Abstract
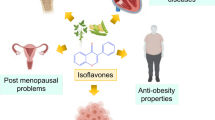
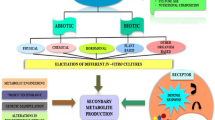
Isoflavones (ISF) are potent secondary metabolites, widely distributed among the members of the Fabaceae family. They have a multitude of biological functions in plants, animals, and humans. Most of the functions exhibited by isoflavones are highly beneficial to human beings. Benefits mainly include lowering cardiovascular diseases, reducing menopausal symptoms, and decreasing the risk of hormone-related cancers. Therefore, this potent compound has emerged as a promising possibility for making medicines and has grabbed the pharmaceutical industry's attention. Nowadays, many isoflavone-based products are manufactured commercially as food supplements, and their demand is drastically increasing globally. Numerous in vitro studies have successfully synthesized value-added metabolites that possess a vast number of industrial applications. Production of isoflavones within the plants depends on several biological and environmental conditions. Hence, in vitro cultures provide an alternate source to recover these compounds independent of environmental factors and limited bio-resources. Plant organ cultures could be highly recommended as a reliable platform for synthesizing plant-based pharmaceutically active isoflavones. Moreover, biotechnological approaches, such as elicitation and precursor feeding, can eventually promote the yield and increase these metabolites' production. This review initially outlines information regarding the source, structure, synthesis, and diverse roles of isoflavones. Later, this paper describes different plant organ cultures for the in vitro production of isoflavones and highlights elicitation strategies as well as metabolic engineering work to enhance production.



Similar content being viewed by others
References
Adler SA, Purup S, Hansen-Møller J et al (2015) Phytoestrogens and their metabolites in bulk-tank milk: effects of farm management and season. PLoS ONE 10:e0127187. https://doi.org/10.1371/journal.pone.0127187
Akashi T, Ishizaki M, Aoki T et al (2005) Isoflavonoid production by adventitious-root cultures of Iris germanica (Iridaceae). Plant Biotechnol 22:207–215. https://doi.org/10.5511/plantbiotechnology.22.207
Algar E, Gutierrez-Mañero FJ, Bonilla A et al (2012) Pseudomonas fluorescens N21.4 metabolites enhance secondary metabolism isoflavones in soybean (Glycine max) calli cultures. J Agric Food Chem 60:11080–11087. https://doi.org/10.1021/jf303334q
Al-Tawaha AM, Seguin P, Smith DL, Beaulieu C (2005) Biotic elicitors as a means of increasing isoflavone concentration of soybean seeds. Ann Appl Biol 146:303–310. https://doi.org/10.1111/j.1744-7348.2005.040106.x
Ames TT, Worden RM (1997) Continuous production of daidzein and genistein from soybean in a magnetofluidized bed bioreactor. Biotechnol Prog 13:336–339. https://doi.org/10.1021/bp970013p
Babaoglu M, Davey MR, Power JB et al (2004) Transformed roots of Lupinus mutabilis: induction, culture and isoflavone biosynthesis. Plant Cell Tissue Organ Cult 78:29–36. https://doi.org/10.1023/B:TICU.0000020386.03780.ea
Bak RO, Gomez-Ospina N, Porteus MH (2018) Gene editing on center stage. Trends Genet 34:600–611. https://doi.org/10.1016/j.tig.2018.05.004
Barta C, Kálai T, Hideg K et al (2004) Differences in the ROS-generating efficacy of various ultraviolet wavelengths in detached spinach leaves. Funct Plant Biol 31:23–28
Blicharski T, Oniszczuk A (2017) Extraction methods for the isolation of isoflavonoids from plant material. Open Chem 15:34–45. https://doi.org/10.1515/chem-2017-0005
Bloemberg GV, Lugtenberg BJ (2001) Molecular basis of plant growth promotion and biocontrol by rhizobacteria. Curr Opin Plant Biol 4:343–350. https://doi.org/10.1016/S1369-5266(00)00183-7
Boonsnongcheep P, Korsangruang S, Soonthornchareonnon N et al (2010) Growth and isoflavonoid accumulation of Pueraria candollei var. candollei and P. candollei var. mirifica cell suspension cultures. Plant Cell Tissue Organ Cult 101:119–126. https://doi.org/10.1007/s11240-010-9668-x
Bouque V, Bourgaud F, Nguyen C, Guckert A (1998) Production of daidzein by callus cultures of Psoralea species and comparison with plants. Plant Cell Tissue Organ Cult 53:35–40. https://doi.org/10.1023/A:1006057211490
Bourgaud F, Bouque V, Guckert A (1999) Production of flavonoids by Psoralea hairy root cultures. Plant Cell Tissue Organ Cult 56:97–104. https://doi.org/10.1023/A:1006206104795
Carroll K (1991) Review of clinical studies on cholesterol-lowering response to soy protein. Am Diet Assoc 91:820–827
Chandrareddy A, Muneyyirci-Delale O, McFarlane SI, Murad OM (2008) Adverse effects of phytoestrogens on reproductive health: a report of three cases. Complement Ther Clin Pract 14:132–135. https://doi.org/10.1016/j.ctcp.2008.01.002
Chen G, Li L (2007) Nutrient consumption and production of isoflavones in bioreactor cultures of Pueraria lobata (Willd). J Environ Biol 28:321–326
Chukwumah YC, Walker LT, Verghese M et al (2007) Comparison of extraction methods for the quantification of selected phytochemicals in peanuts ( Arachis hypogaea ). J Agric Food Chem 55:285–290. https://doi.org/10.1021/jf062148t
D’Angiolillo F, Pistelli L, Noccioli C et al (2014) In vitro cultures of Bituminaria bituminosa: Pterocarpan, Furanocoumarin and Isoflavone production and cytotoxic activity evaluation. Nat Prod Commun. https://doi.org/10.1177/1934578X1400900411
Davis JN, Singh B, Bhuiyan M, Sarkar FH (1998) Genistein-induced upregulation of p21 WAF1, downregulation of cyclin B, and induction of apoptosis in prostate cancer cells. Nutr Cancer 32:123–131. https://doi.org/10.1080/01635589809514730
Deavours BE, Dixon RA (2005) Metabolic engineering of isoflavonoid biosynthesis in alfalfa. Plant Physiol 138:2245–2259. https://doi.org/10.1104/pp.105.062539
Devi MKA, Giridhar P (2014) Isoflavone augmentation in soybean cell cultures is optimized using response surface methodology. J Agric Food Chem 62:3143–3149. https://doi.org/10.1021/jf500207x
Devi MKA, Giridhar P (2015) Variations in physiological response, lipid peroxidation, antioxidant enzyme activities, proline and isoflavones content in soybean varieties subjected to drought stress. Proc Natl Acad Sci India Sect B Biol Sci 85:35–44. https://doi.org/10.1007/s40011-013-0244-0
Devi MKA, Gondi M, Sakthivelu G et al (2009) Functional attributes of soybean seeds and products, with reference to isoflavone content and antioxidant activity. Food Chem 114:771–776. https://doi.org/10.1016/j.foodchem.2008.10.011
Devi MKA, Kumar G, Giridhar P (2020) Effect of biotic and abiotic elicitors on isoflavone biosynthesis during seed development and in suspension cultures of soybean (Glycine max L.). 3 Biotech 10:1–14. https://doi.org/10.1007/s13205-020-2065-1
Dixon RA, Paiva NL (1995) Stress-induced phenylpropanoid metabolism. Plant Cell 7:1085–1097. https://doi.org/10.1105/tpc.7.7.1085
Dixon RA, Harrison MJ, Paiva NL (1995) The isoflavonoid phytoalexin pathway: from enzymes to genes to transcription factors. Physiol Plant 93:385–392. https://doi.org/10.1111/j.1399-3054.1995.tb02243.x
Downie JA, Walker SA (1999) Plant responses to nodulation factors. Curr Opin Plant Biol 2:483–489. https://doi.org/10.1016/S1369-5266(99)00018-7
Du G, Zhao HY, Zhang QW et al (2010a) A rapid method for simultaneous determination of 14 phenolic compounds in Radix puerariae using microwave-assisted extraction and ultra high performance liquid chromatography coupled with diode array detection and time-of-flight mass spectrometry. J Chromatogr A 1217:705–714. https://doi.org/10.1016/j.chroma.2009.12.017
Du H, Huang Y, Tang Y (2010b) Genetic and metabolic engineering of isoflavonoid biosynthesis. Appl Microbiol Biotechnol 86:1293–1312. https://doi.org/10.1007/s00253-010-2512-8
Eldridge AC, Kwolek WF (1983) Soybean isoflavones: effect of environment and variety on composition. J Agric Food Chem 31:394–396. https://doi.org/10.1021/jf00116a052
Engelmann NJ, Reppert A, Yousef G et al (2009) In vitro production of radiolabeled red clover (Trifolium pratense) isoflavones. Plant Cell Tissue Organ Cult 98:147–156. https://doi.org/10.1007/s11240-009-9547-5
Etxeberria U, Fernández-Quintela A, Milagro FI et al (2013) Impact of polyphenols and polyphenol-rich dietary sources on gut microbiota composition. J Agric Food Chem 61:9517–9533. https://doi.org/10.1021/jf402506c
Faraj A, Vasanthan T (2004) Soybean isoflavones: effects of processing and health benefits. Food Rev Int 20:51–75. https://doi.org/10.1081/FRI-120028830
Farquhar C, Marjoribanks J, Lethaby A et al (2005) Long-term hormone therapy for perimenopausal and postmenopausal women. In: Farquhar C (ed) Cochrane database of systematic reviews. John Wiley & Sons, Ltd, Chichester, p CD004143
Farquhar C, Marjoribanks J, Lethaby A et al (2015) Long-term hormone therapy for perimenopausal and postmenopausal women. In: Farquhar C (ed) Cochrane database of systematic reviews. John Wiley & Sons, Ltd, Chichester
Federici E, Touché A, Choquart S et al (2003) High isoflavone content and estrogenic activity of 25 year-old Glycine max tissue cultures. Phytochemistry 64:717–724. https://doi.org/10.1016/S0031-9422(03)00379-0
Fedoreyev SA, Pokushalova TV, Veselova MV et al (2000) Isoflavonoid production by callus cultures of Maackia amurensis. Fitoterapia 71:365–372. https://doi.org/10.1016/S0367-326X(00)00129-5
Filova A (2014) Production of secondary metabolites in plant tissue cultures. Res J Agric Sci 46:236–245
Fotso G, Maher F, Ngnintedo D et al (2015) Three new isoflavonoids with antioxidant properties from Ptycholobium contortum (N.E.Br.) Brummitt (Leguminosae). Phytochem Lett 14:254–259
Franke AA, Lai JF, Halm BM (2014) Absorption, distribution, metabolism, and excretion of isoflavonoids after soy intake. Arch Biochem Biophys 559:24–28. https://doi.org/10.1016/j.abb.2014.06.007
Gao M, Huang W, RoyChowdhury M, Liu C (2007) Microwave-assisted extraction of scutellarin from Erigeron breviscapus Hand-Mazz and its determination by high-performance liquid chromatography. Anal Chim Acta 591:161–166. https://doi.org/10.1016/j.aca.2007.04.004
Georgiev M, Pavlov A, Ilieva M (2004) Rosmarinic acid production by Lavandula vera MM cell suspension: the effect of temperature. Biotechnol Lett 26:855–856. https://doi.org/10.1023/B:BILE.0000025891.64306.16
Georgiev MI, Eibl R, Zhong J-J (2013) Hosting the plant cells in vitro: recent trends in bioreactors. Appl Microbiol Biotechnol 97:3787–3800. https://doi.org/10.1007/s00253-013-4817-x
Gou L, Zhang R, Ma L et al (2016) Multigene synergism increases the isoflavone and proanthocyanidin contents of Medicago truncatula. Plant Biotechnol J 14:915–925. https://doi.org/10.1111/pbi.12445
Graham TL (1995) Cellular biochemistry of phenylpropanoid responses of soybean to infection by phytophthora sojae. Handbook of phytoalexin metabolism and action. CRC Press
Gualtieri G, Bisseling T (2000) The evolution of nodulation. Plant Mol Biol 42:181–194. https://doi.org/10.1023/A:1006396525292
Gueven A, Knorr D (2011) Isoflavonoid production by soy plant callus suspension culture. J Food Eng 103:237–243. https://doi.org/10.1016/j.jfoodeng.2010.10.019
Guillon S, Trémouillaux-Guiller J, Pati PK et al (2006) Hairy root research: recent scenario and exciting prospects. Curr Opin Plant Biol 9:341–346. https://doi.org/10.1016/j.pbi.2006.03.008
Guo-zhen J, Li W (2014) Effect of daidzein on ileum microflora biodiversity in Hy-Line variety brown layers. J Northeast Agric Univ Engl Ed 21:31–36. https://doi.org/10.1016/S1006-8104(15)30017-9
Gutierrez-Gonzalez JJ, Guttikonda SK, Tran L-SP et al (2010) Differential expression of isoflavone biosynthetic genes in soybean during water deficits. Plant Cell Physiol 51:936–948. https://doi.org/10.1093/pcp/pcq065
Hagmann M, Grisebach H (1984) Enzymatic rearrangement of flavanone to isoflavone. FEBS Lett 175:199–202. https://doi.org/10.1016/0014-5793(84)80736-X
Hakamatsuka T, Mori K, Ishida S et al (1998) Purification of 2-hydroyisoflavanone dehydratase from the cell cultures of Pueraria lobata. Phytochemistry 49:497–505
Hammerschmidt R, Dann EK (2007) The role of phytoalexins in plant protection. Novartis Found Symp 223:175–187. https://doi.org/10.1002/9780470515679.ch12
He X-Z, Dixon RA (2000) Genetic manipulation of isoflavone 7- O -methyltransferase enhances biosynthesis of 4′-O-methylated isoflavonoid phytoalexins and disease resistance in Alfalfa. Plant Cell 12:1689–1702. https://doi.org/10.1105/tpc.12.9.1689
Hilakivi-Clarke L, de Assis S (2006) Fetal origins of breast cancer. Trends Endocrinol Metab 17:340–348. https://doi.org/10.1016/j.tem.2006.09.002
Hillman G, Forman J et al (2001) Genistein potentiates the radiation effect on prostate carcinoma cells. Clin Cancer Res 7:382–390
Hoeck JA, Fehr WR, Murphy PA, Welke GA (2000) Influence of genotype and environment on isoflavone contents of soybean. Crop Sci 40:48–51. https://doi.org/10.2135/cropsci2000.40148x
Hoie LH, Guldstrand M, Sjoholm A et al (2007) Cholesterol-lowering effects of a new isolated soy protein with high levels of nondenaturated protein in hypercholesterolemic patients. Adv Ther 24:439–447. https://doi.org/10.1007/BF02849913
Hsu J-T, Hung H-C, Chen C-J et al (1999) Effects of the dietary phytoestrogen biochanin A on cell growth in the mammary carcinoma cell line MCF-7. J Nutr Biochem 10:510–517. https://doi.org/10.1016/S0955-2863(99)00037-6
Humphreys JM, Chapple C (2000) Molecular ‘pharming’ with plant P450s. Trends Plant Sci 5:271–272. https://doi.org/10.1016/S1360-1385(00)01680-0
Januário AH, Lourenço MV, Domézio LA et al (2005) Isolation and structure determination of bioactive isoflavones from callus culture of Dipteryx odorata. Chem Pharm Bull (tokyo) 53:740–742. https://doi.org/10.1248/cpb.53.740
Jeong YJ, An CH, Park SC et al (2018) Methyl jasmonate increases isoflavone production in soybean cell cultures by activating structural genes involved in isoflavonoid biosynthesis. J Agric Food Chem 66:4099–4105. https://doi.org/10.1021/acs.jafc.8b00350
Jia T, An J, Liu Z et al (2017) Salt stress induced soybean GmIFS1 expression and isoflavone accumulation and salt tolerance in transgenic soybean cotyledon hairy roots and tobacco. Plant Cell Tissue Organ Cult 128:469–477. https://doi.org/10.1007/s11240-016-1124-0
Jiang N, Jeon E-H, Pak J-H et al (2010) Increase of isoflavones in soybean callus by Agrobacterium-mediated transformation. Plant Biotechnol Rep 4:253–260. https://doi.org/10.1007/s11816-010-0143-2
Jiang Y, Hu Y, Wang B, Wu T (2014) Bivalent RNA interference to increase isoflavone biosynthesis in Soybean (Glycine max). Braz Arch Biol Technol 57:163–170
Jiao J, Gai Q-Y, Wang W et al (2015) Ultraviolet radiation-elicited enhancement of isoflavonoid accumulation, biosynthetic gene expression, and antioxidant activity in Astragalus membranaceus hairy root cultures. J Agric Food Chem 63:8216–8224. https://doi.org/10.1021/acs.jafc.5b03138
Jin Y, He T, Lu B (2003) Fine scale genetic structure in a wild soybean (Glycine soja) population and the implications for conservation. New Phytol 159:513–519. https://doi.org/10.1046/j.1469-8137.2003.00824.x
Jin H, Yu Y, Quan X, Wu S (2019) Industrial crops & products promising strategy for improving calycosin-7- O - β -D-glucoside production in Astragalus membranaceus adventitious root cultures. Ind Crop Prod 141:111792. https://doi.org/10.1016/j.indcrop.2019.111792
Jooyandeh H (2011) Soy products as healthy and functional foods. Middle-East J Sci Res 7:71–80
Jung W, Yu O, Lau S et al (2000) Identification and expression of isoflavone synthase, the key enzyme for biosynthesis of isoflavones in legumes. Nat Biotechnol 18:208–212
Jung W, Chung I, Heo H (2003) Manipulating isoflavone levels in plants. J Plant Biotechnol 5:149–155
Kanehisa M, Furumichi M, Tanabe M et al (2017) KEGG: new perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res 45:D353–D361. https://doi.org/10.1093/nar/gkw1092
Karuppusamy S (2009) A review on trends in production of secondary metabolites from higher plants by in vitro tissue, organ and cell cultures. J Med Plants Res 3:1222–1239
Karwasara VS, Dixit VK (2012) Culture medium optimization for improved puerarin production by cell suspension cultures of Pueraria tuberosa (Roxb. ex Willd.) DC. In Vitro Cell Dev Biol Plant 48:189–199. https://doi.org/10.1007/s11627-011-9415-y
Kaufman PB, Duke JA, Brielmann H et al (1997) A comparative survey of leguminous plants as sources of the isoflavones, genistein and daidzein: implications for human nutrition and health. J Altern Complement Med 3:7–12. https://doi.org/10.1089/acm.1997.3.7
Kim DH, Kim BG, Lee HJ et al (2005) Enhancement of isoflavone synthase activity by co-expression of P450 reductase from rice. Biotechnol Lett. 27:1291–1294. https://doi.org/10.1007/s10529-005-0221-7
Kim JK, Kim E-H, Park I et al (2014) Isoflavones profiling of soybean [Glycine max (L.) Merrill] germplasms and their correlations with metabolic pathways. Food Chem 153:258–264. https://doi.org/10.1016/j.foodchem.2013.12.066
Kobayashi M, Noguchi H, Sankawa U (1985) Formation of chalcones and isoflavones by callus culture of Glycyrrhiza uralensis with different production patterns. Chem Pharm Bull (tokyo) 33:3811–3816. https://doi.org/10.1248/cpb.33.3811
Kokotkiewicz A, Luczkiewicz M, Kowalski W et al (2013) Isoflavone production in Cyclopia subternata Vogel (honeybush) suspension cultures grown in shake flasks and stirred-tank bioreactor. Appl Microbiol Biotechnol 97:8467–8477. https://doi.org/10.1007/s00253-013-5099-z
Kokotkiewicz A, Bucinski A, Luczkiewicz M (2014) Light and temperature conditions affect bioflavonoid accumulation in callus cultures of Cyclopia subternata Vogel (honeybush). Plant Cell Tissue Organ Cult 118:589–593. https://doi.org/10.1007/s11240-014-0502-8
Korsangruang S, Soonthornchareonnon N, Chintapakorn Y et al (2010) Effects of abiotic and biotic elicitors on growth and isoflavonoid accumulation in Pueraria candollei var. candollei and P. candollei var. mirifica cell suspension cultures. Plant Cell Tissue Organ Cult 103:333–342. https://doi.org/10.1007/s11240-010-9785-6
Kubes J, Skalicky M, Tumova L et al (2019) Vanadium elicitation of Trifolium pratense L. cell culture and possible pathways of produced isoflavones transport across the plasma membrane. Plant Cell Rep 38:657–671. https://doi.org/10.1007/s00299-019-02397-y
Kudou S, Fleury Y, Welti D et al (1991) Malonyl isoflavone glycosides in soybean seeds ( Glycine max Merrill ). Agric Biol Chem 55:2227–2233. https://doi.org/10.1080/00021369.1991.10870966
Kukuła-Koch W, Baj T, Sieniawska E et al (2015) Influence of extraction methods on the recovery of astragaloside IV from the roots of Astragalus mongholicus in Soxhlet- and Twisselmann-type apparatus. Open Chem 13:938–942
Kumar MA, Pammi SSS, Sukanya MS, Giri A (2018) Enhanced production of pharmaceutically important isoflavones from hairy root rhizoclones of Trifolium pratense L. In Vitro Cell Dev Biol Plant 54:94–103. https://doi.org/10.1007/s11627-017-9873-y
Lakshmi MC, Rao LJ, Ravi R, Raghavarao KSMS (2013) Extraction and concentration of isoflavones from soybean (Glycine max). Sep Sci Technol 48:166–174. https://doi.org/10.1080/01496395.2012.674601
Lee SJ, Ahn JK, Kim SH et al (2003) Variation in isoflavone of soybean cultivars with location and storage duration. J Agric Food Chem 51:3382–3389. https://doi.org/10.1021/jf0261405
Lethaby A, Marjoribanks J, Kronenberg F et al (2013) Phytoestrogens for menopausal vasomotor symptoms. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD001395.pub4
Li L, Zhang CR (2006) Production of puerarin and isoflavones in cell suspension cultures of Pueraria lobata (Willd.): Effects of medium supplementation with casein hydrolysate and coconut milk. J Environ Biol 27:21–26
Liu HL, Li L (2002) Cell cultures of Pueraria lobata (Willd.): growth production of isoflavones puerarin. S Afr J Bot 68:542–544. https://doi.org/10.1016/S0254-6299(15)30382-3
Liu HY, Zhang CQ (2008) Effects of daidzein on messenger ribonucleic acid expression of gonadotropin receptors in chicken ovarian follicles. Poult Sci 87:541–545. https://doi.org/10.3382/ps.2007-00274
Liu C-J, Blount JW, Steele CL, Dixon RA (2002) Bottlenecks for metabolic engineering of isoflavone glycoconjugates in Arabidopsis. Proc Natl Acad Sci 99:14578–14583. https://doi.org/10.1073/pnas.212522099
Liu R, Hu Y, Li J, Lin Z (2007) Production of soybean isoflavone genistein in non-legume plants via genetically modified secondary metabolism pathway. Metab Eng 9:1–7. https://doi.org/10.1016/j.ymben.2006.08.003
Liu J, Lan X, Lv S et al (2019) Salicylic acid involved in chilling-induced accumulation of calycosin-7-O-β-d-glucoside in Astragalus membranaceus adventitious roots. Acta Physiol Plant 41:1–9. https://doi.org/10.1007/s11738-019-2909-7
Lourenço PM, de Castro S, Martins TM et al (2002) Growth and proteolytic activity of hairy roots from Centaurea calcitrapa: effect of nitrogen and sucrose. Enzyme Microb Technol 31:242–249. https://doi.org/10.1016/S0141-0229(02)00117-5
Lozovaya VV, Lygin AV, Zernova OV et al (2004) Isoflavonoid accumulation in soybean hairy roots upon treatment with Fusarium solani. Plant Physiol Biochem 42:671–679. https://doi.org/10.1016/j.plaphy.2004.06.007
Lozovaya VV, Lygin AV, Ulanov AV et al (2005) Effect of temperature and soil moisture status during seed development on soybean seed isoflavone concentration and composition. Crop Sci 45:1934–1940. https://doi.org/10.2135/cropsci2004.0567
Lucchesini M, Bertoli A, Mensuali-Sodi A et al (2010) Cytisus aeolicus Guss. ex Lindl. in vitro cultures and genistin production. Open Life Sci 5:111–120. https://doi.org/10.2478/s11535-009-0067-4
Luczkiewicz M, Kokotkiewicz A (2012) Elicitation and permeabilisation affect the accumulation and storage profile of phytoestrogens in high productive suspension cultures of Genista tinctoria. Acta Physiol Plant 34:1–16. https://doi.org/10.1007/s11738-011-0799-4
Luczkiewicz M, Glod D, Baczek T, Bucinski A (2004) LC-DAD UV and LC-MS for the analysis of isoflavones and flavones from in vitro and in vivo biomass of Genista tinctoria L. Chromatographia 60:179–185. https://doi.org/10.1365/s10337-004-0357-y
Luczkiewicz M, Kokotkiewicz A, Glod D (2014) Plant growth regulators affect biosynthesis and accumulation profile of isoflavone phytoestrogens in high-productive in vitro cultures of Genista tinctoria. Plant Cell Tissue Organ Cult 118:419–429. https://doi.org/10.1007/s11240-014-0494-4
Łuczkiewicz M, Głód D (2003) Callus cultures of Genista plants—in vitro material producing high amounts of isoflavones of phytoestrogenic activity. Plant Sci 165:1101–1108
Łuczkiewicz M, Głód D (2005) Morphogenesis-dependent accumulation of phytoestrogenes in Genista tinctoria in vitro cultures. Plant Sci 168:967–979. https://doi.org/10.1016/j.plantsci.2004.11.008
Łuczkiewicz M, Kokotkiewicz A (2005) Co-cultures of shoots and hairy roots of Genista tinctoria L. for synthesis and biotransformation of large amounts of phytoestrogens. Plant Sci 169:862–871. https://doi.org/10.1016/j.plantsci.2005.06.005
Luque-Garcı́a J, de Castro ML (2004) Ultrasound-assisted Soxhlet extraction: an expeditive approach for solid sample treatment. J Chromatogr A 1034:237–242. https://doi.org/10.1016/j.chroma.2004.02.020
Lutz M, Martínez A, Martínez E (2013) Daidzein and genistein contents in seeds of quinoa (Chenopodium quinoa Willd.) from local ecotypes grown in arid Chile. Ind Crop Prod 49:117–121
Mahdavian K, Ghorbanli M, Kh MK (2008) The effects of ultraviolet radiation on the contents of chlorophyll, flavonoid, anthocyanin and proline in Capsicum annuum L. Turk J Bot 32(2008):25–33
Mazur WM, Duke JA, Wähälä K et al (1998) Isoflavonoids and lignans in legumes: nutritional and health aspects in humans. J Nutr Biochem 9:193–200. https://doi.org/10.1016/S0955-2863(97)00184-8
Megías C, Cortés-giraldo I, Alaiz M et al (2016) Isoflavones in chickpea (Cicer arietinum) protein concentrates. J Funct Foods 21:186–192. https://doi.org/10.1016/j.jff.2015.12.012
Meng C, Zhang S, Deng Y-S et al (2015) Overexpression of a tomato flavanone 3-hydroxylase-like protein gene improves chilling tolerance in tobacco. Plant Physiol Biochem 96:388–400. https://doi.org/10.1016/j.plaphy.2015.08.019
Moorby JM, Fraser MD, Theobald VJ et al (2004) The effect of red clover formononetin content on live-weight gain, carcass characteristics and muscle equol content of finishing lambs. Anim Sci 79:303–313. https://doi.org/10.1017/S1357729800090160
Morkunas I, Marczak Ł, Stachowiak J, Stobiecki M (2005) Sucrose-induced lupine defense against Fusarium oxysporum. Plant Physiol Biochem 43:363–373. https://doi.org/10.1016/j.plaphy.2005.02.011
Mortensen A, Kulling SE, Schwartz H et al (2009) Analytical and compositional aspects of isoflavones in food and their biological effects. Mol Nutr Food Res 53:S266–S309. https://doi.org/10.1002/mnfr.200800478
Mun S-C, Mun G-S (2015) Dynamics of phytoestrogen, isoflavonoids, and its isolation from stems of Pueraria lobata (Willd.) Ohwi growing in Democratic People’s Republic of Korea. J Food Drug Anal 23:538–544. https://doi.org/10.1016/j.jfda.2015.04.003
Murphy PA, Barua K, Hauck CC (2002) Solvent extraction selection in the determination of isoflavones in soy foods. J Chromatogr B 777:129–138. https://doi.org/10.1016/S1570-0232(02)00342-2
Nguyen HQ, Hong T, Le T et al (2020) Overexpressing GmCHI1A increases the isoflavone content of transgenic soybean (Glycine max (L.) Merr.) seeds. In Vitro Cell Dev Biol Plant. https://doi.org/10.1007/s11627-020-10076-x
Ohara M, Shimamoto Y (2002) Importance of genetic characterization and conservation of plant genetic resources: the breeding system and genetic diversity of wild soybean (Glycine soja). Plant Species Biol 17:51–58. https://doi.org/10.1046/j.1442-1984.2002.00073.x
Oniszczuk A, Podgórski R (2015) Influence of different extraction methods on the quantification of selected flavonoids and phenolic acids from Tilia cordata inflorescence. Ind Crops Prod 76:509–514. https://doi.org/10.1016/j.indcrop.2015.07.003
Ono NN, Tian L (2011) The multiplicity of hairy root cultures: prolific possibilities. Plant Sci 180:439–446. https://doi.org/10.1016/j.plantsci.2010.11.012
Pandey A, Misra P, Khan MP et al (2014) Co-expression of Arabidopsis transcription factor, AtMYB12, and soybean isoflavone synthase, GmIFS1, genes in tobacco leads to enhanced biosynthesis of isoflavones and flavonols resulting in osteoprotective activity. Plant Biotechnol J 12:69–80. https://doi.org/10.1111/pbi.12118
Park ELT (2016) Isoflavones and biotransformed dihydrodaidzein in hairy roots of Korean wild arrowroot. J Plant Biotechnol 43:125–131
Park YJ, Thwe AA, Li X et al (2015) Triterpene and flavonoid biosynthesis and metabolic profiling of hairy roots, adventitious roots, and seedling roots of Astragalus membranaceus. J Agric Food Chem 63:8862–8869. https://doi.org/10.1021/acs.jafc.5b02525
Pluempanupat S, Kumrungsee N, Pluempanupat WNK, Chavasiri W et al (2013) Laboratory evaluation of Dalbergia oliveri (Fabaceae: Fabales) extracts and isolated isoflavonoids on Aedes aegypti (Diptera: Culicidae) mosquitoes. Ind Crop Prod 44:653–658
Popa D-S, Rusu ME (2017) Isoflavones: vegetable sources, biological activity, and analytical methods for their assessment. Superfood and functional food—the development of superfoods and their roles as medicine. InTech
Pueppke SG (1996) The genetic and biochemical basis for nodulation of legumes by Rhizobia. Crit Rev Biotechnol 16:1–51. https://doi.org/10.3109/07388559609146599
Qi L-W, Yu Q-T, Li P et al (2006) Quality evaluation of Radix astragali through a simultaneous determination of six major active isoflavonoids and four main saponins by high-performance liquid chromatography coupled with diode array and evaporative light scattering detectors. J Chromatogr A 1134:162–169. https://doi.org/10.1016/j.chroma.2006.08.085
Qin WT, Zhang J, Wu HJ et al (2016) Effect of drought stress on biosynthesis of isoflavones in soybean seedling. J Appl Ecol 27(12):3927–3934. https://doi.org/10.13287/j.1001-9332.201612.018
Rasolohery CA, Berger M, Lygin AV et al (2008) Effect of temperature and water availability during late maturation of the soybean seed on germ and cotyledon isoflavone content and composition. J Sci Food Agric 88:218–228. https://doi.org/10.1002/jsfa.3075
Ravindranath MH, Muthugounder S, Presser N, Viswanathan S (2004) Anticancer therapeutic potential of soy isoflavone, genistein. Adv Exp Med Biol, pp 121–165
Reinli K, Block G (1996) Phytoestrogen content of foods—a compendium of literature values. Nutr Cancer 26:123–148. https://doi.org/10.1080/01635589609514470
Reis A, Boutet-Mercey S, Massot S et al (2019) Isoflavone production in hairy root cultures and plantlets of Trifolium pratense. Biotechnol Lett 41:427–442. https://doi.org/10.1007/s10529-018-02640-8
Rostagno MA, Villares A, Guillamón E et al (2009) Sample preparation for the analysis of isoflavones from soybeans and soy foods. J Chromatogr A 1216:2–29. https://doi.org/10.1016/j.chroma.2008.11.035
Rout GR, Samantaray S, Das P (2000) In vitro manipulation and propagation of medicinal plants. Biotechnol Adv 18:91–120. https://doi.org/10.1016/S0734-9750(99)00026-9
Ruelland E, Vaultier M-N, Zachowski A, Hurry V (2009) Cold signaling and cold acclimation in plants. Adv Bot Res, pp 35–150
Saini RK, Akithadevi MK, Giridhar P, Ravishankar GA (2013) Augmentation of major isoflavones in Glycine max L. through the elicitor-mediated approach. Acta Bot Croat 72:311–322. https://doi.org/10.2478/v10184-012-0023-7
Sakthivelu G, Devi MKA, Giridhar P et al (2008) Isoflavone composition, phenol content, and antioxidant activity of soybean seeds from India and Bulgaria. J Agric Food Chem 56:2090–2095. https://doi.org/10.1021/jf072939a
Sansanelli S, Zanichelli D, Filippini A et al (2014) Production of free and glycosylated isoflavones in in vitro soybean (Glycine max L.) hypocotyl cell suspensions and comparison with industrial seed extracts. Plant Cell Tissue Organ Cult 119:301–311. https://doi.org/10.1007/s11240-014-0534-0
Shih C-H, Chen Y, Wang M et al (2008) Accumulation of isoflavone genistin in transgenic tomato plants overexpressing a soybean isoflavone synthase gene. J Agric Food Chem 56:5655–5661. https://doi.org/10.1021/jf800423u
Shimamura M, Akashi T, Sakurai N et al (2007) 2-Hydroxyisoflavanone dehydratase is a critical determinant of isoflavone productivity in hairy root cultures of Lotus japonicus. Plant Cell Physiol 48:1652–1657. https://doi.org/10.1093/pcp/pcm125
Shinde AN, Malpathak N, Fulzele DP (2009a) Studied enhancement strategies for phytoestrogens production in shake flasks by suspension culture of Psoralea corylifolia. Bioresour Technol 100:1833–1839. https://doi.org/10.1016/j.biortech.2008.09.028
Shinde AN, Malpathak N, Fulzele DP (2009b) Enhanced production of phytoestrogenic isoflavones from hairy root cultures of Psoralea corylifolia L. using elicitation and precursor feeding. Biotechnol Bioprocess Eng 14:288–291. https://doi.org/10.1007/s12257-008-0238-6
Shinde AN, Malpathak N, Fulzele DP (2010a) Impact of nutrient components on production of the phytoestrogens daidzein and genistein by hairy roots of Psoralea corylifolia. J Nat Med 64:346–353. https://doi.org/10.1007/s11418-010-0419-4
Shinde AN, Malpathak N, Fulzele DP (2010b) Determination of isoflavone content and antioxidant activity in Psoralea corylifolia L. callus cultures. Food Chem 118:128–132. https://doi.org/10.1016/j.foodchem.2009.04.093
Shirley BW (1996) Flavonoid biosynthesis: ‘new’ functions for an ‘old’ pathway. Trends Plant Sci 1:377–382. https://doi.org/10.1016/S1360-1385(96)80312-8
Sivakumar G (2006) Bioreactor technology: a novel industrial tool for high-tech production of bioactive molecules and biopharmaceuticals from plant roots. Biotechnol J 1:1419–1427. https://doi.org/10.1002/biot.200600117
Skalicky M, Kubes J, Hejnak V et al (2018) Isoflavones production and possible mechanism of their exudation in Genista tinctoria L. suspension culture after treatment with vanadium compounds. Molecules 23:1619. https://doi.org/10.3390/molecules23071619
Smetanska I (2008) Production of secondary metabolites using plant cell cultures. Food biotechnology. Springer Berlin Heidelberg, Berlin, pp 187–228
Smit G, Puvanesarajah V, Carlson R et al (1992) Bradyrhizobium japonicum nodD1 can be specifically induced by soybean flavonoids that do not induce the nodYABCSUIJ operon. J Biol Chem 267:310–318
Speijers MHM, Fraser MD, Theobald VJ, Haresign W (2005) Effects of ensiled forage legumes on performance of store finishing lambs. Anim Feed Sci Technol 120:203–216. https://doi.org/10.1016/j.anifeedsci.2005.02.027
Sreevidya V (2006) Metabolic engineering of rice with soybean isoflavone synthase for promoting nodulation gene expression in rhizobia. J Exp Bot 57:1957–1969. https://doi.org/10.1093/jxb/erj143
Stougaard J (2001) Genetics and genomics of root symbiosis. Curr Opin Plant Biol 4:328–335. https://doi.org/10.1016/S1369-5266(00)00181-3
Subramanian S, Graham MY, Yu O, Graham TL (2005) RNA interference of soybean isoflavone synthase genes leads to silencing in tissues distal to the transformation site and to enhanced susceptibility to Phytophthora sojae. Plant Physiol 137:1345–1353. https://doi.org/10.1104/pp.104.057257
Sukumaran A, McDowell T, Chen L et al (2018) Isoflavonoid-specific prenyltransferase gene family in soybean: GmPT01, a pterocarpan 2-dimethylallyltransferase involved in glyceollin biosynthesis. Plant J 96:966–981. https://doi.org/10.1111/tpj.14083
Thanonkeo S, Panichajakul S (2006) Production of isoflavones, daidzein and genistein in callus cultures of Pueraria candollei Wall. ex Benth. Nutraceutical Funct Food 28:45–53
Theboral J, Sivanandhan G, Subramanyam K et al (2014) Enhanced production of isoflavones by elicitation in hairy root cultures of Soybean. Plant Cell Tissue Organ Cult 117:477–481. https://doi.org/10.1007/s11240-014-0450-3
Thiem B, Kikowska M, Maliński MP et al (2017) Ecdysteroids: production in plant in vitro cultures. Phytochem Rev 16:603–622. https://doi.org/10.1007/s11101-016-9483-z
Thwe A et al (2012) Production of astragaloside and flavones from adventitious root cultures of Astragalus membranaceus var. mongholicus. Plant Omics 5:466–470
Toda K, Takahashi R, Iwashina T, Hajika M (2011) Difference in chilling-induced flavonoid profiles, antioxidant activity and chilling tolerance between soybean near-isogenic lines for the pubescence color gene. J Plant Res 124:173–182. https://doi.org/10.1007/s10265-010-0345-2
Tsao R (2010) Chemistry and biochemistry of dietary polyphenols. Nutrients 2:1231–1246. https://doi.org/10.3390/nu2121231
Tsukamoto C, Shimada S, Igita K et al (1995) Factors affecting isoflavone content in soybean seeds: changes in isoflavones, saponins, and composition of fatty acids at different temperatures during seed development. J Agric Food Chem 43:1184–1192. https://doi.org/10.1021/jf00053a012
Tůmová L, Tůma J (2011) The effect of UV light on isoflavonoid production in Genista tinctoria culture in vitro. Acta Physiol Plant 33:635–640. https://doi.org/10.1007/s11738-010-0566-y
Udomsin O, Yusakul G, Kitisripanya T et al (2020) The deoxymiroestrol and isoflavonoid production and their elicitation of cell suspension cultures of Pueraria candollei var. mirifica: from shake flask to bioreactor. Appl Biochem Biotechnol 190:57–72. https://doi.org/10.1007/s12010-019-03094-y
Udomsuk L, Jarukamjorn K, Tanaka H, Putalun W (2011) Improved isoflavonoid production in Pueraria candollei hairy root cultures using elicitation. Biotechnol Lett 33:369–374. https://doi.org/10.1007/s10529-010-0417-3
van Rhijn P, Vanderleyden J (1995) The rhizobium-plant symbiosis. Microbiol Rev 59:124–142
Veitch NC (2007) Isoflavonoids of the leguminosae. Nat Prod Rep 24:417. https://doi.org/10.1039/b511238a
Veitch NC (2013) Isoflavonoids of the leguminosae. Nat Prod Rep 30:988. https://doi.org/10.1039/c3np70024k
Verpoorte R, Contin A, Memelink J (2002) Biotechnology for the production of plant secondary metabolites. Phytochem Rev 1:13–25. https://doi.org/10.1023/A:1015871916833
Vongsak B, Sithisarn P, Mangmool S et al (2013) Maximizing total phenolics, total flavonoids contents and antioxidant activity of Moringa oleifera leaf extract by the appropriate extraction method. Ind Crops Prod 44:566–571. https://doi.org/10.1016/j.indcrop.2012.09.021
Wang JW, Wu JY (2013) Effective elicitors and process strategies for enhancement of secondary metabolite production in hairy root cultures. Adv Biochem Eng Biotechnol, pp 55–89
Wang M, Guo J, Qi W et al (2013) An effective and green method for the extraction and purification of aglycone isoflavones from soybean. Food Sci Biotechnol 22:705–712. https://doi.org/10.1007/s10068-013-0135-4
Wang X, Liu S, Yin X et al (2019) Soybean seed isoflavones respond differentially to phosphorus applications in low and high phosphorus soils. Nutr Cycl Agroecosyst 113:217–230. https://doi.org/10.1007/s10705-019-09982-3
Xiao CW (2008) Health effects of soy protein and isoflavones in humans. J Nutr 138:1244S-1249S. https://doi.org/10.1093/jn/138.6.1244S
Xiao B-X, Feng L, Cao F-R et al (2016) Pharmacokinetic profiles of the five isoflavonoids from Pueraria lobata roots in the CSF and plasma of rats. J Ethnopharmacol 184:22–29. https://doi.org/10.1016/j.jep.2016.02.027
Xu H, He C (2007) Separation and purification of puerarin with solvent extraction. Sep Purif Technol 56:397–400. https://doi.org/10.1016/j.seppur.2007.06.003
Yang F, Ma Y, Ito Y (2001) Separation and purification of isoflavones from a crude soybean extract by high-speed counter-current chromatography. J Chromatogr A 928:163–170. https://doi.org/10.1016/S0021-9673(01)01144-X
Ye Y-B, Tang X-Y, Verbruggen MA, Su Y-X (2006) Soy isoflavones attenuate bone loss in early postmenopausal Chinese women. Eur J Nutr 45:327–334. https://doi.org/10.1007/s00394-006-0602-2
Yu O, Jung W, Shi J et al (2000) Production of the isoflavones genistein and daidzein in non-legume dicot and monocot tissues. Plant Physiol 124(2):781–794. https://doi.org/10.1104/pp.124.2.781
Zaheer M, Reddy VD, Giri CC (2015) Enhanced daidzin production from jasmonic and acetyl salicylic acid elicited hairy root cultures of Psoralea corylifolia L. (Fabaceae). Nat Prod Res 30:1542–1547. https://doi.org/10.1080/14786419.2015.1054823
Zhang F, Smith DL (1996) Genistein accumulation in soybean (Glycine max [L.] Merr.) root systems under suboptimal root zone temperatures. J Exp Bot 47:785–792. https://doi.org/10.1093/jxb/47.6.785
Zhang H, Yang X, Zhao L, Wang Y (2009a) Ultrasonic assisted extraction of epimedin C from fresh leaves of Epimedium and extraction mechanism. Innov Food Sci Emerg Technol 10:54–60
Zhang HC, Liu JM, Lu HY, Gao SL (2009b) Enhanced flavonoid production in hairy root cultures of Glycyrrhiza uralensis Fisch by combining the over-expression of chalcone isomerase gene with the elicitation treatment. Plant Cell Rep 28:1205–1213. https://doi.org/10.1007/s00299-009-0721-3
Zhang Z-Z, Li X-X, Chu Y-N et al (2012) Three types of ultraviolet irradiation differentially promote expression of shikimate pathway genes and production of anthocyanins in grape berries. Plant Physiol Biochem 57:74–83. https://doi.org/10.1016/j.plaphy.2012.05.005
Zhang P, Du H, Wang J et al (2020) Multiplex CRISPR/Cas9-mediated metabolic engineering increases soya bean isoflavone content and resistance to soya bean mosaic virus. Plant Biotechnol J 18:1384–1395. https://doi.org/10.1111/pbi.13302
Zhou H-Y, Liu C-Z (2006) Microwave-assisted extraction of solanesol from tobacco leaves. J Chromatogr A 1129:135–139. https://doi.org/10.1016/j.chroma.2006.07.083
Acknowledgements
The authors are thankful to the Department of Science and Technology (DST), Ministry of Science and Technology, Government of India, (Reference no: SR/WOS-A/LS-405/2018) for the financial support provided to Vidya Nandakumar to carry out the present work.
Author information
Authors and Affiliations
Contributions
MA: conception and design of the article; NV: wrote the manuscript with the assistance of MA; KS and JH: preparation of tables; KK and JS: conceptualization and preparation of figures; PG, RR, and AU: critical reviewing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Vidya, N., Saravanan, K., Halka, J. et al. An insight into in vitro strategies for bioproduction of isoflavones. Plant Biotechnol Rep 15, 717–740 (2021). https://doi.org/10.1007/s11816-021-00711-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11816-021-00711-3