Abstract
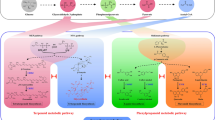
Terpenoid indole alkaloids (TIA) are important pharmaceuticals produced in low amounts through the terpenoid indole alkaloid pathway in Catharanthus roseus. Production of these alkaloids is controlled by biosynthetic and regulatory genes. CrMYC1 is a chief transcription factor that regulates terpenoid indole alkaloid production. In this study, we cloned CrMYC1 and overexpressed it in C. roseus hairy roots to investigate its effect on the production of the TIAs ajmalicine and catharanthine. qRT-PCR results revealed that CrMYC1 expression in transgenic CrMYC1-overexpressing hairy root lines was 3–6 times higher than that of the wild-type hairy root control line. Also, we detected a significant increase in strictosidine β-D-glucosidase (SGD) expression, a critical enzyme in TIA biosynthesis, in the overexpression lines. HPLC analysis showed that CrMYC1 overexpression significantly improved the accumulation of ajmalicine (13 to 14-fold) and catharanthine (3 to 4.4-fold) compared to the control hairy root line. These results provide insight into the role of this understudied transcription factor in TIA pathway regulation and emphasize further research objectives.




Similar content being viewed by others
Data availability
All data are included in this published article and its supplementary information files.
References
Abe H, Yamaguchi-Shinozaki K, Urao T, Iwasaki T, Hosokawa D, Shinozaki K (1997) Role of arabidopsis MYC and MYB homologs in drought-and abscisic acid-regulated gene expression. Plant Cell 9:1859–1868. https://doi.org/10.1105/tpc.9.10.1859
Arafa AS, Ragab AE, Ibrahim AS, Abdel-Mageed WS, Nasr ME (2019) Cloning and overexpression of strictosidine β-D-GLUCOSIDASE GENE SHORT Sequence from Catharanthus roseus in Escherichia coli. Adv Pharm Bull 4:655–661. https://doi.org/10.15171/apb.2019.076
Brown S, Clastre M, Courdavault V, O’Connor SE (2015) De novo production of the plant-derived alkaloid strictosidine in yeast. PNAS 11:3205–3210. https://doi.org/10.1073/pnas.1423555112
Caputi L, Franke J, Farrow SC, Chung K, Payne RM, Nguyen TD, Dang TT, Carqueijeiro IS, Koudounas K, de Bernonville TD, Ameyaw B (2018) Missing enzymes in the biosynthesis of the anticancer drug vinblastine in Madagascar periwinkle. Science 360:1235–1239. https://doi.org/10.1126/science.aat4100
Chatel G, Montiel G, Pré M, Memelink J, Thiersault M, Saint-Pierre B, Doireau P, Gantet P (2003) CrMYC1, a C. roseus elicitor-and jasmonate-responsive bHLH transcription factor that binds the G-box element of the strictosidine synthase gene promoter. J Exp Bot 54:2587–2588. https://doi.org/10.1093/jxb/erg275
Cui L, Ni X, Ji Q, Teng X, Yang Y, Wu C, Zekria D, Zhang D, Kai G (2015) Co-overexpression of geraniol-10-hydroxylase and strictosidine synthase improves anti-cancer drug camptothecin accumulation in Ophiorrhiza pumila. Sci Rep 5:8227–8233. https://doi.org/10.1038/srep08227
Ferreres F, Pereira DM, Valentão P, Oliveira JM, Faria J, Gaspar L, Sottomayor M, Andrade PB (2010) Simple and reproducible HPLC–DAD–ESI-MS/MS analysis of alkaloids in Catharanthus roseus roots. J Pharm Biomed Anal 51:65–69. https://doi.org/10.1016/j.jpba.2009.08.005
Geerlings A, Ibanez MML, Memelink J, van der Heijden R, Verpoorte R (2000) Molecular cloning and analysis of strictosidine β-D-glucosidase, an enzyme in terpenoid indole alkaloid biosynthesis in Catharanthus roseus. J Biol 275:3051–3056. https://doi.org/10.1074/jbc.275.5.3051
Guillon S, Tremouillaux-Guiller J, Pati PK, Rideau M, Gantet P (2006) Harnessing the potential of hairy roots: dawn of a new era. Trends Biotechnol 24:403–409. https://doi.org/10.1016/j.tibtech.2006.07.002
Gutierrez-Valdes N, Häkkinen ST, Lemasson C, Guillet M, Oksman-Caldentey KM, Ritala A, Cardon F (2020) Hairy root cultures a versatile tool with multiple applications. Front Plant Sci 11:1–11. https://doi.org/10.3389/fpls.2020.00033
Hanafy M, Matter M, Asker M, Rady M (2016) Production of indole alkaloids in hairy root cultures of Catharanthus roseus L. and their antimicrobial activity. S Afr J Bot 105:9–18. https://doi.org/10.1016/j.sajb.2016.01.004
Heim MA, Jakoby M, Werber M, Martin C, Weisshaar B, Bailey PC (2003) The basic Helix-Loop-Helix transcription factor family in plants: a genome-wide study of protein structure and functional diversity. Mol Biol Evol 5:735–747. https://doi.org/10.1093/molbev/msg088
Hernandez-Dominguez E, Campos-Tamayo F, Vazquez-Flota F (2004) Vindoline synthesis in in vitro shoot cultures of Catharanthus roseus. Biotechnol Lett 8:671–674. https://doi.org/10.1023/b:bile.0000023028.21985.07
Higo K, Ugawa Y, Iwamoto M, Korenaga T (1999) Plant cis-acting regulatory DNA elements (PLACE) database. Nucleic Acids Res 27:297–300. https://doi.org/10.1093/nar/27.1.297
Jaggi M, Kumar S, Sinha AK (2011) Overexpression of an apoplastic peroxidase gene CrPrx in transgenic hairy root lines of Catharanthus roseus. Appl Microbiol Biotechnol 90:1005–1016. https://doi.org/10.1007/s00253-011-3131-8
Koul M, Lakra NS, Chandra R, Chandra S (2013) Catharanthus roseus and prospects of its endophytes: a new avenue for production of bioactive metabolites. Int J Pharm Sci Res 4:2705–2716. https://doi.org/10.13040/IJPSR.0975-8232.4(7).2705-16
Kumar S, Shilpashree HB, Nagegowda DA (2018) Terpene moiety enhancement by overexpression of geranyl (geranyl) diphosphate synthase and geraniol synthase elevates monomeric and dimeric monoterpene indole alkaloids in transgenic catharanthus roseus. Front Plant Sci 9:942–958. https://doi.org/10.3389/fpls.2018.00942
Kutchan TM, Frick S, Weid M (2008) Engineering plant alkaloid biosynthetic pathways: progress and prospects. Adv Plant Biochem Mol Biol 1:283–310. https://doi.org/10.1016/S1755-0408(07)01010-7
Larionov A, Krause A, Miller WA (2005) standard curve based method for relative real time PCR data processing. BMC Bioinform 6:62–66. https://doi.org/10.1186/1471-2105-6-62
Li CY, Leopold AL, Sander GW, Shanks JV, Zhao L, Gibson SI (2013) The ORCA2 transcription factor plays a key role in regulation of the terpenoid indole alkaloid pathway. BMC Plant Biol 13:155–172. https://doi.org/10.1186/1471-2229-13-155
Li CY, Leopold AL, Sander GW, Shanks JV, Zhao L, Gibson SI (2015) CrBPF1 overexpression alters transcript levels of terpenoid indole alkaloid biosynthetic and regulatory genes. Front Plant Sci. https://doi.org/10.3389/fpls.2015.00818
Liscombe DK, O’Connor SE (2011) A virus-induced gene silencing approach to understanding alkaloid metabolism in Catharanthus roseus. Phytochemistry 72:1969–1977. https://doi.org/10.1016/j.phytochem.2011.07.001
Liu DH, Ren WW, Cui LJ, Zhang LD, Sun XF, Tang KX (2011) Enhanced accumulation of catharanthine and vindoline in Catharanthus roseus hairy roots by overexpression of transcriptional factor ORCA2. Afr J Biotech 10:3260–3268. https://doi.org/10.5897/AJB10.1556
Liu J, Cai J, Wang R, Yang S (2017a) Transcriptional regulation and transport of terpenoid indole alkaloid in Catharanthus roseus: exploration of new research directions. Int J Mol Sci 18:53–73. https://doi.org/10.3390/ijms18010053
Liu J, Gao F, Ren J, Lu X, Ren G, Wang R (2017b) A novel AP2/ERF transcription factor CR1 regulates the accumulation of vindoline and serpentine in Catharanthus roseus. Front Plant Sci 8:2082–2093. https://doi.org/10.3389/fpls.2017.02082
Liu J, Zhu J, Tang L, Wen W, Lv S, Yu R (2014) Enhancement of vindoline and vinblastine production in suspension-cultured cells of Catharanthus roseus by artemisinic acid elicitation. World J Microbiol Biotechnol 30:175–180. https://doi.org/10.1007/s11274-013-1432-z
Liu S, Dang M, Lei Y, Ahmad SS, Khalid M, Kamal MA, Chen L (2020) Ajmalicine and its Analogues against AChE and BuChE for the Management of Alzheimer’s Disease: An In-silico study. Curr Pharm Des 37:4808–4814. https://doi.org/10.2174/1381612826666200407161842
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Maqsood M, Abdul M (2017) Yeast extract elicitation increases vinblastine and vincristine yield in protoplast derived tissues and plantlets in Catharanthus roseus. Rev Bras Farmacogn 27:549–556. https://doi.org/10.1016/j.bjp.2017.05.008
Mehrotra S, Mishra S, Srivastava V (2018) Hairy root cultures for monoterpene indole alkaloid pathway: investigation and biotechnological production. In: Srivastava V, Mehrotra S, Mishra S (eds) Hairy Roots, pp 95–121. Springer, Singapore, https://doi.org/10.1007/978-981-13-2562-5_5
Mishra MR, Srivastava RK, Akhtar N (2018) Enhanced alkaloid production from cell culture system of catharanthus roseus in combined effect of nutrient salts, sucrose and plant growth regulators. J Biotechnol Biomed Sci 4:14–34. https://doi.org/10.14302/issn.2576-6694.jbbs-18-2475
Nammi S, Boini MK, Lodagala SD, Behara RBS (2003) The juice of fresh leaves of Catharanthus roseus Linn. reduces blood glucose in normal and alloxan diabetic rabbits. BMC Complement Altern Med 3:1–4. https://doi.org/10.1186/1472-6882-3-4
Pan YJ, Lin YC, Yu BF, Zu YG, Yu F, Tang ZH (2018) Transcriptomics comparison reveals the diversity of ethylene and methyl-jasmonate in roles of TIA metabolism in Catharanthus roseus. BMC Genomics 1:508–522. https://doi.org/10.1186/s12864-018-4879-3
Patra B, Pattanaik S, Schluttenhofer C, Yuan L (2018) A network of jasmonate-responsive bHLH factors modulate monoterpenoid indole alkaloid biosynthesis in Catharanthus roseus. New Phytol 217:1566–1581. https://doi.org/10.1111/nph.14910
Peebles CA, Sander GW, Hughes EH, Peacock R, Shanks JV, San KY (2011) The expression of 1-deoxy-d-xylulose synthase and geraniol-10-hydroxylase or anthranilate synthase increases terpenoid indole alkaloid accumulation in Catharanthus roseus hairy roots. Metab Eng 13:234–240. https://doi.org/10.1016/j.ymben.2010.11.005
Peebles CA, Hughes EH, Shanks JV, San KY (2009) Transcriptional response of the terpenoid indole alkaloid pathway to the overexpression of ORCA3 along with jasmonic acid elicitation of Catharanthus roseus hairy roots over time. Metab Eng 11:76–86. https://doi.org/10.1016/j.ymben.2008.09.002
Qu Y, Easson ML, Froese J, Simionescu R, Hudlicky T, De Luca V (2015) Completion of the seven-step pathway from tabersonine to the anticancer drug precursor vindoline and its assembly in yeast. PNAS 19:6224–6229. https://doi.org/10.1073/pnas.1501821112
Rai A, Smita SS, Singh AK, Shanker K, Nagegowda DA (2013) Heteromeric and homomeric geranyl diphosphate synthases from Catharanthus roseus and their role in monoterpene indole alkaloid biosynthesis. Mol Plant 6:1531–1549. https://doi.org/10.1093/mp/sst058
Ruiz-May E, Galaz-Avalos RM, Loyola-Vargas VM (2009) Differential secretion and accumulation of terpene indole alkaloids in hairy roots of Catharanthus roseus treated with methyl jasmonate. Mol Biotechnol 41:278–285. https://doi.org/10.1007/s12033-008-9111-2
Salim V, De Luca V (2013) Towards complete elucidation of monoterpene indole alkaloid biosynthesis pathway: catharanthus roseus as a pioneer system. In: Advances in botanical research vol 68, pp 1–37. Academic Press. https://doi.org/10.1016/B978-0-12-408061-4.00001-8
Sharma A, Verma P, Mathur A, Mathur AK (2018a) Genetic engineering approach using early Vinca alkaloid biosynthesis genes led to increased tryptamine and terpenoid indole alkaloids biosynthesis in differentiating cultures of Catharanthus roseus. Protoplasma 255:425–435. https://doi.org/10.1007/s00709-017-1151-7
Sharma A, Verma P, Mathur A, Mathur AK (2018b) Overexpression of tryptophan decarboxylase and strictosidine synthase enhanced terpenoid indole alkaloid pathway activity and antineoplastic vinblastine biosynthesis in Catharanthus roseus. Protoplasma 255:1281–1294. https://doi.org/10.1007/s00709-018-1233-1
Shukla AK, Shasany AK, Gupta MM, Khanuja SP (2006) Transcriptome analysis in Catharanthus roseus leaves and roots for comparative terpenoid indole alkaloid profiles. J Exp Bot 57:3921–3932. https://doi.org/10.1093/jxb/erl146
Shukla AK, Khanuja, SP (2013) Catharanthus roseus: The metabolome that represents a unique reservoir of medicinally important alkaloids under precise genomic regulation. In: Debmalya Barh (Ed) OMICS applications in crop science. pp 345–404. CRC Press, Doi: https://doi.org/10.1201/b16352-11
Siddiqui MJA, Ismail Z, Saidan NH (2011) Simultaneous determination of secondary metabolites from Vinca rosea plant extractives by reverse phase high performance liquid chromatography. Pharmacogn Mag 7:92–96. https://doi.org/10.4103/0973-1296.80662
Stavrinides A, Tatsis EC, Foureau E, Caputi L, Kellner F, Courdavault V, O’Connor SE (2015) Unlocking the diversity of alkaloids in Catharanthus roseus: nuclear localization suggests metabolic channeling in secondary metabolism. Chem Biol 22:336–341. https://doi.org/10.1016/j.chembiol.2015.02.006
Stewart Jr CN, Via LEA (1993) rapid CTAB DNA isolation technique useful for RAPD fingerprinting and other PCR applications. Biotechniques 14:748–50. https://www.ncbi.nlm.nih.gov/pubmed/8512694
Sun J, Peebles CA (2016) Engineering overexpression of ORCA3 and strictosidine glucosidase in Catharanthus roseus hairy roots increases alkaloid production. Protoplasma 253:1255–1264. https://doi.org/10.1007/s00709-015-0881-7
Suttipanta N, Pattanaik S, Kulshrestha M, Patra B, Singh SK, Yuan L (2011) The transcription factor CrWRKY1 positively regulates the terpenoid indole alkaloid biosynthesis in Catharanthus roseus. Plant Physiol 157:2081–2093. https://doi.org/10.1104/pp.111.181834
Thamm AM, Qu Y, De Luca V (2016) Discovery and metabolic engineering of iridoid/secoiridoid and monoterpenoid indole alkaloid biosynthesis. Phytochem Rev 3:339–361. https://doi.org/10.1007/s11101-016-9468-y
Tiong SH, Looi CY, Hazni H, Arya A, Paydar M, Wong WF, Cheah S-C, Mustafa MR, Awang K (2013) Antidiabetic and antioxidant properties of alkaloids from Catharanthus roseus (L.) G. Don Molecules 18:9770–9784. https://doi.org/10.3390/molecules18089770
Van Moerkercke A, Steensma P, Gariboldi I, Espoz J, Purnama PC, Schweizer F, Miettinen K, Vanden Bossche R, De Clercq R, Memelink J (2016) The basic helix-loop-helix transcription factor BIS2 is essential for monoterpenoid indole alkaloid production in the medicinal plant Catharanthus roseus. Plant J 8:3–12. https://doi.org/10.1111/tpj.13230
Verma M, Ghangal R, Sharma R, Sinha AK, Jain M (2014) Transcriptome analysis of Catharanthus roseus for gene discovery and expression profiling. PLoS ONE 9:e103583. https://doi.org/10.1371/journal.pone.0103583
Verma P, Mathur AK, Masood N, Luqman S, Shanker K (2013) Tryptophan overproducing cell suspensions of Catharanthus roseus (L) G. Don and their upscaling in stirred tank bioreactor: detection of a phenolic compound with antioxidant potential. Protoplasma 250:371–380. https://doi.org/10.1007/s00709-012-0423-5
Verma P, Mathur AK, Srivastava A, Mathur A (2012) Emerging trends in research on spatial and temporal organization of terpenoid indole alkaloid pathway in Catharanthus roseus: a literature update. Protoplasma 249:255–268. https://doi.org/10.1007/s00709-011-0291-4
Verpoorte R, Contin A, Memelink J (2002) Biotechnology for the production of plant secondary metabolites. Phytochem Rev 1:13–25. https://doi.org/10.1023/A:1015871916833
Yamada Y, Koyama T, Sato F (2011) Basic helix-loop-helix transcription factors and regulation of alkaloid biosynthesis. Plant Signal Behav 6:1627–1630. https://doi.org/10.4161/psb.6.11.17599
Zhang L, Qing-Hui G, Yuan-Gang Z, Lei Y, Yu-Liang M, Yang L (2014) Simultaneous quantitative determination of five alkaloids in Catharanthus roseus by HPLC-ESI-MS/MS. CJNM 12:786–793. https://doi.org/10.1016/S1875-5364(14)60120-5
Zhao L, Sander GW, Shanks JV (2013) Perspectives of the metabolic engineering of terpenoid indole alkaloids in Catharanthus roseus hairy roots. In: Doran, Pauline M (Ed) Biotechnology of Hairy Root Systems pp 23–54. https://doi.org/10.1007/10_2013_182
Zhu X, Zeng X, Sun C, Chen S (2014) Biosynthetic pathway of terpenoid indole alkaloids in Catharanthus roseus. Front Med 8:285–293. https://doi.org/10.1007/s11684-014-0350-2
Acknowledgements
The authors gratefully acknowledge the financial support of Ferdowsi University of Mashad. We thank Rezgar Faraji Sarabmirza, School of Agriculture Science, University of Tehran, for its assistance with HPLC analysis, and Dr. Benye Liu, Institute of Pharmaceutical Biology, Technische Universität Braunschweig for comments that greatly improved the manuscript.
Author information
Authors and Affiliations
Contributions
SS, AN and FSH contributed to the study conception and design. SS Performed the experiments under AN supervision. The first draft of the manuscript was written by SS. AF contributed to the scientific revision of the investigation.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Human and animal rights statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by Jeong Hee Choi.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sazegari, S., Niazi, A., Shahriari-Ahmadi, F. et al. CrMYC1 contributes to catharanthine and ajmalicine accumulation by regulating the TIA pathway in Catharanthus roseus hairy roots. Hortic. Environ. Biotechnol. 63, 709–717 (2022). https://doi.org/10.1007/s13580-022-00430-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13580-022-00430-w