Abstract
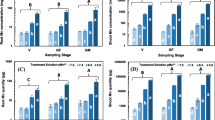
The production of H2O2 in detached rice leaves of Taichung Native 1 (TN1) caused by CdCl2 was investigated. CdCl2 treatment resulted in H2O2 production in detached rice leaves. Diphenyleneiodonium chloride (DPI) and imidazole (IMD), inhibitors of NADPH oxidase (NOX), prevented CdCl2-induced H2O2 production, suggesting that NOX is a H2O2-genearating enzyme in CdCl2-treated detached rice leaves. Phosphatidylinositol 3-kinase inhibitors wortmanin (WM) or LY294002 (LY) inhibited CdCl2-inducted H2O2 production in detached rice leaves. Exogenous H2O2 reversed the inhibitory effect of WM or LY, suggesting that phosphatidylinositol 3-phosphate is required for Cd-induced H2O2 production in detached rice leaves. Nitric oxide donor sodium nitroprusside (SNP) was also effective in reducing CdCl2-inducing accumulation of H2O2 in detached rice leaves. Cd toxicity was judged by the decrease in chlorophyll content. The results indicated that DPI, IMD, WM, LY, and SNP were able to reduce Cd-induced toxicity of detached rice leaves. Twelve-day-old TN1 and Tainung 67 (TNG67) rice seedlings were treated with or without CdCl2. In terms of Cd toxicity (leaf chlorosis), it was observed that rice seedlings of cultivar TN1 are Cd-sensitive and those of cultivar TNG67 are Cd-tolerant. On treatment with CdCl2, H2O2 accumulated in the leaves of TN1 seedlings but not in the leaves of TNG67. Prior exposure of TN1 seedlings to 45oC for 3 h resulted in a reduction of H2O2 accumulation, as well as Cd tolerance of TN1 seedlings treated with CdCl2. The results strongly suggest that Cd toxicity of detached leaves and leaves attached to rice seedlings are due to H2O2 accumulation.









Similar content being viewed by others
Abbreviations
- ASC:
-
Ascorbate
- DAB:
-
3,3-Diaminobenzidine
- DMSO:
-
Dimethyl sulfoxide
- DPI:
-
Diphenyleneiodonium chloride
- HS:
-
Heat shock
- IMD:
-
Imidazole
- LY:
-
294002
- NO:
-
Nitric oxide
- NOX:
-
NADPH oxidase (EC 1.6.99.6)
- PI3K:
-
Phosphatidylinositol 3-kinase
- PI3P:
-
Phosphatidylinostolinositol 3-phosphate
- ROS:
-
Reactive oxygen species
- SNP:
-
Sodium nitroprusside
- TN1:
-
Taichung Native 1
- TNG67:
-
Tainung 67
- WM:
-
Wortmannin
References
Anbar M (1995) Nitric oxide: a synchronizing chemical messenger. Experientia 51:545–550
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399
Auh C-K, Murphy TM (1995) Plasma membrane redox enzyme is involved in the synthesis of \({\text{O}}^{ - }_{2} \) and H2O2 by Phytophthora elicitor-stimulated rose cells. Plant Physiol 107:1241–1247
Beligni MV, Lamattina L (1999) Nitric oxide protects against cellular damage produced by methyl viologen herbicides in potato plants. Nitric Oxide 3:199–208
Bolwell GP, Davies DR, Gerrish C, Auh CK, Murphy TM (1998) Comparative biochemistry of the oxidative burst produced by rose and French bean cells reveals two distinct mechanisms. Plant Physiol 116:1374–1385
Chang CJ, Kao CH (1997) Paraquat toxicity is reduced by polyamines in rice leaves. Plant Growth Regul 22:163–168
Chen SL, Kao CH (1995) Prior temperature exposure affects subsequent Cd-induced ethylene production in rice leaves. Plant Sci 104:135–138
Cho U-H, Seo N-H (2005) Oxidative stress in Arabidopsis thaliana exposed to cadmium is due to hydrogen peroxide accumulation. Plant Sci 168:113–120
Das P, Sammantaray S, Rout GR (1997) Studies on cadmium toxicity: a review. Environ Pollut 98:29–36
Dat JF, Foyer CH, Scott IM (1998) Changes in salicylic acid and antioxidants during induced thermotolerance in mustard seedlings. Plant Physiol 118:1455–1461
Doke N (1983) Generation of superoxide anion by potato tuber protoplasts during the hypersensitive response to hyphal cell wall components of Phytophthora infestans and specific inhibition of the reaction by suppressors of hypersensitivity. Physiol Plant Pathol 23:359–367
Ellson CD, Gobert-Gosse S, Anderson KE, Davidson K, Erdjument-Bromage H, Tempst P, Thring JW, Cooper MA, Lim Z-Y, Holmes AB, Gaffney PRJ, Coadwell J, Chilvers ER, Hawkins PT, Steppheas LR (2001) PtdIns(3)P regulated the neutrophil oxidase complex by binding to the PX domain of p40phox. Nat Cell Biol 3:679–682
Foyer CH, Noctor G (2005) Oxidant and antioxidant signaling in plants: a re-evaluation of the concept of oxidative stress in a physiological context. Plant Cell Environ 28:1056–1071
Groppa MD, Tomaro ML, Benavides MP (2001) Polyamines as protectors against cadmium or copper-induced oxidative damage in sunflower leaf discs. Plant Sci 161:481–488
Hsu YT, Kao CH (2003) Role of abscisic acid in cadmium tolerance of rice (Oryza sativa L.) seedlings. Plant Cell Environ 20:867–874
Hsu YT, Kao CH (2004) Cadmium toxicity is reduced by nitric oxide in rice leaves. Plant Growth Regul 42:227–238
Hsu YT, Kao CH (2007) Cadmium-induced oxidative damage in rice leaves is reduced by polyamines. Plant Soil 291:27–37
Hung KT, Kao CH (2004) Phosphatidylinositol 3-phosphate is required for abscisic acid-induced hydrogen peroxide production in rice leaves. Plant Growth Regul 45:96–101
Hung KT, Kao CH (2005) Nitric oxide counteracts the senescence of rice leaves induced by hydrogen peroxide. Bot Bull Acad Sin 46:21–28
Hung KT, Chang CJ, Kao CH (2002) Paraquat toxicity is reduced by nitric oxide in rice leaves. J Plant Physiol 159:159–166
Hung KT, Hsu YT, Kao CH (2006) Hydrogen peroxide is involved in methyl jasmonate-induced senescence of rice leaves. Physiol Plant 127:293–303
Jana S, Choudhuri MH (1982) Glycolate metabolism of these submerged aquatic angiosperms during aging. Aquat Bot 12:345–354
Joo JH, Yoo HJ, Hwang I, Lee JS, Nam KH, Bae YS (2005) Auxin-induced reactive oxygen species production required the activation of phosphatidyliniaitol 3-kinase. FEBS Lett 579:1243–1248
Jung J-Y, Kim Y-W, Kwak JM, Hwang J-U, Young J, Schroeder JI, Hwang I, Lea Y (2002) Phosphatidyl-inostol 3- and 4-phosphate are required for normal stomatal movements. Plant Cell 147:2397–2412
Kuo MC, Kao CH (2004) Antioxidant enzyme activities are upregulated in response to cadmium in sensitive, but not in tolerant rice (Oryza sativa L.) seedlings. Bot Bull Acad Sin 45:291–299
Lamattina L, Garaia Mata C, Graziano M, Pagnussat G (2003) Nitric oxide: the versatility of an extensive signal molecule. Annu Rev Plant Biol 54:109–136
López-Delgado H, Dat JF, Foyer CH, Scoot IM (1998) Induction of tolerance in potato microplants by acetyl-salicylic acid and H2O2. J Exp Bot 49:713–720
Low PS, Merida JR (1996) The oxidative burst in plant defense: function and signal transduction. Physiol Plant 96:533–542
Mittler R, Vanderauwera S, Gollery M, Van Breusegem F (2004) Tends Plant Sci 9:490–498
Murphy TM, Auh C-K (1996) The superoxide synthases of plasma membrane preparations from cultured rose cells. Plant Physiol 110:621–629
Neill SJ, Desikan R, Hancock J (2002) Hydrogen peroxide signaling. Curr Opin Plant Biol 5:388–395
Neill SJ, Desikan R, Hancock JT (2003) Nitric oxide signalings in plants. New Phytol 159:11–35
Olmos EO, Martínez-Solano JR, Piqueras A, Hellín E (2003) Early steps in the oxidative burst induced by cadmium in cultured tobacco cells (BY-2 line). J Exp Bot 54:291–301
Orozco-Cárdenas ML, Ryan CA (1999) Hydrogen peroxide is generated systemically in plant leaves by wounding and systemin via the octadecanoid pathway. Proc Natl Acad Sci USA 96:6553–6557
Orozco-Cárdenas ML, Ryan CA (2002) Nitric oxide negatively modulate wound signaling in tomato plants. Plant Physiol 130:487–493
Orzech KA, Burke JJ (1988) Heat shock and the protection against metal toxicity in wheat leaves. Plant Cell Environ 11:711–714
Park K-Y, Jung J-Y, Park J, Hwang JU, Kim Y-W, Hwang I, Lee Y (2003) A role for phosphatidylinositol 3-phosphate in abscisic acid-induced reactive oxygen species generation in guard cells. Plant Physiol 132:92–98
Pei Z-M, Murata Y, Benning G, Thomine S, Klüsener B, Allen GJ, Grill E, Schroeder JI (2000) Calcium channels activiated by hydrogen peroxide mediate abscisic acid signaling in guard cells. Nature 406:731–734
Pinton R, Cakmak I, Marschner H (1994) Zinc deficiency enhanced NAD(P)H-dependent superoxide radical production in plasma membrane vesicles isolated from roots of bean plants. J Exp Bot 45:45–50
Piqueras A, Olmos E, Martińez-Solano JR, Hellín E (1999) Cd-induced oxidative burst in tobacco BY2 cells: time course, subcellular location and antioxidant response. Free Radic Res 31:S33–S38
Rodriguez-Serrano M, Romero-Puertas C, Zabalza A, Corpas FJ, Gómez M, del Rio LA, Sandalio LM (2006) Cadmium effect on oxidative metabolism of pea (Pisum sativum L.) roots. Imaging of reactive oxygen species and nitric oxide accumulation in vivo. Plant Cell Environ 291:1532–1544
Romero-Puertas MC, Rodriguez-Serrano M, Corpas FJ, Gómez M, del Rio LA, Sandalio LM (2004) Cadmium-induced subcellular accumulation of \( O^{ - }_{2} \) and H2O2 in pea leaves. Plant Cell Environ 27:1122–1134
Sagi M, Flurhr R (2006) Production of reactive oxygen species by plant NADPH oxidase. Plant Physiol 141:336–340
Sandalio LM, Dalurzo HC, Gómez M, Romero-Puertas MC, del Rio LA (2001) Cadmium-induced changes in the growth and oxidative metabolism of pea plant. J Exp Bot 52:2115–2126
Sauvé S, Norvell WA, McBride M, Hendershot W (2000) Speciation and complexation of cadmium in extracted soil solutions. Environ Sci Technol 34:291–296
Schützendübel A, Schwang P, Teichmann T, Gross K, Langenfeld-Heyer R, Godbold DL, Polle A (2001) Cadmium-induced changes in antioxidative systems, hydrogen peroxide content, and differentiation in scots pine roots. Plant Physiol 127:887–898
Shah K, Kumar RG, Verma S, Dubey RS (2001) Effect of cadmium on lipid peroxidation, superoxide anion generation and activities of antioxidant enzymes in growing rice seedlings. Plant Sci 161:1135–1144
Sharma Y, León J, Roskinn I, Davies KR (1996) Ozone-induced responses in Arabidopsis thaliana: the role of salicylic acid in the accumulation of defense-related transcripts and induced resistance. Proc Natl Acad Sci USA 93:5099–5104
Velikova V, Yordanov I, Edreva S (2000) Oxidative stress and some antioxidant systems in acid rain-treated bean plants. Protective role of exogenous polyamines. Plant Sci 151:59–66
Wagner GJ (1993) Accumulation of cadmium in crop plants and its consequences to human health. Adv Agron 5:173–212
Wintermans JFGM, De Mots A (1965) Spectrophotometric characteristics of chlorophyll a and b and their pheophytins in ethanol. Biochim Biophys Acta 109:448–453
Acknowledgements
This work was supported by a research grant from the National Science Council of the Republic of China (NSC 94-2313-B-002-028).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Fangjie J. Zhao.
Rights and permissions
About this article
Cite this article
Hsu, Y.T., Kao, C.H. Toxicity in leaves of rice exposed to cadmium is due to hydrogen peroxide accumulation. Plant Soil 298, 231–241 (2007). https://doi.org/10.1007/s11104-007-9357-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-007-9357-7