Abstract
Exposure to cadmium results in disturbances in cell homeostasis in all living organisms. The first response to stress factors, including cadmium, is activation of signal transduction pathways that mobilize cell defense mechanisms. The aim of this review is a comparison between the signaling network triggered by Cd in plants and animals. Despite differences in the structure and physiology of plant and animal cells, their cadmium signal transduction pathways share many common elements. These elements include signaling molecules such as ROS, Ca2+ and NO, the involvement of phospholipase C, mitogen-activated protein kinase cascades, and activation of transcription factors. Undoubtedly, both animals and plants also possess specific signaling pathways. In case of animals, Wnt/β-catenin, sonic hedgehog and oestorgen signaling are engaged in the transduction of cadmium signal. Plant specific signal transduction pathways include signaling mediated by plant hormones. The role of ethylene and jasmonic, salicylic and abscisic acid in plant response to cadmium is also discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cadmium is a non-essential heavy metal that is toxic to both plants and animals. Exposure to cadmium results in the generation of reactive oxygen species, membrane leakage, protein and DNA damage, perturbation in cell poliferation, and apoptosis (Deckert 2005; Lehotai et al. 2011; Sobkowiak and Deckert 2004; Vestena et al. 2011; Wang et al. 2011). In the case of mammals, cadmium has a carcinogenic effect (ATSDR 2011). The first response of organisms to the presence of cadmium in their environment is activation of the cell signaling network, which leads to changes in the metabolism indispensible for adaptation to unfavorable conditions. Despite the remarkable differences in the structure and physiology of plant and animal cells, their signal transduction pathways share many common elements. A question that arises is whether signal elements respond in a similar way to Cd exposure in organisms belonging to these two distant taxonomic groups. The aim of the present review is a comparison between the plant and animal signal transduction pathways activated by cadmium. The influence of Cd on signaling mediated by reactive oxygen species (ROS), calcium ions (Ca2+) and nitric oxide (NO), phospholipase C (PLC), mitogen-activated protein kinase (MAPK) cascades, as well as the expression of transcription factors will be reviewed. The last two sections will be dedicated to cadmium signal transduction pathways specific to animal and plant organisms.
Cadmium uptake, accumulation and translocation
In the case of humans, cadmium is mainly absorbed through contaminated water, food or cigarette smoke, although other sources of intoxication are also possible (Mortensen et al. 2011; Yang et al. 2011). An interesting case was described recently in Saudi Arabia. A woman suffered from corneal injury after the usage of traditional eyeliner, which in a toxicology analysis exhibited elevated levels of lead and cadmium (Amry et al. 2011). After absorption, cadmium is transported into the liver, where it binds to small proteins such as glutathione or metallothioneins. Such complexes are released into the blood and might be excreted via urine or faeces, or accumulate in several target tissues, such as the kidneys, liver, pancreas, lungs, and bones (Crews et al. 2000; Thévenod 2009).
In plants, the roots are the main site of cadmium absorption and accumulation, although substantial levels of this heavy metal are also transported to the shoots. The root-to-shoot transfer of cadmium is mediated more by active (symplastic) than passive (apoplasitc) mechanisms (Lu et al. 2009; Vestena et al. 2011). Analysis using X-ray absorption spectroscopy showed that in the stems and leaves, Cd is bound to the oxygen and sulfur groups. Therefore, it is probable that during translocation between different plant organs, cadmium ions form complexes with small O- and S- containing molecules, such as organic acids and phytochelatins. It has been shown that treatment with citrate enhances a plant’s ability to transfer Cd from the roots to shoots (Chen et al. 2003). Moreover, over-expression of the plasma membrane H+-ATPase gene (AHA1) in Arabidopsis thaliana resulted in enhancement of the citrate concentration and secretion, together with an increase in the rate of cadmium root-to-shoot translocation (Hou et al. 2011; Rosa et al. 2004). The putative role of phytochelatins in cadmium transport has also been discussed, as these cysteine containing peptides were recently found in the xylem sap of oilseed rape (Brassica napus) subjected to low cadmium doses (Saathoff et al. 2011).
Cadmium, as a non-essential metal, is most likely taken up by cells through existing transport systems for essential bivalent cations. One of the candidates for cadmium transport are calcium channels. Treatment of Madin-Darby canine kidney (MDCK) cells with an activator of calcium channels, maitotoxin, resulted in enhanced Cd absorption (Olivi and Bessler 2000). Accordingly, pretreatment of Wistar rats with the calcium channel inhibitor verapamil caused a decrease in urinary Cd content, alleviation of cadmium-dependent oxidative stress, and kidney damage. However, verapamil had no effect on renal cortex cadmium content (Xu et al. 2010). Application of calcium channel inhibitors also modulated cadmium uptake in plants. Maize coleoptile segments treated with the calcium channel inhibitors verapamil and lantan chloride exhibited lower cadmium accumulation and diminished cadmium toxicity symptoms. Both calcium channel inhibitors also caused a significant decrease in cadmium concentration in Suaeda salsa roots (Kurtyka et al. 2011; Li et al. 2012). It has been suggested that the wheat transmembrane low-affinity-cation transporter (LCT1), which is known to transport Ca2+, is also engaged in Cd influx into plant cells. Expression of LCT1 in transgenic yeast resulted in a higher susceptibility to cadmium stress and higher Cd uptake (Clemens et al. 1998). However, later research showed that tobacco plants over-expressing the wheat LCT1 gene accumulated less cadmium and exhibited a higher tolerance to this heavy metal than control lines. Therefore, it is unlikely that LCT1 is a main site of cadmium uptake in plant cells (Antosiewicz and Henning 2004).
Other candidates for cadmium influx site are ZIP transporters. These ZTR- and IRT-like proteins are responsible for iron, zinc, and manganese uptake. The expression of ZIP8 and ZIP14 in mouse fetal fibroblast cultures resulted in an increase in intracellular cadmium concentration and accelerated cell damage (Dalton et al. 2004; Girijashanker et al. 2008). Moreover, a connection between the induction of ZIP10 expression and Cd accumulation has been demonstrated in zebrafish (Chauchene et al. 2011). In the case of plants, the ZIP transporters IRT1, ZNT1, and ZNT2 are believed to play a role in cadmium uptake. The 35 S-IRT Arabidospis line, which is characterized by over-expression of the IRT1 gene, accumulated more cadmium in conditions of iron deficiency than in wild type plants (Connolly et al. 2002). The involvement of IRT proteins in cadmium transport is also supported by the fact that rice plants over-expressing IRT1 were characterized by higher cadmium content and exhibited higher sensitivity to this heavy metal than the wild type plants (Lee and An 2009). The ZNT1 and ZNT2 genes isolated from the hyperaccumulator Thlaspi japonicum conferred induced cadmium sensitivity in transgenic yeast (Mizuno et al. 2005).
An alternative candidate for cadmium transport are natural resistance-associated macrophage proteins (Nramp). Injection of human Nramp2, described also as DCT1 or DMT1, to Xenopus oocytes resulted in enhanced cadmium transport. Noteworthy, absorption of the heavy metal was strongly dependent on pH (Okubo et al. 2003). Nramp proteins might be also engaged in cadmium uptake in plants. Characterization of rice Nramp-1 transporter revealed that this protein is located in plasma membrane and that its over-expression results in enhanced accumulation of Cd in rice leaves (Takahashi et al. 2011). Nramp transporters are probably also involved in cadmium vacuolar compartmentalization. There are several facts that support this concept. Firstly, it has been observed in Arabidpsis halleri and Thlaspi caerulescens that Nramp3 and Nramp4 proteins are localized in the vacuole membrane. Secondly, the two mentioned cadmium hyperaccumulators, Arabidpsis halleri and Thlaspi caerulescens, exhibit higher expression levels of Nramp transporters when compared to more sensitive plant species. Lastly, the double mutant Arbidopsis thaliana nramp3nramp4 displays a hypersensitivity to zinc and cadmium without a change in the cellular levels of these heavy metals (Chiang et al. 2006; Oomen et al. 2009). Another transporters which might be involved in vacuolar sequestration of cadmium are P-ATPases. Expression of one P1B-ATPase, AtHMA3, which is found in the vacuolar membrane, increased cadmium tolerance in yeast Cd sensitive Δycf1 mutant (Gravot et al. 2004).
The above-mentioned studies show that cadmium ions are most likely “hitch-hikers” which get into the cell via channels destined for other ions. However, it has been shown that the Ganges ecotype of Thlaspi caerulescens possesses a high affinity transporter for cadmium ions. The author raises the question of the evolutionary meaning of possessing transporters specific for non-essential heavy metals (Lombi et al. 2001).
The role of reactive oxygen species in cadmium signal transduction
An increase in the level of reactive oxygen species is one of the most universal responses to cadmium stress (Hsu and Kao 2007a; Kippler et al. 2012; Kopyra and Gwóźdź 2003; Lehotai et al. 2011; Pytharopoulou et al. 2011; Vestena et al. 2011; Wang et al. 2011). There are several mechanisms that lead to Cd-induced accumulation of ROS. It is postulated that cadmium replaces enzyme-bound-metals that display oxidation-reduction activity such as Fe or Cu. The released metals lead to ROS generation through a Fenton reaction (Casalino et al. 1997). It has been also shown that Cd-induced ROS generation is dependent on the activation of NADPH oxidase in both plant and animal cells. This membrane-bound enzyme produces O •2 , which is then rapidly converted into H202 (Chen et al. 2011b; Cho et al. 2012; Chou et al. 2012; Garnier et al. 2006; Rodríguez-Serrano et al. 2006). Another source of ROS are mitochondria alerted by Cd (Garnier et al. 2006). Cadmium might also lead to a decrease in antioxidant enzyme activity and in this way contribute to an increase in ROS cellular levels (Cho and Seo 2005; Gzyl et al. 2009; Ognjanović et al. 2010; Romero-Puertas et al. 2007). Reactive oxygen species interact with various cellular compounds and lead to their damage. The negative impact of ROS on cellular homeostasis includes changes in membrane permeability, the inactivation of enzymes, and an increased rate in DNA mutation. It has been demonstrated in two cultivars of Arabidopsis thaliana differing in their sensitivity to cadmium that oxidative stress is major contributor to the Cd-dependent phytotoxicity (Cho and Seo 2005). Despite their harmful action, ROS might also play a positive role in response to stress conditions. Some reactive oxygen species act as signaling molecules and in this mode mediate activation of defense mechanisms (Ghouleh et al. 2011; Bhattacharjee 2005). Experiments with the use of microarrays revealed that hydrogen peroxide regulates the expression of 193 genes in human lymphoblastoid TK9 cells, 1404 genes in human colon cancer cells, and 680 genes in Arabidposis seedlings (Briedé et al. 2010; Platel et al. 2010; Wang et al. 2006). It has been recently suggested that the signal can be transduced not only by ROS themselves but also by oxidized fragments of proteins damaged by oxidative stress. The derived peptides could act in more a specific way, as they contain information about the organelle subjected to stressful conditions and the type of ROS produced (Mǿller and Sweetlove 2010).
In terms of response to cadmium stress, the role of reactive oxygen species in cellular signaling in the initiation of apoptotic processes has been most commonly examined. The involvement of ROS in apoptosis has been observed in splenocytes, rat testes, astrocytes and proximal tubular cells, human hepatoma, neuroblastoma and retinal pigment epithelial cells, tomato, tobacco, and Arabidopsis suspension culture (Catterjee et al. 2009; Garnier et al. 2006; Oh and Lim 2006; Kalariya et al. 2009; Kim and Soh 2009; De Michele et al. 2009; Wang et al. 2011; Yakimova et al. 2006; Yang et al. 2008). In animal cells, two apoptosis signaling pathways can be distinguished: the first one engages the activation of caspases and the second one is caspase-independent (Lee and Thévenod 2008). There is evidence that reactive oxygen species are implied in both signaling pathways. The blockage of Cd-induced ROS accumulation in human hepatoma cells by pretreatment with N-acetylcysteine (NAC) resulted in the inhibition of apoptosis through reversion of capsase-8, -3 and -9 activation, accompanied by the inhibition of Bid and Bax protein cleavage and hampered Cyt c release (Oh and Lim 2006). On the other hand, in rat testes subjected to cadmium stress, ROS has been shown to be engaged in poly ADP-ribose polymerase-1 (PARP-1) induction, translocation of the apoptosis-inducing factor (AIF) from mitochondria to nucleus, and, in consequence, initiation of the caspase-independent apoptotic pathway (Kim and Soh 2009). Activation of caspase independent apoptotic processes in response to cadmium exposure has been also demonstrated in rat astrocytes. This process has been mediated by an increase in intracellular calcium levels, which in turn lead to ROS generation (Yang et al. 2008). It seems that proteins which resemble animal caspases also play a role in Cd- induced apoptosis in plants. Treatment of a tomato suspension culture subjected to cadmium stress with human caspase inhibitors caused a significant reduction in the cell death rate. The same experiments showed that the apoptotic signaling activated by CdSO4 in a tomato suspension engages reactive oxygen species, calcium ions, calmodulin, phospholipid signaling, protein kinases, and one of the plant hormones—ethylene (Yakimova et al. 2006). Similar results were obtained in a tobacco suspension culture. Treatment with CdCl2 caused an increase in ROS production, which led to an increased rate of cell death. The generation of reactive oxygen species was dependent on phospholipase C and calmodulin activity (Garnier et al. 2006).
Another cadmium-induced process that is dependent on ROS accumulation is autophagy. This type of programmed cell death consists in the self-digestion of cells. It is still under discussion if autophagy is a cell defense strategy, mode of suicide, or both (Edinger and Thomson 2004). There is evidence that cadmium causes autophagy in both animal and plant cells, and that the process in both cases is mediated by reactive oxygen species (Son et al. 2011; Yang et al. 2009; Zhang and Chen 2010). In mesangial cells, cadmium induces the generation of reactive oxygen species, which in turn activate glycogen synthase kinase-3β (GSK-3β) responsible for the initiation of autophagy (Yang et al. 2009). The sequence of events which lead to mouse epidermal skin cells autophagy consists of ROS generation, phosphorylation of LKB1 and AMPKα proteins, and the formation of LC3-II (Son et al. 2011).
Reactive oxygen species are involved in Cd-induced tumorigenesis. It has been reported that hydrogen peroxide and superoxide anion are required for increased expression of c-fos, c-jun and c-myc proto-oncogenes in BALB/c-3 T3 cells (Joseph et al. 2001). Reactive oxygen species are also engaged in tumor angiogenesis—a critical process for tumor initiation and growth (Jing et al. 2012).
There is crosstalk between ROS signaling and other signal transduction pathways. It has been shown that ROS generation is dependent on Ca2+ signaling in both animal and plant cells (Yang et al. 2008; Rodríguez-Serrano et al. 2006). In rice leaves, Cd-dependent H2O2 accumulation is mediated also by phosphatidylinositol 3-phosphate and nitric oxide (Hsu and Kao 2007b). Reactive oxygen species, on the other hand, work upstream from other signaling elements such as kinases. Experiments conducted with the use of an ROS scavenger have shown that reactive oxygen species activate or/and induce expression of extracellular signal-regulated kinase (ERK), LKB1-AMP-activated kinase (AMPK) and c-Jun N-terminal kinase (JNK) in mammalian cells as well as MPK3 and MPK6 in Arabidopsis cells (Jing et al. 2012; Kalariya et al. 2009; Kim et al. 2005; Liu et al. 2010; Son et al. 2011).
Calcium signaling in response to cadmium
Calcium ions are one of the most important and conserved signal transduction elements found in all living organisms. The mode of action consists in the regulation of protein activity via modifications of its charge and conformation. There is a vast number of calcium sensor proteins, such as highly conserved calmodulin (CAM), plant calmodulin-like proteins (CLM), calcineurin B-like proteins (Hashimoto and Kudla 2011), and Ca2+ dependent protein kinases or neuronal Ca2+ sensors (NCS) specific to animal cells (Clapham 2007). After binding to Ca2+, these calcium sensors are able to interact with other cellular components and modify their structure or/and activity. The targets for calmodulin include kinases, ions transporters, G-proteins, cytoskeleton compounds, and transcription factors. The calcium signal is strictly dependent on the time and spatial course of Ca2+ accumulation. To control calcium action, cells have developed an extended system of Ca2+ compartmentalization, chelation, and excretion (Clapham 2007; Hashimoto and Kudla 2011; Snedden and Fromm 1998).
There are several difficulties in conducting research concerning the involvement of calcium ions in response to cadmium stress. The molecules possess the same charge, and the ion radius Cd2+ and Ca2+ are very similar. Therefore, both Cd2+ and Cd2+ tend to bind to similar proteins, which makes it difficult to find a fluorescent probe specific only for calcium ions. Experiments with the use of calcium channel inhibitors are also complicated because, as has been described in previous sections, cadmium is taken up by cells through the calcium transport systems.
In spite of the previously-mentioned difficulties, there is convincing evidence that cadmium causes an increase in cytosolic calcium levels in various animal cell types (Faurskov and Bjerregaard 2002; Misra et al. 2002; Shankar et al. 1992; Smith et al. 1989; Yamagami et al. 1998; Yang et al. 2008; Ye et al. 2007). The observed Cd-induced intracellular calcium release depends on phospholipase C (PLC) activation and an increase in cellular inositol triphosphate concentration. Interestingly, cadmium ions had no effect on the Ca2+ intracellular levels in human epidermoid carcinoma, rat embryo fibroblasts, and rat aortic smooth muscle cells. These results suggest that the phenomena of calcium mobilization by cadmium ions is dependent on the organism and type of cell (Smith et al. 1989). An increase in intracellular calcium levels is essential for various Cd-induced processes, such as cell transformation, tumorigenesis, and apoptosis. It has been shown in mouse tumor-derived cell lines that accumulation of Ca2+, together with ROS generation, is necessary for activation of c-fos, c-jun and c-myc proto-oncognes (Joseph et al. 2001). Intracellular calcium influx is also engaged in Cd-induced apoptosis processes (Chen et al. 2011a; Wang et al. 2008; Ye et al. 2007). In coronary neurons, the Cd-dependent apoptosis signaling network includes an increase in Ca2+ influx, which leads to the stimulation of CaMPKII. CaMPKII in turn activates subsequent signaling elements—mitogen-activated kinases (MAPKs) and mTOR pathway (Chen et al. 2011a).
The changes in cytosolic calcium levels in response to cadmium treatment in plants seem to depend on the plant species and organ. An increase in cytosolic Ca2+ concentration has been reported in tobacco cells and rice roots, while a significant decrease has been shown in Arabidopsis thaliana root hairs (Fan et al. 2011; Garnier et al. 2006; Yeh et al. 2007). As has been described in previous sections, calcium signaling plays a role in Cd-induced apoptosis in tobacco and in an Arabidposis suspension culture (Garnier et al. 2006; Yakimova et al. 2006). The involvement of calcium ions in plant response to cadmium can be also presumed on the basis of the fact that CdCl2 caused an increase in the expression of genes coding calmodulin-like protein in black night shade and of the calcium-binding protein HvC2d1 in barley plants (Oulhajd et al. 2006; Xu et al. 2009).
Nitric oxide signaling and cadmium stress
Since the discovery that nitric oxide plays an important role in the regulation of the cardiovascular system in the late 1980s, this molecule has had an astonishing career in the biological sciences. Its popularity is reflected by the fact that in 1992 NO was chosen as the Molecule of the Year by the leading scientific journal Science (Hasanuzzaman et al. 2010). Nitric oxide is a signaling molecule that can act directly through regulation of protein activity by S-nitrosylation and nitrotyrosylation, or indirectly by modification of other signaling pathways, such as protein kinases or calcium signaling (Krasylenko et al. 2010). Among the processes which are controlled by nitric oxide signaling in animals, cell proliferation, embryogenesis, cardiovascular tension maintenance, neurotransmission, immune defense, apoptosis, and the regeneration of lower metazoans can be listed (Colasanti et al. 2010; Moncada and Higgs 2006). In plants, NO also regulates a vast number of processes, such as seed germination, plant growth, tissue differentiation, flowering, seed maturation, senesces, the initiation of programmed cell death, as well as responses to biotic and abiotic stress factors (Hasanuzzaman et al. 2010; Krasylenko et al. 2010). There are some discrepancies concerning the pattern of NO accumulation in response to Cd. It has been suggested that short-time cadmium treatment causes the induction of NO production, while longer treatment periods lead to a decrease in the level of nitric oxide. Such NO production kinetics have been observed in mouse peritoneal macrophages characterized by an increase in NO concentration 6, 18 and 24 h after cadmium treatment and depletion in prolonged treatment (Ramirez et al. 1999). A similar effect has been observed in two independent experiments performed on pea roots. During short-term exposure to Cd ions (24 and 48 h of treatment), an enhancement in NO production was observed, while in plants treated for 14 days, the levels of nitric oxide where lower than in the control (Lehotai et al. 2011; Rodríguez-Serrano et al. 2006). However, there is evidence that NO production can be inhibited even shortly after exposure to cadmium, and, conversely, elevated nitric oxide amounts were observed in organisms treated with Cd for relatively long time periods (Groppa et al. 2008; Mahmood et al. 2009; Xiong et al. 2009). Thus, it seems that the pattern of nitric oxide accumulation in reaction to cadmium depends on the organism, cell type, metal concentration, and treatment duration (for reference see Table 1). There are also conflicting data concerning the involvement of nitric oxide in plant defense against cadmium stress. On the one hand, there is evidence that NO can diminish Cd-induced oxidative stress by stimulating the antioxidant system. This molecule has been also shown to promote hemicelluloses and pectin synthesis, which may facilitate cadmium immobilization (Arasimowicz-Jelonek et al. 2011). On the other hand, nitric oxide amplifies cadmium uptake in Arabidopsis and tobacco BY-2 cells (Besson-Bard et al. 2009; Ma et al. 2010). Moreover, nitric oxide causes the nitrosilation of phytochelatins and participates in the initiation of apoptosis (Arasimowicz-Jelonek et al. 2011). It is still under discussion whether these effects promote cadmium tolerance or, conversely, lead to the augmentation of Cd toxicity.
Animal cells are also affected by changes in NO concentration. A lowering of NO concentration in response to cadmium might lead to disturbances in various processes, such as angiogenesis and immune defense (Kolluru et al. 2006; Thévenod 2009). Nitric oxide might also play a protective role during cadmium stress. The NO-releasing prodrug V-PYRRO/NO alleviates Cd cytotoxicity in rat liver cells, most probably through the induction of metallothioneine (MT) synthesis. The chemical agent was also shown to inhibit c-Jun N-terminal kinase (JNK1/2). As the activation of JNK1/2 is connected with induction of apoptosis, NO released from V-PYRRO/NO might arrest the Cd-induced cell death in rat liver (Qu et al. 2005).
The role of phospholipase C in response to cadmium action
Phospholipase C is a membrane-bound enzyme that catalyses the hydrolysis of phosphoinositol 4,5-biphosphate (PIP2). In the reaction, two second messengers, diacyloglycerol (DAG) and inositol 1,4,5 triphosphate (IP3), are formed. An accumulation of IP3 leads to the release of Ca2+ from intracellular stores, while DAG functions as an activator for protein kinase C (PKC). There are several isoformes of PLC, among which the δ isoenyzme is present in fungi, plants and animals (Ochocka and Pawelczyk 2003).
As has been described in the section concerning calcium signaling, the results of several experiments imply that in animal cells, cadmium stimulates phospholipase C, which leads to an increase in IP3 levels and the release of Ca2+ (Faurskov and Bjerregaard 2002; Misra et al. 2002; Smith et al. 1989; Yamagami et al. 1998). The activation of phospholipase C might also lead to the accumulation of DAG and the induction of protein kinase C activity. In fact, an increase in PKC activity in response to cadmium treatment has been reported in the freshwater crab Sinoptoman yangtsekiense (Li et al. 2011). Studies with the use of protein kinase C inhibitors showed that PKC plays a significant role in various processes induced by cadmium action. These processes include accumulation of the transcription factor Nrf2 in the nucleus of astorcytoma cells, perturbations in calcium homeostasis in osteasarcoma cells, actin gluthionylation, and protein carbonylation in the haemocytes of the mussel Mytilus galloprovincialis (Dailianis et al. 2009; Lawal and Ellis 2011; Long 1997).
Phospholipase C might perform protective functions under cadmium stress, as a transgenic fibroblast that overexpressed the PLCβ-1 gene exhibited greater viability under cadmium treatment when compared to control cells (Lee et al. 2000). Moreover, PLC is engaged in the stimulation of plants under low cadmium doses. Barley seedlings treated with Cd in a concentration of 5 × 10−8 M exhibited higher levels of chlorophyll and cytokinins. The application of various inhibitors showed that this effect was mediated by DAG, PKC, and MAPK (Kovács et al. 2009). In a tomato suspension culture, on the other hand, PLC has been shown to mediate Cd-induced cell death. Application of the PLC inhibitor neomycin significantly decreased the number of dead cells (Yakimova et al. 2006).
MAPK cascades and Cd2+
Mitogen-activated protein kinases (MAPKs) form a conserved signal transduction mechanism that can be found in all eukaryotic organisms. The MAPK cascade consists of at least three elements: MAPKKK, MAPKK, and MAPK. The activation of MAPKKK leads to the phosphorylation of MAPKK, which in turn phosphorylates MAPK. MAPKs have the ability to phosphotylate, and in this way regulate the activity of various cellular components, such as other protein kinases, proteins associated with cytoskeleton, and transcription factors. In the Arabidopsis genome, 60 MAPKKKs, 10 MAPKKs, and 20 MAPKs have been found. Taking into account the fact that MAPKKKs can interact with various MAPKKs, and different MAPKKs can activate various MAPKs, the mitogen-activated protein kinase cascades form an extensive and complicated signaling network. MAPK cascades are engaged in the regulation of various processes, including the response to stress factors (Chang and Karin 2001; Nakagami et al. 2005).
In mammalian cells, MAPKKs (also called MEKs or MKKs) work upstream from MAPKs, which include extracellular signal-related kinase (ERK), Jun amino-terminal kinase (JNK), and p38 proteins (Chang and Karin 2001). The majority of reports state that Cd activates all of the above-mentioned MAPKs (Chen et al. 2008; Jing et al. 2012; Jung et al. 2008; Kalariya et al. 2009; Kim et al. 2005; Park et al. 2009; Valbonesi et al. 2008). However, in experiments performed on human embryonic kidney cells, the activity of JNK was stimulated only at higher cadmium concentrations (50 μM), while treatment with low Cd doses (0.5 μM) caused a decrease in its activity (Hao et al. 2009). Moreover, in microcultures of limb bud cells isolated from mouse embryos, cadmium caused phosphorylation of JNK, while the level of phosphorylated ERK and p38 remained unchanged (Liu and Kapron 2010). There is extensive evidence that activation of MAPKs in response to cadmium is mediated by reactive oxygen species. The application of ROS scavengers to human retinal pigment epithelial cells treated with Cd reversed the Cd-induced phosphorylation of JNK, ERK1/2 and p38 (Kalariya et al. 2009). A connection between ROS generation and the activation of JNK and/or ERK has been also observed in mouse macropaghes and in human neuronal and lung epithelial cells exposed to Cd (Chen et al. 2008; Jing et al. 2012; Kim et al. 2005). Moreover, both H2O2 and Cd caused a similar pattern of JNK, ERK, and p38 activation in extravillous trophoblast cells (Valbonesi et al. 2008).
The nomenclature of plant mitogen-activated protein kinase network might be confusing, although some attempts to clarify the system has been made. Generally, it has been proposed to adopt name MPKs for MAPKs and MKKs for MAPKKs. Plant MPKs include tobacco SIPK, WIPK and NTF, alfalfa SAMK and SIMK and rice BWMK kinases. Some examples of plant MKKs are tobacco MEK and SIPK and alfalfa PRKK and SIMKK (Ichimura and MAPK Group 2002). It has been shown that various components of plant MAPK cascades are stimulated by cadmium. Exposure to Cd resulted in the activation of MPK3 and MPK6 in Arabidposis as well as of SIMK, MMK2, MMK3, and SAMK in alfalfa (Jonak et al. 2004; Liu et al. 2010). Moreover, rice plants subjected to cadmium stress exhibited elevated expression of the OsBWMK1, OsMSRMK2, and OsEDR1 genes (Agrawal et al. 2002, 2003; Kim et al. 2003). The response of MAPK cascades to cadmium action is very rapid, as the induction of gene coding MAPKKK has been observed as early as 15 min after the application of Cd, while the enhanced phosphorylation of MAPKs has been noted even after 10 min of cadmium treatment (Jonak et al. 2004; Kim et al. 2003). It is probable that, just like in animals, the activation of MAPKs in plants is mediated by reactive oxygen species. Pre-treatment of Arabidopsis plants with the ROS scavenger, gluthatione, resulted in the attenuation of Cd-induced MPK3 and MPK6 phosphorylation (Liu et al. 2010). Similar results were obtained in experiments performed on rice. Cadmium increased the activity of 40 kDa and 42 kDa MAPKs. This effect was reversed by pre-treatment with a ROS scavenger, as well as with an NADPH oxidase inhibitor, suggesting that the activation of MAPK was dependent on ROS generated by this membrane-bound enzyme. Stimulation of MAPKS was also connected with calcium dependent protein-kinases (CDPK), phosphoinositol 3-kinase (PI3), and the functional state of mitochondria (Yeh et al. 2007).
Cadmium-induced transcription factors
The activation of signaling pathways leads to changes in the activity of transcription factors (TFs), which in turn regulate the expression of various genes. It has been calculated that animals possess approximately 860 different transcription factors per species which form 4.65 % of the genomic content. In the case of plants, these numbers come to 590 and 2.12 % respectively. Transcription factors are classified into several families, frequently named after their DNA binding domains (Charoensawan et al. 2010).
In animal cells, several genes induced by heavy metals possess specific sequences in their promoter, called metal responsive elements (MRE). These sequences are recognized by the MTF-1 transcription factor (metal-regulatory or MRE-binding transcription factor). The DNA-binding ability of MTF-1 is dependent on the concentration of zinc ions. It has been suggested that other heavy metals, including cadmium, regulate MTF-1 activity through the release of zinc ions from intracellular stores (Lichtlen and Schaffner 2001). Another hypothesis states that several factors, such as zinc, cadmium, hydrogen peroxide, or heat shock, mediate the transfer of MTF-1 from cytoplasm to nucleus (Lichtlen and Schaffner 2001). Indeed, treatment of mouse hepatoma cells with cadmium increased the amount of MTF-1 in the nucleus and decreased its concentration in cytoplasm (Smirnova et al. 2000). A similar phenomenon has been observed in the case of another transcription factor, Nrf2. Treatment of rat liver and mouse hepatoma cells with CdCl2 resulted in an increase in Nrf2 levels. The induced TF was found mainly in the nucleus (Casalino et al. 2007; Stewart et al. 2003). Accumulation of Nrf2 under Cd treatment is most probably dependent on the ability of cadmium to stabilize this protein and prolong its half-life (Stewart et al. 2003). Nrf2 plays a role in Cd-dependent stimulation of oxygenase-1 and cytochrome p4502A5 (Abu-Bakar et al. 2004; Alam et al. 2000).
A sequence similar to animal MRE, called PvSr2, has been identified in beans. Transgenic tobacco plants into which the reporter GUS transgene was introduced under the control of the PvSR2 promoter, exhibited an elevated expression of the GUS gene in response to Cu2+, Zn2+, Hg2+, and Cd2+ treatments (Qi et al. 2007). However, to our knowledge no transcription factor associated with the above-mentioned MRE sequence has been identified so far. It is, however, known that in plants, TFs belonging to the AP2/EREBP, MYB, WRKY, and bZIP families are involved in the response to cadmium action (DalCorso et al. 2010). BrCdR15 isolated from Brassica juncea is an example of a transcriptional factor belonging to the bZIP family that is induced by cadmium. Tobacco and Arabidposis plants that overexpressed BjCdR15 exhibited a higher tolerance to Cd and a higher Cd content in their shoots. The fact that this TF regulates the synthesis of phytochelatines and metal transporters suggests that BjCdR15 confers cadmium tolerance through regulation of cadmium uptake and root-to-shoot translocation (Farinati et al. 2010). Comprehensive analysis of the expression profiles of 163 transcription factors belonging to the MYB family in Arabidposis thaliana showed that approximately 20 % of them respond to cadmium treatment (Yanhui et al. 2006). Another source of information about transcription factors engaged in transduction of the cadmium signal are global analyses of gene expression. Analysis of the expression profiles of genes regulated by Cd showed that this metal induces genes which code OsDREB1A, OsDREB1B, and WKRY09 in rice. Another experiment showed that in Arabidopsis thaliana genes which code transcription factors containing ATAF, DREB2A, bZIP and WRKY motifs were induced by treatment with cadmium ions. At the same time, several transcription factors belonging to the AP2/ERF superfamily were down-regulated by Cd in Solanum turvom (Ogawa et al. 2009; Suzuki et al. 2001; Yamaguchi et al. 2009)
Animal specific signaling
One of the characteristic effects of cadmium action, that is specific to animal organisms, is induction of cell proliferation which leads formation of cancer tumors (Deckert 2005). It has been suggested that the carcinogenic effect of Cd is connected with its ability to mimick Wnt/β-catenin signaling. In mammalian cells β-catenin plays dual role—it performs structural functions by bridging E-cadherin to α-catenin in adherens junctions and acts as signaling molecule that can regulate expression of various genes including proto-oncogenes. The Wnt/β-catenin signaling includes several steps: binding of Wnt to receptor, inactivation of β-catenin “destruction” complex, increase in cytoplasmic and nuclear β-catenin levels and finally interaction of β-catenin with T-cell specific factors/lymphoid enhancer binding factor (TCF/LEF), which leads to the activation of target genes (Berthon et al. 2012; Thévenod 2009; Weisberg et al. 2003). Several studies show that cadmium causes loss of adherens junctions integrity which leads to the translocation of β-catenin from membrane to cytoplasm and nuclei (Chakraborty et al. 2010; Prozialeck et al. 2003; Thompson et al. 2008). Translocation of β-catenin to nucleus results in formation of β-catenin/TCF4 complexes. These complexes are engaged in activation of Wnt signaling target genes, c-myc, cyclin D1 and Abcd1a (Chakraborty et al. 2010). In contrast to the described studies, experiments performed on mice showed that prenatal cadmium treatment caused down-regulation of Wnt/β-catenin signaling pathways in thymus of the offspring. This effect was exhibited by the decrease in Wnt10b gene expression, increase in cytoplasmic phospho-β-catenin, predestined for degradation, and decrease in active β-catenin in nucleus (Hanson et al. 2010).
Sonic hedgehog (shh) signaling is an example of another specific animal signaling pathways alerted by cadmium. The Shh receptors consist of two transmembrane proteins: patched (ptc) and smoothened (smo). Binding of Shh protein to the receptor results in abolishment of ptc inhibitory effect on smo. Activated smo transduces the Shh signal to other signaling elements such as Gil proteins (Benson et al. 2004). Two independent experiments showed that prenatal cadmium treatment causes decrease in Shh signaling in mice fetuses and thymus of mice offspring. Interestingly both studies imply that, despite the observed decrease in Shh signaling, Shh and Gil protein levels were unchanged. It is suggested that the decrease in Shh signaling pathway is responsible for developmental anomalies such as postaxial ectrodactyly and abnormal thymocyte development (Hanson et al. 2010; Scott et al. 2005).
The influence of cadmium on oestrogen signaling has been recently profoundly reviewed in Silva et al. 2012. Generally, it has been demonstrated in several studies performed on human breast cancer cell lines, that cadmium is able to bind to membrane and nuclear oestrogen receptors (ER) and activate oestrogen response. This response includes activation of AKT and ERK1/2, induction of c-myc, c-jun and c-fos proto-oncogenes and stimulation of cell proliferation (Silva et al. 2012).
Specific plant response
Plants also possess regulatory mechanisms that are specific to this taxonomic group. These mechanisms include signaling mediated by plant hormones (McSteen and Zhao 2008). Ethylene is frequently called a plant stress hormone, as an increase in its levels has been observed under various unfavorable conditions, including cadmium stress (Arteca and Arteca 2007; Rodríguez-Serrano et al. 2006). Masood et al. 2012 showed that an increase in ethylene concentration in mustard plants subjected to Cd is correlated with an increase in 1-aminocyclopropane-1-carboxylic acid synthase activity (ACS). ACS is a key enzyme in the ethylene synthesis pathway (Masood et al. 2012). Several functions in plant response to cadmium stress are ascribed to ethylene action. This hormone mediates Cd-induced accumulation of H2O2. Bean and onion plants treated with CdCl2 and STS, an inhibitor of ethylene perception, showed a significant decrease in H2O2 levels when compared with plants treated only with CdCl2 (Maksymiec 2011). A tomato mutant with the antisense ACS gene also exhibited a reduction in ethylene synthesis accompanied by reduced hydrogen peroxide levels under cadmium stress (Liu et al. 2008). Ethylene is engaged in the induction of apoptosis. Application of the ethylene synthesis inhibitor AVG to tomato suspension cultures subjected to CdSO4 resulted in a decreased number of dead cells (Yakimova et al. 2006). This plant hormone also has a protective effect against cadmium toxicity on photosynthesis. Treatment of mustard plants with the ethylene donor, ethephon, alleviated the inhibitory effect of cadmium on the activity of Rubisco—a key enzyme in the photosynthesis process (Masood et al. 2012).
Among plant hormones, in addition to ethylene, salicylic (SA), jasmonic (JA), and absicis acid (ABA) might play a role in cadmium signal transduction. Accumulation of salicylic acid under cadmium stress has been noted in pea, maize and Arabidopsis plants (Krantev et al. 2008; Rodríguez-Serrano et al. 2006; Zawoznik et al. 2007). The majority of reports state that SA protects plants against the toxic effects of cadmium. Exogenous application of SA caused alleviation of Cd-induced oxidative stress, which was most probably connected with stimulation of the antioxidant system (Krantev et al. 2008; Panda and Patra 2007; Zhang et al. 2011). It is possible that salicylic acid also activates other ROS scavenging mechanisms, as in mustard a SA-dependent decrease in Cd-induced H2O2 levels was accompanied by inhibition of the activity of antioxidant enzymes (Ahmed et al. 2011). Interestingly, experiments performed on rice imply that the protective role of SA is dependent on SA-induced generation of hydrogen peroxide. Therefore, SA might play a biphasic role in ROS metabolism. On one hand it mediates H2O2 accumulation through stimulation of NADPH oxidase activity, on the other hand the SA-dependent H2O2 production is essential for activation of antioxidant enzymes (Chao et al. 2010). The role of SA in ROS generation is also supported by the fact that SA-deficient Arabidopsis mutants exhibited decreased levels of H2O2 and diminished lipid peroxidation in response to CdCl2 when compared with the wild type plants. Moreover, transgenic plants showed, in contrast to the wild type, induction of guaiacol proxidase and catalase, and a slighter reduction in other antioxidant enzyme activity under cadmium treatment (Zawoznik et al. 2007). Salicylic acid seems to play a protective role in photosynthesis. Pea, maize, and wheat plants pretreated with SA and subjected to Cd stress showed a diminished reduction in chlorophyll content and/or photosynthetic enzymes activity (Krantev et al. 2008; Moussa and El-Gamal 2010a; Popova et al. 2009). However, in castor beans exposed to Cd, pretreatment with SA aggravated the toxic effect of this heavy metal on growth and photosynthetic parameters (Liu et al. 2011). These reports suggest that the mode of SA action depends on the concentration of salicylic acid and the plant’s susceptibility to this hormone.
Elevated levels of jasmonic acid has been observed in pea, runner bean, and Arabidopsis plants treated with cadmium (Maksymiec et al. 2005; Rodríguez-Serrano et al. 2006). It is suggested that JA has a protective effect against Cd action at lower concentrations (Maksymie and Krupa 2002; Noriega et al. 2012). However, at higher concentrations, it may induce changes usually observed under heavy metal stress, such as growth reduction, chlorophyll degradation, and inhibition of various photosynthetic parameters (Maksymie and Krupa 2002). Jasmonic acid might also interact with ROS signaling—it has been shown to mediate the generation of reactive oxygen species in Arabidopsis plants treated with CdSO4 (Maksymiec and Krupa 2006).
The response of abscisic acid to cadmium is not so obvious. A decrease in ABA content has been observed in wheat plants treated with 400 and 1000 μM of Cd, while an increase in the hormone’s content has been reported in two rice cultivars subjected to 500 μM Cd. Interestingly, ABA induction was greater in Cd-tolerant rice cultivar (Hsu and Kao 2003; Moussa and El-Gamal 2010b). Exogenous addition of ABA resulted in enhanced tolerance to cadmium stress and a decrease in uptake of this heavy metal (Hsu and Kao 2003). Another piece of evidence for the protective role of abscisic acid against cadmium stress has been provided by a comparison of the reaction to CdSO4 of wild type Arabidopsis plants and ABA-deficient plants. The experiments showed that the mutants were more sensitive to Cd (Sharma and Kumar 2002). These facts suggest that ABA might be involved in signaling events that lead to a decrease in Cd accumulation or in the activation of other defense mechanisms against heavy metal stress. However, the exact role of ABA in cadmium signal transduction is still unknown.
Conclusions
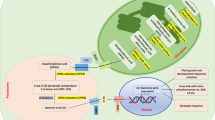
On the basis of the facts described above, it can be concluded that animal and plant cadmium signal transduction networks share many resemblances. Therefore, it is probable that at least some elements involved in cadmium sensing, are evolutionary conserved. These elements include mechanism of cadmium uptake, engagement of phospholipases and MAP kinases and involvement of ROS, Ca2+ and NO signaling molecules. It is also probable that both animals and plants posses in their genome metal responsive elements, that are engaged in regulation of expression of Cd-responsive genes. Except the common response, animals and plants posses also specific signaling pathways activated by cadmium. In case of animals Wnt/β-catenin, sonic hedgehog and oestrogen signaling can be enumerated. In plants, hormones such as ethylene and jasmonic, salicylic and abscisic acid are engaged in the transduction of cadmium signal. It can be therefore concluded, that cadmium influences most of the main animal and plant signaling pathways, the common as well as the specific ones.
Abbreviations
- ABA:
-
Abscisic acid
- AIF:
-
Apoptosis-inducing factor
- ACS:
-
1-aminocyclopropane-1-carboxylic acid synthase
- AMPK:
-
LKB1-AMP-activated kinase
- AVG:
-
Aminoethoxyvinylglycine
- CAM:
-
Calmodulin
- CBL:
-
Calcineurin B-like protein
- CDPK:
-
Calcium dependent protein-kinase
- CLM:
-
Calmodulin-like protein
- DAG:
-
Diacyloglycerol
- ERK:
-
Extracellular signal-regulated kinase
- GSK-3β:
-
Glycogen synthase kinase-3β
- IP3 :
-
Inositol 1,4,5 triphosphate
- JA:
-
Jasmonic acid
- JNK:
-
c-Jun N-terminal kinase
- LCT1:
-
Low-affinity-cation transporter
- LEF:
-
Lymphoid enhancer binding factor
- MAPK:
-
Mitogen-activated protein kinase
- MAPKK:
-
Mitogen-activated protein kinase kinase
- MAPKKK:
-
Mitogen-activated protein kinase kinase kinase
- MDCK:
-
Madin-Darby canine kidney
- MRE:
-
Metal responsive element
- MT:
-
Methallothioneine
- MTF:
-
Metal-regulatory transcription factor, MRE-binding factor
- NAC:
-
N-acetylcysteine
- NCS:
-
Neuronal calcium sensor
- NO:
-
Nitric oxide
- Nramp:
-
Resistance-associated macrophage proteins
- ROS:
-
Reactive oxygen species
- PAPR-1:
-
Poly ADP-ribose polymerase-1
- PI3 :
-
Phosphoinositol 3-kinase
- PIP3 :
-
Phosphoinositol 4,5 biphosphate
- PLC:
-
Phospholipase C
- PKC:
-
Protein kinase C
- SA:
-
Salicylic acid
- Shh:
-
Sonic hedgehog
- STS:
-
Silver thiosulphate
- TCF:
-
T-cell specific factor
- TF:
-
Transcription factor
References
Abu-Bakar A, Satarug S, Marks GC, Lang MA, Moore MR (2004) Acute cadmium chloride administration induces hepatic renal CYP2A5 mRNA, protein and activity in mouse: involvement of transcription factor Nrf2. Toxicol Lett 148:199–210
Agrawal GK, Rakwal R, Iwahashi H (2002) Isolation of novel rice (Oryza sativa L.) multiple stress responsive MAP kinase gene, OsMSRMK2, whose mRNA accumulates rapidly in response to environmental cues. Biochem Biophys Res Commun 294:1009–1016
Agrawal GK, Tamogami S, Iwahashi H, Agrawal VP, Rakwal R (2003) Transient regulation of jasmonic acid-inducible rice MAP kinase gene (OsBWMK1) by diverse biotic and abiotic stresses. Plant Physiol Bioch 41:355–361
Ahmed P, Nabi G, Ashraf M (2011) Cadmium-induced oxidative damage in mustard [Brassica juncea (L) Czern.&Coss.] can be alleviated by salicylic acid. S Afr J Bot 77:36–44
Alam J, Wicks C, Stewart D, Gong P, Touchard C, Otterbein S, Choi AMK, Burow ME, Tou J (2000) Mechanism of heme oxygenase-1 gene activation by cadmium in MCF-7 mammary epithelial cells. J Biol Chem 275:27694–27702
Amry AL, Al-Saikhan F, Ayoubi A (2011) Toxic effect of cadmium found in eyeliner to the eye of a 21 year old Saudi woman: a case report. Saudi Pharm J 19:269–272
Antosiewicz DM, Henning J (2004) Overexpression of LCT1 in tobacco enhances the protective action of calcium against cadmium toxicity. Environ Pollut 129:237–245
Arasimowicz-Jelonek M, Floryszak-Wieczorek J, Gwóźdź AE (2011) The message of nitric oxide in cadmium challenged plants. Plant Sci 181:612–620
Arteca RNI, Arteca JM (2007) Heavy-metal- induced ethylene production in Arabidopsis thaliana. J Plant Physiol 164:1480–1488
ATSDR 2011. Agency for Toxic Substances and Disease Registry, U.S. Toxicological Profile for Cadmium. Department of Health and Human Service, Public Health Service, Centre for Disease Control, Atlanta, GA, U.S.A. http://www.atsdr.cdc.gov/
Barroso JB, Corpas FJ, Carreras A, Rodriguez-Serrano M, Estaban FJ, Fernández-Ocaña A, Chaki M, Romero-Puertas MC, Valderrama R, Sandalio LM, del Rio LD (2006) Localization of S-nitrosoglutathione and expression of S-nitrosoglutathione reductase in pea plants under cadmium stress. J Exp Bot 57:1785–1793
Benson RA, Lowrey JA, Lamb JR, Howie SEM (2004) The Notch and Sonic hedgehog signalling pathways in immunity. Mol Immunol 41:715–725
Berthon A, Martinez A, Bertherat J, Val P (2012) Wnt/β-catenin signalling in adrenal physiology and tumor development. Mol Cell Endocrinol 351:87–95
Besson-Bard A, Gravot A, Richaud P, Auroy P, Duc C, Gaymard F, Taconnat L, Renou J-P, Pugin A, Wendehenne D (2009) Nitric oxide contributes to cadmium toxicity in Arabidopsis by promoting cadmium accumulation in roots and by up-regulating genes related to cadmium uptake. Plant Physiol 149:1302–1315
Bhattacharjee S (2005) Reactive oxygen species and oxidative burst: roles in stress, senescence and signal transduction in plants. Curr Sci 89:1113–1121
Briedé JJ, van Delft JMH, de Kok TMCM, van Herwijnen MHM, Maas LM, Gottshalk RWH, Kleinjans JCS (2010) Global gene expression analysis reveals differences in cellular responses to hydroxyl- and superoxide anion radical-induced oxidative stress in Caco-2 cells. Toxicol Sci 114:193–203
Casalino E, Sblano C, Landriscina C (1997) Enzyme activity alteration by cadmium administration to rats: the possibility of iron involvement in lipid peroxidation. Arch Biochem Biophys 346:171–179
Casalino E, Calzaretti G, Landriscina M, Sblano C, Fabiano A, Landriscina C (2007) The Nrf2 transcription factor contributes to the induction of to the acute cadmium and manganese intoxicated rats: comparison with the toxic effect on NAD(P)H:quinone reductase. Toxicology 237:24–34
Catterjee S, Kundu S, Sengupta S, Bhattacharyya A (2009) Divergence to apoptosis from ROS induced cell cycle arrest: effect of cadmium. Mutat Res 663:22–31
Chakraborty PK, Lee W-K, Molitor M, Wolff NA, Thévenod F (2010) Cadmium induces Wnt signaling to upregulate proliferation and survival genes in sub-confluent kidney proximal tubule cells. Mol Cancer 9:102
Chang L, Karin M (2001) Mammalian MAP kinase rolifera cascades. Nature 410:37–40
Chao Y-Y, Chen C-Y, Huang W-D, Kao CH (2010) Salicylic acid-mediated hydrogen peroxide accumulation and protection against Cd toxicity in rice leaves. Plant Soil 329:327–337
Charoensawan V, Wilson D, Teichman SA (2010) Genomic repertoires of DNA-binding transcription factors across the tree of life. Nucleic Acids Res 38:7264–7377
Chauchene L, Banni M, Kerkeni A, Saȉd K, Messaoudi I (2011) Cadmium-induced ovarian pathophysiology is mediated by change in gene expression pattern of zinc transporters in zebrafish (Danio rerio). Chem-Biol Interact 193:172–179
Chen L, Liu L, Huang S (2008) Cadmium activates the mitogen-activated protein kinase (MAPK) pathways via induction of reactive oxygen species and inhibition of protein phosphatases 2A and 5. Free Radic Biol Med 45:1035–1044
Chen S, Xu Y, Xu B, Guo M, Zhang Z, Liu L, Ma H, Chen Z, Luo Y, Huang S, Chen L (2011a) CaMKII is involved in cadmium activation of MAPK and mTOR pathways leading to neuronal cell death. J Neurochem 119:1108–1118
Chen S, Xu B, Liu L, Luo Y, Zhou H, Chen W, Shen T, Han X, Kontes CD, Huang S (2011b) Cadmium induction of reactive oxygen species activates the mTOR pathways, leading to neuronal cell death. Free Radic Biol Med 50:624–632
Chen YX, Lin Q, Luo YM, He YF, Zhen SJ, Yu YL, Tian GM, Wong MH (2003) The role of citric acid on the phytoremediation of heavy metal contaminated soil. Chemosphere 50:807–811
Chiang H-C, Lo J-C, Yeh K-C (2006) Genes associated with heavy metal tolerance and accumulation in Zn/Cd hyperaccumlator Arabidopsis halleri: a genomic survey with cDNA microarray. Environ Sci Tech 40:6792–6798
Cho S-C, Chao Y-Y, Hong C-Y, Kao CH (2012) The role of hydrogen peroxide in cadmium-inhibited root growth of rice seedlings. Plant Growth Regul 66:27–35
Cho U-H, Seo N-H (2005) Oxidative stress in Arabidopsis thaliana exposed to cadmium is due to hydrogen peroxide accumulation. Plant Sci 168:113–120
Chou T-S, Chao Y-Y, Kao CH (2012) Involvement of hydrogen peroxide in heat shock- and cadmium-induced expression of ascorbate peroxidase and glutathione reductase in leaves of rice seedlings. J Plant Physiol 169:478–486
Clapham DE (2007) Calcium signaling. Cell 131:1047–1058
Clemens S, Antosiewicz DM, Ward JW, Schachtman DP, Schroeder JI (1998) The plant cDNA LCT1 mediates the uptake of calcium and cadmium in yeast. Proc Natl Acad Sci USA 95:12043–12048
Colasanti M, Persichini T, And VG (2010) Nitric oxide pathway in lower metazoans. Nitric Oxide-Biol Chem 23:94–100
Connolly EL, Fett JP, Guerinot ML (2002) Expression of the IRT1 metal transporter is controlled by metals at the levels of transcript and protein accumulation. Plant Cell 14:1347–1357
Crews MH, Owen LM, Langford N, Fairweather-Tait SJ, Fox TE, Hubbard L, Phillips D (2000) Use of the stable isotope 106Cd for studying dietry cadmium absorption in humans. Toxicol Lett 112–113:201–207
Dailianis S (2009) Production of superoxide and nitric oxide generation in haemocytes of mussel Mytilus galloprovincialis (Lmk.) after exposure to cadmium: a possible involvement of Naţ/Hţ exchanger in the induction of cadmium toxic effects. Fish Shellfish Immun 27:446–453
Dailianis S, Patetsini E, Kaloyianni M (2009) The role of signaling molecules on actin glutathionylation and protein carbonylation induced by cadmium in haemocytes of mussel Mytilus galloprovincialis (Lmk). J Exp Biol 212:3612–3620
DalCorso G, Farinati S, Furini A (2010) Regulatory networks of cadmium stress in plants. Plant Signal Behav 5(6):663–667
Dalton TP, He L, Wang B, Miller ML, Jin L, Stringer KF, Chang X, Baxter CS, Nebert DW (2004) Identification of mouse SLC39A8 as the transporter responsible for cadmium-induced toxicity in testis. PNAS 102:3401–3406
De Michele R, Vurro E, Rigo C, Costa A, Elviri L, di Valentin M, Careri M, Zottini M, di Toppi LS, Schiavo FL (2009) Nitric oxide is involved in cadmium-induced cell death in Arabidposis suspension cultures. Plant Physiol 150:217–228
Deckert J (2005) Cadmium toxicity in plants: is there any analogy to its carcinogenic effect in mammalian cells? Biometals 18:275–481
Edinger AL, Thomson CB (2004) Death by design: apoptosis, necrosis and autophagy. Curr Opin Cell Biol 16:663–669
Fan J-L, Wei X-Z, Wan L-C, Zhang L-Y, Zhao X-Q, Liu W-Z, Hao H-Q, Zhang H-Y (2011) Disarrangement of actin filaments and Ca2+ gradient by CdCl2 alerts cell wall construction in Arabidopsis thaliana root hairs by inhibiting vesicular trafficking. J Plant Physiol 168:1157–1167
Farinati S, DalCorso G, Verotto S, Furini A (2010) The Brassica juncea BjCdR15, an ortholog of Arabidopsis TGA3, is a regulator of cadmium uptake, transport and accumulation in shoots and confers cadmium tolerance in transgenic plants. New Phytol 185:974–978
Faurskov B, Bjerregaard HF (2002) Evidence for cadmium mobilization of intracellular calcium through divalent cation receptor in renal distal epithelial A6 cells. Pflug Arch Eur J Phy 445:40–50
Garnier L, Simon-Plas F, Thuleau P, Agnel J-P, Blein J-P, Ranjeva R, Montillet J-L (2006) Cadmium affects tobacco cells by a series of three waves of reactive oxygen species that contribute to cytotoxicity. Plant Cell Environ 29:1956–1969
Ghouleh IA, Khoo NKH, Knaus UG, Griendling KK, Touyz RM, Tannickal VJ, Barchowsky A, Nauseef WM, Kelley EE, Bauer PM, Darley-Usmar V, Shiva S, Cifuents-Pagado E, Freeman BA, Galdwin MT, Pagano PJ (2011) Oxidases and peroxidases in cardiovascular and lung disease: new concept in reactive oxygen species signaling. Free Radic Biol Med 51:1271–1288
Girijashanker K, He L, Soleimani M, Reed JM, Li H, Liu Z, Wang B, Dalton TP, Nebert DW (2008) Slc39a14 gene encodes ZIP14, a metal/bicarbonate symporter: similarities to the ZIP8 transporter. Mol Pharmacol 73:1413–1423
Gravot A, Lieutaud A, Verret F, Auroy P, Vavasseur A, Richaud P (2004) AtHMA3, a plant P1B-ARPase, functions as a Cd/Pb transporter in yeast. FEBS Lett 561:22–28
Groppa MD, Rosales EP, Iannone MF, Benavides MP (2008) Nitric oxide, polyamines and Cd-induced phytotoxicity in wheat roots. Phytochemistry 69:2609–2615
Gzyl J, Rymer K, Gwóźdź EA (2009) Differential response of antioxidant enzymes to cadmium stress in tolerant and sensitive cell line of cucumber (Cucumis sativus L.). Acta Biochim Pol 56:723–727
Hanson ML, Brundage KM, Schafer R, Tou JC, Barnett JB (2010) Prenatal cadmium exposure dysregulates sonic hedgehog and Wnt/β-catenin signaling in the thymus resulting in alerted thymocyte development. Toxicol Appl Pharm 242:136–145
Hao C, Hao W, Wei X, Xing L, Jiang J, Shang L (2009) The role of MAPK in the biphasic dose–response phenomenon induced by cadmium and mercury in HEK293 cells. Toxicol in Vitro 23:660–666
Hasanuzzaman M, Hossain MA, Fujita M (2010) Physiological and biochemical mechanisms of nitric oxide induced abiotic stress tolerance in plants. Am J Plant Physiol 5:295–324
Hashimoto K, Kudla J (2011) Calcium decoding mechanisms in plants. Biochimie 93:2054–2059
Hassoun EA, Stohs SJ (1996) Cadmium-induced production of superoxide anion and nitric oxide, DNA single strand breaks and lactate dehydrogenase leakage in J17774A.1 cells cultures. Toxicology 112:219–226
Hou L, Shi W, Wie W, Shen H (2011) Cadmium uptake, translocation and tolerance in AHA1OX Arabidopsis thaliana. Biol Trace Res 139:228–240
Hsu YT, Kao CH (2007a) Heat shock-mediated H2O2 accumulation and protection against Cd toxicity in rice seedlings. Plant Soil 300:137–147
Hsu YT, Kao CH (2007b) Toxicity in leaves of rice exposed to cadmium is due to hydrogen peroxide accumulation. Plant Soil 298:231–241
Hsu YT, Kao CH (2003) Role of abscisic acid in cadmium tolerance of rice (Oryza sativa L.) seedlings. Plant Cell Environ 26:867–874
Ichimura K, MAPK Group (2002) Mitogen-activated protein kinase cascades in plants: a new nomenclature. Trends Plant Sci 7:301–308
Jing Y, Liu L-Z, Jiang Y, Zhu Y, Guo NL, Barnett G, Rojanasakul Y, Agani F, Jiang B-H (2012) Cadmium induced HIF-1 and VEGF expression through ROS, ERK, and AKT signaling pathways and induced malignant transformation of human bronchial epithelial cells. Toxicol Sci 125:10–19
Jonak C, Nakagami H, Hirt H (2004) Heavy metal stress. Activation of distinct mitogen-activated protein kinase pathways by copper and cadmium. Plant Physiol 136:3276–3283
Joseph P, Muchnok TK, Klishis ML, Robert RJ, Antonini JM, Whong W-Z, Ong T-M (2001) Cadmium-induced cell transformation and tumorgenesis are associated with transcriptional activation of of c-fos, c-jun, c-myc proto-ocnogens: Role of cellular calcium and reactive oxygen species. Toxicol Sci 61:295–303
Jung Y-S, Joeng E-M, Park EK, Kim Y-M, Sohn S, Lee SH, Baik EJ, Moon C-H (2008) Cadmium induces apoptotic cell death through p38 MAPK in brain microvessel endothelial cells. Eur J Pharmac 578:11–18
Kalariya NM, Wills NK, Ramana KV, Srivastava SK, van Kuijk FJGM (2009) Cadmium-induced apoptotic death of human retinal pigment epithelial cells is mediated by MAPK pathway. Exp Eye Res 89:494–502
Kim J-A, Agrawal GK, Rakwal R, Han K-S, Kim K-N, Yun C-H, Heu S, Park S-Y, Lee Y-H, Jwa N-S (2003) Molecular cloning and mRNA expression analysis of novel rice (Oryza sativa L.) MAPK kinase kinase, OsEDR1, an ortholog of Arabidopsis AtEDR1, reveal its role in defense/stress signaling pathways and development. Bioch Bioph Res Co 300:868–876
Kim J, Kim SH, Johnson VJ, Sharma RP (2005) Extracellular signal-regulated kinase-signaling-dependent G2/M arrest and cell death in murine macrophages by cadmium. Environ Toxicol Chem 24:3069–3077
Kim J, Soh J (2009) Cadmium-induced apoptosis is mediated by the translocation of AIF to the nucleus of rat testes. Toxicol Lett 188:45–51
Kippler M, Hossain MB, Lindh C, Morre SE, Kabir I, Vahter M, Broberg K (2012) Early life low-level cadmium exposure is positively associated with increased oxidative stress. Environ Res 112:164–170
Kolluru GK, Tamilarasan KP, Priya SG, Durgha NP, Chetterjee S (2006) Cadmium induced endothelial disfunction: consequence of defective migratory pattern of endothelial cells in association with poor nitric oxide availability under cadmium challenge. Cell Biol Int 30:427–438
Kopyra M, Gwóźdź EA (2003) Nitric oxide stimulates seed germination and counteracts the inhibitory effect of heavy metals and salinity on root growth of Lupinus luteus. Plant Physiol Bioch 41:1011–1017
Kopyra M, Stachoń-Wilk M, Gwóźdź AE (2006) Effects of exogenous nitric oxide on the antioxidant capacity of cadmium-treated soybean cell suspension. Acta Physiol Plant 28:525–536
Kovács E, Nyitrai P, Czövek P, Òvári M, Keresztes À (2009) Investigation into the mechanism of stimulation by low-dose concentration stressors in barley seedlings. J Plant Physiol 166:72–79
Krantev A, Yordanova R, Janda T, Szalai G, Popova L (2008) Treatment with salicylic acid decreases the effect of cadmium on photosynthesis in maize plants. J Plant Physiol 165:920–931
Krasylenko YA, Yemets AI, Blume YB (2010) Functional role of nitric oxide in plants. Rus J Plant Physl 57:451–461
Kurtyka R, Kita A, Karcz W (2011) Fusicoccim counteracts the toxic effect of cadmium on the growth of maize coleptile segments. Arch Environ Contam Toxicol 61:568–577
Lawal AO, Ellis EM (2011) Nrf2-mediated adaptive response to cadmium-induced toxicity involves protein kinase C delta in human 1321N1 astrocytoma cells. Environ Toxicol Phar 32:54–62
Lee S, An G (2009) Over-expression of OsIRT1 leads to increased iron and zinc accumulation in rice. Plant Cell Environ 32:408–416
Lee W-K, Thévenod F (2008) Novel roles for ceramides, calpaines and caspases in kidney proximal tubule cell apoptosis: Lessons from in vitro cadmium toxicity studies. Biochem Pharmacol 76:1323–1332
Lee YH, Kim S-Y, Kim J-R, Yoh K-T, Baek S-H, Kim MJ, Ryu MH, Suh PG, Kim J-H (2000) Overexpression of phospholipase Cβ-protects NIH3T3 cells from oxidative stress-induced cell death. Life Sci 67:827–837
Lehotai N, Petö A, Bajkán S, Erdei L, Tari I, Kolbert Z (2011) In vivo and in situ visualization of early physiological events induces by heavy metals in pea root meristem. Acta Physiol Plant 33:2199–2207
Li L, Liu X, Peijnenburg WJGM, Zhao J, Chen X, Yu J, Wu H (2012) Pathways of cadmium fluxes in the root of halophyte Suaeda salsa. Ecotox Environ Safe 75:1–7
Li R, Zhou Y, Wang L, Ren G (2011) Low-molecular-weight-chitosane ameliorates cadmium-induced toxicity in freshwater crab, Sinopotamon yangtsekiense. Ecotox Environ Safe 74:1164–1170
Lichtlen P, Schaffner W (2001) Putting its fingers on stressful situations: the heavy-metal regulatory transcription factor MTF-1. Bioessays 23:1010–1017
Liu C, Guo J, Cui Y, Lű T, Zhang X, Shi G (2011) Effects of cadmium and salicylic acid on growth, spectral reflectance and photosynthesis of castor bean seedlings. Plant Soil 344:131–141
Liu K, Shen LI, Sheng J (2008) Improvement in cadmium tolerance in tomato seedlings with antisense DNA for 1-aminocyklopropane-1-carboxylate synthase. J Plant Nutr 31:809–827
Liu J, Kapron CM (2010) Differential induction of MAP kinase signaling pathways by cadmium in primary cultures of mouse embryo limb bud cells. Reprod Toxicol 29:286–291
Liu X-M, Kim KE, Kim K-C, Nguyen XC, Han HJ, Jung MS, Kim HS, Kim SH, Parl HC, Yun D-J, Chung WS (2010) Cadmium activates Arabidopsis MPK3 and MPK6 via accumulation of reactive oxygen species. Phytochemistry 71:614–618
Lombi E, Zhao FJ, Young SD, Sacchi GA (2001) Physiological evidence for high-affinity cadmium transporter highly expressed in a Thlapsi caerulescens ecotype. New Phytol 149:53–60
Long JG (1997) Cadmium perturbs calcium homeostasis in rat osteosarcoma (ROS17/2.8) cells: a possible role of protein kinase C. Toxicol Lett 91:91–97
Lu LL, Tian SK, Yang XE, Li TQ, He ZL (2009) Cadmium uptake and xylem loading are active processes in the hyperaccumulator Sedum alfredi. J Plant Physiol 166:579–587
Ma W, Xu W, Xu H, Chen Y, He Z, Ma M (2010) Nitric oxide modulates cadmium influx during cadmium-induced programmed cell death in tobacco BY-2 cells. Planta 232:325–335
Mahmood T, Gupta KJ, Kaiser WM (2009) Cadmium stress stimulates nitric oxide production in wheat roots. Pak J Bot 41:1285–1290
Maksymie W, Krupa Z (2002) Jasmonic acid and heavy metals in Arabidopsis thaliana – a similar physiological response to both stressors? J Plant Physiol 159:509–515
Maksymiec W, Krupa Z (2006) The effect of short term exposition to Cd, excess of Cu ions and jasmonate on oxidative stress appearing in Arabisopsis thaliana. Environ Exp Bot 57:187–194
Maksymiec W, Wianowska D, Dawidowicz AL, Radkiewicz S, Mardarowicz M, Krupa Z (2005) The levels of jasmonic acid in Arabidopsis thaliana and Phaseolus coccineus plants under heavy metals stress. J Plant Physiol 182:1338–1346
Maksymiec W (2011) Effect of jasmonate and some other signaling factors on bean and onion growth during the initial phase of cadmium action. Biol Plant 55:112–118
Masood A, Iqbal NI, Khan NA (2012) Role of ethylene in alleviation of cadmium-induced photosynthetic capacity inhibition by sulphur in mustard. Plant Cell Environ 35:524–533
Misra UK, Gawdi G, Akabani G, Pizzo SV (2002) Cadmium-induced DNA synthesis and cell proliferation in macrophages: the role of intracellular calcium and signal transduction mechanisms. Cell Signal 14:327–340
Mizuno T, Usui K, Horie K, Nosaka S, Mizuno N, Obata H (2005) Cloning of three ZIP/Nramp transporter genes from Ni hyperaccumulator Thlaspi japonicum and their Ni2+ -transport abilities. Plant Physiol Bioch 43:793–801
McSteen P, Zhao Y (2008) Plant hormones and signaling: common themes and new developments. Dev Cell 14:467–473
Moncada S, Higgs EA (2006) The discovery of nitric oxide and its role in vascular biology. Brit J Pharmacol 147:S193–S201
Mortensen ME, Wong L-Y, Osterloch JD (2011) Smoking status and urine cadmium above levels associated with subclinical renal effects in U.S. adults without chronic kidney disease. Int J Hyg Environ Health 214:305–310
Moussa HR, El-Gamal SM (2010a) Effect of salicylic acid pretreatment on cadmium toxicity in wheat. Biol Plant 54:315–320
Moussa HR, El-Gamal SM (2010b) Role of salicylic acid in regulation of cadmium toxicity in wheat (Triticum aesativum L.). J Plant Nutr 33:1460–1471
Mǿller MI, Sweetlove LJ (2010) ROS signalling – specifity is required. Trends Plant Sci 15:370–374
Nakagami H, Pitzscke A, Hirt H (2005) Emerging MAP kinase pathways in plant stress signaling. Trends Plant Sci 10:339–346
Noriega G, Cruz DS, Batlle A, Tomaro M, Balestrasse K (2012) Heme oxygenase is involved in the protection exerted by jasmonic acid against cadmium stress in soybean roots. J Plant Growth Reg 31:79–89
Ochocka A-M, Pawelczyk T (2003) Isozymes delta of phosphoinositide-specific phospholipase C and their role in signal transduction in the cell. Acta Biochim Pol 50:1097–1110
Ogawa I, Nakanishi H, Mori S, Nishizawa NK (2009) Time course analysis of gene regulation under cadmium stress in rice. Plant Soil 325:97–108
Ognjanović BI, Marković SD, Dordević NZ, Trbojević IS, Štajn AŠ, Saičić ZS (2010) Cadmium-induced lipid peroxidation and changes in antioxidant defense system in the rat testes: protective role of coenzyme Q10 and Vitamin E. Reprod Toxicol 29:191–197
Oh S-H, Lim S-C (2006) A rapid and transient ROS generation by cadmium triggers apoptosis via caspase-dependent pathway in HepG2 cells and this is inhibited through N-acetylcysteine-mediated catalase upregulation. Toxicol Appl Pharm 212:212–223
Okubo M, Yamada K, Hosoyamada M, Shibasaki T, Endou H (2003) Cadmium transport by human Nramp2 expressed in Xenopus laevis oocytes. Toxicol Appl Pharm 187:162–167
Olivi L, Bessler J (2000) Maitotoxin stimulates Cd influx in Madin-Darby kidney cells by activating Ca-permeable cation channels. Cell Calcium 27:187–193
Oomen RJF, Wu J, Leliévre F, Blanchet S, Richaud P, Barbier-Brygoo H, Aarts MGM, Thomine S (2009) Functional characterization of NRAMP3 and NRAMP4 from the metal hyperaccumulator Thlaspi caerulenscens. New Phytol 181:637–650
Oulhajd A, Kuschk P, Humback K (2006) Heavy metal stress and leaf senescence induce the barley gene HvC2d1 encoding a calcium-dependent novel C2 domain-like protein. New Phytol 170:261–273
Park SL, Kim Y-M, Ahn J-H, Lee SH, Baik EJ, Moon C-H, Jung Y-S (2009) Cadmium stimulates the expression of vascular cell adhesion molecule-1 (VCAM-1) via p38 mitogen-activated protein kinase MAPK and JNK activation in cerebrovascular endothelial cells. J Pharmacol Sci 110:405–409
Panda SK, Patra HK (2007) Effect of salicylic acid potentates cadmium-induced oxidative damage in Oryza sativa L. leaves. Acta Physiol Plant 29:567–575
Platel A, Gervais V, Sajot N, Nesslany F, Marzin D, Claude N (2010) Study of gene expression profile in TK6 human cells exposed to DNA-oxidizing agents. Mutat Res 689:21–49
Popova LP, Maslenkova LT, Yordanova RY, Ivanova AP, Krantev AP, Szalai G, Janda T (2009) Exogenous treatment with salicylic acid attenuates cadmium toxicity in pea seedlings. Plant Physiol Bioch 47:224–231
Prozialeck WC, Lamar PC, Lynch SM (2003) Cadmium alerts the localization of N-cadherin, and β-catenin in the proximal tubule epithelium. Toxicol Appl Pharm 189:180–195
Pytharopoulou S, Grintzalis K, Sazakli E, Leotsindis M, Georgiou CD, Kalpaxis DL (2011) Translation responses and oxidative stress of mussels experimentally exposed to Hg, Cu and Cd: pattern does not fit at all. Aquat Toxicol 105:157–165
Qi X, Zhang Y, Chai T (2007) Characterization of a novel plant promoter specifically induced by heavy metal and identification of the promoter regions conferring heavy metal responsiveness. Plant Physiol 143:50–59
Qu W, Liu J, Fuquay R, Shimoda R, Sakurai T, Saaverda JE, Keefer LK, Waalkes MP (2005) The nitric oxide prodrug, V-PYRRO/NO, protects against cadmium toxicity and apoptosis at the cellular level. Nitric Oxide-Biol Chem 12:114–120
Ramirez DC, Martinez LD, Marchevsky E, Gimenez MS (1999) Biphasic effect of cadmium in non-cytotoxic conditions on the secretion of nitric oxide from peritoneal macrophages. Toxicology 139:167–177
Ramirez DC, Gimenez MS (2003) Induction of redox changes, inducible nitric oxide synthase and cyclooxygenase-2 by chronic cadmium exposure in mouse peritoneal macrophages. Toxicol Lett 142:121–132
Rodríguez-Serrano M, Romero-Puertas MC, Zabalza A, Corpas F, Gómez M, del Río LA, Sandalio LM (2006) Cadmium effect on oxidative metabolism of pea (Pisum sativum L.) roots. Imaging of reactive oxygen species and nitric oxide accumulation in vivo. Plant Cell Environ 29:1532–1544
Rodríguez-Serrano M, Romero-Puertas MC, Pazmiño DM, Testillano PS, Risueño MC, del Rio LD, Sandalio LM (2009) Cellular response of pea plants to cadmium toxicity: cross talk between reactive oxygen species, nitric oxide, and calcium. Plant Physiol 150:229–243
Romero-Puertas MC, Corpas F, Rodríguez-Serrano M, Gómez M, del Río LA, Sandalio LM (2007) Differential expression and regulation of antioxidant enzymes by cadmium in pea plants. J Plant Physiol 164:1346–1357
Rosa G, Peralta-Videa JR, Montes M, Parsons JG, Cano-Aguilera I, Gardea-Torresdey JL (2004) Cadmium uptake and translocation in tumbleweed (Salsola kali), a potential Cd-hyperaccumulator desert plant species: ICP/OES and XAS studies. Chemosphere 55:1159–1168
Saathoff AJ, Ahner B, Spanswick RM, Walker LP (2011) Detection of phytochelatin in the xylem sap of Brassica napus. Environ Eng Sci 28:103–111
Silva N, Peiris-John R, Wickremasinghe R, Senanayake H, Sathiakumar N (2012) Cadmium a metalloestorgen: are we convinced? J Appl Toxicol 32:318–322
Scott WJ Jr, Schreiner CM, Goetz JA, Robbins D, Bel SM (2005) Cadmium-induced postaxial forelimb ectrodactyly: association with alerted sonic hedgehog signaling. Reprod Toxicol 19:479–485
Shankar VS, Bax CM, Alam AS, Bax BE, Huang CL, Zaidi M (1992) The osteoclast Ca2+ receptor is highly sensitive to activation by transition metal cations. Biochem Biophys Res Co 187:913–918
Sharma SS, Kumar V (2002) Responses of wild type and abscisic acid mutants of Arabidopsis thaliana to cadmium. J Plant Physiol 159:1323–1327
Smirnova IV, Bittel DC, Ravindra R, Jiang H, Andrews GK (2000) Zinc and cadmium can promote rapid nuclear translocation of metal response element-binding transcription factor-1. J Biol Chem 31:9377–9384
Smith JB, Dwyer SD, Smith L (1989) Cadmium evokes inositol polyphosphate formation and calcium mobilization. J Biol Chem 264:7115–7118
Snedden WA, Fromm H (1998) Calmodulin, calmodulin-related proteins and plant responses to the environment. Trends Plant Sci 3:299–304
Sobkowiak R, Deckert J (2004) The effect of cadmium on cell cycle control in suspension culture cells of soybean. Acta Physiol Plant 26:335–344
Son Y-O, Wang X, Hitron JA, Zhang Z, Cheng S, Budhraja A, Ding S, Lee J-C, Shi X (2011) Cadmium induces autophagy through ROS-dependent activation of the LKB1-AMPK signaling in skin epidermal cells. Toxicol Appl Pharm 255:287–296
Stewart D, Killen E, Naquin R, Alam S, Alam J (2003) Degradation of transcription factor Nrf2 via the ubiquitin-proteosome pathway and stabilization by cadmium. J Biol Chem 24:2396–2402
Suzuki N, Koizumi N, Sano H (2001) Screening of cadmium-responsive genes in Arabidopsis thaliana. Plant Cell Environ 24:1177–1188
Takahashi R, Ishimura Y, Senoura T, Shimo H, Ishiwaka S, Arao T, Nakanishi H, Nishizawa NK (2011) The OsNRAMP1 iron transporter is involved in Cd accumulation in rice. J Exp Bot 62:4843–4850
Thévenod F (2009) Cadmium and cellular signaling cascades: to be or not to be? Toxicol Appl Pharm 238:221–239
Thompson J, Wong L, Lau PS, Bannigan J (2008) Adherens junction breakdown in the periderm following cadmium administration in the chick embryo: distribution of cadherins and associated molecules. Reprod Toxicol 25:39–46
Valbonesi P, Ricci L, Franzellittti S, Bondi C, Fabbri E (2008) Effect of cadmium on MAPK signaling pathway and HSP70 expression in human trophoblast cell line. Placenta 2009:725–733
Valentovičová K, Haluškova L, Huttová J, Mistrík I, Tamás L (2010) Effect of cadmium on diaphorase activity and nitric oxide production in barley root tips. J Plant Physiol 167:10–14
Vestena S, Cambraia J, Ribeiro C, Oliveira JA, Oliva MA (2011) Cadmium induced oxidative stress and antioxidative enzyme response in Water Hyacinth and Salvinia. Braz J Plant Physiol 23:131–139
Wang P-C, Du Y-Y, An G-Y, Zhou Y, Miao C, Song C-P (2006) Analysis of global expression profile of Arabidposis genes under abscisic acid and H2O2 application. J Integr Plant Biol 48:62–74
Wang SH, Shih YL, Ko WC, Wei YH, Shih CM (2008) Cadmium-induced autophagy and apoptosis are mediated by calcium signaling pathways. Cell Mol Life Sci 65:3640–3652
Wang L, Wang H, Li J, Chen D, Liu Z (2011) Simultaneous effect of lead and cadmium on primary cultures of rat proximal tubular cells: interaction of apoptosis and oxidative stress. Arch Environ Contam Toxicol 61:500–511
Weisberg M, Joseph P, Hale B, Beyersmann D (2003) Molecular and cellular mechanisms of cadmium carcinogenesis. Toxicology 192:95–117
Xiong J, Lu H, Lu K, Duan Y, An L, Xhu C (2009) Cadmium decreases crown root number by decreasing endogenous nitric oxide, which is indispensable for crown root primordial initiation in rice seedlings. Planta 230:599–610
Xu J, Yin H, Wang W, Mi Q, Liao X, Li X (2009) Identification of Cd-responsive genes of Solanum nigrum seedlings through differential display. Plant Mol Biol Rep 27:563–569
Xu B, Xu Z-F, Deng Y, Yang J-H (2010) Protective effects of Chlorpromazine and Verapamil against cadmium-induced kidney damage in vivo. Exp Toxicol Path 62:27–34
Yakimova ET, Kapchina-Toteva VM, Laarhoven L-J, Harren FM, Woltering EJ (2006) Involvement of ethylene and lipid signaling in cadmium-induced cell death in tomato suspension cultures. Plant Physiol Bioch 44:581–589
Yamagami K, Nishimura S, Sorimachi M (1998) Cd2+ and Co2+ at micromolar concentrations mobilize Ca2+ via the generation of 1,4,5-triphosphate in bovine chromaffin cells. Brain Res 798:316–319
Yamaguchi H, Fukuoka H, Arao T, Ohyama A, Nunome T, Miyatake K, Negoro S (2009) Gene expression analysis in cadmium-stressed roots of low cadmium-accumulating solanaceous plant, Solanum torvum. J Expl Bot 61:423–437
Yang C-S, Tzou B-C, Liu YP, Tsai M-J, Shye SQ, Tzeng S-F (2008) Inhibition of cadmium induced oxidative injury in rat primery astrocytes by the addition of antioxidants and the reduction of intracellular calcium. J Cell Biochem 103:825–834
Yang L-Y, Wu K-H, Chiu W-T, Wang S-H, Shih C-M (2009) The cadmium-induced death of mesangial cells results in nephrotoxicity. Autophagy 4(5):571–572
Yang Y, Li F, Bi X, Sun L, Liu T, Jin Z, Liu C (2011) Lead, zinc and cadmium in vegetables/crops in a zinc smelting region and its potential human toxicity. Bull Environ Contam Toxicol 87:586–590
Yanhui C, Xiaoyuan Y, Kun H, Meihua L, Jigang L, Zhaofeng G, Zhiqiang L, Yunfei Z, Xiaoxiao W, Xiaoming Q, Yunping S, Li Z, Xiaohui D, Jingchu L, Xing-Wang D, Zhangliang C, Hongya G, Li-Jia Q (2006) The MYB transcription factor superfamily of Arabidopsis: expression analysis and phylogenetic comparison with the rice MYB family. Plant Mol Biol 60:107–124
Ye J-L, Mao W-P, Wu A-L, Zhang N-N, Zhang C, Yu Y-J, Zhou L, Wei C-L (2007) Cadmium-induced apoptosis in human normal liver L-02 cells by acting on mitochondria and regulating Ca2+ signals. Environ Toxicol Phar 24:45–54
Yeh C-M, Chien P-S, Huang H-J (2007) Distinct signaling pathways for induction of MAP kinase activities by cadmium and copper in rice roots. J Exp Bot 58:659–671
Zawoznik MS, Groppa MD, Tomaro ML, Benavides MP (2007) Endogenous salicylic acid potentiates cadmium-induced oxidative stress in Arabidopsis thaliana. Plant Sci 173:190–197
Zhang F, Zhang H, Xia Y, Wang G, Xu L, Shen Z (2011) Exogenous application of salicylic acid alleviates cadmium toxicity and reduces hydrogen peroxide accumulation in root apoplasts of Phaseoulus aureus and Vicia sativa. Plant Cell Rep 30:1475–1483
Zhang W, Chen W (2010) Autophagy induction upon reactive oxygen species in Cd-stressed Arabidopsis thaliana. Proc SPIE 7568:75681Y
Open Access
This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License (https://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Chmielowska-Bąk, J., Deckert, J. A common response to common danger? Comparison of animal and plant signaling pathways involved in cadmium sensing. J. Cell Commun. Signal. 6, 191–204 (2012). https://doi.org/10.1007/s12079-012-0173-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12079-012-0173-3