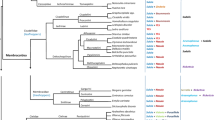
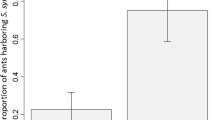
Abstract
Insects can establish a variety of symbiotic associations with bacteria that can have a significant impact on their evolutionary ecology. Some bacterial lineages are particularly pervasive as symbiotic associates. This is the case of the Sodalis genus, whose members have established independent, maternally transmitted symbioses in diverse insect taxa. The first members of the genus were isolated and studied some thirty years ago in tsetse flies, where they evolved as heritable facultative symbionts. Since then, numerous symbiotic associations involving members of the genus have been documented, some of which have evolved into strictly host-dependent mutualistic associations. The genus also includes members circulating freely in the environment, which can be pathogenic, have extensive metabolic capabilities and constitute a potential reservoir of new insect symbionts. In this review, we cover more than thirty years of literature to highlight how the diversity of the Sodalis genus described so far embodies the different degrees of host dependence and anatomical integration that bacteria can experience over the course of their evolution with insects. We discuss the propensity of Sodalis bacteria to embrace an endosymbiotic lifestyle, how this feature can be used to understand the nascent stages of bacterial endosymbiosis, and how Sodalis bacteria can be used to address fundamental and applied research issues. Throughout the review, emphasis is placed on research gaps that need to be filled to better address these aspects. We also draw attention to previously overlook facets of the genus that deserve further investigation, such as the potential role of Sodalis bacteria in wood digestion in certain insects, or the nature of their interaction with plants.



Similar content being viewed by others
References
Adeolu M, Alnajar S, Naushad S, Gupta S R (2016) Genome-based phylogeny and taxonomy of the Enterobacteriales: proposal for Enterobacterales Ord. nov. divided into the families Enterobacteriaceae, Erwiniaceae fam. nov., Pectobacteriaceae fam. nov., Yersiniaceae fam. nov., Hafniaceae fam. nov., Morganellaceae fam. nov., and Budviciaceae fam. Nov. Int J Syst Evol Microbiol 66:5575–5599. https://doi.org/10.1099/ijsem.0.001485
Akman L, Yamashita A, Watanabe H et al (2002) Genome sequence of the endocellular obligate symbiont of tsetse flies, Wigglesworthia glossinidia. Nat Genet 32:402–407. https://doi.org/10.1038/ng986
Aksoy S (1995) Wigglesworthia gen. nov. and Wigglesworthia glossinidia sp. nov., Taxa Consisting of the Mycetocyte-Associated, primary endosymbionts of tsetse flies. Int J Syst Bacteriol 45:848–851. https://doi.org/10.1099/00207713-45-4-848
Aksoy S, Chen X, Hypsa V (1997) Phylogeny and potential transmission routes of midgut-associated endosymbionts of tsetse (Diptera:Glossinidae). Insect Mol Biol 6:183–190. https://doi.org/10.1111/j.1365-2583.1997.tb00086.x
Aksoy S, Weiss B, Attardo G (2008) Paratransgenesis Applied for Control of Tsetse transmitted sleeping sickness. In: Aksoy S (ed) Transgenesis and the management of Vector-Borne Disease. Springer, New York, NY, pp 35–48
Aksoy E, Telleria EL, Echodu R et al (2014) Analysis of multiple tsetse fly populations in Uganda reveals limited diversity and species-specific gut microbiota. Appl Environ Microbiol 80:4301–4312. https://doi.org/10.1128/AEM.00079-14
Alam U, Medlock J, Brelsfoard C et al (2011) Wolbachia Symbiont Infections induce strong cytoplasmic incompatibility in the tsetse fly Glossina morsitans. PLOS Pathog 7:e1002415. https://doi.org/10.1371/journal.ppat.1002415
Alam U, Hyseni C, Symula RE et al (2012) Implications of Microfauna-Host Interactions for Trypanosome Transmission Dynamics in Glossina fuscipes fuscipes in Uganda. Appl Environ Microbiol. https://doi.org/10.1128/AEM.00806-12
Alickovic L, Johnson KP, Boyd BM (2021) The reduced genome of a heritable symbiont from an ectoparasitic feather feeding louse. BMC Ecol Evol 21:108. https://doi.org/10.1186/s12862-021-01840-7
Ankrah N, Chouaia B, Douglas A (2018) The cost of metabolic interactions in Symbioses between insects and Bacteria with reduced genomes. https://doi.org/10.1128/mBio.01433-18. mBio 9:
Anselme C, Pérez-Brocal V, Vallier A et al (2008) Identification of the Weevil immune genes and their expression in the bacteriome tissue. BMC Biol 6:43. https://doi.org/10.1186/1741-7007-6-43
Arp A, Munyaneza JE, Crosslin JM et al (2014) A global comparison of Bactericera cockerelli (Hemiptera: Triozidae) microbial communities. Environ Entomol 43:344–352. https://doi.org/10.1603/EN13256
Attardo GM, Lohs C, Heddi A et al (2008) Analysis of milk gland structure and function in Glossina morsitans: milk protein production, symbiont populations and fecundity. J Insect Physiol 54:1236–1242. https://doi.org/10.1016/j.jinsphys.2008.06.008
Attardo GM, Scolari F, Malacrida A (2020) Bacterial symbionts of Tsetse flies: relationships and functional interactions between tsetse flies and their symbionts. In: Kloc M (ed) Symbiosis: Cellular, Molecular, Medical and Evolutionary aspects. Springer International Publishing, Cham, pp 497–536
Balmand S, Lohs C, Aksoy S, Heddi A (2013) Tissue distribution and transmission routes for the tsetse fly endosymbionts. J Invertebr Pathol 112:S116–S122. https://doi.org/10.1016/j.jip.2012.04.002
Baumann P (2005) Biology of Bacteriocyte-Associated endosymbionts of Plant Sap-Sucking insects. Annu Rev Microbiol 59:155–189. https://doi.org/10.1146/annurev.micro.59.030804.121041
Beard CB, O’Neill SL, Mason P et al (1993) Genetic transformation and phylogeny of bacterial symbionts from tsetse. Insect Mol Biol 1:123–131. https://doi.org/10.1111/j.1365-2583.1993.tb00113.x
Belda E, Moya A, Bentley S, Silva FJ (2010) Mobile genetic element proliferation and gene inactivation impact over the genome structure and metabolic capabilities of Sodalis glossinidius, the secondary endosymbiont of tsetse flies. BMC Genomics 11:449. https://doi.org/10.1186/1471-2164-11-449
Boyd BM, Allen JM, Koga R et al (2016) Two bacterial Genera, Sodalis and Rickettsia, Associated with the seal louse Proechinophthirus fluctus (Phthiraptera: Anoplura). Appl Environ Microbiol 82:3185–3197. https://doi.org/10.1128/AEM.00282-16
Bruner-Montero G, Wood M, Horn HA et al (2021) Symbiont-mediated Protection of Acromyrmex Leaf-Cutter ants from the Entomopathogenic Fungus Metarhizium anisopliae. https://doi.org/10.1128/mBio.01885-21. mBio
Burke G, Fiehn O, Moran N (2010) Effects of facultative symbionts and heat stress on the metabolome of pea aphids. ISME J 4:242–252. https://doi.org/10.1038/ismej.2009.114
Caspi-Fluger A, Inbar M, Mozes-Daube N et al (2011) Horizontal transmission of the insect symbiont Rickettsia is plant-mediated. Proc R Soc B Biol Sci 279:1791–1796. https://doi.org/10.1098/rspb.2011.2095
Channumsin M, Ciosi M, Masiga D et al (2018) Sodalis glossinidius presence in wild tsetse is only associated with presence of trypanosomes in complex interactions with other tsetse-specific factors. BMC Microbiol 18:163. https://doi.org/10.1186/s12866-018-1285-6
Chaput G, Ford J, DeDiego L et al (2022) Sodalis Ligni strain 159R isolated from an anaerobic lignin-degrading Consortium. Microbiol Spectr 10:e0234621. https://doi.org/10.1128/spectrum.02346-21
Chari A, Oakeson KF, Enomoto S et al (2015) Phenotypic characterization of Sodalis praecaptivus sp. nov., a close non-insect-associated member of the Sodalis-allied lineage of insect endosymbionts. Int J Syst Evol Microbiol 65:1400–1405. https://doi.org/10.1099/ijs.0.000091
Charles H, Heddi A, Rahbe Y (2001) A putative insect intracellular endosymbiont stem clade, within the Enterobacteriaceae, infered from phylogenetic analysis based on a heterogeneous model of DNA evolution. Comptes Rendus Académie Sci - Ser III - Sci Vie 324:489–494. https://doi.org/10.1016/S0764-4469(01)01328-2
Chellappan M, Ranjith MT (2021) Metagenomic approaches for insect symbionts. In: Omkar (ed) Microbial approaches for Insect Pest Management. Springer, Singapore, pp 271–313
Chong RA, Park H, Moran NA (2019) Genome evolution of the Obligate Endosymbiont Buchnera aphidicola. Mol Biol Evol 36:1481–1489. https://doi.org/10.1093/molbev/msz082
Chrudimský T, Husník F, Nováková E, Hypša V (2012) Candidatus Sodalis melophagi sp. nov.: phylogenetically Independent comparative model to the tsetse fly Symbiont Sodalis glossinidius. PLoS ONE 7:e40354. https://doi.org/10.1371/journal.pone.0040354
Clayton AL, Oakeson KF, Gutin M et al (2012) A Novel Human-infection-derived bacterium provides insights into the Evolutionary origins of Mutualistic insect–bacterial Symbioses. PLOS Genet 8:e1002990. https://doi.org/10.1371/journal.pgen.1002990
Conord C, Despres L, Vallier A et al (2008) Long-term Evolutionary Stability of Bacterial endosymbiosis in Curculionoidea: additional evidence of Symbiont replacement in the Dryophthoridae Family. Mol Biol Evol 25:859–868. https://doi.org/10.1093/molbev/msn027
Cooper WR, Horton DR, Swisher-Grimm K et al (2022) Bacterial endosymbionts of Bactericera maculipennis and three mitochondrial haplotypes of B. cockerelli (Hemiptera: Psylloidea: Triozidae). Environ Entomol 51:94–107. https://doi.org/10.1093/ee/nvab133
Dale C, Maudlin I (1999) Sodalis gen. nov. and Sodalis glossinidius sp. nov., a microaerophilic secondary endosymbiont of the tsetse fly Glossina morsitans morsitans. Int J Syst Bacteriol 49 Pt 1:267–275. https://doi.org/10.1099/00207713-49-1-267
Dale C, Welburn SC (2001) The endosymbionts of tsetse flies: manipulating host-parasite interactions. Int J Parasitol 31:628–631. https://doi.org/10.1016/s0020-7519(01)00151-5
Dale C, Plague GR, Wang B et al (2002) Type III secretion systems and the evolution of mutualistic endosymbiosis. Proc Natl Acad Sci 99:12397–12402. https://doi.org/10.1073/pnas.182213299
Dale C, Jones T, Pontes M (2005) Degenerative evolution and functional diversification of type-III secretion systems in the insect endosymbiont Sodalis glossinidius. Mol Biol Evol 22:758–766. https://doi.org/10.1093/molbev/msi061
De Vooght L, Caljon G, Stijlemans B et al (2012) Expression and extracellular release of a functional anti-trypanosome Nanobody® in Sodalis glossinidius, a bacterial symbiont of the tsetse fly. Microb Cell Factories 11:23. https://doi.org/10.1186/1475-2859-11-23
De Vooght L, Caljon G, De Ridder K, Van Den Abbeele J (2014) Delivery of a functional anti-trypanosome Nanobody in different tsetse fly tissues via a bacterial symbiont, Sodalis glossinidius. Microb Cell Factories 13:156. https://doi.org/10.1186/s12934-014-0156-6
De Vooght L, Caljon G, Van Hees J, Van Den Abbeele J (2015) Paternal transmission of a secondary symbiont during mating in the Viviparous Tsetse fly. Mol Biol Evol 32:1977–1980. https://doi.org/10.1093/molbev/msv077
De Vooght L, Van Keer S, Van Den Abbeele J (2018) Towards improving tsetse fly paratransgenesis: stable colonization of Glossina morsitans morsitans with genetically modified Sodalis. BMC Microbiol 18:165. https://doi.org/10.1186/s12866-018-1282-9
Dedeine F, Vavre F, Fleury F et al (2001) Removing symbiotic Wolbachia bacteria specifically inhibits oogenesis in a parasitic wasp. Proc Natl Acad Sci 98:6247–6252. https://doi.org/10.1073/pnas.101304298
Dennis JW, Durkin SM, Horsley Downie JE et al (2014) Sodalis glossinidius prevalence and trypanosome presence in tsetse from Luambe National Park, Zambia. Parasit Vectors 7:378. https://doi.org/10.1186/1756-3305-7-378
Dhami MK, Buckley TR, Beggs JR, Taylor MW (2013) Primary symbiont of the ancient scale insect family Coelostomidiidae exhibits strict cophylogenetic patterns. Symbiosis 61:77–91. https://doi.org/10.1007/s13199-013-0257-8
Douglas AE (2014) Molecular dissection of nutrient exchange at the insect-microbial interface. Curr Opin Insect Sci 4:23–28. https://doi.org/10.1016/j.cois.2014.08.007
Drew GC, Stevens EJ, King KC (2021) Microbial evolution and transitions along the parasite–mutualist continuum. Nat Rev Microbiol 19:623–638. https://doi.org/10.1038/s41579-021-00550-7
Dyer NA, Lawton SP, Ravel S et al (2008) Molecular phylogenetics of tsetse flies (Diptera: Glossinidae) based on mitochondrial (COI, 16S, ND2) and nuclear ribosomal DNA sequences, with an emphasis on the palpalis group. Mol Phylogenet Evol 49:227–239. https://doi.org/10.1016/j.ympev.2008.07.011
El Yamlahi Y, Bel Mokhtar N, Maurady A et al (2023) Characterization of the Bacterial Profile from Natural and Laboratory Glossina populations. Insects 14:840. https://doi.org/10.3390/insects14110840
Elston KM, Leonard SP, Geng P et al (2022) Engineering insects from the endosymbiont out. Trends Microbiol 30:79–96. https://doi.org/10.1016/j.tim.2021.05.004
Emms DM, Kelly S (2019) OrthoFinder: phylogenetic orthology inference for comparative genomics. Genome Biol 20:238. https://doi.org/10.1186/s13059-019-1832-y
Engel P, Moran NA (2013) The gut microbiota of insects – diversity in structure and function. FEMS Microbiol Rev 37:699–735. https://doi.org/10.1111/1574-6976.12025
Enomoto S, Chari A, Clayton AL, Dale C (2017) Quorum sensing attenuates virulence in Sodalis praecaptivus. Cell Host Microbe 21:629–636e5. https://doi.org/10.1016/j.chom.2017.04.003
Farikou O, Njiokou F, Mbida Mbida JA et al (2010) Tripartite interactions between tsetse flies, Sodalis glossinidius and trypanosomes–an epidemiological approach in two historical human African Trypanosomiasis foci in Cameroon. Infect Genet Evol J Mol Epidemiol Evol Genet Infect Dis 10:115–121. https://doi.org/10.1016/j.meegid.2009.10.008
Feldhaar H (2011) Bacterial symbionts as mediators of ecologically important traits of insect hosts. Ecol Entomol 36:533–543. https://doi.org/10.1111/j.1365-2311.2011.01318.x
Ferrarini MG, Dell’Aglio E, Vallier A et al (2022) Efficient compartmentalization in insect bacteriomes protects symbiotic bacteria from host immune system. Microbiome 10:156. https://doi.org/10.1186/s40168-022-01334-8
Ferrarini MG, Vallier A, Vincent-Monégat C et al (2023) Coordination of host and endosymbiont gene expression governs endosymbiont growth and elimination in the cereal weevil Sitophilus spp. 2023.04.03.535335
Fourie A, Venter SN, Slippers B, Fourie G (2023) Pantoea bathycoeliae sp. nov and Sodalis sp. are core gut microbiome symbionts of the two-spotted stink bug. Front Microbiol 14
Frago E, Zytynska SE, Fatouros NE (2020) Chapter Four - Microbial symbionts of herbivorous species across the insect tree. In: Oliver KM, Russell JA (eds) Advances in Insect Physiology. Academic Press, pp 111–159
Fukatsu T, Koga R, Smith WA et al (2007) Bacterial endosymbiont of the Slender Pigeon Louse, Columbicola columbae, Allied to Endosymbionts of Grain Weevils and Tsetse flies. https://doi.org/10.1128/AEM.01131-07. Appl Environ Microbiol
Galambos N, Vincent-Monegat C, Vallier A et al (2023) Cereal Weevil’s antimicrobial peptides. At the Crosstalk between Development, Endosymbiosis and Immune Response, Immunology
Ganesan R, Janke RS, Kaltenpoth M, Flórez LV (2023) Colonization dynamics of a defensive insect ectosymbiont. Biol Lett 19:20230100. https://doi.org/10.1098/rsbl.2023.0100
Garber AI, Kupper M, Laetsch DR et al (2021) The evolution of interdependence in a four-way mealybug symbiosis. Genome Biol Evol 13:evab123. https://doi.org/10.1093/gbe/evab123
Gatehouse LN, Sutherland P, Forgie SA et al (2012) Molecular and histological characterization of primary (Betaproteobacteria) and secondary (Gammaproteobacteria) endosymbionts of three mealybug species. Appl Environ Microbiol. https://doi.org/10.1128/AEM.06340-11
Geiger A, Cuny G, Frutos R (2005) Two tsetse fly species, Glossina palpalis gambiensis and glossina morsitans morsitans, carry genetically distinct populations of the secondary Symbiont Sodalis glossinidius. Appl Environ Microbiol 71:8941–8943. https://doi.org/10.1128/AEM.71.12.8941-8943.2005
Geiger A, Ponton F, Simo G (2015) Adult blood-feeding tsetse flies, trypanosomes, microbiota and the fluctuating environment in sub-saharan Africa. ISME J 9:1496–1507. https://doi.org/10.1038/ismej.2014.236
Ghosh S, Sela N, Kontsedalov S et al (2020) An Intranuclear Sodalis-Like Symbiont and Spiroplasma Coinfect the Carrot psyllid, Bactericera Trigonica. Psylloidea) Microorganisms 8:692. https://doi.org/10.3390/microorganisms8050692. Hemiptera
Gil R, Belda E, Gosalbes MJ et al (2008) Massive presence of insertion sequences in the genome of SOPE, the primary endosymbiont of the rice weevil Sitophilus oryzae. Int Microbiol Off J Span Soc Microbiol 11:41–48
Goodhead I, Blow F, Brownridge P et al (2020) Large-scale and significant expression from pseudogenes in Sodalis glossinidius – a facultative bacterial endosymbiont. Microb Genomics 6:e000285. https://doi.org/10.1099/mgen.0.000285
Grossi AA, Tian C, Ren M et al (2024) Co-phylogeny of a hyper-symbiotic system: endosymbiotic bacteria (Gammaproteobacteria), chewing lice (Insecta: Phthiraptera) and birds (Passeriformes). Mol Phylogenet Evol 190:107957. https://doi.org/10.1016/j.ympev.2023.107957
Grünwald S, Pilhofer M, Höll W (2010) Microbial associations in gut systems of wood- and bark-inhabiting longhorned beetles [Coleoptera: Cerambycidae]. Syst Appl Microbiol 33:25–34. https://doi.org/10.1016/j.syapm.2009.10.002
Gruwell ME, Hardy NB, Gullan PJ, Dittmar K (2010) Evolutionary relationships among primary endosymbionts of the Mealybug Subfamily Phenacoccinae (Hemiptera: Coccoidea: Pseudococcidae). Appl Environ Microbiol. https://doi.org/10.1128/AEM.01354-10
Hall RJ, Flanagan LA, Bottery MJ et al (2019) A tale of three species: adaptation of Sodalis glossinidius to Tsetse Biology, Wigglesworthia Metabolism, and Host Diet. https://doi.org/10.1128/mBio.02106-18. mBio
Hall RJ, Thorpe S, Thomas GH, Wood AJ (2020) Simulating the evolutionary trajectories of metabolic pathways for insect symbionts in the genus Sodalis. Microb Genomics 6:mgen000378. https://doi.org/10.1099/mgen.0.000378
Hamidou Soumana I, Berthier D, Tchicaya B et al (2013) Population dynamics of Glossina palpalis gambiensis symbionts, Sodalis glossinidius, and Wigglesworthia glossinidia, throughout host-fly development. Infect Genet Evol 13:41–48. https://doi.org/10.1016/j.meegid.2012.10.003
Hao Z, Kasumba I, Lehane MJ et al (2001) Tsetse immune responses and trypanosome transmission: implications for the development of tsetse-based strategies to reduce Trypanosomiasis. Proc Natl Acad Sci U S A 98:12648–12653. https://doi.org/10.1073/pnas.221363798
Heddi A, Charles H, Khatchadourian C et al (1998) Molecular characterization of the principal symbiotic Bacteria of the Weevil Sitophilus oryzae: a peculiar G + C content of an endocytobiotic DNA. J Mol Evol 47:52–61. https://doi.org/10.1007/PL00006362
Heddi A, Grenier A-M, Khatchadourian C et al (1999) Four intracellular genomes direct weevil biology: Nuclear, mitochondrial, principal endosymbiont, and Wolbachia. Proc Natl Acad Sci 96:6814–6819. https://doi.org/10.1073/pnas.96.12.6814
Heyworth ER, Smee MR, Ferrari J (2020) Aphid Facultative symbionts Aid Recovery of their Obligate Symbiont and their host after heat stress. Front Ecol Evol 8
Hosokawa T, Kaiwa N, Matsuura Y et al (2015) Infection prevalence of Sodalis symbionts among stinkbugs. Zool Lett 1:5. https://doi.org/10.1186/s40851-014-0009-5
Hrusa G, Farmer W, Weiss BL et al (2015) TonB-Dependent Heme Iron Acquisition in the tsetse fly Symbiont Sodalis glossinidius. Appl Environ Microbiol. https://doi.org/10.1128/AEM.04166-14
Husnik F, McCutcheon JP (2016) Repeated replacement of an intrabacterial symbiont in the tripartite nested mealybug symbiosis. Proc Natl Acad Sci 113:E5416–E5424. https://doi.org/10.1073/pnas.1603910113
Jackson R, Monnin D, Patapiou PA et al (2022) Convergent evolution of a labile nutritional symbiosis in ants. ISME J 16:2114–2122. https://doi.org/10.1038/s41396-022-01256-1
Jousselin E, Cœur d’Acier A, Vanlerberghe-Masutti F, Duron O (2013) Evolution and diversity of Arsenophonus endosymbionts in aphids. Mol Ecol 22:260–270. https://doi.org/10.1111/mec.12092
Kaiwa N, Hosokawa T, Kikuchi Y et al (2010) Primary gut symbiont and secondary, Sodalis-Allied Symbiont of the Scutellerid Stinkbug Cantao Ocellatus. Appl Environ Microbiol. https://doi.org/10.1128/AEM.00421-10
Kaiwa N, Hosokawa T, Kikuchi Y et al (2011) Bacterial symbionts of the Giant Jewel Stinkbug Eucorysses Grandis (Hemiptera: Scutelleridae). Zoolog Sci 28:169–174. https://doi.org/10.2108/zsj.28.169
Kaiwa N, Hosokawa T, Nikoh N et al (2014) Symbiont-supplemented maternal investment underpinning host’s ecological adaptation. Curr Biol 24:2465–2470. https://doi.org/10.1016/j.cub.2014.08.065
Kallu SA, Ndebe J, Qiu Y et al (2023) Prevalence and Association of Trypanosomes and Sodalis glossinidius in Tsetse flies from the Kafue National Park in Zambia. Trop Med Infect Dis 8:80. https://doi.org/10.3390/tropicalmed8020080
Kanté Tagueu S, Farikou O, Njiokou F, Simo G (2018) Prevalence of Sodalis glossinidius and different trypanosome species in Glossina palpalis palpalis caught in the Fontem sleeping sickness focus of the southern Cameroon. Parasite Paris Fr 25:44. https://doi.org/10.1051/parasite/2018044
Kanyile SN, Engl T, Heddi A, Kaltenpoth M (2023) Endosymbiosis allows Sitophilus oryzae to persist in dry conditions. Front Microbiol 14
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol 30:772–780. https://doi.org/10.1093/molbev/mst010
Kaur R, Shropshire JD, Cross KL et al (2021) Living in the endosymbiotic world of Wolbachia: a centennial review. Cell Host Microbe 29:879–893. https://doi.org/10.1016/j.chom.2021.03.006
Keller CM, Kendra CG, Bruna RE et al (2021) Genetic modification of Sodalis species by DNA transduction. mSphere 6. https://doi.org/10.1128/msphere.01331-20
Kendra CG, Keller CM, Bruna RE, Pontes MH (2020) Conjugal DNA transfer in Sodalis glossinidius, a maternally inherited Symbiont of Tsetse flies. mSphere 5:e00864–e00820. https://doi.org/10.1128/mSphere.00864-20
Kikuchi Y, Tada A, Musolin DL et al (2016) Collapse of insect gut symbiosis under simulated climate change. mBio 7:e01578–e01516. https://doi.org/10.1128/mBio.01578-16
Koga R, Moran NA (2014) Swapping symbionts in spittlebugs: evolutionary replacement of a reduced genome symbiont. ISME J 8:1237–1246. https://doi.org/10.1038/ismej.2013.235
Koga R, Bennett GM, Cryan JR, Moran NA (2013) Evolutionary replacement of obligate symbionts in an ancient and diverse insect lineage. Environ Microbiol 15:2073–2081. https://doi.org/10.1111/1462-2920.12121
Koga R, Moriyama M, Onodera-Tanifuji N et al (2022) Single mutation makes Escherichia coli an insect mutualist. Nat Microbiol 7:1141–1150. https://doi.org/10.1038/s41564-022-01179-9
Landmann F (2019) The Wolbachia endosymbionts. Microbiol Spectr. https://doi.org/10.1128/microbiolspec.BAI-0018-2019
Lefèvre C, Charles H, Vallier A et al (2004) Endosymbiont phylogenesis in the dryophthoridae weevils: evidence for bacterial replacement. Mol Biol Evol 21:965–973. https://doi.org/10.1093/molbev/msh063
Lindh JM, Lehane MJ (2011) The tsetse fly Glossina fuscipes fuscipes (Diptera: Glossina) harbours a surprising diversity of bacteria other than symbionts. Antonie Van Leeuwenhoek 99:711–720. https://doi.org/10.1007/s10482-010-9546-x
Lo W-S, Huang Y-Y, Kuo C-H (2016) Winding paths to simplicity: genome evolution in facultative insect symbionts. FEMS Microbiol Rev 40:855–874. https://doi.org/10.1093/femsre/fuw028
Login FH, Balmand S, Vallier A et al (2011) Antimicrobial peptides keep insect endosymbionts under control. Science 334:362–365. https://doi.org/10.1126/science.1209728
Maire J, Vincent-Monégat C, Balmand S et al (2019) Weevil pgrp-lb prevents endosymbiont TCT dissemination and chronic host systemic immune activation. Proc Natl Acad Sci U S A 116:5623–5632. https://doi.org/10.1073/pnas.1821806116
Maire J, Parisot N, Galvao Ferrarini M et al (2020) Spatial and morphological reorganization of endosymbiosis during metamorphosis accommodates adult metabolic requirements in a weevil. Proc Natl Acad Sci 117:19347–19358
Maltz MA, Weiss BL, O’Neill M et al (2012) OmpA-mediated biofilm formation is essential for the commensal bacterium Sodalis glossinidius to colonize the tsetse fly gut. Appl Environ Microbiol 78:7760–7768. https://doi.org/10.1128/AEM.01858-12
Malulu DJ, Nyingilili HS, Edward D et al (2023) Interactions among Sodalis, Glossina pallidipes salivary gland hypertrophy virus and trypanosomes in wild Glossina pallidipes. Int J Trop Insect Sci 43:1649–1657. https://doi.org/10.1007/s42690-023-01062-y
Manzano-Marín A, Coeur d’acier A, Clamens A-L et al (2018) A Freeloader? The highly eroded yet large genome of the Serratia symbiotica Symbiont of Cinara Strobi. Genome Biol Evol 10:2178–2189. https://doi.org/10.1093/gbe/evy173
Manzano-Marín A, Coeur d’acier A, Clamens A-L et al (2023) Co-obligate symbioses have repeatedly evolved across aphids, but partner identity and nutritional contributions vary across lineages. Peer Community J 3. https://doi.org/10.24072/pcjournal.278
Manzano-Marı́n A, D’acier AC, Clamens A-L et al (2020) Serial horizontal transfer of vitamin-biosynthetic genes enables the establishment of new nutritional symbionts in aphids’ di-symbiotic systems. ISME J 14:259–273. https://doi.org/10.1038/s41396-019-0533-6
Masson F, Zaidman-Rémy A, Heddi A (2016) Antimicrobial peptides and cell processes tracking endosymbiont dynamics. Philos Trans R Soc B Biol Sci 371:20150298. https://doi.org/10.1098/rstb.2015.0298
Matsuura Y, Hosokawa T, Serracin M et al (2014) Bacterial symbionts of a devastating coffee plant pest, the stinkbug Antestiopsis Thunbergii (Hemiptera: Pentatomidae). Appl Environ Microbiol 80:3769–3775. https://doi.org/10.1128/AEM.00554-14
Matsuura Y, Moriyama M, Łukasik P et al (2018) Recurrent symbiont recruitment from fungal parasites in cicadas. Proc Natl Acad Sci 115:E5970–E5979. https://doi.org/10.1073/pnas.1803245115
Matthew CZ, Darby AC, Young SA et al (2005) The rapid isolation and growth dynamics of the tsetse symbiont Sodalis glossinidius. FEMS Microbiol Lett 248:69–74. https://doi.org/10.1016/j.femsle.2005.05.024
Maudlin I, Welburn SC, Mehlitz D (1990) The relationship between rickettsia-like-organisms and trypanosome Infections in natural populations of tsetse in Liberia. Trop Med Parasitol off Organ Dtsch Tropenmedizinische Ges Dtsch Ges Tech Zusammenarbeit GTZ 41:265–267
McCutcheon JP, Moran NA (2012) Extreme genome reduction in symbiotic bacteria. Nat Rev Microbiol 10:13–26. https://doi.org/10.1038/nrmicro2670
McGowan S, Sebaihia M, Jones S et al (1995) Carbapenem antibiotic production in Erwinia carotovora is regulated by CarR, a homologue of the LuxR transcriptional activator. Microbiology 141:541–550. https://doi.org/10.1099/13500872-141-3-541
Medlock J, Atkins KE, Thomas DN et al (2013) Evaluating paratransgenesis as a potential control strategy for African Trypanosomiasis. PLoS Negl Trop Dis 7:e2374. https://doi.org/10.1371/journal.pntd.0002374
Mfopit YM, Engel JS, Chechet GD et al (2023) Molecular detection of Sodalis glossinidius, Spiroplasma species and Wolbachia endosymbionts in wild population of tsetse flies collected in Cameroon, Chad and Nigeria. BMC Microbiol 23:260. https://doi.org/10.1186/s12866-023-03005-6
Michalkova V, Benoit JB, Weiss BL et al (2014) Vitamin B6 generated by Obligate Symbionts is critical for maintaining Proline Homeostasis and Fecundity in Tsetse flies. Appl Environ Microbiol 80:5844–5853. https://doi.org/10.1128/AEM.01150-14
Moloo SK (1993) The distribution of Glossina species in Africa and their natural hosts. Int J Trop Insect Sci 14:511–527. https://doi.org/10.1017/S1742758400014211
Monnin D, Jackson R, Kiers ET et al (2020) Parallel evolution in the integration of a co-obligate aphid symbiosis. Curr Biol. https://doi.org/10.1016/j.cub.2020.03.011
Moran NA, McCutcheon JP, Nakabachi A (2008) Genomics and evolution of heritable bacterial symbionts. Annu Rev Genet, 42, 165–190. https://doi.org/10.1146/annurev.genet.41.110306.130119
Moran NA, Bennett GM (2014) The tiniest tiny genomes. Annu Rev Microbiol 68:195–215. https://doi.org/10.1146/annurev-micro-091213-112901
Motyka A, Zoledowska S, Sledz W, Lojkowska E (2017) Molecular methods as tools to control plant Diseases caused by Dickeya and Pectobacterium spp: a minireview. New Biotechnol 39:181–189. https://doi.org/10.1016/j.nbt.2017.08.010
Munoz MM, Spencer N, Enomoto S et al (2020) Quorum sensing sets the stage for the establishment and vertical transmission of Sodalis praecaptivus in tsetse flies. PLOS Genet 16:e1008992. https://doi.org/10.1371/journal.pgen.1008992
Niepoth N, Ellers J, Henry LM (2018) Symbiont interactions with non-native hosts limit the formation of new symbioses. BMC Evol Biol 18:27. https://doi.org/10.1186/s12862-018-1143-z
Nováková E, Hypša V (2007) A new Sodalis lineage from bloodsucking fly Craterina melbae (Diptera, Hippoboscoidea) originated independently of the tsetse flies symbiont Sodalis glossinidius. FEMS Microbiol Lett 269:131–135. https://doi.org/10.1111/j.1574-6968.2006.00620.x
Nováková E, Hypša V, Klein J et al (2013) Reconstructing the phylogeny of aphids (Hemiptera: Aphididae) using DNA of the obligate symbiont Buchnera aphidicola. Mol Phylogenet Evol 68:42–54. https://doi.org/10.1016/j.ympev.2013.03.016
Nováková E, Husník F, Šochová E, Hypša V (2015) Arsenophonus and Sodalis symbionts in Louse flies: an analogy to the Wigglesworthia and Sodalis System in Tsetse flies. Appl Environ Microbiol 81:6189–6199. https://doi.org/10.1128/AEM.01487-15
Oakeson KF, Gil R, Clayton AL et al (2014) Genome degeneration and adaptation in a nascent stage of symbiosis. Genome Biol Evol 6:76–93. https://doi.org/10.1093/gbe/evt210
Oliver KM, Degnan PH, Burke GR, Moran NA (2010) Facultative symbionts in Aphids and the horizontal transfer of ecologically important traits. Annu Rev Entomol 55:247–266. https://doi.org/10.1146/annurev-ento-112408-085305
Pais R, Lohs C, Wu Y et al (2008) The Obligate Mutualist Wigglesworthia glossinidia influences Reproduction, digestion, and immunity processes of its host, the tsetse fly. Appl Environ Microbiol. https://doi.org/10.1128/AEM.00741-08
Perreau J, Moran NA (2022) Genetic innovations in animal–microbe symbioses. Nat Rev Genet 23:23–39. https://doi.org/10.1038/s41576-021-00395-z
Plarre R (2013) An attempt to reconstruct the natural and cultural history of the granary weevil, Sitophilus granarius (Coleoptera: Curculionidae). EJE 107:1–11. https://doi.org/10.14411/eje.2010.001
Pons I, Renoz F, Noël C, Hance T (2019) Circulation of the Cultivable Symbiont Serratia symbiotica in Aphids is mediated by plants. Front Microbiol 10
Pons I, González Porras MÁ, Breitenbach N et al (2022a) For the road: calibrated maternal investment in light of extracellular symbiont transmission. Proc R Soc B Biol Sci 289:20220386. https://doi.org/10.1098/rspb.2022.0386
Pons I, Scieur N, Dhondt L et al (2022b) Pervasiveness of the symbiont Serratia symbiotica in the aphid natural environment: distribution, diversity and evolution at a multitrophic level. FEMS Microbiol Ecol 98:fiac012. https://doi.org/10.1093/femsec/fiac012
Pontes MH, Dale C (2006) Culture and manipulation of insect facultative symbionts. Trends Microbiol 14:406–412. https://doi.org/10.1016/j.tim.2006.07.004
Pontes MH, Dale C (2011) Lambda red-mediated genetic modification of the insect endosymbiont Sodalis glossinidius. Appl Environ Microbiol 77:1918–1920. https://doi.org/10.1128/AEM.02166-10
Pontes MH, Babst M, Lochhead R et al (2008) Quorum sensing primes the oxidative stress response in the Insect Endosymbiont, Sodalis glossinidius. PLoS ONE 3:e3541. https://doi.org/10.1371/journal.pone.0003541
Ratcliffe NA, Furtado Pacheco JP, Dyson P et al (2022) Overview of paratransgenesis as a strategy to control pathogen transmission by insect vectors. Parasit Vectors 15:112. https://doi.org/10.1186/s13071-021-05132-3
Ratledge C, Dover LG (2000) Iron Metabolism in pathogenic Bacteria. Annu Rev Microbiol 54:881–941. https://doi.org/10.1146/annurev.micro.54.1.881
Renoz F, Ambroise J, Bearzatto B et al (2022) The Di-Symbiotic systems in the aphids Sipha maydis and periphyllus lyropictus provide a contrasting picture of recent Co-obligate Nutritional endosymbiosis in Aphids. Microorganisms 10:1360. https://doi.org/10.3390/microorganisms10071360
Rio RVM, Lefevre C, Heddi A, Aksoy S (2003) Comparative Genomics of insect-symbiotic Bacteria: influence of host environment on Microbial Genome Composition. Appl Environ Microbiol 69:6825–6832. https://doi.org/10.1128/AEM.69.11.6825-6832.2003
Rio RVM, Wu Y, Filardo G, Aksoy S (2006) Dynamics of multiple symbiont density regulation during host development: tsetse fly and its microbial flora. Proc R Soc B Biol Sci 273:805–814. https://doi.org/10.1098/rspb.2005.3399
Rio RVM, Symula RE, Wang J et al (2012) Insight into the transmission biology and species-specific functional capabilities of tsetse (Diptera: glossinidae) obligate symbiont Wigglesworthia. mBio 3:e00240–e00211. https://doi.org/10.1128/mBio.00240-11
Roma JS, D’Souza S, Somers PJ et al (2019) Thermal stress responses of Sodalis glossinidius, an indigenous bacterial symbiont of hematophagous tsetse flies. PLoS Negl Trop Dis 13:e0007464. https://doi.org/10.1371/journal.pntd.0007464
Rosas-Pérez T, de León AV-P, Rosenblueth M et al (2017) The Symbiome of Llaveia Cochineals (Hemiptera: Coccoidea: Monophlebidae) Includes a Gammaproteobacterial Cosymbiont Sodalis TME1 and the Known Candidatus Walczuchella monophlebidarum. IntechOpen
Rubin BER, Sanders JG, Turner KM et al (2018) Social behaviour in bees influences the abundance of Sodalis (Enterobacteriaceae) symbionts. R Soc Open Sci 5:180369. https://doi.org/10.1098/rsos.180369
Runyen-Janecky LJ, Brown AN, Ott B et al (2010) Regulation of High-Affinity Iron Acquisition homologues in the tsetse fly Symbiont Sodalis glossinidius. J Bacteriol 192:3780–3787. https://doi.org/10.1128/JB.00161-10
Runyen-Janecky LJ, Scheutzow JD, Farsin R et al (2022) Heme-induced genes facilitate endosymbiont (Sodalis glossinidius) colonization of the tsetse fly (Glossina Morsitans) midgut. PLoS Negl Trop Dis 16:e0010833. https://doi.org/10.1371/journal.pntd.0010833
Salem H, Bauer E, Kirsch R et al (2017) Drastic genome reduction in an Herbivore’s Pectinolytic Symbiont. Cell 171:1520–1531e13. https://doi.org/10.1016/j.cell.2017.10.029
Santos-Garcia D, Silva FJ, Morin S et al (2017) The All-Rounder Sodalis: a New Bacteriome-Associated Endosymbiont of the lygaeoid bug Henestaris Halophilus (Heteroptera: Henestarinae) and a critical examination of its evolution. Genome Biol Evol 9:2893–2910. https://doi.org/10.1093/gbe/evx202
Shaida SS, Weber JS, Gbem TT et al (2018) Diversity and phylogenetic relationships of Glossina populations in Nigeria and the Cameroonian border region. BMC Microbiol 18:180. https://doi.org/10.1186/s12866-018-1293-6
Simonet P, Duport G, Gaget K et al (2016) Direct flow cytometry measurements reveal a fine-tuning of symbiotic cell dynamics according to the host developmental needs in aphid symbiosis. Sci Rep 6:1–13. https://doi.org/10.1038/srep19967
Simonet P, Gaget K, Balmand S et al (2018) Bacteriocyte cell death in the pea aphid/Buchnera symbiotic system. Proc Natl Acad Sci 115:E1819–E1828. https://doi.org/10.1073/pnas.1720237115
Sloan DB, Moran NA (2012) Genome reduction and co-evolution between the primary and secondary bacterial symbionts of psyllids. Mol Biol Evol 29:3781–3792. https://doi.org/10.1093/molbev/mss180
Smith CL, Weiss BL, Aksoy S, Runyen-Janecky LJ (2013a) Characterization of the Achromobactin Iron Acquisition Operon in Sodalis glossinidius. Appl Environ Microbiol 79:2872–2881. https://doi.org/10.1128/AEM.03959-12
Smith WA, Oakeson KF, Johnson KP et al (2013b) Phylogenetic analysis of symbionts in feather-feeding lice of the genus Columbicola: evidence for repeated symbiont replacements. BMC Evol Biol 13:109. https://doi.org/10.1186/1471-2148-13-109
Snyder AK, Rio RVM (2015) Wigglesworthia morsitans Folate (vitamin B9) biosynthesis contributes to tsetse host fitness. Appl Environ Microbiol 81:5375–5386. https://doi.org/10.1128/AEM.00553-15
Snyder AK, Deberry JW, Runyen-Janecky L, Rio RVM (2010) Nutrient provisioning facilitates homeostasis between tsetse fly (Diptera: Glossinidae) symbionts. Proc Biol Sci 277:2389–2397. https://doi.org/10.1098/rspb.2010.0364
Snyder AK, McMillen CM, Wallenhorst P, Rio RVM (2011) The phylogeny of Sodalis-like symbionts as reconstructed using surface-encoding loci. FEMS Microbiol Lett 317:143–151. https://doi.org/10.1111/j.1574-6968.2011.02221.x
Šochová E, Husník F, Nováková E et al (2017) Arsenophonus and Sodalis replacements shape evolution of symbiosis in louse flies. PeerJ 5:e4099. https://doi.org/10.7717/peerj.4099
Stoy KS, Gibson AK, Gerardo NM, Morran LT (2020) A need to consider the evolutionary genetics of host–symbiont mutualisms. J Evol Biol 33:1656–1668. https://doi.org/10.1111/jeb.13715
Su Y, Lin H-C, Teh LS et al (2022) Rational engineering of a synthetic insect-bacterial mutualism. Curr Biol 32:3925–3938e6. https://doi.org/10.1016/j.cub.2022.07.036
Sudakaran S, Kost C, Kaltenpoth M (2017) Symbiont Acquisition and replacement as a source of Ecological Innovation. Trends Microbiol 25:375–390. https://doi.org/10.1016/j.tim.2017.02.014
Tláskal V, Pylro VS, Žifčáková L, Baldrian P (2021) Ecological divergence within the Enterobacterial Genus Sodalis: from insect symbionts to inhabitants of decomposing Deadwood. Front Microbiol 12
Toh H, Weiss BL, Perkin SAH et al (2006) Massive genome erosion and functional adaptations provide insights into the symbiotic lifestyle of Sodalis glossinidius in the tsetse host. Genome Res 16:149–156. https://doi.org/10.1101/gr.4106106
Toju H, Fukatsu T (2011) Diversity and Infection prevalence of endosymbionts in natural populations of the chestnut weevil: relevance of local climate and host plants. Mol Ecol 20:853–868. https://doi.org/10.1111/j.1365-294X.2010.04980.x
Toju H, Hosokawa T, Koga R et al (2010) Candidatus Curculioniphilus Buchneri, a novel clade of bacterial endocellular symbionts from weevils of the Genus Curculio. Appl Environ Microbiol 76:275–282. https://doi.org/10.1128/AEM.02154-09
Toju H, Tanabe AS, Notsu Y et al (2013) Diversification of endosymbiosis: replacements, co-speciation and promiscuity of bacteriocyte symbionts in weevils. ISME J 7:1378–1390. https://doi.org/10.1038/ismej.2013.27
Trappeniers K, Matetovici I, Van Den Abbeele J, De Vooght L (2019) The tsetse fly displays an attenuated Immune response to its secondary Symbiont, Sodalis glossinidius. Front Microbiol 10
Tsagmo Ngoune JM, Reveillaud J, Sempere G et al (2019) The composition and abundance of bacterial communities residing in the gut of Glossina palpalis palpalis captured in two sites of southern Cameroon. Parasit Vectors 12:151. https://doi.org/10.1186/s13071-019-3402-2
Vigneron A, Kaltenpoth M (2022) Symbiosis: creating a tractable intracellular insect-microbe association. Curr Biol CB 32:R943–R946. https://doi.org/10.1016/j.cub.2022.08.011
Vigneron A, Masson F, Vallier A et al (2014) Insects recycle endosymbionts when the benefit is over. Curr Biol 24:2267–2273. https://doi.org/10.1016/j.cub.2014.07.065
Visick KL, Foster J, Doino J et al (2000) Vibrio fischeri lux genes play an important role in colonization and development of the host light organ. J Bacteriol 182:4578–4586. https://doi.org/10.1128/jb.182.16.4578-4586.2000
Vooght LD, Ridder KD, Hussain S et al (2022) Targeting the tsetse-trypanosome interplay using genetically engineered Sodalis glossinidius. PLOS Pathog 18:e1010376. https://doi.org/10.1371/journal.ppat.1010376
Wamwiri FN, Alam U, Thande PC et al (2013) Wolbachia, Sodalis and trypanosome co-infections in natural populations of Glossina Austeni and Glossina pallidipes. Parasit Vectors 6:232. https://doi.org/10.1186/1756-3305-6-232
Wang J, Brelsfoard C, Wu Y, Aksoy S (2013) Intercommunity effects on microbiome and GpSGHV density regulation in tsetse flies. J Invertebr Pathol 112:S32–S39. https://doi.org/10.1016/j.jip.2012.03.028
Weeks AR, Turelli M, Harcombe WR et al (2007) From parasite to Mutualist: Rapid Evolution of Wolbachia in Natural populations of Drosophila. PLOS Biol 5:e114. https://doi.org/10.1371/journal.pbio.0050114
Weiss BL, Mouchotte R, Rio RVM et al (2006) Interspecific transfer of bacterial endosymbionts between tsetse fly species: Infection establishment and effect on host fitness. Appl Environ Microbiol 72:7013–7021. https://doi.org/10.1128/AEM.01507-06
Weiss BL, Wu Y, Schwank JJ et al (2008) An insect symbiosis is influenced by bacterium-specific polymorphisms in outer-membrane protein A. Proc Natl Acad Sci 105:15088–15093. https://doi.org/10.1073/pnas.0805666105
Welburn SC, Maudlin I (1999) Tsetse–trypanosome interactions: rites of passage. Parasitol Today 15:399–403. https://doi.org/10.1016/S0169-4758(99)01512-4
Welburn SC, Maudlin I, Ellis DS (1987) In vitro cultivation of rickettsia-like-organisms from Glossina spp. Ann Trop Med Parasitol 81:331–335. https://doi.org/10.1080/00034983.1987.11812127
Welburn SC, Arnold K, Maudlin I, Gooday GW (1993) Rickettsia-like organisms and chitinase production in relation to transmission of trypanosomes by tsetse flies. Parasitology 107(Pt 2):141–145. https://doi.org/10.1017/s003118200006724x
Whittle M, Barreaux AMG, Bonsall MB et al (2021) Insect-host control of obligate, intracellular symbiont density. Proc R Soc B Biol Sci 288:20211993. https://doi.org/10.1098/rspb.2021.1993
Wicker C (1983) Differential vitamin and choline requirements of symbiotic and aposymbiotic s. oryzae (coleoptera: curculionidae). https://doi.org/10.1016/0300-9629(83)90311-0
Wicker C, Nardon P (1982) Development responses of symbiotic and aposymbiotic weevils Sitophilus oryzae L. (Coleoptera, curculionidae) to a diet supplemented with aromatic amino acids. https://doi.org/10.1016/0022-1910(82)90008-7
Zaidman-Rémy A, Vigneron A, Weiss BL, Heddi A (2018) What can a weevil teach a fly, and reciprocally? Interaction of host immune systems with endosymbionts in Glossina and Sitophilus. BMC Microbiol 18:150. https://doi.org/10.1186/s12866-018-1278-5
Zhang W, Wang J, Huang Z et al (2023) Symbionts in Hodgkinia-free cicadas and their implications for co-evolution between endosymbionts and host insects. Appl Environ Microbiol 0:e01373–e01323. https://doi.org/10.1128/aem.01373-23
Zientz E, Dandekar T, Gross R (2004) Metabolic Interdependence of Obligate Intracellular Bacteria and their insect hosts. Microbiol Mol Biol Rev 68:745–770. https://doi.org/10.1128/MMBR.68.4.745-770.2004
Zug R, Hammerstein P (2015) Bad guys turned nice? A critical assessment of Wolbachia mutualisms in arthropod hosts. Biol Rev 90:89–111. https://doi.org/10.1111/brv.12098
Zytynska SE, Tighiouart K, Frago E (2021) Benefits and costs of hosting facultative symbionts in plant-sucking insects: a meta-analysis. Mol Ecol 30:2483–2494. https://doi.org/10.1111/mec.15897
Acknowledgements
This study was financially supported by the National Fund for Scientific Research, FNRS grant no. 1B374.21. François Renoz acknowledges support from the Japan Society for the Promotion of Science (JSPS) - Postdoctoral Fellowships for Research in Japan [Fellowship Number PE22052]. The authors thank Prof. Thierry Hance for his valuable comments. This paper is publication BRC411 of the Biodiversity Research Centre (Université Catholique de Louvain).
Author information
Authors and Affiliations
Contributions
FR and IP wrote the paper. HA performed the phylogenomic analyses and made manuscript revisions. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Renoz, F., Arai, H. & Pons, I. The genus Sodalis as a resource for understanding the multifaceted evolution of bacterial symbiosis in insects. Symbiosis 92, 187–208 (2024). https://doi.org/10.1007/s13199-023-00966-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13199-023-00966-0