Abstract
Due to its clear inherited backgrounds as well as simple and diverse genetic manipulation systems, Bacillus subtilis is the key Gram-positive model bacterium for studies on physiology and metabolism. Furthermore, due to its highly efficient protein secretion system and adaptable metabolism, it has been widely used as a cell factory for microbial production of chemicals, enzymes, and antimicrobial materials for industry, agriculture, and medicine. In this mini-review, we first summarize the basic genetic manipulation tools and expression systems for this bacterium, including traditional methods and novel engineering systems. Secondly, we briefly introduce its applications in the production of chemicals and enzymes, and summarize its advantages, mainly focusing on some noteworthy products and recent progress in the engineering of B. subtilis. Finally, this review also covers applications such as microbial additives and antimicrobials, as well as biofilm systems and spore formation. We hope to provide an overview for novice researchers in this area, offering them a better understanding of B. subtilis and its applications.
Similar content being viewed by others
Introduction
Bacillus subtilis is an aerobic, Gram-positive soil bacterium, which has been widely used for the production of heterologous proteins [1]. It secretes numerous enzymes to degrade a variety of substrates, enabling the bacterium to survive in a continuously changing environment. This species and some of its close relatives have excellent protein secretion ability, making them important hosts for the production of medicinal proteins and industrial enzymes. For these reasons, it has been widely used to produce heterologous proteins. Moreover, it has excellent physiological characteristics and highly adaptable metabolism, which makes it easy to cultivate on cheap substrates. Accordingly, B. subtilis grows fast and the fermentation cycle is shorter, usually, around 48 h, while the fermentation cycle of Saccharomyces cerevisiae is around 180 h [2, 3]. Furthermore, excellent expression systems with good genetic stability are available for this organism, and it has no strong codon preference. Different from Escherichia coli, B. subtilis has a single cell membrane, which facilitates protein secretion, simplifies downstream processing, and reduces the process costs. Finally, this species is generally recognized as safe (GRAS) [4, 5].
Over the decades of research, many different tools for genetic modification of B. subtilis have been developed, including the classical counter-selection marker strategies and recently developed clustered regularly interspaced short palindromic repeats (CRISPR)-Cas9/Cpf1 based tool box. Its diverse protein secretion systems, as well as the recently developed artificial promoter and ribosome binding site (RBS) libraries are also helpful in the production of extracellular enzymes. The newly discovered expression cassette integration (MEXI) method based on the mariner transposon can produce knock-in mutants with higher levels of intracellular GFP and extracellular AprE expression than the commonly used amyE integration method [6], thus improving the production of heterologous proteins. In addition to being an excellent host in bioreactors, B. subtilis is an ideal multifunctional probiotic, with great potential for preventing the growth of pathogenic bacteria and enhancing nutrient assimilation [7]. B. subtilis is also commonly used as an industrial cell factory, for the production of vitamins, inositol, acetoin, hyaluronan, and other chemicals. Its clear inherited backgrounds and well-developed gene manipulation tools enabled the reconstruction of its cellular metabolism, and the availability of public knockout collections makes them attractive as metabolic engineering hosts [8]. Yang Gu et al. redesigned the central carbon and redox metabolism of B. subtilis with a new “push-pull promote” approach, through which they manipulated the central carbon metabolism, eliminated the metabolic overflows, and achieved high production of N-acetylglucosamine (GlcNAc) [9]. In agriculture, studies have shown that adding an appropriate amount of B. subtilis can significantly improve the humus and carbon content of compost, thus improving soil quality and promoting crop growth [10]. B. subtilis can also form complex biofilms, which can be used as living biological materials for the production of many functional biomaterials, such as surface growth factors, antibiotics, lysozyme, and antimicrobial peptides for medical materials.
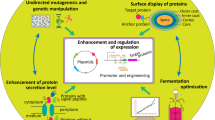
In this paper, we reviewed recent progress in the metabolic engineering and protein expression systems, as well as industrial, agricultural, and biomaterial applications of B. subtilis. Finally, we analyzed the factors that hinder the further application of this strain and discussed the reasons. This review provides a reference for researchers who want to gain a general understanding of B. subtilis and its various applications (Fig. 1).
Application of B. subtilis for genetic engineering, production of industrial chemicals or enzymes, agriculture, medicine and biomaterials. The CRISPR/cas9 tool has been widely used in the genetic engineering of B. subtilis. The bacterium can be used to produce various industrial enzymes, such as α-amylase, xylanase, lichenase, lipase, cellulase, or pectinase. It can also be used to produce various chemicals, such as riboflavin, menaquinone-7, inositol, or N-acetylglucosamine. In agriculture, it can be used as a feed additive. Biofilms of B. subtilis can be used as a biomaterial in 3D printing. In medicine, B. subtilis can be used to produce vaccines
Genetic manipulation of Bacillus subtilis
As B. subtilis was selected as a model bacterium, simple and efficient genetic tools have been developed in the past decades. Classical genome modification relies on the insertion of a selectable marker, usually an antibiotic resistance gene, into the chromosome of the target strain [11]. The most commonly used scarless genetic manipulations systems for B. subtilis rely on counter-selectable markers (CSM) [12], while other methods include site-specific recombination systems (SSR) [13], and the recently developed CRISPR-Cas9 system [14].
CSM are often used for the markerless construction of engineered strains and have been used to construct Bacillus cell factories for various industrial applications. Selectable markers can generally be divided into positive and negative selection markers, whereby the former are most commonly antibiotic-resistance markers. In this classical approach, antibiotic-resistant strains are selected on appropriate agar plates (Fig. 2). In addition to the genomically integrated markers, Jeong et al. constructed a synthetic gene circuit consisting of a plasmid-based selection system, in which the Pxyl-lacI and neomycin resistant gene (neo) are integrated into the genome, while a Pspac-chloramphenicol (cat) resistant cassette and xylR gene are on the plasmid. In the first recombination, Pxyl-lacI and neo are integrated into the genome as a selectable marker. When xylose is added to the medium, the lacI gene is expressed then the chloramphenicol resistant gene is repressed. Consequently, the cell will survive only when the Pxyl-lacI and neo are deleted through a second round of recombination. Finally, the plasmid can be removed after several rounds of culture without chloramphenicol [15]. This is a highly efficient method for genome engineering in B. subtilis, and it avoids the introduction of a selectable marker into the genome or the tightly controlled expression of a toxic gene. Other counter-selectable markers commonly used in B. subtilis include upp, blaI, araR, and hewI [11]. Fabret et al. used the upp gene, which encodes uracil phosphoribosyltransferase as a counter-selection marker to achieve the transmission of unlabeled point mutations, in-frame deletions and large numbers of deletions on the chromosome [16]. Brans et al. developed another method to knock out a single gene and introduce a new gene by combining the use of blaI, an antibiotic resistance gene, which encodes a repressor of the Bacillus licheniformis BlaP β-lactamase, with a conditional lysine-auxotrophic B. subtilis strain [17]. However, CSM-based strategies require host pre-modification and have a low success rate due to the leaky expression of the CSM.
Schematic overview of genome editing methods based on counter-selectable markers. Left: genome editing (gene knockout as an example) with two integration steps. Step 1, an exogenous artificial DNA (plasmid or fragment) with up- and downstream homologous sequences is integrated into the genome, replacing the target gene. The recombinant clone can be selected under condition A. Step 2, under the selection condition B, the clone obtained in step 1 deleted the selectable marker and repressor/toxin gene through a self-recombination with the DR (direct repeats). Right: Composition of the selectable elements, the selectable marker A, toxin gene/repressor, and up- and downstream homologous sequences of the target gene can be constructed as fragments or on a plasmid. Examples of selectable markers A include: cat (chloramphenicol), phleo (phleomycin), or spe (spectinomycin). Examples of toxic genes include: upp, pyrF, or mazF. Examples of repressor genes include: xylR, blaI, araR, or lacI. The repressor can inhibit the expression of the selectable marker B, which can be integrated into the genome or a plasmid
Site-specific recombination (SSR) systems are powerful tools for precise excision of DNA fragments. These systems, such as FLP/FRT [18] and Cre/loxP [13], have much higher recombination efficiency than the endogenous recombination systems, making them an ideal tool for many genetic manipulations. By combining a mutated Cre/lox system with the long segment fusion PCR method [19], Yan et al. developed a rapid and accurate B. subtilis genome engineering tool that allows operations such as targeted gene inactivation, long-fragment deletion, and in-frame deletion of target genes [13].
In recent years, the application of the CRISPR-related (Cas) system in B. subtilis has further enriched the gene editing toolbox. The CRISPR locus is first transcribed into a precursor CRISPR ribonucleic acid (pre-crRNA), which is then cut into small RNA units under the action of Cas protein or endonuclease. These small RNA units are mature crRNAs that contain spacer sequences and partial repeat sequences. Maturation of the crRNAs of type II CRISPR/Cas systems requires not only the participation of Cas9 and RNase, but also the guidance of a tracrRNA [20]. Mature crRNAs and tracrRNA form double-stranded RNA structures through complementary base pairing. The resulting duplexes bind Cas9 protein to form a targeted cutting complex, specifically cutting foreign sequences to achieve the goal of identifying and eliminating invading foreign genes such as plasmids and viruses [21,22,23]. At present, there are three kinds of CRISPR/Cas9-based genome editing strategies widely used in B. subtilis. (1) The single-plasmid based system, in which Cas9, a single guide RNA (gRNA), donor DNA, and other elements are assembled into the same carrier skeleton, wherein Cas9 protein and gRNA are respectively expressed from inducible or strong constitutive promoters; (2) The two-plasmid-based system is more flexible than the single plasmid system. In this system, Cas9, gRNA, and donor DNA are assembled on two different plasmids, which are respectively used to produce Cas9 protein and deliver the gRNA transcription module and donor DNA template; (3) The chromosomally integrated system is more stable and effective than the first two systems, but it requires the use of engineered strains. The Cas9 was integrated into the genome, and then araE/R initiation subsystem was used to construct a multi-gRNA delivery vector [22]. Furthermore, this CRISPR-Cas9 toolkit was extended to CRISPR interference (CRISPRi) for transcriptional-level regulation [21]. The CRISPRi system is composed of a deactivated Cas9 (dCas9) protein and gRNA, enabling the targeting of dCsa9 to any target gene on the genome under the guidance of gRNA to inhibit its transcription without inducing a double-strand break, which can be applied to gene repression in metabolic engineering [24]. So et al. developed a CRISPR-derived genome engineering technique to efficiently generate large genomic deletions in B. subtilis without the introduction of counter-selectable markers such as antibiotic-resistance genes, which had previously limited the application of B. subtilis in food engineering [25]. This method has wide applicability for various types of site-directed mutagenesis in B. subtilis [24, 26, 27]. However, CRISPR/Cas9 has low efficiency in multi-gene editing. Liu et al. recently developed a CRISPR/Cas9n-mediated genome editing system, using Cas9n to exchange the natural Cas9 of existing constructs for B. subtilis and for iterative editing of the genome [28]. This system is more effective than CRISPR/Cas9 in various types of gene modification and shows higher efficiency for large genomic deletions or multiplex gene editing. In terms of multi-gene editing and regulation, the newly developed CRISPR/Cpf1 system is the most powerful tool in B. subtilis. Compared with CRISPR/Cas9, CRISPR/Cpf1 has higher targeting specificity and can be used for gene editing in human cells, plant cells and many bacteria, while also offering a higher efficiency of multiplex gene editing [29]. In fact, the system provides up to 100% efficiency of double in-frame knockouts, enables the introduction of multiple point mutations (up to six) with 100% efficiency, and can be used to simultaneously activate and/or inhibit multiple genes [27].
Gene expression in Bacillus subtilis
To enhance and appropriately adjust gene expression levels in B. subtilis, it is essential to study the promoters that regulate transcription levels [30]. Inducible and constitutive promoters are usually applied for the expression of heterologous genes in B. subtilis [31]. In addition to the promoters, the protein expression level is also influenced by the strength of the ribosome binding site, while plasmids with different copy numbers also provide choices for adjusting the level of gene expression [31]. Different genes have specific expression features, and their expression must be adjusted to a level appropriate for a specific metabolic pathway, which necessitates the use of promoters with different strengths for engineering purposes [32]. Researchers have broadened the scope of target gene transcription levels by constructing promoter libraries. In a recent study, Liu et al. constructed a synthetic promoter library with an intensity gradient by analyzing microarray transcriptome data of B. subtilis 168, which can be used for extensive fine-tuning of genetic pathways in B. subtilis, facilitating strain engineering and synthetic biology [33]. To enable the efficient and accurate co-expression of multiple genes in metabolic networks, a recent review discussed the construction promoter libraries by site-directed mutagenesis [34], such as error-prone PCR, saturation mutagenesis and directional design. RBS sequences can be used to fine-tune gene expression at the translational level. When inducible promoters are used, different ribosomal binding sites can be used to fine-tune the dynamic range of gene expression. In addition, a proteolysis tag can be used to control the degradation rate of a protein at the post-translational level [35]. A synthetic gene expression toolbox consisting of promoter libraries, RBS libraries, and different proteolytic tags can realize gene regulation with a dynamic range of 5 orders of magnitude [36]. Other elements other than the promoter and RBS are also used to adjust gene expression in B. subtilis. Tian et al. conducted and statistical analyzed 96 rationally selected N-terminal coding sequences (NCSs) from B. subtilis, which influenced gene expression at the translation level. They found that NCS substitution is more efficient and convenient than promoter substitution for gene expression improvement [37]. Naseri et al. discussed the construction of complex libraries and combinatorial optimization strategies. However, the examples shown in their review indicate that E.coli and S. cerevisiae are used as host more often than B. subtilis [38]. Therefore, future studies should focus on developing new tools for this important Gram-positive model bacterium.
Protein secretion systems of Bacillus subtilis
Bacillus subtilis has a strong capacity for protein expression and secretion, which has led to its wide use in the production of industrial enzyme preparations. In addition to the abundant promoters and plasmid expression systems described above, B. subtilis also has an efficient protein secretion system to meet the needs of the secretion of various proteins. There are three classical protein secretion pathways in B. subtilis, the general protein secretion pathway (Sec), the twin-arginine translocation pathway (Tat), and the ATP-binding cassette (ABC) transporters [39] (Fig. 3). The Sec pathway is the main transport channel, which can transport a large number of exported proteins. The essential elements of the Sec pathway are the signal recognition particle (SRP), Sec translocase, type I signal peptidase, and chaperones. After the synthesis of the precursor protein, there are two routes through the Sec pathway. In the first one, the signal peptide is recognized by the signal recognition particle (SRP) with the help of a cytoplasmic chaperone, and then transferred to the membrane to bind with FstY, the receptor protein of the SRP, and transported to the channel of the Sec translocase complex (translocation channel). The second route maintains the precursor protein’s translocation ability by preventing its complete folding using intracellular chaperones, and the it is then transfer to the Sec translocase complex. Next, the N-terminal signal peptide sequence is cut off by signal peptidases. Finally, the translocating protein is folded in the extracellular space with the aid of extracellular chaperones [30, 40]. In contrast to the Sec pathway, which relies on unfolded substrates, the Tat pathway transports tightly folded proteins that contain a conserved twin-arginine motif in the signal peptide sequences. Before translocation, the precursors fold in the cytoplasm with the help of cofactors, and are then excreted through the Tat protein complex (Tat translocase) utilizing energy from the pH gradient across the cytoplasmic membrane. After translocation, the type I signal peptidase processes the signal peptide, and finally, the folded mature proteins are secreted out of the cell. ATP-binding cassette (ABC) transporters contain two transmembrane domains (TMD) that define substrate binding sites, and two soluble nucleotide binding domains (NBD) that act as motor domains [30, 39]. They are relatively specific for their substrates and can export or import various molecules (ions, amino acids, peptides, antibiotics, polysaccharides, proteins, etc.) [39].
In addition to the three classical protein secretion pathways mentioned above, researchers also found that many non-classical secretion pathways are used to secrete non-classical proteins lacking any known signal peptides or secretion motifs [41]. Wang et al. used four typical non-classical secretory proteins as signals to direct the export of the nucleoskeletal-like protein (Nsp) from B. subtilis cells. Two of them were able to guide the export of alkaline phosphatase (PhoA), and one of them was able to guide the secretion of the thermostable reporter protein β-galactosidase (BgaB) [42]. Chen et al. used d-psicose 3-epimerase (RDPE) to directly secrete two of five foreign proteins from other bacteria. The fusion proteins not only retained the corresponding enzymatic or biological activities, but also had the activity of RDPE [43] (Fig. 3).
Industrial application of chemicals produced by Bacillus subtilis
The industrial application of B. subtilis has developed rapidly in the last decades, and it has become the major microbial cell factory for many industrial products [44, 45], including enzymes [46], heterologous proteins [31], antibiotics [47], vitamins [48], and amino acids [26]. Chemicals produced by B. subtilis also play an important role in various fields, such as food, feed, cosmetics, chemicals, and pharmaceuticals. Here, we will focus on vitamins and other chemicals produced by B. subtilis, among which the vitamin B2, vitamin K, scyllo-inositol, hyaluronic acid, and N-acetylglucosamine will be discussed in detail. Table 1 also lists some other representative chemicals produced by B. subtilis.
Vitamins as high-value products
In a recent review, microbial cell factories for the production of B vitamins were described in detail [48]. B. subtilis can be used to produce vitamins B1, B2, B5, B6, and B7, and many production strains were constructed by metabolic engineering or screened and selected from mutant libraries [48]. Vitamin B2 is one of the most successful microbial fermentation products on an industrial scale [49]. Also known as riboflavin (RF), Vitamin B2 is the precursor of flavin mononucleotide and flavin adenine dinucleotide [50], and it is widely used for its antioxidant, immunity enhancing, anticancer, as well as food and feed enhancing effects [51]. Metabolic engineering strategies based on the riboflavin metabolic pathway of B. subtilis are usually based on optimization of the central carbon metabolism, overexpression and deregulation of the RF synthesis and purine biosynthesis pathways, as well as blocking the synthesis of by-products [52]. The precursor supply in the RF biosynthesis pathway can be enhanced by redirecting the carbon flux through the PPP (pentose phosphate pathway) from the EMP (Embden-Meyerhof-Parnas) pathway, and increasing the expression of purine biosynthesis genes (pur operon), thus increasing GTP production [53, 54]. The yield of riboflavin in fed-batch fermentation using B. subtilis reached up to 826.52 mg/L [54]. The excellent RF productivity of B. subtilis can also be attributed to its excellent productivity of chemicals sharing the same precursors, such as ribose, purine nucleoside, and folic acid. In addition, the yield of RF can be increased by improving the host characteristics. Recent studies found that the introduction of heat shock proteins from thermophilic bacteria can improve the heat resistance and osmotic tolerance of B. subtilis, enabling it to ferment at a higher temperature, thereby shortening the fermentation time and improving the RF titer [55].
In addition to B vitamins, B. subtilis can also produce menaquinone-7 (MK-7), a member of the valuable vitamin K2 family, which can promote blood coagulation and osteogenic ability, and is also known for other nutraceutical and pharmacological properties. In the fermentation process of MK-7, glycerol, glucose, sucrose, or starch are used as carbon sources, and yeast extract, peptone, sodium nitrate, or soybean peptone as nitrogen sources [56]. Using ethanol to extract MK-7 directly from the cells after fermentation produced a MK-7 yield of 1.47 mg/g [57]. Wu and Ahn optimized the medium components via a three-step response surface methodology (RSM) approach, and the yield of vitamin K increased to 71.95 ± 1.00 mg/L [58]. Yang et al. found that the expression of menA, dxs, dxr, yacM, yacN and glpD constitutes bottlenecks for MK-7 production. Knocking out dhbB can promote the production of MK-7, and it was further improved by adopting high-density fermentation technology [59]. In addition, MK-7 is an important component of the microbial membrane, where it plays an important role in the process of electron transport and oxidative phosphorylation. Cui et al. regulated the membrane composition and electron transport by co-expression of the cell membrane protein tatAD-CD and menaquinol-cytochrome lyase qcrA-C, which increased the titer of MK-7 to 410 mg/L in shake flasks [60].
Scyllo-inositol (SI)
Scyllo-inositol (SI), a stereoisomer of inositol, is being investigated as a potential therapeutic agent for Alzheimer’s disease. However, this compound is far less abundant than its analog myo-inositol (MI), which is the most abundant inositol stereoisomer in nature and can be obtained from rice bran. Consequently, many studies have investigated strategies to convert MI into SI using microorganisms. Tanaka et al. constructed a strain with modified inositol metabolism by deleting all genes related to inositol metabolism, and overexpressing the key enzymes, IolG and IolW, in B. subtilis, resulting in a cell factory that can convert MI into SI with increased efficiency [61]. Through the genetic modification of inositol metabolism and phytase secretion pathway of B. subtilis, the signal peptide of B. subtilis was optimized, the main MI transporter, IolT, was overexpressed, and the substrate absorption was improved. At the same time, the pntAB gene of E. coli was introduced to improve the NADPH production, which improves the activity of inositol dehydrogenase, IolW. These modifications successfully increased the conversion efficiency of MI to 30 g/L/48 h [62].
Hyaluronic acid (HA)
Hyaluronic acid (HA) is a high-value glycosaminoglycan, which is widely used in the biomedical, pharmaceutical, cosmetic, and food industries. Notably, HA preparations with different molecular weight show different effects [63]. Peng Jin et al. downregulated the glycolysis pathway by co-expressing identified committed genes (tuaD, gtaB, glmU, glmM, and glmS). They used the ribosome binding site engineering strategy to regulate the translational level of hyaluronidase and optimized the HA synthesis pathway, which led to the specific production of low-molecular-weight HA [64]. Li et al. recently reported an engineered strain of B. subtilis that can produce HA with different molecular weights and titers at different temperatures. They found that when the biomass increased, the molecular weight of the produced HA decreased while the titer increased [63].
N-acetylglucosamine (GlcNAc)
In addition to naturally occurring products, scientists are also introducing new pathways into B. subtilis to produce new chemicals. N-acetylglucosamine is an acetylated amine derivative of glucose, which plays an important role in the maintenance and repair of cartilage and joint tissue function. Liu et al. divided the GlcNAc biosynthesis network into a glycolysis module, a GlcNAc biosynthesis module, and a peptidoglycan biosynthesis module. By applying modular pathway engineering, they increased the production of GlcNAc in B. subtilis from 1.85 to 31.65 g/L [65]. Then, by knocking out acetolactate synthase (AlsS) and acetolactate decarboxylase (AlsD), the formation of the neutral byproduct acetoin was reduced, and the carbon flux from fructose-6-phosphate toward the GlcNAc synthesis pathway was increased. Consequently, the titer and yield of GlcNAc increased to 48.9 g/L and 0.32 g/g glucose, respectively [66]. The catabolism of GlcNAc in B. subtilis is regulated by GlcN6P. Recently, Liu et al. built a coupled ADC system consisting of a GlcN6P-responsive biosensor and CRISPRi. Using this system, a genetic feedback circuit was constructed to fine-tune the metabolic flow toward GlcNAc synthesis and competing modules, which increased the titer of GlcNAc in a 15-L fed-batch bioreactor to 131.6 g/L [67].
Enzymes produced by Bacillus subtilis
Due to its rapid growth on inexpensive substrates, strong protein secretion ability, non-pathogenicity, and favorable downstream processing, B. subtilis has become an ideal expression host for the production of various industrial enzymes. According to incomplete statistics, enzymes produced using B. subtilis account for 50% of the total enzyme market [44]. Many enzymes have been successfully expressed in B. subtilis, including amylases, xylanases, lichenase, β-galactosidase [68], cellulases [69], alkaline serine proteases [42], and many others. These enzymes play important roles in the food, feed, detergent, textile, leather, paper, and pharmaceutical industries [46]. Due to its GRAS status, proteases from B. subtilis can be used in various food applications, such as soybean hydrolysate preparation, meat tenderization, casein hydrolysate preparation, milk coagulation, and food waste treatment [70]. Here, we will introduce illustrative examples of engineered B. subtilis factories for the production of enzymes such as amylases, xylanases, and lichenases.
Alpha-amylase
Alpha-amylase (EC 3.2.1.1) catalyzes the cleavage of α-1,4-glucosidic bonds, releasing glucose from starch. It is widely used in the textile and paper industries, and B. subtilis is a major host for the production of heterologous α-amylases [71]. Ma et al. used atmospheric and room temperature plasma mutagenesis followed by a novel screening method, and combined with a fermentation optimization strategy, significantly improved the yield of alkaline amylase in B. subtilis 168 [72]. Other studies focused on regulating protein transport and transcription levels by integrating signal peptides and promoter engineering [73]. PrsA is an effective folding catalyst for proteins expressed in B. subtilis. Overexpression of native PrsA from B. subtilis can improve the yield of amylase, but for heterologous amylase, co-expressing the cognate prsA gene had a better effect. QuesadaGanuza et al. developed a new recombinant PrsA variant, which not only increased the yield of amylase in B. subtilis, but also relieved the secretion pressure of the strain [74]. By optimizing the signal peptide and overexpression partner, followed by error-prone PCR and high throughput screening technology to screen improved mutants, the α-amylase activity was improved to 9201.1 U/mL [75].
Xylanases
Xylanases (EC. 3.2.1.8) are enzymes that catalyze the hydrolysis of β-1,4 glycosidic linkages of xylans, releasing oligosaccharides and disaccharides containing reducing sugars and xylose [76]. They have significant application value in biotechnology and can be used to modify lignocellulosic materials. Xylanases are used in animal feed manufacturing, the paper and textile industries, and biofuel production. Commercial xylanases are mainly produced in B. subtilis. Sanchez-Alponti et al. created and characterized single mutants individually replacing five residues in the mesophilic xylanase of B. subtilis with homologous residues from thermophilic enzymes. When the five mutants were combined in a random combinatorial library, a double mutant with improved specific activity and thermal stability was obtained [77]. Yardimci and Cekmecelioglu used Box–Behnken response surface methodology to optimize xylanase production in a co-culture of B. subtilis and Kluyveromyces marxianus, which improved the xylanase yield 4.4-fold (reached 49.5 IU/mL) compared to the initial un-optimized single culture [76].
Lichenase
Lichenase (EC. 3.2.1.73) is a mixed linked β-glucan (MLG) endo-hydrolase found in both microorganisms and plants, which has become a focus of studies on the feasibility of biofuel production [78]. However, due to its poor thermal stability, it is not suitable for biocatalytic biomass conversion. Wang et al. used SpyTag/SpyCatcher-mediated cyclization and non-chromatographic ITC purification to obtain cyclo-lichenase with good heat resistance, which is superior to linear lichenase [78]. In addition, in terms of the catalytic activity of the enzyme, studies have shown that the assembly of subunits induced by the collagen triple helix domain and essential oligomers, such as comp and foldon, can cause target enzyme trimerization, thus improving the activity of lichenase [79].
Application of Bacillus subtilis in agriculture, biomaterials and medicine
Because it is a non-pathogenic probiotic, B. subtilis is often used as a microbial additive to improve intestinal function in animals. It was found promote animal growth and prevent diseases [80]. It can be manufactured in the form of endospores, which then enter the intestinal tract of animals and quickly reactivate to secrete highly active proteases, as lipases and amylases in the upper intestinal tract, which is helpful to degrade complex carbohydrates in plant feed. Furthermore, B. subtilis can produce polypeptides that have an antagonistic effect against intestinal pathogens, effectively improving the digestibility of feed. Additionally, it can be used in water bioremediation and prevent diseases in aquaculture organisms such as shrimp and fish [7]. Because B. subtilis is an aerobic bacterium, it contributes to the anaerobic environment by consuming oxygen in the intestines, which in turn promotes the reproduction of dominant bacteria in the intestine, maintaining the ecological balance of intestines.
B. subtilis can secrete a variety of low molecular weight antimicrobial peptides and bacteriocins, such as surfactin [81], bacilysin [82], and subtilin [83], which have potential value in biomedical engineering, food and agriculture [84]. Antimicrobial peptides from B. subtilis are promising therapeutic tools, because of their broad activity and rapid killing activity against a variety of pathogens. With the increasing problems of microbial resistance due to the non-rational use of conventional antibiotics, antimicrobial peptides will play a more significant role in the treatment of bacterial infections [85]. Antimicrobial peptides also have the advantages of safety and environmental friendliness. They are widely used as feed additives in agriculture and animal husbandry to enhance animal fiber digestion and intestinal health. B. subtilis can improve the balance of intestinal flora and has the potential to improve intestinal health and food absorption efficiency. Studies have shown that adding B. subtilis spores to dairy cattle feed can improve milk and protein production [86]. Furthermore, adding B. subtilis to the diet of laying hens can improve their performance as well as the shell quality of eggs produced by aged laying hens [87].
Biofilms are structured communities of tightly associated cells that constitute the predominant state of bacterial growth in natural and human-made environments [88]. Biofilms can be used to produce living materials with self-healing functions, which are desirable for many products [89]. B. subtilis can form complex and robust biofilms and is a good model strain for studying biofilm formation. Recent studies have shown that biofilms based on the TasA amyloid protein mechanism in B. subtilis exhibit viscoelastic behavior of hydrogels. Studies have shown that biofilms can be accurately fabricated into various three-dimensional (3D) microstructures through 3D-printing and microencapsulation technology [90]. Compared with chemical materials, this artificial living material has metabolic activity, self-renewal ability, and programmability [89]. In addition, the ability of B. subtilis to form biofilms promotes the synthesis of quorum-sensing pentapeptide and nitric oxide (NO), which can delay the aging of the host [91]. Biofilm formation improves the ability of microorganisms to metabolize nutrients and produce chemicals, and can be used to improve the stability of fermentation processes. Recent studies show that a biofilm reactor can promote the extracellular secretion of MK-7 [92].
When faced with starvation, B. subtilis produces endospores that can survive for a long time in a state of anabiosis. And when nutrients are available, it germinate again [93]. This characteristic is conducive to the successful expression of heterologous antigens or increasing the relative activity, thermostability, pH stability, and reusability of enzymes on the surface of spores. The enormous stress resistance of endospores can be coopted to improve the stability and promote the reusability of enzymes in complex environments [94]. In addition, spore surface display technology has recently been successfully applied in the production of protein polymers, vaccines and industrial enzymes [95]. Enzymes such as lipase and chitinase have been successfully expressed on the surface of endospores [94].
Conclusions and future perspectives
As the main model species of Gram-positive bacteria, B. subtilis has a broad array of mature genetic tools, promoters, and plasmid expression systems, which can be used in metabolic engineering, protein expression, and synthetic biology. It can produce chemicals, enzymes, and other industrial bio-products, but also be used as a platform for vaccine preparation or a feed additive in agriculture. Moreover, it is also an ideal model for studying biofilm formation and other physiological characteristics of attached cells. This paper reviewed the advantages of B. subtilis as a chassis cell from several aspects, including genetic manipulation, and heterologous gene expression, as well as its application in industry, agriculture, and medicine.
However, although B. subtilis has various attractive applications, it remains far less studied than its Gram-negative counterpart, E. coli. This remains true even in the present era with rich methodology and rapid tool development. One major bottleneck in the application of new methods attribute to the lower efficiency of plasmids construction in B. subtilis compared with E. coli. To solve this, future engineered strains of B. subtilis can be developed to allow direct plasmid construction. Conversely, the high recombination rate of B. subtilis has some advantages for the development of genome editing tools.
Our goal was to provide reads with a general understanding of the characteristics of B. subtilis, so one can take advantage of its features for certain applications or research purposes. Of course, as more and more scientists and engineers contribute studies on B. subtilis, more technologies, tools, and methods will be applied to B. subtilis in the future, and the potential of this bacterium for scientific and industrial applications will be enhanced further.
Availability of data and supporting materials
Not applicable.
Abbreviations
- RBS:
-
Ribosome binding site
- GlcNAc:
-
N-acetylglucosamine
- CSM:
-
Counter-selectable marker
- SSR:
-
Site-specific recombination system
- CRISPR:
-
Clustered regularly interspaced short palindromic repeats
- CRISPRi:
-
CRISPR interference
- Sec:
-
Secretion pathway
- Tat:
-
Twin-arginine translocation pathway
- ABC:
-
ATP-binding cassette
- SRP:
-
Signal recognition particle
- TMD:
-
Transmembrane domain
- NBD:
-
Nucleotide binding domain
- Nsp:
-
Nucleoskeletal-like protein
- PhoA:
-
Alkaline phosphatase
- BgaB:
-
β-Galactosidase
- RDPE:
-
d-Psicose 3-epimerase
- RF:
-
Riboflavin
- PP pathway:
-
Pentose phosphate pathway
- MK-7:
-
Menaquinone-7
- RSM:
-
Response surface methodology
- SI:
-
scyllo-inositol
- MI:
-
myo-Inositol
- HA:
-
Hyaluronic acid
- AlsS:
-
Acetolactate synthase
- AlsD:
-
Acetolactate decarboxylase
- MLG:
-
Mixed linked β-glucan
- 3D:
-
Three-dimensional
References
Earl AM, Losick R, Kolter R. Ecology and genomics of Bacillus subtilis. Trends Microbiol. 2008;16:269–75.
Chen J, Zhu Y, Fu G, Song Y, Jin Z, Sun Y, Zhang D. High-level intra- and extra-cellular production of d-psicose 3-epimerase via a modified xylose-inducible expression system in Bacillus subtilis. J Ind Microbiol Biotechnol. 2016;43:1577–91.
Aslankoohi E, Rezaei MN, Vervoort Y, Courtin CM, Verstrepen KJ. Glycerol production by fermenting yeast cells is essential for optimal bread dough fermentation. PLoS ONE. 2015;10:e0119364.
Westers L, Westers H, Quax WJ. Bacillus subtilis as cell factory for pharmaceutical proteins: a biotechnological approach to optimize the host organism. Biochim Biophys Acta. 2004;1694:299–310.
Zweers JC, Barak I, Becher D, Driessen AJ, Hecker M, Kontinen VP, Saller MJ, Vavrova L, van Dijl JM. Towards the development of Bacillus subtilis as a cell factory for membrane proteins and protein complexes. Microb Cell Fact. 2008;7:10.
Jeong DE, So Y, Park SY, Park SH, Choi SK. Random knock-in expression system for high yield production of heterologous protein in Bacillus subtilis. J Biotechnol. 2018;266:50–8.
Olmos J, Acosta M, Mendoza G, Pitones V. Bacillus subtilis, an ideal probiotic bacterium to shrimp and fish aquaculture that increase feed digestibility, prevent microbial diseases, and avoid water pollution. Arch Microbiol. 2020;202:427–35.
Gu Y, Xu X, Wu Y, Niu T, Liu Y, Li J, Du G, Liu L. Advances and prospects of Bacillus subtilis cellular factories: From rational design to industrial applications. Metab Eng. 2018;50:109–21.
Gu Y, Lv X, Liu Y, Li J, Du G, Chen J, Rodrigo LA, Liu L. Synthetic redesign of central carbon and redox metabolism for high yield production of N-acetylglucosamine in Bacillus subtilis. Metab Eng. 2019;51:59–69.
Duan M, Zhang Y, Zhou B, Qin Z, Wu J, Wang Q, Yin Y. Effects of Bacillus subtilis on carbon components and microbial functional metabolism during cow manure-straw composting. Bioresour Technol. 2020;303:122868.
Dong H, Zhang D. Current development in genetic engineering strategies of Bacillus species. Microb Cell Fact. 2014;13:63.
Bai H, Deng A, Liu S, Cui D, Qiu Q, Wang L, Yang Z, Wu J, Shang X, Zhang Y, Wen T. A novel tool for microbial genome editing using the restriction-modification system. ACS Synth Biol. 2018;7:98–106.
Yan X, Yu HJ, Hong Q, Li SP. Cre/lox system and PCR-based genome engineering in Bacillus subtilis. Appl Environ Microbiol. 2008;74:5556–62.
Altenbuchner J. Editing of the Bacillus subtilis genome by the CRISPR-Cas9 system. Appl Environ Microbiol. 2016;82:5421–7.
Jeong DE, Park SH, Pan JG, Kim EJ, Choi SK. Genome engineering using a synthetic gene circuit in Bacillus subtilis. Nucleic Acids Res. 2015;43:e42.
Fabret C, Ehrlich SD, Noirot P. A new mutation delivery system for genome-scale approaches in Bacillus subtilis. Mol Microbiol. 2002;46:25–36.
Brans A, Filee P, Chevigne A, Claessens A, Joris B. New integrative method to generate Bacillus subtilis recombinant strains free of selection markers. Appl Environ Microbiol. 2004;70:7241–50.
Chen PT, Shaw JF, Chao YP, David Ho TH, Yu SM. Construction of chromosomally located T7 expression system for production of heterologous secreted proteins in Bacillus subtilis. J Agric Food Chem. 2010;58:5392–9.
Shevchuk NA, Bryksin AV, Nusinovich YA, Cabello FC, Sutherland M, Ladisch S. Construction of long DNA molecules using long PCR-based fusion of several fragments simultaneously. Nucleic Acids Res. 2004;32:e19.
Schilling T, Dietrich S, Hoppert M, Hertel R. A CRISPR-Cas9-based toolkit for fast and precise in vivo genetic engineering of Bacillus subtilis phages. Viruses. 2018;10:241.
Westbrook AW, Moo-Young M, Chou CP. Development of a CRISPR-Cas9 tool kit for comprehensive engineering of Bacillus subtilis. Appl Environ Microbiol. 2016;82:4876.
Hong KQ, Liu DY, Chen T, Wang ZW. Recent advances in CRISPR/Cas9 mediated genome editing in Bacillus subtilis. World J Microbiol Biotechnol. 2018;34:153.
van der Oost J, Westra ER, Jackson RN, Wiedenheft B. Unravelling the structural and mechanistic basis of CRISPR-Cas systems. Nat Rev Microbiol. 2014;12:479–92.
Dong X, Li N, Liu Z, Lv X, Shen Y, Li J, Du G, Wang M, Liu L. CRISPRi-guided multiplexed fine-tuning of metabolic flux for enhanced lacto-N-neotetraose production in Bacillus subtilis. J Agric Food Chem. 2020;68:2477–84.
So Y, Park SY, Park EH, Park SH, Kim EJ, Pan JG, Choi SK. A highly efficient CRISPR-Cas9-mediated large genomic deletion in Bacillus subtilis. Front Microbiol. 2017;8:1167.
Wang C, Cao Y, Wang Y, Sun L, Song H. Enhancing surfactin production by using systematic CRISPRi repression to screen amino acid biosynthesis genes in Bacillus subtilis. Microb Cell Fact. 2019;18:90.
Wu Y, Liu Y, Lv X, Li J, Du G, Liu L. CAMERS-B: CRISPR/Cpf1 assisted multiple-genes editing and regulation system for Bacillus subtilis. Biotechnol Bioeng. 2020;117:1817–25.
Liu D, Huang C, Guo J, Zhang P, Chen T, Wang Z, Zhao X. Development and characterization of a CRISPR/Cas9n-based multiplex genome editing system for Bacillus subtilis. Biotechnol Biofuels. 2019;12:197.
Zhu H, Liang C. CRISPR-DT: designing gRNAs for the CRISPR-Cpf1 system with improved target efficiency and specificity. Bioinformatics. 2019;35:2783–9.
Song Y, Nikoloff JM, Zhang D. Improving protein production on the level of regulation of both expression and secretion pathways in Bacillus subtilis. J Microbiol Biotechnol. 2015;25:963–77.
Cui W, Han L, Suo F, Liu Z, Zhou L, Zhou Z. Exploitation of Bacillus subtilis as a robust workhorse for production of heterologous proteins and beyond. World J Microbiol Biotechnol. 2018;34:145.
Blazeck J, Alper HS. Promoter engineering: recent advances in controlling transcription at the most fundamental level. Biotechnol J. 2013;8:46–58.
Liu D, Mao Z, Guo J, Wei L, Ma H, Tang Y, Chen T, Wang Z, Zhao X. Construction, model-based analysis, and characterization of a promoter library for fine-tuned gene expression in Bacillus subtilis. ACS Synth Biol. 2018;7:1785–97.
Jin LQ, Jin WR, Ma ZC, Shen Q, Cai X, Liu ZQ, Zheng YG. Promoter engineering strategies for the overproduction of valuable metabolites in microbes. Appl Microbiol Biotechnol. 2019;103:8725–36.
Guiziou S, Sauveplane V, Chang HJ, Clerte C, Declerck N, Jules M, Bonnet J. A part toolbox to tune genetic expression in Bacillus subtilis. Nucleic Acids Res. 2016;44:7495–508.
Liu Y, Liu L, Li J, Du G, Chen J. Synthetic biology toolbox and chassis development in Bacillus subtilis. Trends Biotechnol. 2019;37:548–62.
Tian R, Liu Y, Chen J, Li J, Liu L, Du G, Chen J. Synthetic N-terminal coding sequences for fine-tuning gene expression and metabolic engineering in Bacillus subtilis. Metab Eng. 2019;55:131–41.
Naseri G, Koffas MAG. Application of combinatorial optimization strategies in synthetic biology. Nat Commun. 2020;11:2446.
Ling Lin F, Zi Rong X, Wei Fen L, Jiang Bing S, Ping L, Chun Xia H. Protein secretion pathways in Bacillus subtilis: implication for optimization of heterologous protein secretion. Biotechnol Adv. 2007;25:1–12.
Zhang K, Su L, Wu J. Recent advances in recombinant protein production by Bacillus subtilis. Annu Rev Food Sci Technol. 2020;11:295–318.
Zhao L, Chen J, Sun J, Zhang D. Multimer recognition and secretion by the non-classical secretion pathway in Bacillus subtilis. Sci Rep. 2017;7:44023.
Wang JP, Yeh CM, Tsai YC. Improved subtilisin YaB production in Bacillus subtilis using engineered synthetic expression control sequences. J Agric Food Chem. 2006;54:9405–10.
Chen J, Zhao L, Fu G, Zhou W, Sun Y, Zheng P, Sun J, Zhang D. A novel strategy for protein production using non-classical secretion pathway in Bacillus subtilis. Microb Cell Fact. 2016;15:69.
Schallmey M, Singh A, Ward OP. Developments in the use of Bacillus species for industrial production. Can J Microbiol. 2004;50:1–17.
Liu Y, Li J, Du G, Chen J, Liu L. Metabolic engineering of Bacillus subtilis fueled by systems biology: recent advances and future directions. Biotechnol Adv. 2017;35:20–30.
van Dijl J, Hecker M. Bacillus subtilis: from soil bacterium to super-secreting cell factory. Microb Cell Fact. 2013;12:3.
Cherukuri PK, Songkiatisak P, Ding F, Jault JM, Xu XN. Antibiotic drug nanocarriers for probing of multidrug ABC membrane transporter of Bacillus subtilis. ACS Omega. 2020;5:1625–33.
Acevedo-Rocha CG, Gronenberg LS, Mack M, Commichau FM, Genee HJ. Microbial cell factories for the sustainable manufacturing of B vitamins. Curr Opin Biotechnol. 2019;56:18–29.
Revuelta JL, Ledesma-Amaro R, Lozano-Martinez P, Diaz-Fernandez D, Buey RM, Jimenez A. Bioproduction of riboflavin: a bright yellow history. J Ind Microbiol Biotechnol. 2017;44:659–65.
Wang G, Shi T, Chen T, Wang X, Wang Y, Liu D, Guo J, Fu J, Feng L, Wang Z, Zhao X. Integrated whole-genome and transcriptome sequence analysis reveals the genetic characteristics of a riboflavin-overproducing Bacillus subtilis. Metab Eng. 2018;48:138–49.
Suwannasom N, Kao I, Pruss A, Georgieva R, Baumler H. Riboflavin: the health benefits of a forgotten natural vitamin. Int J Mol Sci. 2020;21:950.
Shi T, Wang Y, Wang Z, Wang G, Liu D, Fu J, Chen T, Zhao X. Deregulation of purine pathway in Bacillus subtilis and its use in riboflavin biosynthesis. Microb Cell Fact. 2014;13:101.
Liu S, Hu W, Wang Z, Chen T. Production of riboflavin and related cofactors by biotechnological processes. Microb Cell Fact. 2020;19:31.
Shi S, Chen T, Zhang Z, Chen X, Zhao X. Transcriptome analysis guided metabolic engineering of Bacillus subtilis for riboflavin production. Metab Eng. 2009;11:243–52.
Wang J, Wang W, Wang H, Yuan F, Xu Z, Yang K, Li Z, Chen Y, Fan K. Improvement of stress tolerance and riboflavin production of Bacillus subtilis by introduction of heat shock proteins from thermophilic bacillus strains. Appl Microbiol Biotechnol. 2019;103:4455–65.
Berenjian A, Mahanama R, Talbot A, Biffin R, Regtop H, Valtchev P, Kavanagh J, Dehghani F. Efficient media for high menaquinone-7 production: response surface methodology approach. Nat Biotechnol. 2011;28:665–72.
Fang Z, Wang L, Zhao G, Liu H, Wei H, Wang H, Ni W, Zheng Z, Wang P. A simple and efficient preparative procedure for menaquinone-7 from Bacillus subtilis (natto) using two-stage extraction followed by microporous resins. Process Biochem. 2019;83:183–8.
Wu WJ, Ahn BY. Statistical optimization of medium components by response surface methodology to enhance menaquinone-7 (Vitamin K(2)) production by Bacillus subtilis. J Microbiol Biotechnol. 2018;28:902–8.
Yang S, Cao Y, Sun L, Li C, Lin X, Cai Z, Zhang G, Song H. Modular pathway engineering of Bacillus subtilis to promote de novo biosynthesis of menaquinone-7. ACS Synth Biol. 2019;8:70–81.
Cui S, Xia H, Chen T, Gu Y, Lv X, Liu Y, Li J, Du G, Liu L. Cell membrane and electron transfer engineering for improved synthesis of menaquinone-7 in Bacillus subtilis. iScience. 2020;23:100918.
Tanaka K, Natsume A, Ishikawa S, Takenaka S, Yoshida KI. A new-generation of Bacillus subtilis cell factory for further elevated scyllo-inositol production. Microb Cell Fact. 2017;16:67.
Yoshida KI, Ishikawa S. Production of scyllo-Inositol: conversion of rice bran into a promising disease-modifying therapeutic agent for Alzheimer’s disease. J Nutr Sci Vitaminol. 2019;65:139–42.
Li Y, Li G, Zhao X, Shao Y, Wu M, Ma T. Regulation of hyaluronic acid molecular weight and titer by temperature in engineered Bacillus subtilis. 3 Biotech. 2019;9:225.
Jin P, Kang Z, Yuan P, Du G, Chen J. Production of specific-molecular-weight hyaluronan by metabolically engineered Bacillus subtilis 168. Metab Eng. 2016;35:21–30.
Liu Y, Zhu Y, Li J, Shin HD, Chen RR, Du G, Liu L, Chen J. Modular pathway engineering of Bacillus subtilis for improved N-acetylglucosamine production. Metab Eng. 2014;23:42–52.
Ma W, Liu Y, Shin HD, Li J, Chen J, Du G, Liu L. Metabolic engineering of carbon overflow metabolism of Bacillus subtilis for improved N-acetyl-glucosamine production. Bioresour Technol. 2018;250:642–9.
Wu Y, Chen T, Liu Y, Tian R, Lv X, Li J, Du G, Chen J, Ledesma-Amaro R, Liu L. Design of a programmable biosensor-CRISPRi genetic circuits for dynamic and autonomous dual-control of metabolic flux in Bacillus subtilis. Nucleic Acids Res. 2020;48:996–1009.
Watzlawick H, Altenbuchner J. Multiple integration of the gene ganA into the Bacillus subtilis chromosome for enhanced beta-galactosidase production using the CRISPR/Cas9 system. AMB Express. 2019;9:158.
Lan Thanh Bien T, Tsuji S, Tanaka K, Takenaka S, Yoshida K. Secretion of heterologous thermostable cellulases in Bacillus subtilis. J Gen Appl Microbiol. 2014;60:175–82.
Patel AR, Mokashe NU, Chaudhari DS, Jadhav AG, Patil UK. Production optimisation and characterisation of extracellular protease secreted by newly isolated Bacillus subtilis AU-2 strain obtained from Tribolium castaneum gut. Biocatal Agric Biotechnol. 2019;19:101122.
Yan S, Wu G. Bottleneck in secretion of alpha-amylase in Bacillus subtilis. Microb Cell Fact. 2017;16:124.
Ma Y, Shen W, Chen X, Liu L, Zhou Z, Xu F, Yang H. Significantly enhancing recombinant alkaline amylase production in Bacillus subtilis by integration of a novel mutagenesis-screening strategy with systems-level fermentation optimization. J Biol Eng. 2016;10:13.
Yang H, Ma Y, Zhao Y, Shen W, Chen X. Systematic engineering of transport and transcription to boost alkaline alpha-amylase production in Bacillus subtilis. Appl Microbiol Biotechnol. 2020;104:2973–85.
Quesada-Ganuza A, Antelo-Varela M, Mouritzen JC, Bartel J, Becher D, Gjermansen M, Hallin PF, Appel KF, Kilstrup M, Rasmussen MD, Nielsen AK. Identification and optimization of PrsA in Bacillus subtilis for improved yield of amylase. Microb Cell Fact. 2019;18:158.
Yao D, Su L, Li N, Wu J. Enhanced extracellular expression of Bacillus stearothermophilus α-amylase in Bacillus subtilis through signal peptide optimization, chaperone overexpression and α-amylase mutant selection. Microb Cell Fact. 2019;18:69.
Yardimci GO, Cekmecelioglu D. Assessment and optimization of xylanase production using co-cultures of Bacillus subtilis and Kluyveromyces marxianus. 3 Biotech. 2018;8:290.
Alponti JS, Fonseca Maldonado R, Ward RJ. Thermostabilization of Bacillus subtilis GH11 xylanase by surface charge engineering. Int J Biol Macromol. 2016;87:522–8.
Wang J, Wang Y, Wang X, Zhang D, Wu S, Zhang G. Enhanced thermal stability of lichenase from Bacillus subtilis 168 by SpyTag/SpyCatcher-mediated spontaneous cyclization. Biotechnol Biofuels. 2016;9:79.
Wang X, Ge H, Zhang D, Wu S, Zhang G. Oligomerization triggered by foldon: a simple method to enhance the catalytic efficiency of lichenase and xylanase. BMC Biotechnol. 2017;17:57.
Lee NK, Kim WS, Paik HD. Bacillus strains as human probiotics: characterization, safety, microbiome, and probiotic carrier. Food Sci Biotechnol. 2019;28:1297–305.
Kim PI, Ryu J, Kim YH, Chi Y-T. Production of biosurfactant lipopeptides iturin A, fengycin and surfactin A from Bacillus subtilis CMB32 for control of colletotrichum gloeosporioides. J Microbiol Biotechnol. 2010;20:138–45.
Afsharmanesh H, Perez-Garcia A, Zeriouh H, Ahmadzadeh M, Romero D. Aflatoxin degradation by Bacillus subtilis UTB1 is based on production of an oxidoreductase involved in bacilysin biosynthesis. Food Control. 2018;94:48–55.
Corvey C, Stein T, Düsterhus S, Karas M, Entian K-D. Activation of subtilin precursors by Bacillus subtilis extracellular serine proteases subtilisin (AprE), WprA, and Vpr. Biochem Biophys Res Commun. 2003;304:48–54.
Stein T. Bacillus subtilis antibiotics: structures, syntheses and specific functions. Mol Microbiol. 2005;56:845–57.
Sumi CD, Yang BW, Yeo IC, Hahm YT. Antimicrobial peptides of the genus Bacillus: a new era for antibiotics. Can J Microbiol. 2015;61:93–103.
Souza VL, Lopes NM, Zacaroni OF, Silveira VA, Pereira RAN, Freitas JA, Almeida R, Salvati GGS, Pereira MN. Lactation performance and diet digestibility of dairy cows in response to the supplementation of Bacillus subtilis spores. Livestock Science. 2017;200:35–9.
Abdelqader A, Irshaid R, Al-Fataftah AR. Effects of dietary probiotic inclusion on performance, eggshell quality, cecal microflora composition, and tibia traits of laying hens in the late phase of production. Trop Anim Health Prod. 2013;45:1017–24.
Pisithkul T, Schroeder JW, Trujillo EA, Yeesin P, Stevenson DM, Chaiamarit T, Coon JJ, Wang JD, Amador-Noguez D. Metabolic remodeling during biofilm development of Bacillus subtilis. MBio. 2019;10:e00623–19.
Huang J, Liu S, Zhang C, Wang X, Pu J, Ba F, Xue S, Ye H, Zhao T, Li K, et al. Programmable and printable Bacillus subtilis biofilms as engineered living materials. Nat Chem Biol. 2019;15:34–41.
Balasubramanian S, Aubin-Tam ME, Meyer AS. 3D printing for the fabrication of biofilm-based functional living materials. ACS Synth Biol. 2019;8:1564–7.
Kovacs AT, Dragos A. Evolved biofilm: review on the experimental evolution studies of Bacillus subtilis pellicles. J Mol Biol. 2019;431:4749–59.
Mahdinia E, Demirci A, Berenjian A. Biofilm reactors as a promising method for vitamin K (menaquinone-7) production. Appl Microbiol Biotechnol. 2019;103:5583–92.
Setlow P. Spores of Bacillus subtilis: their resistance to and killing by radiation, heat and chemicals. J Appl Microbiol. 2006;101:514–25.
Lin P, Yuan H, Du J, Liu K, Liu H, Wang T. Progress in research and application development of surface display technology using Bacillus subtilis spores. Appl Microbiol Biotechnol. 2020;104:2319–31.
Chen H, Ullah J, Jia J. Progress in Bacillus subtilis spore surface display technology towards environment, vaccine development, and biocatalysis. J Mol Microbiol Biotechnol. 2017;27:159–67.
Zhou K, Zou R, Zhang C, Stephanopoulos G, Too HP. Optimization of amorphadiene synthesis in bacillus subtilis via transcriptional, translational, and media modulation. Biotechnol Bioeng. 2013;110:2556–61.
Huang J, Du Y, Xu G, Zhang H, Zhu F, Huang L, Xu Z. High yield and cost-effective production of poly(γ-glutamic acid) with Bacillus subtilis. Eng Life Sci. 2011;11:291–7.
Dai J, Wang Z, Xiu ZL. High production of optically pure (3R)-acetoin by a newly isolated marine strain of Bacillus subtilis CGMCC 13141. Bioprocess Biosyst Eng. 2019;42:475–83.
Licona-Cassani C, Lara AR, Cabrera-Valladares N, Escalante A, Hernandez-Chavez G, Martinez A, Bolivar F, Gosset G. Inactivation of pyruvate kinase or the phosphoenolpyruvate: sugar phosphotransferase system increases shikimic and dehydroshikimic acid yields from glucose in Bacillus subtilis. J Mol Microbiol Biotechnol. 2014;24:37–45.
Fu J, Huo G, Feng L, Mao Y, Wang Z, Ma H, Chen T, Zhao X. Metabolic engineering of Bacillus subtilis for chiral pure meso-2,3-butanediol production. Biotechnol Biofuels. 2016;9:90.
Qi H, Li S, Zhao S, Huang D, Xia M, Wen J. Model-driven redox pathway manipulation for improved isobutanol production in Bacillus subtilis complemented with experimental validation and metabolic profiling analysis. PLoS One. 2014;9:e93815.
Jin P, Zhang L, Yuan P, Kang Z, Du G, Chen J. Efficient biosynthesis of polysaccharides chondroitin and heparosan by metabolically engineered Bacillus subtilis. Carbohydr Polym. 2016;140:424–32.
Acknowledgements
This work was supported by the National Key R&D Program of China (2018YFA0901600), the Tianjin Science Fund for Distinguished Young Scholars (17JCJQJC45300), and the Science and Technology Service Network (STS) Initiative of the Chinese Academy of Sciences (CAS) (KFJ-STS-ZDTP-065).
Funding
This study was funded by National Key R&D Program of China (2018YFA0901600), the Tianjin Science Fund for Distinguished Young Scholars (17JCJQJC45300), Science and Technology Service Network (STS) Initiative of the Chinese Academy of Sciences (CAS) (KFJ-STS-ZDTP-065).
Author information
Authors and Affiliations
Contributions
All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Su, Y., Liu, C., Fang, H. et al. Bacillus subtilis: a universal cell factory for industry, agriculture, biomaterials and medicine. Microb Cell Fact 19, 173 (2020). https://doi.org/10.1186/s12934-020-01436-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12934-020-01436-8