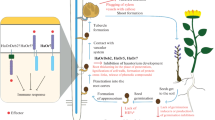
Abstract
The breeding of sunflower (Helianthus annuus L.) for resistance to downy mildew (caused by the oomycete Plasmopara halstedii Farl. Berl. & de Toni) is reviewed in this work under the scope of its sustainability and efficiency. When sunflower turned into an oilseed crop, resistance to the disease was included in its initial breeding strategies. Subsequent development of genomic tools allowed a significant expansion of the knowledge on the diversity of its genetic resistance and its application to the genetic control of the disease. Simultaneously to genetic improvements, and as a consequence of the close interaction between the pathogen and its host plant, an enormous variety of pathotypes has been described in all the sunflower-growing areas worldwide. Thus, the genetic control of sunflower downy mildew is an active research field subjected to continuous evolution and challenge. In practice, genetic resistance constitutes the base tier of Integrated Pest Management against sunflower downy mildew. The second tier is composed of elements related to crop management: rotation, removal of volunteer plants, sowing date, tillage. Biological control alternatives and resistance inducers could also provide additional restraint. Finally, the top tier includes chemical treatments that should only be used when necessary and if the more basal Integrated Pest Management elements fail to keep pathogen populations under the economic threshold.

Similar content being viewed by others
Availability of data and material
All data and materials referred to in this review are available either open access or upon request to the corresponding author of the manuscript.
References
Ahmed S, Tourvieille de Labrouhe D, Delmotte F (2012) Emerging virulence arising from hybridisation facilitated by multiple introductions of the sunflower downy mildew pathogen Plasmopara halstedii. Fungal Genet Biol 49:847–855
Albourie JM, Tourvieille J, Tourvieille de Labrouhe D (1998) Resistance to metalaxyl in isolates of the sunflower pathogen Plasmopara halstedii. Eur J Plant Pathol 104:235–242
As-sadi F, Carrere S, Gascuel Q, Hourlier T, Rengel D, Le Paslier MC, Bordat A, Boniface MC, Brunel D, Gouzy J, Godiard L, Vincourt P (2011) Transcriptomic analysis of the interaction between Helianthus annuus and its obligate parasite Plasmopara halstedii shows single nucleotide polymorphisms in CRN sequences. BMC Genomics 12:498. http://www.biomedcentral.com/1471-2164/12/498
Bachlava E, Radwan OE, Abratti G, Tang S, Gao W, Heesacker AF, Bazzalo ME, Zambelli A, Leon AJ, Knapp SJ (2011) Downy mildew (Pl8 and Pl14) and rust (RAdv) resistance genes reside in close proximity to tandemly duplicated clusters of non-TIR-like NBS-LRR-encoding genes on sunflower chromosomes 1 and 13. Theor Appl Genet 122:1211–1221
Badouin H, Gouzy J et al (2017) The sunflower genome provides insights into oil metabolism, flowering and Asterid evolution. Nature 546(1):148. https://doi.org/10.1038/nature22380
Baldini M, Danuso F, Turi M, Sandra M, Raranciuc S (2008) Main factors influencing downy mildew (Plasmopara halstedii) infection in high-oleic sunflower hybrids in northern Italy. Crop Prot 27:590–599
Bán R, Kovács A, Perczel M, Körösi K, Turóczi G (2014) First report on the increased distribution of pathotype 704 of Plasmopara halstedii in Hungary. Plant Dis 98(6):844. https://doi.org/10.1094/PDIS-09-13-0920-PDN
Bartlett DW, Clough JM, Godwin JR, Hall AA, Hamer M, Parr-Dobrzanski B (2002) The strobilurin fungicides. Pest Manag Sci 58:649–662
Bazzalo ME, Huguet N, Romano MC, Bock F, Piubello S, Quiroz F, Erreguerrena I, Zuil S, Bertero A (2016) History and present state of downy mildew in Argentina. In: International sunflower association (ed) proceedings of the 19th international sunflower conference. Edirne, Turkey, May 29-June 3 2016. International Sunflower Association, Paris, France, vol. 2, pp 800–804
Bert PF, Tourvieille de Labrouhe D, Philippon J, Mouzeyar S, Jouan I, Nicolas P, Vear F (2001) Identification of a second linkage group carrying genes controlling resistance to downy mildew (Plasmopara halstedii) in sunflower (Helianthus annuus L.). Theor Appl Genet 103:992–997
Bertero de Romano A, Romano C, Bulos M, Altieri E, Sala C (2010) A new gene for resistance to downy mildew in sunflower. In: International sunflower association (ed) proceedings of the international symposium “Sunflower breeding on resistance to diseases”. Krasnodar, Russia, June 23–24 2010. All-Russia Research Institute of Oil Crops by V. S. Pustovoit (VNIIMK), Krasnodar, Russia, pp 141–146
Bouzidi MF, Badaoui S, Cambon F, Vear F, De Labrouhe DT, Nicolas P, Mouzeyar S (2002) Molecular analysis of a major locus for resistance to downy mildew in sunflower with specific PCR-based markers. Theor Appl Genet 104:592–600
Carson ML (1981) New race of Plasmopara halstedii virulent on resistant sunflowers in South Dakota. Plan Dis 65:842–843
Cohen Y (1994) 3-Aminobutyric acid induces systemic resistance against Peronospora tabacina. Physiol Mol Plant Pathol 44:273–288
Cohen Y, Coffey MD (1986) Systemic fungicides and the control of oomycetes. Annu Rev Phytopathol 24:311–338
Covarelli L, Tosi L (2006) Presence of sunflower downy mildew in an integrated weed control field trial. Phytopathol 154:281–285
Davidse LC, van den Berg-Velthuis GCM, Mantel BC, Jespers ABK (1991) Phenylamides and Phytophthora. In: Lucas JA, Shattock RC, Shaw DS, Cooke LR (eds) Phytophthora. British Mycological Society, Cambridge, pp 349–360
Delmotte F, Giresse X, Richard-Cervera S, M’Baya J, Vear F, Tourvieille J, Walser P, Tourvieille de Labrouhe D (2008) Single nucleotide polymorphisms reveal multiple introductions into France of Plasmopara halstedii, the plant pathogen causing sunflower downy mildew. Infect Genet Evol 8:534–540
Diaz de la Guardia M, Garcia-Baudin C, Montes-Agusti F, Romero-Munoz F (1981) “Pemir”, variedad española de girasol con resistencia al mildiu. An Inst Nac Invest Agrar 15:13–22
Drabkova Trojanova Z, Sedlarova M, Pospıchalova R, Lebeda A (2018) Pathogenic variability of Plasmopara halstedii infecting sunflower in the Czech Republic. Plant Pathol 67:136–144. https://doi.org/10.1111/ppa.12722
Dußle CM, Hahn V, Knapp SJ, Bauer E (2004) PlArg from Helianthus argophyllus is unlinked to other known downy mildew resistance genes in sunflower. Theor Appl Genet 109:1083–1086
Enns H, Dorrell DG, Hoes HA, Chubb WO (1970) Sunflower research – a progress report. In: International sunflower association (ed) proceedings of the 4th international sunflower conference. Memphis TN, USA, June 23–25 1970. International Sunflower Association, Paris, France, pp 162–167
Er Y, Özer N, Katırcıoğlu YZ (2021) In vivo anti-mildew activity of essential oils against downy mildew of sunflower caused by Plasmopara halstedii. Eur J Plant Pathol 161:619–627. https://doi.org/10.1007/s10658-021-02347-z
Fages J, Lux B (1991) Identification of bacteria isolated from roots of sunflower cultivated in a French soil. Can J Microbiol 37:971–974
FAOSTAT, 2020. https://www.fao.org/faostat/es/#data/QCL. Last accessed Oct. 19, 2021
Fernández-Martínez JM, Domínguez Giménez J (1978) Estudios genéticos de la resistencia al mildiu en girasol (Helianthus annuus L.) Ann INIA Ser Prod Veg 8:105–111.
Fick GN, Miller JF (1997) Sunflower Breeding. In: Schneiter AA (ed) Sunflower technology and production. CSSA, SSSA publishers, Madison WI, ASA, pp 395–439
Fick GN, Zimmer DE (1974) RHA 271, RHA 273, and RHA 274: sunflower parental lines for producing downy mildew resistant hybrids. Farm Res ND Agric Exp Stn Fargo 32(2):7–9
Fick GN, Zimmer DE (1975) Linkage tests among genes for six qualitative characters in sunflowers. Crop Sci 15:777–779
Fick GN, Kinman ML, Zimmer DE (1975) Registration of RHA-273 and RHA-274 sunflower parental lines. Crop Sci 15:106
Franchel J, Bouzidi MF, Bronner G, Vear F, Nicolas P, Mouzeyar S (2013) Positional cloning of a candidate gene for resistance to the sunflower downy mildew, Plasmopara halstedii race 300. Theor Appl Genet 126:359–367. https://doi.org/10.1007/s00122-012-1984-6
Fuchs A (1988) Implications of steroisomerism in agricultural fungicides. In: Ariëns EJ, van Rensen JJS, Welling W (eds) Stereoselectivity of pesticides: biological and chemical problems. Elsevier science Publishers, Amsterdam, pp 203–209
García G, Gulya TJ (1991) Sunflower downy mildew race distribution in North Dakota and Minnesota. In: National sunflower association (ed) Proceedings of the 13th sunflower research workshop, Fargo ND, USA, Jan 10–11 1991. National Sunflower Association, Bismarck ND, USA, pp 3–5
Gascuel Q, Martinez Y, Boniface MC, Vear F, Pichon M, Godiard L (2015) The sunflower downy mildew pathogen Plasmopara halstedii. Mol Plant Pathol 16(2):109–122. https://doi.org/10.1111/mpp.12164
Gedil MA, Slabaugh MB, Berry S, Johnson R, Michelmore R, Miller J, Gulya T, Knapp SJ (2001) Candidate disease resistance genes in sunflower cloned using conserved nucleotide-binding site motifs: genetic mapping and linkage to the downy mildew resistance gene Pl1. Genome 44(2):205–212
Giresse X, Tourvieille de Labrouhe D, Richard-Cervera S, Delmotte F (2007) Twelve polymorphic expressed sequence tags-derived markers for Plasmopara halstedii, the causal agent of sunflower downy mildew. Mol Ecol Notes 7:1363–1365. https://doi.org/10.1111/j.1471-8286.2007.01887.x
Goossen PG, Sackston WE (1968) Transmission, and biology of sunflower downy mildew. Can J Bot 46:5–10
Görlach J, Volrath S, KnaufBeiter G, Hengy G, Beckhove U, Kogel KH, Oostendorp M, Staub T, Ward E, Kessmann H, Ryals J (1996) Benzothiadiazole, a novel class of inducers of systemic acquired resistance, activates gene expression and disease resistance in wheat. Plant Cell 8:629–643
Guatimosim E, Pinto HJ, Pereira OL, Fuga CAG, Vieira BS, Barreto RW (2015) Pathogenic mycobiota of the weeds Bidens pilosa and Bidens subalternans. Trop Plant Pathol 40:298–317. https://doi.org/10.1007/s40858-015-0040-x
Gulya TJ, Urs RR (1985) A new race of sunflower downy mildew. Phytopathol 75:1339
Gulya TJ, Sackston WE, Viranyi F, Masirevic S, Rashid KY (1991) New races of the sunflower downy mildew pathogen (Plasmopara halstedii) in Europe and North and South America. J Phytopathol 132:303–311
Gulya TJ, Rashid KY, Marisevic S (1997) Sunflower diseases. In: Schneiter AA (ed) Sunflower technology and production. CSSA, SSSA publishers, Madison WI, ASA, pp 263–379
Gulya TJ, de Labrouhe DT, Masirevic S, Penaud A, Rashid K, Viranyi F (1998) Proposal for standardized nomenclature and identification of races of Plasmopara halstedii (sunflower downy mildew). In: Gulya T, Vear F (eds) Proceedings of the ISA symposium III Sunflower downy mildew. Fargo, ND, USA, Jan 13–14 1998. International Sunflower Association, Paris, France, pp 130–136
Gulya TJ, Draper M, Harbour J, Holen C, Knodel J, Lamey A, Mason P (1999) Metalaxyl resistance in sunflower downy mildew in North America. In: National sunflower association (ed) Proceedings of the 21st Sunflower research workshop. Fargo ND, USA, Jan 12–13 1999. National Sunflower Association, Bismarck ND, USA, pp 2–7
Hebbar P, Berge O, Heulin T, Singh SP (1991) Bacterial antagonists of sunflower (Helianthus annuus L.) fungal pathogens. Plant Soil 133:131–140
Hulke BS, Miller JF, Gulya TJ, Vick BA (2010) Registration of the oilseed sunflower genetic stocks HA 458, HA 459, and HA 460 possessing genes for resistance to downy mildew. J Plant Regist 4:93–97
Humann RM, Johnson KD et al (2019) Evaluation of oxathiapiprolin for the management of sunflower downy mildew. Plant Dis 103(10):2498–2504
Iliescu H (1980) La lutte contre le mildiou du tournesol par des traîtements chimiques. In: Servicio de publicaciones agrarias (ed) Resúmenes de la IX Conferencia internacional del girasol Vol. I, Torremolinos, Malaga, Spain, June 8–13 1980. Vol. I. Jomagar, Madrid, Spain, pp 152–161
Intelmann F, Spring O (2002) Analysis of total DNA by minisatellite and simple sequence repeat primers for the use of population studies in Plasmopara halstedii. Can J Microbiol 48:555–559
Jaber LR (2015) Grapevine leaf tissue colonization by the fungal entomopathogen Beauveria bassiana s.l. and its effect against downy mildew. Biocontrol 60:103–112
Jaber LR, Ownley BH (2018) Can we use entomopathogenic fungi as endophytes for dual biological control of insect pests and plant pathogens? Biol Control 116:36–45
Jan CC, Tan AS, Gulya TJ (1991) Genetics of downy mildew race 2 resistance derived from two wild Helianthus annuus accessions. In: National sunflower association (ed) Proceedings of the 13th Sunflower research workshop. Fargo ND, USA, Jan 10–11 1991. National Sunflower Association, Bismarck ND, USA, p 125
Kandel H, Endres G, Buetow R (2020) Sunflower production guide. North Dakota State University Extension, Fargo ND, USA. www.ndsu.edu/extension
Kinman ML (1970) New Developments in the USDA and State Experiment Station Sunflower Breeding Programs. In: International sunflower association (ed) Proceedings of the 4th International sunflower conference. Memphis TN, USA, June 23–25 1970. International Sunflower Association, Paris, France, pp 181–183
Kogel KH, Beckhove U, Dreschers J, Münch S, Romme Y (1994) Acquired resistance in barley. Plant Physiol 106:1269–1277
Körösi K, Lázár N, Virányi F (2009) Resistance to downy mildew in sunflower induced by chemical activators. Acta Phytopathologica 44:1–9
Körösi K, Bán R, Barna B, Virányi F (2011) Biochemical and molecular changes in downy mildew-infected sunflower triggered by resistance inducers. J Phytopathol 159:471–478. https://doi.org/10.1111/j.1439-0434.2011.01787.x
Lawson WR, Goulter KC, Henry RJ, Kong GA, Kochman JK (1998) Marker-assisted selection for two rust resistance genes in sunflower. Mol Breed 4:227–234
Leclercq P (1969) Une sterilite male cytoplasmique chez Ie tournesol. Ann Amelior Plant 19:99–106
Leclercq P, Cauderon Y, Dauge M (1970) Selection pour la resistance au mildiou du tournesol à partir d’hybrides topinambour x tournesol. Ann Amelior Plant 20(3):363–373
Limpert E, Müller K (1994) Designation of pathotypes of plant-pathogens. J Phytopathol 140(4):346–358. https://doi.org/10.1111/j.1439-0434.1994.tb00617.x
Liu Z, Gulya TJ, Seiler GJ, Vick BA, Jan CC (2012) Molecular mapping of the Pl16 downy mildew resistance gene from HA-R4 to facilitate marker-assisted selection in sunflower. Theor Appl Genet 125:121–131
Liu Z, Zhang L, Ma GJ, Seiler GJ, Jan CC, Qi LL (2019) Molecular mapping of the downy mildew and rust resistance genes in a sunflower germplasm line TX16R. Mol Breed 39:19
Ljubich A, Gulya TJ, Miller JF (1988) A new race of sunflower downy mildew in North America. Phytopathol 78:1580
Ma GJ, Markell SG, Song QJ, Qi LL (2017) Genotyping-by-sequencing targeting of a novel downy mildew resistance gene Pl20 from wild Helianthus argophyllus in sunflower (Helianthus annuus L.). Theor Appl Genet 130:1519–1529
Martín-Sanz A, Rueda S, García-Carneros AB, Molinero-Ruiz L (2020) First report of a new highly virulent pathotype of sunflower downy mildew (Plasmopara halstedii) overcoming the Pl8 resistance gene in Europe. Plant Dis 104(2):597. https://doi.org/10.1094/PDIS-07-19-1425-PDN
Melero-Vara JM, García-Baudín C, López-Herrera CJ, Jiménez-Díaz RM (1982) Control of sunflower downy mildew with metalaxyl. Plant Dis 66:132–135
Menzler-Hokkanen I (2006) Socioeconomic significance of biological control. In: Eilenberg G, Menzler-Hokkanen I (eds) An ecological and societal approach to biological control. Springer, Dordrecht, pp 13–25
Miller JF (1986) Registration of five nonoilseed sunflower restorer germplasm lines. Crop Sci 26:1265
Miller JF (1992) Update on inheritance of sunflower characteristics. In: International sunflower association (ed) Proc 13th Int Sunf Conf, Pisa, Italy, Sept 7–11, 1992. International Sunflower Association, Paris, France, pp 905–945
Miller JF, Gulya TJ (1984) Sources and inheritance of resistance to race 3 downy mildew in sunflower. Helia 7:17–20
Miller JF, Gulya TJ (1987) Inheritance of resistance to race 3 downy mildew in sunflower. Crop Sci 27:210–212
Miller JF, Gulya TJ (1991) Inheritance of resistance to race 4 of downy mildew derived from interspecific crosses in sunflower. Crop Sci 31:40–43
Miranda-Fuentes P, Garcia-Carneros AB, Montilla-Carmona AM, Molinero-Ruiz L (2020) Evidence of soil-located competition as the cause of the reduction of sunflower verticillium wilt by entomopathogenic fungi. Plant Pathol 69:1492–1503
Miranda-Fuentes P, García-Carneros A, Molinero-Ruiz L (2021) Updated characterization of races of Plasmopara halstedii and entomopathogenic fungi as endophytes of sunflower plants in axenic culture. Agronomy 11:268. https://doi.org/10.3390/agronomy11020268
Molinero-Ruiz L (2019) Recent advances on the characterization and control of sunflower soilborne pathogens under climate change conditions. OCL 26(1):2. https://doi.org/10.1051/ocl/2018046
Molinero-Ruiz L, García-de-Tejada JA (2021) Genes y productos en el control del mildiu de girasol. In: Asociación española del girasol (ed) Presentaciones de la XXXI reunión anual de AEG, Sanlúcar de Barrameda, España, Nov 25 2021
Molinero-Ruiz ML, Melero-Vara JM, Dominguez J (2002) Inheritance of resistance to race 330 of Plasmopara halstedii in three sunflower (Helianthus annuus L.) lines. Plant Breed 121 (1):61–65. https://doi.org/10.1046/j.1439-0523.2002.00654.x
Molinero-Ruiz ML, Melero-Vara JM, Dominguez J (2003a) Inheritance of resistance to two races of sunflower downy mildew (Plasmopara halstedii) in two Helianthus annuus L. lines. Euphytica 131:47–51. https://doi.org/10.1023/A:1023063726185
Molinero-Ruiz ML, Melero-Vara JM, Gulya TJ, Dominguez J (2003b) First report of resistance to metalaxyl in downy mildew of sunflower caused by Plasmopara halstedii in Spain. Plant Dis 87:749
Molinero-Ruiz ML, Cordón-Torres MM, Martínez-Aguilar J, Melero-Vara JM, Domínguez J (2008) Resistance to metalaxyl and to metalaxyl-M in populations of Plasmopara halstedii causing downy mildew in sunflower. Can J Plant Pathol 30:97–105
Molinero-Ruiz L, Delavault P, Pérez-Vich B, Pacureanu-Joita M, Bulos M, Altieri E, Dominguez J (2015) History of the race structure of Orobanche cumana and the breeding of sunflower for resistance to the parasitic weed: a review. Spanish J Agri Res 13 (4): e10R01, https://doi.org/10.5424/sjar/2015134-8080
Mouzeyar S, Tourvieille de Labrouhe D, Vear F (1993) Histopathological studies of resistance of sunflower (Helianthus annuus L.) to downy mildew (Plasmopara halstedii). J Phytopathol 139:289–297
Mouzeyar S, Tourvieille de Labrouhe D, Vear F (1994) Effect of host-race combination on resistance of sunflower, Helianthus annuus L., to downy mildew Plasmopara halstedii. J Phytopathol 141:249–258
Mouzeyar S, Roeckel-Crevet P, Gentzbittel L, Philippon J, Tourvieille D, Vear F, Nicolas P (1995) RFLP and RAPD mapping of the sunflower Pl1 locus for resistance to Plasmopara halstedii race 1. Theor Appl Gent 91:733–737
Mulpuri S, Liu Z, Feng J, Gulya TJ, Jan CC (2009) Inheritance and molecular mapping of a downy mildew resistance gene, Pl13 in cultivated sunflower (Helianthus annuus L.). Theor Appl Genet 119:795–803
Panchenco Y (1966) Sunflower production and breeding in the USSR. In: International sunflower association (ed) Proceedings of the 2nd International sunflower conference, Morden, Manitoba, Canada, August 17–18, 1966. International Sunflower Association, Paris, France, pp 15–29
Pasteris RJ, Hanagan MA et al (2016) Discovery of oxathiapiprolin, a new oomycete fungicide that targets an oxysterol binding protein. Bioorg Med Chem 24:354–361
Pecrix Y, Penouilh-Suzette C, Muños S, Vear F, Godiard L (2018) Ten broad spectrum resistances to downy mildew physically mapped on the sunflower genome. Front Plant Sci 9:1780. https://doi.org/10.3389/fpls.2018.01780
Pecrix Y, Buendia L, Penouilh-Suzette C, Maréchaux M, Legrand L, Bouchez O, Rengel D, Gouzy J, Cottret L, Vear F, Godiard L (2019) Sunflower resistance to multiple downy mildew pathotypes revealed by recognition of conserved effectors of the oomycete Plasmopara halstedii. Plant J. https://doi.org/10.1111/tpj.14157
Pustovoit GV (1966) Distant (interspecific) hybridization of sunflowers in the USSR. In: International sunflower association (ed) Proceedings of the 2nd International sunflower conference, Morden, Manitoba, Canada, august 17–18, 1966. International Sunflower Association, Paris, France, pp 82–95
Putt ED (1978) History and present world status. In: Carter JF (ed) Sunflower science and technology. ASA, Madison WI, pp 4–29
Qi L, Ma G (2020) Marker-assisted gene pyramiding and the reliability of using SNP markers located in the recombination suppressed regions of sunflower (Helianthus annuus L.). Genes 11:10. https://doi.org/10.3390/genes11010010
Qi LL, Long YM, Jan CC, Ma GJ, Gulya TJ (2015) Pl17 is a novel gene independent of known downy mildew resistance genes in the cultivated sunflower (Helianthus annuus L.). Theor Appl Genet 128:757–767
Qi LL, Foley ME, Cai XW, Gulya TJ (2016) Genetics and mapping of a novel downy mildew resistance gene, Pl18, introgressed from wild Helianthus argophyllus into cultivated sunflower (Helianthus annuus L.). Theor Appl Genet 129(4):741–752. https://doi.org/10.1007/s00122-015-2662-2
Qi LL, Ma GJ, Li X, Seiler GJ (2019) Diversification of the downy mildew resistance gene pool by introgression of a new gene, Pl35, from wild Helianthus argophyllus into oilseed and confection sunflowers (Helianthus annuus L.) Theor Appl Genet 132(9):2553–2565. https://doi.org/10.1007/s00122-019-03370-9
Radwan O, Gandhi S, Heesacker A, Whitaker B, Taylor C, Plocik A, Kesseli R, Kozik A, Michelmore RW, Knapp SJ (2008) Genetic diversity and genomic distribution of homologs encoding NBSLRR disease resistance proteins in sunflower. Mol Genet Genomics 280:111–125
Radwan O, Bouzidi MF, Mouzeyar S (2011) Molecular characterization of two types of resistance in sunflower to Plasmopara halstedii, the causal agent of downy mildew. Phytopathol 101:970–979
Rahim M, Jan CC, Gulya TJ (2002) Inheritance of resistance to sunflower downy mildew races 1, 2 and 3 in cultivated sunflower. Plant Breed 121:57–60
Roeckel-Drevet P, Gagne G, Mouzeyar S, Gentzbittel L, Philippon J, Nicolas P, De Labrouhe DT, Vear F (1996) Colocation of downy mildew (Plasmopara halstedii) resistance genes in sunflower (Helianthus annuus L.). Euphytica 91:225–228
Roeckel-Drevet P, Tourvieille J, Gulya TJ, Charmet G, Nicolas P, Tourvieille de Labrouhe D (2003) Molecular variability of sunflower downy mildew, Plasmopara halstedii, from different continents. Can J Microbiol 49:492–502
Sackston WE (1992a) On a treadmill: breeding sunflowers for resistance to disease. Annu Rev Phytopathol 30:529–551
Sackston WE (1992b) Cotyledon limited infection (CLI) and leaf disk immersion (LDI) inoculation of sunflower by downy mildew (Plasmopara halstedii). In: International sunflower association (ed) Proc 13th Int Sunf Conf, Pisa, Italy, Sept 7–11, 1992. International Sunflower Association, Paris, France, pp 840–848
Sackston WE (1993) Biological control of downy mildew of sunflower. Phytopathol 83:247
Sedlářová M, Pospíchalová R, Drábková Trojanová Z, Bartůšek T, Slobodianová L, Lebeda A (2016) First report of Plasmopara halstedii new races 705 and 715 on sunflower from the Czech Republic – short communication. Plant Protect Sci 52(3):182–187. https://doi.org/10.17221/7/2016-PPS
Seiler GJ (1991) Registration of 13 downy mildew tolerant interspecific sunflower germplasm lines derived from wild annual species. Crop Sci 31:1714–1716
Seiler GJ, Qi LL, Marek LF (2017) Utilization of sunflower crop wild relatives for cultivated sunflower improvement. Crop Sci 57:1083–1011
Semelczi-Kovacs A (1975) Acclimatization and dissemination of the sunflower in Europe. Acta Ethnogr Acad Sci Hung 24:47–88
Spring O, Zipper R (2006) Evidence for asexual genetic recombination in sunflower downy mildew, Plasmopara halstedii. Mycol Res 110:657–663
Spring O, Gomez-Zeledon J, Hadziabdic D, Trigiano RN, Thines M, Lebeda A (2018) Biological characteristics and assessment of virulence diversity in pathosystems of economically important biotrophic oomycetes. Crit Rev Plant Sci 37(6):439–495. https://doi.org/10.1080/07352689.2018.1530848
Sudisha J, Niranjana SR, Sukanya SL, Girijamba R, Lakshmi Devi N, Shekar Shetty H (2010) Relative efficacy of strobilurin formulations in the control of downy mildew of sunflower. J Pest Sci 83:461–470. https://doi.org/10.1007/s10340-010-0316-3
Sukul P, Spiteller M (2000) Metalaxyl: persistence, degradation, metabolism and analytical methods. Rev Envir Contam Toxicol 164:1–26
Talukder ZI, Ma G, Hulke BS, Jan CC, Qi L (2019) Linkage mapping and genome-wide association studies of the Rf gene cluster in sunflower (Helianthus annuus L.) and their distribution in world sunflower collections. Front Genet 10:216. https://doi.org/10.3389/fgene.2019.00216
Thines M, Komjáti H, Spring O (2005) Exceptional length of ITS in Plasmopara halstedii is due to multiple repetitions in the ITS-2 region. Eur J Plant Pathol 112:395–398
Tosi L, Beccari L (2007) A new race, 704, of Plasmopara helianthi pathogen of sunflower downy mildew in Italy. Plant Dis 91(4):463. https://doi.org/10.1094/PDIS-91-4-0463B
Tosi L, Giovannetti M, Zazzerini A, Sbrana C (1993) Interactions between Plasmopara halstedii and arbuscular mycorrhizal fungi in sunflower seedlings susceptible and resistant to downy mildew. Phytopathol Medit 32:106–114
Tourvieille de Labrouhe D, Lafon S, Walser P, Raulic I (2000) Une nouvelle race de Plasmopara halstedii, agent du mildiou du tournesol. OCL 7(5):404–405. https://doi.org/10.1051/ocl.2000.0404
Tourvieille de Labrouhe D, Walser P, Serre F, Roche S, Vear F (2008) Relations between spring rainfall and infection of sunflower by Plasmopara halstedii (downy mildew). In: International sunflower Association (ed) Proceedings of the 17th International sunflower conference. Córdoba, Spain, June 8–12 2008. International Sunflower Association, Paris, France, pp 97–102
Tourvieille de Labrouhe D, Walser P, Jolivot D, Roche S, Serre F, Leguillon M, Delmotte F, Bordat A, Godiard L, Vincourt P, Vear F (2012) Proposal for improvement of sunflower downy mildew race nomenclature. In: International sunflower association (ed) Proceedings of the 18th International sunflower conference. Mar del Plata, Argentina, February 26-March 1 2012. International Sunflower Association, Paris, France, pp 322–327
Vear F (1974) Studies on resistance to downy mildew in sunflowers (Helianthus annuus L.). In: Vranceanu A (ed) Proceedings of the 6th International sunflower conference, Bucharest, Romania, July 22–24, 1974. International Sunflower Association, Paris, France, pp 297–302
Vear F (2016) Changes in sunflower breeding over the last fifty years. OCL 23(2):D202. https://doi.org/10.1051/ocl/2016006
Vear F, Leclercq P (1971) Deux nouveaux gènes de résistance au mildiou du tournesol. Ann Amélior Plantes 21(3):251–255
Vear F, Philippon J (1978) Réaction de certains génotypes de tournesol résistants au mildiou (Plasmopara helianthi) au test de résistance sur la plantule. Ann Amélior Plantes 28(3):327–332
Vear F, Gentzbittel L, Philippon J, Mouzeyar S, Mestries E, Roeckel-Drevet P, Tourvieille de Labroube D, Nicolas P (1997) The genetics of resistance to five races of downy mildew (Plasmopara halstedii) in sunflower (Helianthus annuus L.). Theor Appl Genet 95:584–589
Vear F, Seriveys H, Petit A, Serre F, Boudon JP, Roche S, Walser P, de Labrouhe DT (2008) Origins of major genes for downy mildew resistance in sunflower. In: Velasco L (ed) Proceedings of 17th International Sunflower Conference, Cordoba, Spain, june 8–12 2008. Junta de Andalucía, Consejería de Agricultura y Pesca, Sevilla, Spain, pp 125–130
Vega FE (2018) The use of fungal entomopathogens as endophytes in biological control: a review. Mycologia 110:4–30
Vincourt P, As-Sadi F, Bordat A, Langlade NB, Gouzy J, Pouilly N, Lippi Y, Serre F, Godiard L, De Labrouhe DT, Vear F (2012) Consensus mapping of major resistance genes and independent QTL for quantitative resistance to sunflower downy mildew. Theor Appl Genet 125:909–920
Virányi F, Gulya TJ (1995) Inter-isolate variation for virulence in Plasmopara halstedii (sunflower downy mildew) from Hungary. Plant Pathol 44:619–624
Virányi F, Gulya TJ, Labrouhe de Tourvieille D (2015) Recent changes in the pathogenic variability of Plasmopara halstedii (sunflower downy mildew) populations from different continents. Helia 38(63):149–162. https://doi.org/10.1515/helia-2015-0009
Vrânceanu V (1970) Advances in sunflower breeding in Romania. In: International sunflower association (ed) Proceedings of the 4th International sunflower conference, Memphis TN, USA, june 23–25 1970. International Sunflower Association, Paris, France, pp 136–148
Vrânceanu A, Stoenescu F (1970) Immunity to sunflower downy mildew due to a single dominant gene. Probl Agric 2:34–40
Vrânceanu A, Pirvu N, Iliescu H (1978) A new race of the fungus Plasmopara halstedii Novot. identified in Romania. In: International sunflower association (ed), Proceedings of the 8th International sunflower conference, July 23–27 1978, Minneapolis MN, USA, pp 328–333
Vrânceanu A, Pirvu N, Stoenescu FM (1981) New sunflower downy mildew resistance genes and their management. Helia 4:23–27
Wiggins TE, Jager BJ (1993) Mode of action of the new methoxyacrylate antifungal agent ICIA5504. Biochem Soc Trans 22:68S
Yu JK, Tang S, Slabaugh MB, Heesacker A, Cole G, Herring MJ, Soper J, Han F, Chu WC, Webb DM, Thompson L, Edwards KJ, Berry S, Leon A, Olungu C, Maes N, Knapp SJ (2003) Towards a saturated molecular genetic linkage map for cultivated sunflower. Crop Sci 43:367–387
Zhang M, Liu Z, Jan CC (2016) Molecular mapping of a rust resistance gene R14 in cultivated sunflower line PH3. Mol Breed 36:32
Zhang ZW, Ma GJ, Zhao J, Markell SG, Qi LL (2017) Discovery and introgression of the wild sunflower-derived novel downy mildew resistance gene Pl19 in confection sunflower (Helianthus annuus L.). Theor Appl Genet 130:29–39
Zimmer DE (1974) Physiological specialization between races of Plasmopara halstedii in America and Europe. Phytopathol 64:1465–1467
Zimmer DE (1975) Some biotic and climatic factors influencing sporadic occurrence of sunflower downy mildew. Phytopathol 65:751–754
Zimmer DE, Kinman ML (1972) Downy mildew resistance in cultivated sunflower and its inheritance. Crop Sci 12:749–751
Funding
No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Not applicable.
Corresponding author
Ethics declarations
Conflicts of interest
The author has no competing interests to declare that are relevant to the content of this article.
Additional information
Communicated by Dragana Miladinović.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Molinero-Ruiz, L. Sustainable and efficient control of sunflower downy mildew by means of genetic resistance: a review. Theor Appl Genet 135, 3757–3771 (2022). https://doi.org/10.1007/s00122-022-04038-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-022-04038-7