Abstract
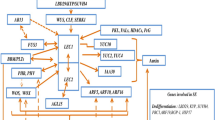
Somatic embryogenesis (SE), the process through which already differentiated cells reverse their developmental programme and become embryogenic, requires drastic changes in the transcriptome of the explant cells. Among the various factors that underlie this developmental switch, genes encoding transcription factors (TFs), which constitute the sequence-specific DNA-binding proteins, are widely accepted as playing a central function in the gene expression regulation. In recent years, intensive analysis of the global transcriptomes of plant cells that are undergoing embryogenic transition and the use of Arabidopsis (a model in plant genomics) in studies on the genetic control of SE have substantially contributed to the identification of SE regulators. A survey of SE-associated transcriptomes illustrated the combinational effects of stress and hormone signalling that are related to the in vitro environment that is imposed during a culture. Accordingly, among the TFs that are considered to be essential in SE induction, those that are involved in stress and hormone plant responses and especially flower development were found to be most frequent. This chapter provides a comprehensive review of the current knowledge about the TFs that are involved in the induction of SE in plant explants that are cultured in vitro. In addition to a general characterisation of the TF transcriptomes that are associated with SE induction in different plants, the individual TF genes with documented functions in the regulation of SE are presented with a special reference to their possible targets and the TF-controlled molecular mechanisms that underlie SE induction.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Aida M, Beis D, Heidstra R et al (2004) The PLETHORA genes mediate patterning of the Arabidopsis root stem cell niche. Cell 119:109–120. doi:10.1016/j.cell.2004.09.018
Aker J, de Vries SC (2008) Plasma membrane receptor complexes. Plant Physiol 147:1560–1564. doi:10.1104/pp.108.120501
Alemanno L, Devic M, Niemenak N et al (2008) Characterization of leafy cotyledon1-like during embryogenesis in Theobroma cacao L. Planta 227:853–866. doi:10.1007/s00425-007-0662-4
Arroyo-Herrera A, Gonzalez AK, Moo RC et al (2008) Expression of WUSCHEL in Coffea canephora causes ectopic morphogenesis and increases somatic embryogenesis. Plant Cell Tiss Org 94:171–180. doi:10.1007/s11240-008-9401-1
Bai B, Su YH, Yuan J et al (2013) Induction of somatic embryos in Arabidopsis requires local YUCCA expression mediated by the down-regulation of ethylene biosynthesis. Mol Plant 6(4):1247–1260. doi:10.1093/mp/sss154
Belide S, Zhou XR, Kennedy Y et al (2013) Rapid expression and validation of seed-specific constructs in transgenic LEC2 induced somatic embryos of Brassica napus. Plant Cell Tiss Org 113:543–553. doi:10.1007/s11240-013-0295-1
Belmonte MF, Kirkbride RC, Stone SL et al (2013) Comprehensive developmental profiles of gene activity in regions and subregions of the Arabidopsis seed. Proc Natl Acad Sci USA 110(5):E435–E444. doi:10.1073/pnas.1222061110
Blilou I, Xu J, Wildwater M et al (2005) The PIN auxin efflux facilitator network controls growth and patterning in Arabidopsis roots. Nature 433:39–44. doi:10.1038/nature03184
Bouchabké-Coussa O, Obellianne M, Linderme D et al (2013) Wuschel overexpression promotes somatic embryogenesis and induces organogenesis in cotton (Gossypium hirsutum L.) tissues cultured in vitro. Plant Cell Rep 32:675–686. doi:10.1007/s00299-013-1402-9
Boutilier K, Offringa R, Sharma VK et al (2002) Ectopic expression of BABY BOOM triggers a conversion from vegetative to embryonic growth. Plant Cell 14:1737–1749. doi:10.1105/tpc.001941
Braybrook SA, Stone SL, Park S et al (2006) Genes directly regulated by LEAFY COTYLEDON2 provide insight into the control of embryo maturation and somatic embryogenesis. Proc Natl Acad Sci (USA) 103:3468–3473. doi:10.1073/pnas.0511331103
Braybrook SA, Harada JJ (2008) LECs go crazy in embryo development. Trends Plant Sci 13:624–630. doi:10.1016/j.tplants.2008.09.008
Casson SA, Lindsey K (2006) The turnip mutant of Arabidopsis reveals that LEAFY COTYLEDON1 expression mediates the effects of auxin and sugars to promote embryonic cell identity. Plant Physiol 42(2):526–541. doi:10.1104/pp.106.080895
Chakrabarty D, Trivedi KP, Shri M et al (2010) Differential transcriptional expression following thidiazuron-induced callus differentiation developmental shifts in rice. Plant Biol 12:46–59. doi:10.1111/j.1438-8677.2009.00213.x
Causier B, Ashworth M, Guo W et al (2012) The TOPLESS interactome: a framework for gene repression in Arabidopsis. Plant Physiol 158:423–438. doi:10.1104/pp.111.186999
Che P, Love TM, Frame BR et al (2006) Gene expression patterns during somatic embryo development and germination in maize Hi II callus cultures. Plant Mol Biol 62:1–14. doi:10.1007/s11103-006-9013-2
Chen M, Xuan L, Wang Z et al (2014) TRANSPARENT TESTA8 inhibits seed fatty acid accumulation by targeting several seed development regulators in Arabidopsis. Plant Physiol 165:905–916. doi:10.1104/pp.114.235507
Curaba J, Moritz T, Blervaque R et al (2004) AtGA3ox2, a key gene responsible for bioactive gibberellin biosynthesis, is regulated during embryogenesis by LEAFY COTYLEDON2 and FUSCA3 in Arabidopsis. Plant Physiol 136:3660–3669. doi:10.1104/pp.104.047266
Deng W, Luo K, Li Z et al (2009) A novel method for induction of plant regeneration via somatic embryogenesis. Plant Sci 177:43–48. doi:10.1016/j.plantsci.2009.03.009
Dodeman VL, Ducreux G, Kreis M (1997) Zygotic versus somatic embryogenesis. J Exp Bot 48:1493–1509. doi:10.1093/jxb/48.8.1493
Dubos C, Stracke R, Grotewold E et al (2010) MYB transcription factors in Arabidopsis. Trends Plant Sci 15:573–581. doi:10.1016/j.tplants.2010.06.005
Elliott RC, Betzner AS, Huttner E et al (1996) AINTEGUMENTA, an APETALA2-like gene of Arabidopsis with pleiotropic roles in ovule development and floral organ growth. Plant Cell 8:155–168. doi:10.1105/tpc.8.2.155
El Ouakfaoui S, Schnell J, Abdeen A et al (2010) Control of somatic embryogenesis and embryo development by AP2 transcription factors. Plant Mol Biol 74:313–326. doi:10.1007/s11103-010-9674-8
Fambrini M, Durante C, Cionini G et al (2006) Characterization of LEAFY COTYLEDON1-LIKE gene in Helianthus annuus and its relationship with zygotic and somatic embryogenesis. Dev Genes Evol 216:253–264. doi:10.1007/s00427-005-0050-7
Fehér A, Pasternak TP, Dudits D (2003) Transition of somatic plant cells to an embryogenic state. Plant Cell Tiss Org 74:201–228. doi:10.1023/A:1024033216561
Fehér A (2015) Somatic embryogenesis—stress-induced remodeling of plant cell fate. Biochim Biophys Acta 1849:385–402. doi:10.1016/j.bbagrm.2014.07.005
Fernandez DE, Wang CT, Zheng Y et al (2015) The MADS-domain factors AGL15 and AGL18, along with SVP and AGL24, are necessary to block floral gene expression during the vegetative phase. Plant Physiol 165(4):1591–1603. doi:10.1104/pp.114.242990
Franco-Zorrilla JM, López-Vidriero I, Carrasco JL et al (2014) DNA-binding specificities of plant transcription factors and their potential to define target genes. Proc Natl Acad Sci (USA) 111(6):2367–2372. doi:10.1073/pnas.1316278111
Friml J, Vieten A, Sauer M et al (2003) Efflux-dependent auxin gradients establish the apical-basal axis of Arabidopsis. Nature 426:147–153. doi:10.1038/nature02085
Gaj MD (2004) Factors influencing somatic embryogenesis induction and plant regeneration with particular reference to Arabidopsis thaliana (L.) Heynh. Plant Growth Regul 43:27–47. doi:10.1023/B:GROW.0000038275.29262.fb
Gaj MD, Zhang S, Harada JJ et al (2005) LEAFY COTYLEDON genes are essential for induction of somatic embryogenesis of Arabidopsis. Planta 222:977–988. doi:10.1007/s00425-005-0041-y
Gaj MD, Trojanowska A, Ujczak A et al (2006) Hormone-response mutants of Arabidopsis thaliana (L.) Heynh. impaired in somatic embryogenesis. Plant Growth Regul 49:189–197. doi:10.1007/s10725-006-9104-8
Galinha C, Hofhuis H, Luijten M et al (2007) PLETHORA proteins as dose-dependent master regulators of Arabidopsis root development. Nature 449:1053–1057. doi:10.1038/nature06206
Gao MJ, Lydiate DJ, Li X et al (2009) Repression of seed maturation genes by a trihelix transcriptional repressor in Arabidopsis seedlings. Plant Cell 21:54–71. doi:10.1105/tpc.108.061309
Garcês HM, Champagne CE, Townsley BT et al (2007) Evolution of asexual reproduction in leaves of the genus Kalanchoë. Proc Natl Acad Sci (USA) 104:15578–15583. doi:10.1073/pnas.0704105104
Gazzarrini S, Tsuchiya Y, Lumba S et al (2004) The transcription factor FUSCA3 controls development timing in Arabidopsis through the hormones gibberellin and abscisic acid. Develop Cell 7:373–385. doi:10.1016/j.devcel.2004.06.017
Gliwicka M, Nowak K, Cieśla E et al (2012) Expression of seed storage product genes (CRA1 and OLEO4) in embryogenic cultures of somatic tissues of Arabidopsis. Plant Cell Tiss Org 109:235–245. doi:10.1007/s11240-011-0089-2
Gliwicka M, Nowak K, Balazadeh S et al (2013) Extensive modulation of the transcription factor transcriptome during somatic embryogenesis in Arabidopsis thaliana. PLoS ONE 8(7):e69261. doi:10.1371/journal.pone.0069261
Go YS, Kim H, Kim HJ et al (2014) Arabidopsis cuticular wax biosynthesis is negatively regulated by the DEWAX gene encoding an AP2/ERF-type transcription factor. Plant Cell 26:1666–1680. doi:10.1105/tpc.114.123307
Gordon SP, Chickarmane VS, Ohno C et al (2009) Multiple feedback loops through cytokinin signaling control stem cell number within the Arabidopsis shoot meristem. Proc Natl Acad Sci (USA) 106:16529–16534. doi:10.1073/pnas.0908122106
Guo F, Liu C, Xia H et al (2013) Induced expression of AtLEC1 and AtLEC2 differentially promotes somatic embryogenesis in transgenic tobacco plants. PLoS ONE 8:e71714. doi:10.1371/journal.pone.0071714
Habets MEJ, Offringa R (2014) PIN-driven polar auxin transport in plant developmental plasticity: a key target for environmental and endogenous signals. New Phytol 203:362–377. doi:10.1111/nph.12831
Hacker A, Groß-Hardt R, Geiges B et al (2004) Expression dynamics of WOX genes mark cell fate decisions during early embryonic patterning in Arabidopsis thaliana. Development 131:657–668. doi:10.1242/dev.00963
Harada JJ (2001) Role of Arabidopsis LEAFY COTYLEDON genes in seed development. J Plant Physiol 158:405–409. doi:10.1078/0176-1617-00351
Harding EW, Tang W, Nichols KW et al (2003) Expression and maintenance of embryogenic potential is enhanced through constitutive expression of AGAMOUS-Like 15. Plant Physiol 133:653–663. doi:10.1104/pp.103.023499
He C, Huang H, Xu L (2013) Mechanisms guiding Polycomb activities during gene silencing in Arabidopsis thaliana. Front Plant Sci 4:454. doi:10.3389/fpls.2013.00454
Hecht V, Vielle-Calzada JP, Hartog MV et al (2001) The Arabidopsis SOMATIC EMBRYOGENESIS RECEPTOR-LIKE KINASE1 gene is expressed in developing ovules and embryos and enhances embryogenic competence in culture. Plant Physiol 127:803–816. doi:10.1104/pp.010324
Hill K, Wang H, Perry SE (2008) A transcriptional repression motif in the MADS factor AGL15 is involved in recruitment of histone deactylase complex components. Plant J 53:172–185. doi:10.1111/j.1365-313X.2007.03336.x
Hirakawa Y, Kondo Y, Fukuda H (2010) TDIF peptide signaling regulates vascular stem cell proliferation via the WOX4 homeobox gene in Arabidopsis. Plant Cell 22:2618–2629. doi:10.1105/tpc.110.076083
Horstman A, Willemsen V, Boutilier K et al (2014) AINTEGUMENTA-LIKE proteins: hubs in a plethora of networks. Trends Plant Sci 19:146–157. doi:10.1016/j.tplants.2013.10.010
Horstman A (2015) BABBY BOOM-induced somatic embryogenesis in Arabidopsis. Dissertation, Wegeningen Univeristy
Horstman A, Fukuoka H, Muino JM et al (2015) AIL and HDG proteins act antagonistically to control cell proliferation. Development 142:454–464. doi:10.1242/dev.117168
Hosp J, Maraschin SDF, Touraev A et al (2007) Functional genomics of microspore embryogenesis. Euphytica 158:275–285. doi:10.1007/s10681-006-9238-9
Ikeda M, Umehara M, Kamada H (2006) Embryogenesis-related genes; its expression and roles during somatic and zygotic embryogenesis in carrot and Arabidopsis. Plant Biotechnol 23:153–161. doi:http://doi.org/10.5511/plantbiotechnology.23.153
Ikeda M, Mitsuda N, Ohme-Takagi M (2009) Arabidopsis WUSCHEL is a bifunctional transcription factor that acts as a repressor in stem cell regulation and as an activator in floral patterning. Plant Cell 21:3493–3505. doi:10.1105/tpc.109.069997
Imin N, Goffard N, Nizamidin M et al (2008) Genome-wide transcriptional analysis of super-embryogenic Medicago truncatula explant cultures. BMC Plant Biol 8:110–124. doi:10.1186/1471-2229-8-110
Iwase A, Mitsuda N, Koyama T et al (2011) The AP2/ERF transcription factor WIND1 controls cell dedifferentiation in Arabidopsis. Curr Biol 21:508–514. doi:10.1016/j.cub.2011.02.020
Jia H, Suzuki M, McCarty DR (2014) Regulation of the seed to seedling developmental phase transition by the LAFL and VAL transcription factor networks. WIREs Dev Biol 3:135–145. doi:10.1002/wdev.126
Jiménez VM (2005) Involvement of plant hormones and plant growth regulators on in vitro somatic embryogenesis. Plant Growth Regul 47:91–110. doi:10.1007/s10725-005-3478-x
Jin F, Hu L, Yuan D et al (2014) Comparative transcriptome analysis between somatic embryos (SEs) and zygotic embryos in cotton: evidence for stress response functions in SE development. Plant Biotechnol J 12:161–173. doi:10.1111/pbi.12123
Junker A, Monke G, Rutten T et al (2012) Elongation-related functions of LEAFY COTYLEDON1 during the development of Arabidopsis thaliana. Plant J 71:427–442. doi:10.1111/j.1365-313X.2012.04999.x
Junker A, Bäumlein H (2012) Multifunctionality of the LEC1 transcription factor during plant development. Plant Signal Behav 7:1718–1720. doi:10.4161/psb.22365
Jürgens G (2001) Apical-basal pattern formation in Arabidopsis embryogenesis. EMBO J 20:3609–3616. doi:10.1093/emboj/20.14.3609
Kagaya Y, Toyoshima R, Okuda R et al (2005) LEAFY COTYLEDON1 controls seed storage protein genes through its regulation of FUSCA3 and ABSCISIC ACID INSENSITIVE3. Plant Cell Physiol 46:399–406. doi:10.1093/pcp/pci048
Kamada H, Ishikawa K, Saga H et al (1993) Induction of somatic embryogenesis in carrot by osmotic stress. Plant Tiss Cult Lett 10:38–44. doi:http://doi.org/10.5511/plantbiotechnology1984.10.38
Karami O, Saidi A (2010) The molecular basis for stress-induced acquisition of somatic embryogenesis. Mol Biol Rep 37:2493–2507. doi:10.1007/s11033-009-9764-3
Karlova R, Boeren S, Russinova E et al (2006) The Arabidopsis SOMATIC EMBRYOGENESIS RECEPTOR-LIKE KINASE1 protein complex includes BRASSINOSTEROID-INSENSITIVE1. Plant Cell 18:626–638. doi:10.1105/tpc.105.039412
Klucher KM, Chow H, Reiser L et al (1996) The AINTEGUMENTA gene of Arabidopsis required for ovule and female gametophyte development is related to the floral homeotic gene APETALA2. Plant Cell 8:137–153. doi:10.1105/tpc.8.2.137
Ledwoń A, Gaj MD (2009) LEAFY COTYLEDON2 gene expression and auxin treatment in relation to embryogenic capacity of Arabidopsis somatic cells. Plant Cell Rep 28:1677–1688. doi:10.1007/s00299-009-0767-2
Ledwoń A, Gaj MD (2011) LEAFY COTYLEDON1, FUSCA3 expression and auxin treatment in relation to somatic embryogenesis induction in Arabidopsis. Plant Growth Regul 65:157–167. doi:10.1007/s10725-011-9585-y
Legrand S, Hendricks T, Gilbert JL et al (2007) Characterization of expressed sequence tags obtained by SSH during somatic embryogenesis in Cichorium intybus L. BMC Plant Biol 7:27. doi:10.1186/1471-2229-7-27
Leibfried A, To JP, Busch W et al (2005) WUSCHEL controls meristem function by direct regulation of cytokinin-inducible response regulators. Nature 438:1172–1175. doi:10.1038/nature04270
Lotan T, Ohto M, Yee KM et al (1998) Arabidopsis LEAFY COTYLEDON1 is sufficient to induce embryo development in vegetative cells. Cell 93:1195–1205. doi:10.1016/S0092-8674(00)81463-4
Low ET, Alias H, Boon SH et al (2008) Oil palm (Elaeis guineensis Jacq.) tissue culture ESTs: Identifying genes associated with callogenesis and embryogenesis. BMC Plant Biol 8:62. doi:10.1186/1471-2229-8-62
Lumba S, Tsuchiya Y, Delmas F et al (2012) The embryonic leaf identity gene FUSCA3 regulates vegetative phase transitions by negatively modulating ethylene-regulated gene expression in Arabidopsis. BMC Biol 10:8. doi:10.1186/1741-7007-10-8
Mantiri FR, Kurdyukov S, Lohar DP et al (2008a) The transcription factor MtSERF1 of the ERF subfamily identified by transcriptional profiling is required for somatic embryogenesis induced by auxin plus cytokinin in Medicago truncatula. Plant Physiol 146:1622–1636. doi:10.1104/pp.107.110379
Mantiri FR, Kurdyukov S, Chen SK et al (2008b) The transcription factor MtSERF1 may function as a nexus between stress and development in somatic embryogenesis in Medicago truncatula. Plant Signal Behav 3:498–500. doi:10.4161/psb.3.7.6049
Mayer KFX, Schoof H, Haecker A et al (1998) Role of WUSCHEL in regulating stem cell fate in the Arabidopsis shoot meristem. Cell 95:805–815. doi:10.1016/S0092-8674(00)81703-1
Möller B, Weijers D (2009) Auxin control of embryo patterning. Cold Spring Harb Perspect Biol 1:a001545. doi:10.1101/cshperspect.a001545
Mu J, Tan H, Zheng Q et al (2008) LEAFY COTYLEDON1 is a key regulator of fatty acid biosynthesis in Arabidopsis. Plant Physiol 148:1042–1054. doi:10.1104/pp.108.126342
Mu J, Tan H, Hong S et al (2013) Arabidopsis transcription factor genes NF-YA1, 5, 6, and 9 play redundant roles in male gametogenesis, embryogenesis, and seed development. Mol Plant 6:188–201. doi:10.1093/mp/sss061
Muday GK, Rahman A, Binder BM (2012) Auxin and ethylene: collaborators or competitors? Trends Plant Sci 17:181–195. doi:10.1016/j.tplants.2012.02.001
Nakano T, Suzuki K, Fujimura T et al (2006) Genome-wide analysis of the ERF gene family in Arabidopsis and rice. Plant Physiol 140:411–432. doi:10.1104/pp.105.073783
Nic-Can GI, Lopez-Torres A, Barredo-Pool F et al (2013) New insights into somatic embryogenesis: LEAFY COTYLEDON1, BABY BOOM1 and WUSCHEL-RELATED HOMEOBOX4 are epigenetically regulated in Coffea canephora. PLoS ONE 8:e72160. doi:10.1371/journal.pone.0072160
Nowak K, Wójcikowska B, Gaj MD (2015) ERF022 impacts the induction of somatic embryogenesis in Arabidopsis through the ethylene-related pathway. Planta 241:967–985. doi:10.1007/s00425-014-2225-9
Nowak K, Gaj MD (2016) Stress-related function of bHLH109 in somatic embryo induction in Arabidopsis. J Plant Physiol 193:119–126. doi:10.1016/j.jplph.2016.02.012
Palovaara J, Hallberg H, Stasolla C et al (2010) Comparative expression pattern analysis of WUSCHEL related homeobox 2 (WOX2) and WOX8⁄9 in developing seeds and somatic embryos of the gymnosperm Picea abies. New Phytol 188:122–135. doi:10.1111/j.1469-8137.2010.03336.x
Passarinho P, Ketelaar T, Xing M et al (2008) BABY BOOM target genes provide diverse entry points into cell proliferation and cell growth pathways. Plant Mol Biol 68:225–237. doi:10.1007/s11103-008-9364-y
Patnaik D, Mahalakshmi A, Khurana P (2005) Effect of water stress and heavy metals on induction of somatic embryogenesis in wheat leaf base cultures. Ind J Exp Biol 43:740–745
Perry SE, Lehti MD, Fernandez DE (1999) The MADS-domain protein AGAMOUS-like 15 accumulates in embryonic tissues with diverse origins. Plant Physiol 120:121–129. doi:10.1104/pp.120.1.121
Pinon V, Prasada K, Grigga SP et al (2013) Local auxin biosynthesis regulation by PLETHORA transcription factors controls phyllotaxis in Arabidopsis. Proc Natl Acad Sci (USA) 110:1107–1112. doi:10.1073/pnas.1213497110
Piyatrakul P, Putranto RA, Martin F et al (2012) Some ethylene biosynthesis and AP2/ERF genes reveal a specific pattern of expression during somatic embryogenesis in Hevea brasiliensis. BMC Plant Biol 12:244–256. doi:10.1186/1471-2229-12-244
Rademacher EH, Möller B, Lokerse AS et al (2011) A cellular expression map of the Arabidopsis AUXIN RESPONSE FACTOR gene family. Plant J 68:597–606. doi:10.1111/j.1365-313X.2011.04710.x
Rademacher EH, Lokerse AS, Schlereth A et al (2012) Different auxin response machineries control distinct cell fates in the early plant embryo. Develop Cell 22:211–222. doi:10.1016/j.devcel.2011.10.026
Riechmann JL, Heard J, Martin G et al (2000) Arabidopsis transcription factors: Genome-wide comparative analysis among eukaryotes. Science 290:2105–2110. doi:10.1126/science.290.5499.2105
Rensing SA, Lang D, Schumann E et al (2005) EST sequencing from embryogenic Cyclamen persicum cell cultures identifies a high proportion of transcripts homologous to plant genes involved in somatic embryogenesis. J Plant Growth Regul 24:102–115. doi:10.1007/s00344-005-0033-y
Rolland F, Moore B, Sheen J (2002) Sugar sensing and signaling in plants. Plant Cell 14:S185-S205. doi:10.1105/tpc.010455
Sarkar AK, Luijten M, Miyashima S et al (2007) Conserved factors regulate signaling in Arabidopsis thaliana shoot and root stem cell organizers. Nature 446:811–814. doi:10.1038/nature05703
Sato A, Yamamoto KT (2008) Overexpression of the non-canonical Aux/IAA genes causes auxin-related aberrant phenotypes in Arabidopsis. Physiol Plant 133:397–405. doi:10.1111/j.1399-3054.2008.01055.x
Serivichyaswat P, Ryu HS, Kim W et al (2015) Expression of the floral repressor miRNA156 is positively regulated by the AGAMOUS-like proteins AGL15 and AGL18. Mol Cells 38:259–266. doi:10.14348/molcells.2015.2311
Sharma SK, Millam S, Hedley PE et al (2008) Molecular regulation of somatic embryogenesis in potato: an auxin led perspective. Plant Mol Biol 68:185–201. doi:10.1007/s11103-008-9360-2
Singla B, Akhilesh K, Jitendra PT et al (2007) Analysis of expression profile of selected genes expressed during auxin-induced somatic embryogenesis in leaf base system of wheat (Triticum aestivum) and their possible interactions. Plant Mol Biol 65:677–692. doi:10.1007/s11103-007-9234-z
Slattery M, Voutev R, Ma L et al (2013) Divergent transcriptional regulatory logic at the intersection of tissue growth and developmental patterning. PLoS Genet 9:e1003753. doi:10.1371/journal.pgen.1003753
Slattery M, Zhou T, Yang L et al (2014) Absence of a simple code: how transcription factors read the genome. Trends Biochem Sci 39:381–399. doi:10.1016/j.tibs.2014.07.002
Smaczniak C, Immink RGH, Angenent GC et al (2012) Developmental and evolutionary diversity of plant MADS-domain factors: insights from recent studies. Development 139:3081–3098. doi:10.1242/dev.074674
Smith ZR, Long JA (2010) Control of Arabidopsis apical-basal embryo polarity by antagonistic transcription factors. Nature 464:423–426. doi:10.1038/nature08843
Solís-Ramos LY, González-Estrada T, Nahuath-Dzib S et al (2009) Overexpression of WUSCHEL in Capsicum chinense causes ectopic morphogenesis. Plant Cell Tiss Org 96:279–287. doi:10.1007/s11240-008-9485-7
Stepanova AN, Yun J, Likhacheva AV et al (2007) Multilevel interactions between ethylene and auxin in Arabidopsis roots. Plant Cell 19:2169–2185. doi:10.1105/tpc.107.052068
Stone SL, Kwong LW, Yee KM et al (2001) LEAFY COTYLEDON2 encodes a B3 domain transcription factor that induces embryo development. Proc Natl Acad Sci (USA) 98:11806–11811. doi:10.1073/pnas.201413498
Stone SL, Braybrook SA, Paula SL et al (2008) Arabidopsis LEAFY COTYLEDON2 induces maturation traits and auxin activity: implications for somatic embryogenesis. Proc Natl Acad Sci (USA) 105:3151–3156. doi:10.1073/pnas.0712364105
Su N, He K, Jiao Y et al (2007) Distinct reorganization of the genome transcription associates with organogenesis of somatic embryo, shoots, and roots in rice. Plant Mol Biol 63:337–349. doi:10.1007/s11103-006-9092-0
Su YH, Zhao XY, Liu YB et al (2009) Auxin-induced WUS expression is essential for embryonic stem cell renewal during somatic embryogenesis in Arabidopsis. Plant J 59:448–460. doi:10.1111/j.1365-313X.2009.03880.x
Su YH, Liu YB, Bai B et al (2015) Establishment of embryonic shoot–root axis is involved in auxin and cytokinin response during Arabidopsis somatic embryogenesis. Front Plant Sci Plant Evol Develop 5:792. doi:10.3389/fpls.2014.00792
Sundaram S, Kertbundit S, Shakirov EV et al (2013) Gene networks and chromatin and transcriptional regulation of the phaseolin promoter in Arabidopsis. Plant Cell 25:2601–2617. doi:10.1105/tpc.113.112714
Takada S, Takada N, Yoshida A (2013) ATML1 promotes epidermal cell differentiation in Arabidopsis shoots. Development 140:1919–1923. doi:10.1242/dev.094417
Tang X, Bian S, Tang M et al (2012) MicroRNA–mediated repression of the seed maturation program during vegetative development in Arabidopsis. PLoS Genet 8:e1003091. doi:10.1371/journal.pgen.1003091
Thakare D, Tang W, Hill K et al (2008) The MADS-domain transcriptional regulator AGAMOUS-LIKE15 promotes somatic embryo development in Arabidopsis and soybean. Plant Physiol 146:1663–1672. doi:10.1104/pp.108.115832
Thibaud-Nissen F, Shealy RT, Khanna A et al (2003) Clustering of microarray data reveals transcript patterns associated with somatic embryogenesis in soybean. Plant Physiol 132:118–136. doi:10.1104/pp.103.019968
To A, Valon C, Savino G et al (2006) A network of local and redundant gene regulation governs Arabidopsis seed maturation. Plant Cell 18:1642–1651. doi:10.1105/tpc.105.039925
Tsuwamoto R, Pokoi S, Takahata Y (2010) Arabidopsis EMBRYOMAKER encoding an AP2 domain transcription factor plays a key role in developmental change from vegetative to embryonic phase. Plant Mol Biol 73:481–492. doi:10.1007/s11103-010-9634-3
Tzafrir I, Pena-Muralla R, Dickerman A et al (2004) Identification of genes required for embryo development in Arabidopsis. Plant Physiol 135:1206–1220. doi:10.1104/pp.104.045179
Ueda M, Zhang Z, Laux T (2011) Transcriptional activation of Arabidopsis axis patterning genes WOX8/9 links zygote polarity to embryo development. Develop Cell 20:264–270. doi:10.1016/j.devcel.2011.01.009
van der Graaff E, Laux T, Rensing SA (2009) The WUS homeobox-containing (WOX) protein family. Genome Biol 10:248–259. doi:10.1186/gb-2009-10-12-248
Vanneste S, Friml J (2009) Auxin: a trigger for change in plant development. Cell 136:1005–1016. doi:10.1016/j.cell.2009.03.001
Venkatesh K, Rani AR, Baburao N et al (2009) Effect of auxins and auxin polar transport inhibitor (TIBA) on somatic embryogenesis in groundnut (Arachis hypogaea L.). Afr J Plant Sci 3:288–293
Wang H, Caruso LV, Downie AB et al (2004) The embryo MADS domain protein AGAMOUS-Like15 directly regulates expression of a gene encoding an enzyme involved in gibberellin metabolism. Plant Cell 16:1206–1219. doi:10.1105/tpc.021261
Wang XC, Niu QW, Teng C et al (2008) Overexpression of PGA37/MYB118 and MYB115 promotes vegetative-to-embryonic transition in Arabidopsis. Cell Res 19:224–235. doi:10.1038/cr.2008.276
Wang F, Perry SE (2013) Identification of direct targets of FUSCA3, a key regulator of Arabidopsis seed development. Plant Physiol 161:1251–1264. doi:10.1104/pp.112.212282
Wenzel CL, Schuetz M, Yu Q et al (2007) Dynamics of MONOPTEROS and PIN-FORMED1 expression during leaf vein pattern formation in Arabidopsis thaliana. Plant J 49:387–398. doi:10.1111/j.1365-313X.2006.02977.x
West AG, Shore P, Sharrocks AD (1997) DNA binding by MADS-box transcription factors: a molecular mechanism for differential DNA bending. Mol Cell Biol 17:2876–2887. doi:10.1128/MCB.17.5.2876
Wickramasuriya AM, Dunwell JM (2015) Global scale transcriptome analysis of Arabidopsis embryogenesis in vitro. BMC Genom 16:301. doi:10.1186/s12864-015-1504-6
Wiśniewska A, Grabowska A, Pietraszewska-Bogiel A et al (2012) Identification of genes up-regulated during somatic embryogenesis of cucumber. Plant Physiol Biochem 50:54–64. doi:10.1016/j.plaphy.2011.09.017
Wójcikowska B, Jaskóła K, Gąsiorek P et al (2013) LEAFY COTYLEDON2 (LEC2) promotes embryogenic induction in somatic tissue of Arabidopsis via YUCCA-mediated auxin biosynthesis. Planta 238:425–440. doi:10.1007/s00425-013-1892-2
Wójcikowska B, Gaj MD (2015) LEAFY COTYLEDON2-mediated control of the endogenous hormone content: implications for the induction of somatic embryogenesis in Arabidopsis. Plant Cell Tiss Org 121:255–258. doi:10.1007/s11240-014-0689-8
Wu XL, Dabi T, Weigel D (2005) Requirement of homeobox gene STIMPY/WOX9 for Arabidopsis meristem growth and maintenance. Curr Biol 15:436–440. doi:10.1016/j.cub.2004.12.079
Wu G, Poethig RS (2006) Temporal regulation of shoot development in Arabidopsis thaliana by miR156 and its target SPL3. Development 133:3539–3547. doi:10.1242/dev.02521
Wu X, Li F, Zhang C et al (2009) Differential gene expression of cotton cultivar CCRI24 during somatic embryogenesis. J Plant Physiol 166:1275–1283. doi:10.1016/j.jplph.2009.01.012
Yamamoto A, Kagaya Y, Usui H et al (2010) Diverse roles and mechanisms of gene regulation by the Arabidopsis seed maturation master regulator FUS3 revealed by microarray analysis. Plant Cell Physiol 51:2031–2046. doi:10.1093/pcp/pcq162
Yang X, Zhang X, Yuan D et al (2012) Transcript profiling reveals complex auxin signaling pathway and transcription regulation involved in dedifferentiation and redifferentiation during somatic embryogenesis in cotton. BMC Plant Biol 12:110. doi:10.1186/1471-2229-12-110
Yazawa K, Takahata K, Kamada H (2004) Isolation of the gene encoding Carrot leafy cotyledon1 and expression analysis during somatic and zygotic embryogenesis. Plant Physiol Biochem 42:215–223. doi:10.1016/j.plaphy.2003.12.003
Zavattieri MA, Frederico AM, Lima M et al (2010) Induction of somatic embryogenesis as an example of stress-related plant reactions. Elect J Biotechnol 13:1–9. doi:10.2225/vol13-issue1-fulltext-4
Zhang Y, Cao G, Qu JL et al (2009) Involvement of an R2R3-MYB transcription factor gene AtMYB118 in embryogenesis in Arabidopsis. Plant Cell Rep 28:337–346. doi:10.1007/s00299-008-0644-4
Zhang Y, Clemens A, Maximova SN et al (2014) The Theobroma cacao B3 domain transcription factor TcLEC2 plays a dual role in control of embryo development and maturation. BMC Plant Biol 14:106. doi:10.1186/1471-2229-14-106
Zhang Y, Li B, Huai D et al (2015) The conserved transcription factors, MYB115 and MYB118, control expression of the newly evolved benzoyloxy glucosinolate pathway in Arabidopsis thaliana. Front Plant Sci 6:1–19. doi:10.3389/fpls.2015.00343
Zhao Y, Christensen SK, Fankhauser C et al (2001) A role for flavin monooxygenase-like enzymes in auxin biosynthesis. Science 291:306–309. doi:10.1126/science.291.5502.306
Zhao B, Ge L, Liang R et al (2009) Members of miR-169 family are induced by high salinity and transiently inhibit the NF-YA transcription factor. BMC Mol Biol 10:29–41. doi:10.1186/1471-2199-10-29
Zheng Q, Zheng Y, Perry SE (2013) AGAMOUS-Like15 promotes somatic embryogenesis in Arabidopsis and soybean in part by the control of ethylene biosynthesis and response. Plant Physiol 161:2113–2127. doi:10.1104/pp.113.216275
Zheng W, Zhang X, Yang Z et al (2014) AtWuschel promotes formation of the embryogenic callus in Gossypium hirsutum. PLoS ONE 9:e87502. doi:10.1371/journal.pone.0087502
Zheng Q, Perry SE (2014) Alterations in the transcriptome of soybean in response to enhanced somatic embryogenesis promoted by orthologs of AGAMOUS-Like15 and AGAMOUS-Like18. Plant Physiol 164:1365–1377. doi:10.1104/pp.113.234062
Zhu S, Wang J, Ye J et al (2014a) Isolation and characterization of LEAFY COTYLEDON 1-LIKE gene related to embryogenic competence in Citrus sinensis. Plant Cell Tiss Org 119:1–13. doi:10.1007/s11240-014-0509-1
Zhu T, Moschou PN, Alvarez JM et al (2014b) WUSCHEL-RELATED HOMEOBOX 8/9 is important for proper embryo patterning in the gymnosperm Norway spruce. J Exp Bot 65(22):6543–6552. doi:10.1093/jxb/eru371
Zuo J, Niu QW, Frugis G et al (2002a) The WUSCHEL gene promotes vegetative- to -embryonic transition in Arabidopsis. Plant J 30:349–359. doi:10.1046/j.1365-313X.2002.01289.x
Zuo J, Niu QW, Ikeda Y et al (2002b) Marker-free transformation: increasing transformation frequency by the use of regeneration-promoting genes. Curr Opin Biotech 13:173–180. doi:10.1016/S0958-1669(02)00301-4
Acknowledgements
This work was supported by a research grant of the National Centre for Science in Poland (Grant N 2013/09/B//NZ2/03233)
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Nowak, K., Gaj, M.D. (2016). Transcription Factors in the Regulation of Somatic Embryogenesis. In: Loyola-Vargas, V., Ochoa-Alejo, N. (eds) Somatic Embryogenesis: Fundamental Aspects and Applications. Springer, Cham. https://doi.org/10.1007/978-3-319-33705-0_5
Download citation
DOI: https://doi.org/10.1007/978-3-319-33705-0_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-33704-3
Online ISBN: 978-3-319-33705-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)