase
- 7-DLH:
-
7-deoxyloganic acid hydroxylase
- ADH:
-
alcohol dehydrogenase
- ASα:
-
anthranilate synthase alpha subunit
- BPF:
-
box P-binding factor
- CPR:
-
cytochrome P450 reductase
- CRSDH4H:
-
desacetoxyvindoline 4-hydroxylase
- D4H:
-
desacetoxyvindoline 4-hydroxylase
- DAT:
-
deacetylvindoline acetyltransferase
- DMAPP:
-
dimethylallyl pyrophosphate
- DXR:
-
1-deoxy-d-xylulose-5-phosphate reductoisomerase
- DXS:
-
1-deoxy-d-xylulose 5-phosphate synthase
- G10H:
-
geraniol 10-hydroxylase
- GBF:
-
G-box binding factor
- GPPS:
-
geranyl diphosphate synthase
- IO:
-
iridoid oxidase
- IPP:
-
isopentenyl pyrophosphate
- IS:
-
iridoid synthase
- JA:
-
jasmonic acid
- LAMT:
-
loganic acid methyltransferase
- MAT:
-
minovincinine 19-hydroxy-O-acetyltransferase
- MEP:
-
2-C-Methyl-d-erythritol 4-phosphate
- MYC2:
-
MYC2 transcription factor
- ORCAs:
-
octadecanoid responsive Catharanthus AP2-domain proteins
- Prx1:
-
peroxidase
- SGD:
-
strictosidine beta-glucosidase
- SLS:
-
secologanin synthase
- STR:
-
strictosidine synthase
- T16H:
-
tabersonine 16-hydroxylase
- T19H:
-
tabersonine 19-hydroxylase
- TDC:
-
tryptophan decarboxylase
- TIA:
-
terpenoid indole alkaloid
- WRKY1:
-
WRKY transcription factor 1
- ZCT:
-
transcription factor IIIA-type zinc finger family
References
Bhadra R, Vani S, Shanks JV (1993) Production of indole alkaloids by selected hairy root lines of Catharanthus roseus. Biotechnol Bioeng 41:581–592
De Geyter N, Gholami A, Goormachtig S, Goossens A (2012) Transcriptional machineries in jasmonate-elicited plant secondary metabolism. Trends Plant Sci 17:349–359
Dostal V, Libusova L (2014) Microtubule drugs: action, selectivity, and resistance across the kingdoms of life. Protoplasma 251:991–1005
El-Sayed M, El-Sayed M, Verpoorte R (2007) Catharanthus terpenoid indole alkaloids: biosynthesis and regulation. Phytochem Rev 6:277
Fulzele DP, Heble MR (1994) Large-scale cultivation of Catharanthus roseus cells—production of ajmalicine in a 20-1-airlift bioreactor. J Biotechnol 35:1–7
Gelvin SB (2003) Agrobacterium-mediated plant transformation: the biology behind the “gene-jockeying” tool. Microbiol Mol Biol Rev 67:16–37
Gendenshtein É, Mikhailenko LA (1964) Antiarrhythmic action of serpentine in some experimental disturbances of the auricular and ventricular rhythm. Bull Exp Biol Med 57:446–449
Giddings L-A, Liscombe D, Hamilton J, Childs K, DellaPenna D, Buell CR, O’Connor S (2011) A stereoselective hydroxylation step of alkaloid biosynthesis by a unique cytochrome P450 in Catharanthus roseus. J Biol Chem 286:16751–16757
Glenn WS, Runguphan W, O’Connor SE (2013) Recent progress in the metabolic engineering of alkaloids in plant systems. Curr Opin Biotechnol 24:354–365
Hughes EH, Hong SB, Shanks JV, San KY, Gibson SI (2002) Characterization of an inducible promoter system in Catharanthus roseus hairy roots. Biotechnol Prog 18:1183–1186
Hughes EH, Hong SB, Gibson SI, Shanks JV, San KY (2004a) Expression of a feedback-resistant anthranilate synthase in Catharanthus roseus hairy roots provides evidence for tight regulation of terpenoid indole alkaloid levels. Biotechnol Bioeng 86:718–727
Hughes EH, Hong SB, Gibson SI, Shanks JV, San KY (2004b) Metabolic engineering of the indole pathway in Catharanthus roseus hairy roots and increased accumulation of tryptamine and serpentine. Metab Eng 6:268–276
Jordan MA, Thrower D, Wilson L (1991) Mechanism of inhibition of cell-proliferation by vinca alkaloids. Cancer Res 51:2212–2222
Li CY, Leopold AL, Sander GW, Shanks JV, Zhao L, Gibson SI (2013) The ORCA2 transcription factor plays a key role in regulation of the terpenoid indole alkaloid pathway. Bmc Plant Biol 13:155
Liscombe DK, O’Connor SE (2011) A virus-induced gene silencing approach to understanding alkaloid metabolism in Catharanthus roseus. Phytochemistry 72:1969–1977
Luijendijk TJC, Stevens LH, Verpoorte R (1998) Purification and characterisation of strictosidine beta-D-glucosidase from Catharanthus roseus cell suspension cultures. Plant Physiol Biochem 36:419–425
Makhzoum A, Petit-Paly G, St Pierre B, Bernards MA (2011) Functional analysis of the DAT gene promoter using transient Catharanthus roseus and stable Nicotiana tabacum transformation systems. Plant Cell Rep 30:1173–1182
Memelink J, Gantet P (2007) Transcription factors involved in terpenoid indole alkaloid biosynthesis in Catharanthus roseus. Phytochem Rev 6:353–362
Menke FLH, Parchmann S, Mueller MJ, Kijne JW, Memelink J (1999) Involvement of the octadecanoid pathway and protein phosphorylation in fungal elicitor-induced expression of terpenoid indole alkaloid biosynthetic genes in Catharanthus roseus. Plant Physiol 119:1289–1296
Mérillon JM, Doireau P, Guillot A, Chénieux JC, Rideau M (1986) Indole alkaloid accumulation and tryptophan decarboxylase activity in Catharanthus roseus cells cultured in three different media. Plant Cell Rep 5:23–26
Miettinen K et al (2014) The seco-iridoid pathway from Catharanthus roseus. Nat Commun 5:1–11
Morgan JA, Shanks JV (1999) Inhibitor studies of tabersonine metabolism in C. roseus hairy roots. Phytochemistry 51:61–68
Morgan JA, Barney CS, Penn AH, Shanks JV (2000) Effects of buffered media upon growth and alkaloid production of Catharanthus roseus hairy roots. Appl Microbiol Biotechnol 53:262–265
Ngan VK, Bellman K, Hill BT, Wilson L, Jordan MA (2001) Mechanism of mitotic block and inhibition of cell proliferation by the semisynthetic vinca alkaloids vinorelbine and its newer derivative vinflunine. Mol Pharmacol 60:225–232
O’Connor SE, Maresh JJ (2006) Chemistry and biology of monoterpene indole alkaloid biosynthesis. Nat Prod Rep 23:532–547
Pan QF et al (2012) Overexpression of ORCA3 and G10H in Catharanthus roseus plants regulated alkaloid biosynthesis and metabolism revealed by NMR-metabolomics. PLoS ONE 7:1–14
Pasquali G, Goddijn OJM, de Waal A, Verpoorte R, Schilperoort RA, Hoge JHC, Memelink J (1992) Coordinated regulation of two indole alkaloid biosynthetic genes from Catharanthus roseus by auxin and elicitors. Plant Mol Biol 18:1121
Peebles CAM, Hong SB, Gibson SI, Shanks JV, San KY (2005) Transient effects of overexpressing anthranilate synthase alpha and beta subunits in Catharanthus roseus hairy roots. Biotechnol Prog 21:1572–1576
Peebles CAM, Hong SB, Gibson SI, Shanks JV, San KY (2006) Effects of terpenoid precursor feeding on Catharanthus roseus hairy roots over-expressing the alpha or the alpha and beta subunits of anthranilate synthase. Biotechnol Bioeng 93:534–540
Peebles CAM, Hughes EH, Shanks JV, San KY (2009) Transcriptional response of the terpenoid indole alkaloid pathway to the overexpression of ORCA3 along with jasmonic acid elicitation of Catharanthus roseus hairy roots over time. Metab Eng 11:76–86
Peebles CAM, Sander GW, Hughes EH, Peacock R, Shanks JV, San KY (2011) The expression of 1-deoxy-D-xylulose synthase and geraniol-10-hydroxylase or anthranilate synthase increases terpenoid indole alkaloid accumulation in Catharanthus roseus hairy roots. Metab Eng 13:234–240
Rowinsky EK, Donehower RC (1991) The clinical pharmacology and use of antimicrotubule agents in cancer chemotherapeutics. Pharmacol Ther 52:35–84
Shalel-Levanon S, San KY, Bennett GN (2005) Effect of oxygen, and ArcA and FNR regulators on the expression of genes related to the electron transfer chain and the TCA cycle in Escherichia coli. Metab Eng 7:364–374
Shukla AK, Shasany AK, Verma RK, Gupta MM, Mathur AK, Khanuja SPS (2010) Influence of cellular differentiation and elicitation on intermediate and late steps of terpenoid indole alkaloid biosynthesis in Catharanthus roseus. Protoplasma 242:35–47
Suttipanta N, Pattanaik S, Gunjan S, Xie CH, Littleton J, Yuan L (2007) Promoter analysis of the Catharanthus roseus geraniol 10-hydroxylase gene involved in terpenoid indole alkaloid biosynthesis. Biochim Biophys Acta Gene Struct Expr 1769:139–148
Suttipanta N, Pattanaik S, Kulshrestha M, Patra B, Singh S, Yuan L (2011) The transcription factor CrWRKY1 positively regulates the terpenoid indole alkaloid biosynthesis in Catharanthus roseus. Plant Physiol 157:2081–2093
van der Fits L, Memelink J (2000) ORCA3, a jasmonate-responsive transcriptional regulator of plant primary and secondary metabolism. Science 289:295–297
van der Fits L, Memelink J (2001) The jasmonate-inducible AP2/ERF-domain transcription factor ORCA3 activates gene expression via interaction with a jasmonate-responsive promoter element. Plant J 25:43–53
van der Heijden R, Jacobs DI, Snoeijer W, Hallared D, Verpoorte R (2004) The Catharanthus alkaloids: pharmacognosy and biotechnology. Curr Med Chem 11:607–628
Verma P, Mathur AK, Srivastava A, Mathur A (2012) Emerging trends in research on spatial and temporal organization of terpenoid indole alkaloid pathway in Catharanthus roseus: a literature update. Protoplasma 249:255–268
Viale PH (2013) The continuing impact of oncology drug shortages. J Adv Pract Oncol 4:81
Wang Q, Yuan F, Pan QF, Li MY, Wang GF, Zhao JY, Tang KX (2010) Isolation and functional analysis of the Catharanthus roseus deacetylvindoline-4-O-acetyltransferase gene promoter. Plant Cell Rep 29:185–192
Wasternack C (2014) Action of jasmonates in plant stress responses and development applied aspects. Biotechnol Adv 32:31–39
Zhao J, Hu Q, Guo YQ, Zhu WH (2001) Effects of stress factors, bioregulators, and synthetic precursors on indole alkaloid production in compact callus clusters cultures of Catharanthus roseus. Appl Microbiol Biotechnol 55:693–698
Zhu X, Zeng X, Sun C, Chen S (2014) Biosynthetic pathway of terpenoid indole alkaloids in Catharanthus roseus. Front Med 8:285–293
Acknowledgments
The authors express our gratitude to Dr. Ka-Yiu San at Rice University for providing A. rhizogenes 15834 strain and plasmids pTA7002/ORCA3 and pUCGALA. The authors would like to thank Dr. Nam-Hai Chua at the Rockefeller University for providing the inducible promoter plasmid (pTA7002) and Dr. Jacqueline V Shanks at Iowa State University for providing the lochnericine and hörhammericine standards. This work was supported by Colorado State University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Peter Nick
Rights and permissions
About this article
Cite this article
Sun, J., Peebles, C.A.M. Engineering overexpression of ORCA3 and strictosidine glucosidase in Catharanthus roseus hairy roots increases alkaloid production. Protoplasma 253, 1255–1264 (2016). https://doi.org/10.1007/s00709-015-0881-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-015-0881-7