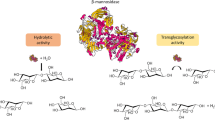
Abstract
Mannan is a predominant constituent of cork hemicellulose and is widely distributed in various plant tissues. β-Mannanase is the principal mannan-degrading enzyme, which breaks down the β-1,4-linked mannosidic bonds in mannans in an endo-acting manner. Microorganisms are a valuable source of β-mannanase, which exhibits catalytic activity in a wide range of pH and temperature, making it highly versatile and applicable in pharmaceuticals, feed, paper pulping, biorefinery, and other industries. Here, the origin, classification, enzymatic properties, molecular modification, immobilization, and practical applications of microbial β-mannanases are reviewed, the future research directions for microbial β-mannanases are also outlined.



Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Adiguzel A, Nadaroglu H, Adiguzel G (2015) Purification and characterization of β-mannanase from Bacillus pumilus (M27) and its applications in some fruit juices. J Food Sci Technol 52:5292–5298. https://doi.org/10.1007/s13197-014-1609-y
Adiguzel G, Sonmez Z, Adiguzel A, Nadaroglu H (2016) Purification and characterization of a Thermostable endo-beta-1,4 mannanase from Weissella viridescens LB37 and its application in fruit juice clarification. Eur Food Res Technol 242:769–776. https://doi.org/10.1007/s00217-015-2584-x
Barati R (2015) Application of nanoparticles as fluid loss control additives for hydraulic fracturing of tight and ultra-tight hydrocarbon-bearing formations. J Nat Gas Sci Eng 27:1321–1327. https://doi.org/10.1016/j.jngse.2015.03.028
Blibech M, Ghorbel RE, Fakhfakh I, Ntarima P, Piens K, Bacha AB, Chaabouni SE (2010) Purification and characterization of a low Molecular Weight of β-Mannanase from Penicillium Occitanis Pol6. Appl Biochem Biotechnol 160:1227–1240. https://doi.org/10.1007/s12010-009-8630-z
Cai H, Shi P, Luo H, Bai Y, Huang H, Yang P, Yao B (2011a) Acidic β-mannanase from Penicillium pinophilum C1: Cloning, characterization and assessment of its potential for animal feed application. J Biosci Bioeng 112:551–557. https://doi.org/10.1016/j.jbiosc.2011.08.018
Cai H, Shi P, Huang H, Luo H, Bai Y, Yang P, Meng K, Yao B (2011b) An acidic β-mannanase from Penicillium sp. C6: gene cloning and over-expression in Pichia pastoris. World J Microbiol Biotechnol 27:2813–2819. https://doi.org/10.1007/s11274-011-0759-6
Chai SY, Abu Bakar FD, Mahadi NM, Murad AMA (2016) A thermotolerant Endo-1,4-β-mannanase from Trichoderma virens UKM1: Cloning, recombinant expression and characterization. J Mol Catal B-Enzym 125:49–57. https://doi.org/10.1016/j.molcatb.2015.12.011
Chandra MRS, Lee YS, Park IH, Zhou Y, Kim KK, Choi YL (2011) Isolation, purification and characterization of a thermostable β-mannanase from Paenibacillus sp. DZ3. J Korean Soc Appl Biol Chem 54:325–331. https://doi.org/10.3839/jksabc.2011.052
Chauhan PS, Puri N, Sharma P, Gupta N (2012) Mannanases: microbial sources, production, properties and potential biotechnological applications. Appl Microbiol Biotechnol 93:1817–1830. https://doi.org/10.1007/s00253-012-3887-5
Chauhan PS, George N, Sondhi S, Puri N, Gupta N (2014a) An overview of purification strategies for microbial mannanases. Int J Pharm Bio Sci 5:176–192
Chauhan PS, Sharma P, Puri N, Gupta N (2014b) Purification and characterization of an alkali-thermostable β-mannanase from Bacillus nealsonii PN-11 and its application in mannooligosaccharides preparation having prebiotic potential. Eur Food Res Technol 238:927–936. https://doi.org/10.1007/s00217-014-2170-7
Chauhan PS, Tripathi SP, Sangamwar AT, Puri N, Sharma P, Gupta N (2015) Cloning, molecular modeling, and docking analysis of alkali-thermostable β-mannanase from Bacillus nealsonii PN-11. Appl Microbiol Biotechnol 99:8917–8925. https://doi.org/10.1007/s00253-015-6613-2
Chen CY, Huang YC, Yang TY, Jian JY, Chen WL, Yang CH (2016) Degradation of Konjac Glucomannan by Thermobifida fusca thermostable β-mannanase from yeast transformant. Int J Biol Macromol 82:1–6. https://doi.org/10.1016/j.ijbiomac.2015.10.008
Chen C, Li K, Li T, Li J, Liu Q, Yin H (2023a) Identification and characterization of a Novel Mannanase from Klebsiella grimontii. Bioengineering 10:1230. https://doi.org/10.3390/bioengineering10101230
Chen X, Tian Z, Zhou H, Zhou G, Cheng H (2023b) Enhanced enzymatic performance of β-Mannanase immobilized on Calcium Alginate beads for the generation of Mannan Oligosaccharides. Foods 12:3089. https://doi.org/10.3390/foods12163089
Cheng L, Duan S, Feng X, Zheng K, Yang Q, Liu Z (2016) Purification and characterization of a thermostable β-Mannanase from Bacillus subtilis BE-91: potential application in Inflammatory diseases. Biomed Res Int 2016:6380147. https://doi.org/10.1155/2016/6380147
Chopade P, Fontenelle L, Reddy BR, Coria B (2015) Novel stabilized enzyme Breaker for Fracturing Applications. SPE Prod Oper Symp 1–9. https://doi.org/10.2118/173583-MS
Cong H, Ma Z, Hu M, Han J, Wang X, Han Y, Li Y, Sun G (2022) Surface gelatin-coated β-Mannanase-immobilized lignin for delayed release of β-Mannanase to Remediate Guar-based fracturing fluid damage. ACS Omega 7:11722–11730. https://doi.org/10.1021/acsomega.1c06817
Couturier M, Féliu J, Bozonnet S, Roussel A, Berrin JG (2013) Molecular engineering of fungal GH5 and GH26 beta-(1,4)-mannanases toward improvement of enzyme activity. PLoS ONE 8:e79800. https://doi.org/10.1371/journal.pone.0079800
Couturier M, Touvrey-Loiodice M, Terrapon N, Drula E, Buon L, Chirat C, Henrissat B, Helbert W (2022) Functional exploration of the glycoside hydrolase family GH113. PLoS ONE 17:e0267509. https://doi.org/10.1371/journal.pone.0267509
David A, Singh Chauhan P, Kumar A, Angural S, Kumar D, Puri N, Gupta N (2018) Coproduction of protease and mannanase from Bacillus nealsonii PN-11 in solid state fermentation and their combined application as detergent additives. Int J Biol Macromol 108:1176–1184. https://doi.org/10.1016/j.ijbiomac.2017.09.037
Dawood A, Ma K (2020) Applications of Microbial β-Mannanases. Front Bioeng Biotech 8:598630. https://doi.org/10.3389/fbioe.2020.598630
Dhawan S, Kaur J (2007) Microbial mannanases: an overview of production and applications. Crit Rev Biotechnol 27:197–216. https://doi.org/10.1080/07388550701775919
Dhawan S, Singh R, Kaur R, Kaur J (2016) A β-mannanase from Paenibacillus sp.: optimization of production and its possible prebiotic potential. Biotechnol Appl Biochem 63:669–678. https://doi.org/10.1002/bab.1419
Dhiman S, Srivastava B, Singh G, Khatri M, Arya SK (2020) Immobilization of mannanase on sodium alginate-grafted-β-cyclodextrin: an easy and cost effective approach for the improvement of enzyme properties. Int J Biol Macromol 156:1347–1358. https://doi.org/10.1016/j.ijbiomac.2019.11.175
Do BC, Dang TT, Berrin JG, Haltrich D, To KA, Sigoillot JC, Yamabhai M (2009) Cloning, expression in Pichia pastoris, and characterization of a thermostable GH5 mannan endo-1,4-beta-mannosidase from Aspergillus niger BK01. Microb Cell Fact 8:59. https://doi.org/10.1186/1475-2859-8-59
Eom GT, Oh JY, Park JH, Jegal J, Song JK (2016) Secretory production of enzymatically active endo-β-1,4-mannanase from Bacillus subtilis by ABC exporter in Escherichia coli. Process Biochem 51:999–1005. https://doi.org/10.1016/j.procbio.2016.05.004
Ergün BG, Çalık P (2016) Lignocellulose degrading extremozymes produced by Pichia pastoris: current status and future prospects. Bioproc Biosyst Eng 39:1–36. https://doi.org/10.1007/s00449-015-1476-6
Fu X, Huang X, Liu P, Lin L, Wu G, Li C, Feng C, Hong Y (2010) Cloning and characterization of a novel mannanase from Paenibacillus sp. BME-14. J Microbiol Biotechnol 20:518–524
Gao J, Zheng H, Wang X, Li Y (2023) Characterization of a novel GH26 β-mannanase from Paenibacillus polymyxa and its application in the production of mannooligosaccharides. Enzyme Microb Technol 165:110197. https://doi.org/10.1016/j.enzmictec.2023.110197
Guo S, Tang JJ, Wei DZ, Wei W (2014) Construction of a shuttle vector for protein secretory expression in Bacillus subtilis and the application of the mannanase functional heterologous expression. J Microbiol Biotechnol 24:431–439. https://doi.org/10.4014/jmb.1311.11009
Hakamada Y, Ohkubo Y, Ohashi S (2014) Purification and characterization of β-mannanase from Reinekea sp. KIT-YO10 with transglycosylation activity. Biosci Biotech Biochem 78:722–728. https://doi.org/10.1080/09168451.2014.895658
Haq IU, Shakoor S, Nawaz A, Arshad Y, Mukhtar H (2021) Purification and characterization of Mannanase from Aspergillus Awamori for Fruit Juice Clarification. Protein Pept Lett 28:459–468. https://doi.org/10.2174/0929866527666200916142305
Hu K, Li CX, Pan J, Ni Y, Zhang XY, Xu JH (2014) Performance of a new thermostable mannanase in breaking guar-based fracturing fluids at high temperatures with little premature degradation. Appl Biochem Biotechnol 172:1215–1226. https://doi.org/10.1007/s12010-013-0484-8
Huang JW, Chen CC, Huang CH, Huang TY, Wu TH, Cheng YS, Ko TP, Lin CY, Liu JR, Guo RT (2014) Improving the specific activity of β-mannanase from Aspergillus Niger BK01 by structure-based rational design. Biochim Biophys Acta 1844:663–669. https://doi.org/10.1016/j.bbapap.2014.01.011
Ishii J, Okazaki F, Djohan AC, Hara KY, Asai-Nakashima N, Teramura H, Andriani A, Tominaga M, Wakai S, Kahar P, Yopi, Prasetya B, Ogino C, Kondo A (2016) From mannan to bioethanol: cell surface co-display of β-mannanase and β-mannosidase on yeast Saccharomyces cerevisiae. Biotechnol Biofuels 9:188. https://doi.org/10.1186/s13068-016-0600-4
Jana UK, Suryawanshi RK, Prajapati BP, Soni H, Kango N (2018) Production optimization and characterization of mannooligosaccharide generating β-mannanase from aspergillus oryzae. Bioresource Technol 268:308–314. https://doi.org/10.1016/j.biortech.2018.07.143
Jana UK, Suryawanshi RK, Prajapati BP, Kango N (2021) Prebiotic mannooligosaccharides: synthesis, characterization and bioactive properties. Food Chem 342:128328. https://doi.org/10.1016/j.foodchem.2020.128328
Ji H, Cao H, Zhao L, Na R, Ping W, Ge J, Zhao D (2022) The response surface optimization of β-mannanase produced by Weissella cibaria F1 and its potential in juice clarification. Prep Biochem Biotech 52:1151–1159. https://doi.org/10.1080/10826068.2022.2033993
Jiang Z, Wei Y, Li D, Li L, Chai P, Kusakabe I (2006) High-level production, purification and characterization of a thermostable β-mannanase from the newly isolated Bacillus subtilis WY34. Carbohydr Polym 66:88–96. https://doi.org/10.1016/j.carbpol.2006.02.030
Jin Y, Petricevic M, John A, Raich L, Jenkins H, Portela De Souza L, Cuskin F, Gilbert HJ, Rovira C, Goddard-Borger ED, Williams SJ, Davies GJ (2016) A β-Mannanase with a Lysozyme-Like fold and a Novel Molecular Catalytic mechanism. ACS Cent Sci 2:896–903. https://doi.org/10.1021/acscentsci.6b00232
Kaira GS, Panwar D, Kapoor M (2016) Recombinant endo-mannanase (ManB-1601) production using agro-industrial residues: development of economical medium and application in oil extraction from copra. Bioresource Technol 209:220–227. https://doi.org/10.1016/j.biortech.2016.02.133
Kalidas NR, Saminathan M, Ismail IS, Abas F, Maity P, Islam SS, Manshoor N, Shaari K (2017) Structural characterization and evaluation of prebiotic activity of oil palm kernel cake mannanoligosaccharides. Food Chem 234:348–355. https://doi.org/10.1016/j.foodchem.2017.04.159
Kansoh AL, Nagieb ZA (2004) Xylanase and mannanase enzymes from Streptomyces galbus NR and their use in biobleaching of softwood kraft pulp. Antonie Van Leeuwenhoek 85:103–114. https://doi.org/10.1023/b:Anto.0000020281.73208.62
Katsimpouras C, Dimarogona M, Petropoulos P, Christakopoulos P, Topakas E (2016) A thermostable GH26 endo-β-mannanase from Myceliophthora Thermophila capable of enhancing lignocellulose degradation. Appl Microbiol Biotechnol 100:8385–8397. https://doi.org/10.1007/s00253-016-7609-2
Ko H, Kang HK, Moturi J, Ingale SL, Kim J (2021) Supplementation of enzyme cocktail in chickens diet is an effective approach to increase the utilization of nutrient in wheat-based diets. J Anim Sci Technol 63:69–76. https://doi.org/10.5187/jast.2021.e1
Kong H, Jiang X, Wang Y, Huang H, Luo H (2020) Fusion expression of β-mannanase Man5A and xylanase Tlxyn11B in Pichia pastoris. Chin J Biotechnol 36:1849–1858. https://doi.org/10.13345/j.cjb.200051
Kumagai Y, Usuki H, Yamamoto Y, Yamasato A, Arima J, Mukaihara T, Hatanaka T (2011) Characterization of calcium ion sensitive region for β-mannanase from Streptomyces Thermolilacinus. Biochim Biophys Acta 1814:1127–1133. https://doi.org/10.1016/j.bbapap.2011.04.017
Li J, Wei X, Tang C, Wang J, Zhao M, Pang Q, Wu M (2014) Directed modification of the aspergillus usamii β-mannanase to improve its substrate affinity by in silico design and site-directed mutagenesis. J Ind Microbiol Biotechnol 41:693–700. https://doi.org/10.1007/s10295-014-1406-7
Li YX, Yi P, Wang NN, Liu J, Liu XQ, Yan QJ, Jiang ZQ (2017) High level expression of β-mannanase (RmMan5A) in Pichia pastoris for partially hydrolyzed guar gum production. Int J Biol Macromol 105:1171–1179. https://doi.org/10.1016/j.ijbiomac.2017.07.150
Li YX, Yi P, Liu J, Yan QJ, Jiang ZQ (2018) High-level expression of an engineered β-mannanase (mRmMan5A) in Pichia pastoris for manno-oligosaccharide production using steam explosion pretreated palm kernel cake. Bioresource Technol 256:30–37. https://doi.org/10.1016/j.biortech.2018.01.138
Li YX, Wang NN, Yan QJ, Hua XH, Liu Y, Jiang ZQ (2022) A novel neutral thermophilic β-mannanase from Malbranchea cinnamomea for controllable production of partially hydrolyzed konjac powder. Appl Microbiol Biotechnol 106:1919–1932. https://doi.org/10.1007/s00253-022-11832-2
Liao H, Li S, Zheng H, Wei Z, Liu D, Raza W, Shen Q, Xu Y (2014) A new acidophilic thermostable endo-1,4-β-mannanase from Penicillium Oxalicum GZ-2: cloning, characterization and functional expression in Pichia pastoris. BMC Biotechnol 14:90. https://doi.org/10.1186/s12896-014-0090-z
Lin J, Zou Y, Ma C, She Q, Liang Y, Chen Z, Ge X (2015) Heterologous expression of Mannanase and developing a new reporter gene system in Lactobacillus casei and Escherichia coli. PLoS ONE 10:e0142886. https://doi.org/10.1371/journal.pone.0142886
Liu Q, Yang P, Luo H, Shi P, Huang H, Meng K, Yao B (2012) A Novel endo-1,4-β-Mannanase from Bispora antennata with Good Adaptation and Stability over a broad pH range. Appl Biochem Biotechnol 166:1442–1453. https://doi.org/10.1007/s12010-011-9537-z
Liu HX, Gong JS, Li H, Lu ZM, Li H, Qian JY, Xu ZH, Shi JS (2015) Biochemical characterization and cloning of an endo-1,4-β-mannanase from Bacillus subtilis YH12 with unusually broad substrate profile. Process Biochem 50:712–721. https://doi.org/10.1016/j.procbio.2015.02.011
Liu S, Cui T, Song Y (2020a) Expression, homology modeling and enzymatic characterization of a new β-mannanase belonging to glycoside hydrolase family 1 from Enterobacter aerogenes B19. Microb Cell Fact 19:142. https://doi.org/10.1186/s12934-020-01399-w
Liu Z, Ning C, Yuan M, Yang S, Wei X, Xiao M, Fu X, Zhu C, Mou H (2020b) High-level expression of a thermophilic and acidophilic β-mannanase from Aspergillus kawachii IFO 4308 with significant potential in mannooligosaccharide preparation. Bioresource Technol 295:122257. https://doi.org/10.1016/j.biortech.2019.122257
Luo H, Wang Y, Wang H, Yang J, Yang Y, Huang H, Yang P, Bai Y, Shi P, Fan Y, Yao B (2009) A novel highly acidic β-mannanase from the acidophilic fungus Bispora sp. MEY-1: gene cloning and overexpression in Pichia pastoris. Appl Microbiol Biotechnol 82:453–461. https://doi.org/10.1007/s00253-008-1766-x
Luo Z, Miao J, Li G, Du Y, Yu X (2017) A recombinant highly thermostable β-Mannanase (ReTMan26) from thermophilic Bacillus subtilis (TBS2) expressed in Pichia pastoris and its pH and temperature Stability. Appl Biochem Biotechnol 182:1259–1275. https://doi.org/10.1007/s12010-017-2397-4
Lv J, Chen Y, Pei H, Yang W, Li Z, Dong B, Cao Y (2013) Cloning, expression, and characterization of β-mannanase from Bacillus subtilis MAFIC-S11 in Pichia pastoris. Appl Biochem Biotechnol 169:2326–2340. https://doi.org/10.1007/s12010-013-0156-8
Magengelele M, Hlalukana N, Malgas S, Rose SH, van Zyl WH, Pletschke BI (2021) Production and in vitro evaluation of prebiotic manno-oligosaccharides prepared with a recombinant aspergillus Niger endo-mannanase, Man26A. Enzyme Microb Technol 150:109893. https://doi.org/10.1016/j.enzmictec.2021.109893
Malgas S, van Dyk JS, Pletschke BI (2015) A review of the enzymatic hydrolysis of mannans and synergistic interactions between β-mannanase, β-mannosidase and α-galactosidase. World J Microbiol Biotechnol 31:1167–1175. https://doi.org/10.1007/s11274-015-1878-2
Montiel MD, Hernández M, Rodríguez J, Arias ME (2002) Evaluation of an endo-beta-mannanase produced by Streptomyces Ipomoea CECT 3341 for the biobleaching of pine kraft pulps. Appl Microbiol Biotechnol 58:67–72. https://doi.org/10.1007/s00253-001-0866-7
Moreira LR, Filho EX (2008) An overview of mannan structure and mannan-degrading enzyme systems. Appl Microbiol Biotechnol 79:165–178. https://doi.org/10.1007/s00253-008-1423-4
Nguyen HM, Pham ML, Stelzer EM, Plattner E, Grabherr R, Mathiesen G, Peterbauer CK, Haltrich D, Nguyen TH (2019) Constitutive expression and cell-surface display of a bacterial β-mannanase in Lactobacillus plantarum. Microb Cell Fact 18:76. https://doi.org/10.1186/s12934-019-1124-y
Pangsri P, Piwpankaew Y, Ingkakul A, Nitisinprasert S, Keawsompong S (2015) Characterization of mannanase from Bacillus circulans NT 6.7 and its application in mannooligosaccharides preparation as prebiotic. SpringerPlus 4:771. https://doi.org/10.1186/s40064-015-1565-7
Panwar D, Kaira GS, Kapoor M (2017) Cross-linked enzyme aggregates (CLEAs) and magnetic nanocomposite grafted CLEAs of GH26 endo-β-1,4-mannanase: improved activity, stability and reusability. Int J Biol Macromol 105:1289–1299. https://doi.org/10.1016/j.ijbiomac.2017.07.154
Parvizpour S, Razmara J, Ramli AN, Md Illias R, Shamsir MS (2014) Structural and functional analysis of a novel psychrophilic β-mannanase from Glaciozyma Antarctica PI12. J Comput Aided Mol Des 28:685–698. https://doi.org/10.1007/s10822-014-9751-1
Patel K, Dudhagara P (2020) Compatibility testing and enhancing the pulp bleaching process by hydrolases of the newly isolated thermophilic Isoptericola variabilis strain UD-6. Biocatal Biotransfor 38:144–160. https://doi.org/10.1080/10242422.2019.1711067
Pradeep GC, Cho SS, Choi YH, Choi YS, Jee JP, Seong CN, Yoo JC (2016) An extremely alkaline mannanase from Streptomyces sp. CS428 hydrolyzes galactomannan producing series of mannooligosaccharides. World J Microbiol Biotechnol 32:84. https://doi.org/10.1007/s11274-016-2040-5
Rana M, Kumar A, Kumar D, Mazumder K, Gupta N (2020) Isolation and characterization of mannanolytic bacteria for the production of prebiotic mannooligosaccharides from copra meal. J Adv Sci Res 11:117–123
Rana M, Kumar D, Angural S, Warmoota R, Mazumder K, Gupta N (2023a) Hyperproduction of a bacterial mannanase and its application for production of bioactive mannooligosaccharides from agro-waste. Process Biochem 124:13–23. https://doi.org/10.1016/j.procbio.2022.10.035
Rana M, Jassal S, Yadav R, Sharma A, Puri N, Mazumder K, Gupta N (2023b) Functional β-mannooligosaccharides: sources, enzymatic production and application as prebiotics. Crit Rev Food Sci Nutr 19:1–18. https://doi.org/10.1080/10408398.2023.2222165
Regmi S, Pradeep GC, Choi YH, Choi YS, Choi JE, Cho SS, Yoo JC (2016) A multi-tolerant low molecular weight mannanase from Bacillus sp. CSB39 and its compatibility as an industrial biocatalyst. Enzyme Microb Technol 92:76–85. https://doi.org/10.1016/j.enzmictec.2016.06.018
Roque BM, Reyes GC, Tewoldebrhan TA, Apphuamy J, Lee JJ, Seo S, Kebreab E (2019) Exogenous β-mannanase supplementation improved immunological and metabolic responses in lactating dairy cows. J Dairy Sci 102:4198–4204. https://doi.org/10.3168/jds.2018-15568
Sadaqat B, Sha C, Dar MA, Dhanavade MJ, Sonawane KD, Mohamed H, Shao W, Song Y (2022) Modifying thermostability and reusability of Hyperthermophilic Mannanase by immobilization on glutaraldehyde cross-linked Chitosan beads. Biomolecules 12:999. https://doi.org/10.3390/biom12070999
Sakai K, Mochizuki M, Yamada M, Shinzawa Y, Minezawa M, Kimoto S, Murata S, Kaneko Y, Ishihara S, Jindou S, Kobayashi T, Kato M, Shimizu M (2017) Biochemical characterization of thermostable β-1,4-mannanase belonging to the glycoside hydrolase family 134 from aspergillus oryzae. Appl Microbiol Biotechnol 101:3237–3245. https://doi.org/10.1007/s00253-017-8107-x
Sakai K, Kimoto S, Shinzawa Y, Minezawa M, Suzuki K, Jindou S, Kato M, Shimizu M (2018) Characterization of pH-tolerant and thermostable GH 134 β-1,4-mannanase SsGH134 possessing carbohydrate binding module 10 from Streptomyces sp. NRRL B-24484. J Biosci Bioeng 125:287–294. https://doi.org/10.1016/j.jbiosc.2017.10.009
Sathitkowitchai W, Ayimbila F, Nitisinprasert S, Keawsompong S (2022) Selection of pretreatment method and mannanase enzyme to improve the functionality of palm kernel cake. J Biosci Bioeng 134:301–306. https://doi.org/10.1016/j.jbiosc.2022.06.016
Seesom W, Thongket P, Yamamoto T, Takenaka S, Sakamoto T, Sukhumsirichart W (2017) Purification, characterization, and overexpression of an endo-1,4-β-mannanase from thermotolerant Bacillus sp. SWU60. World J Microbiol Biotechnol 33:53. https://doi.org/10.1007/s11274-017-2224-7
Sharma K, Dhillon A, Goyal A (2018) Insights into structure and reaction mechanism of beta-mannanases. Curr Protein Pept Sci 19:34–47. https://doi.org/10.2174/1389203717666161013115724
Shi P, Yuan T, Zhao J, Huang H, Luo H, Meng K, Wang Y, Yao B (2011) Genetic and biochemical characterization of a protease-resistant mesophilic β-mannanase from Streptomyces sp. S27. J Ind Microbiol Biotechnol 38:451–458. https://doi.org/10.1007/s10295-010-0789-3
Shimizu M, Kaneko Y, Ishihara S, Mochizuki M, Sakai K, Yamada M, Murata S, Itoh E, Yamamoto T, Sugimura Y, Hirano T, Takaya N, Kobayashi T, Kato M (2015) Novel β-1,4-Mannanase belonging to a New Glycoside Hydrolase Family in Aspergillus nidulans. J Biol Chem 290:27914–27927. https://doi.org/10.1074/jbc.M115.661645
Shukor H, Abdeshahian P, Al-Shorgani NK, Hamid AA, Rahman NA, Kalil MS (2016) Saccharification of polysaccharide content of palm kernel cake using enzymatic catalysis for production of biobutanol in acetone-butanol-ethanol fermentation. Bioresource Technol 202:206–213. https://doi.org/10.1016/j.biortech.2015.11.078
Singh S, Singh G, Khatri M, Kaur A, Arya SK (2019) Thermo and alkali stable β-mannanase: characterization and application for removal of food (mannans based) stain. Int J Biol Macromol 134:536–546. https://doi.org/10.1016/j.ijbiomac.2019.05.067
Soni H, Kango N (2013) Hemicellulases in lignocellulose biotechnology: recent patents. Recent Pat Biotechnol 7:207–218. https://doi.org/10.2174/18722083113076660011
Soni H, Rawat HK, Ahirwar S, Kango N (2017) Screening, statistical optimized production, and application of β-mannanase from some newly isolated fungi. Eng Life Sci 17:392–401. https://doi.org/10.1002/elsc.201600136
Souza M, Eeckhaut V, Goossens E, Ducatelle R, Van Nieuwerburgh F, Poulsen K, Baptista AAS, Bracarense A, Van Immerseel F (2023) Guar gum as galactomannan source induces dysbiosis and reduces performance in broiler chickens and dietary β-mannanase restores the gut homeostasis. Poult Sci 102:102810. https://doi.org/10.1016/j.psj.2023.102810
Srivastava PK, Kapoor M (2017) Production, properties, and applications of endo-β-mannanases. Biotechnol Adv 35:1–19. https://doi.org/10.1016/j.biotechadv.2016.11.001
Srivastava PK, Appu Rao GAR, Kapoor M (2016) Metal-dependent thermal stability of recombinant endo-mannanase (ManB-1601) belonging to family GH 26 from Bacillus sp. CFR1601. Enzyme Microb Technol 84:41–49. https://doi.org/10.1016/j.enzmictec.2015.12.010
Sun X, Xue X, Li M, Gao F, Hao Z, Huang H, Luo H, Qin L, Yao B, Su X (2017) Efficient coproduction of Mannanase and Cellulase by the Transformation of a Codon-Optimized Endomannanase Gene from Aspergillus Niger into Trichoderma reesei. J Agric Food Chem 65:11046–11053. https://doi.org/10.1021/acs.jafc.7b05114
Suzuki K, Michikawa M, Sato H, Yuki M, Kamino K, Ogasawara W, Fushinobu S, Kaneko S (2018) Purification, cloning, functional expression, structure, and characterization of a thermostable β-Mannanase from Talaromyces Trachyspermus B168 and its efficiency in production of mannooligosaccharides from Coffee Wastes. J Appl Glycosci 65:13–21. https://doi.org/10.5458/jag.jag.JAG-2017_018
Tailford LE, Ducros VM, Flint JE, Roberts SM, Morland C, Zechel DL, Smith N, Bjørnvad ME, Borchert TV, Wilson KS, Davies GJ, Gilbert HJ (2009) Understanding how diverse beta-mannanases recognize heterogen eous substrates. Biochemistry 48:7009–7018. https://doi.org/10.1021/bi900515d
Takasuka TE, Acheson JF, Bianchetti CM, Prom BM, Bergeman LF, Book AJ, Currie CR, Fox BG (2014) Biochemical properties and atomic resolution structure of a proteolytically processed β-mannanase from cellulolytic Streptomyces sp. SirexAA-E PloS One 9:e94166. https://doi.org/10.1371/journal.pone.0094166
Tang CD, Li JF, Wei XH, Min R, Gao SJ, Wang JQ, Yin X, Wu MC (2013a) Fusing a carbohydrate-binding module into the aspergillus usamii β-mannanase to improve its thermostability and cellulose-binding capacity by in silico design. PLoS ONE 8:e64766. https://doi.org/10.1371/journal.pone.0064766
Tang CD, Guo J, Li JF, Wei XH, Hu D, Gao SJ, Yin X, Wu MC (2013b) Enhancing expression level of an acidophilic β-mannanase in Pichia pastoris by double vector system. Ann Microbiol 64:561–569. https://doi.org/10.1007/s13213-013-0689-7
Tang CD, Shi HL, Tang QH, Zhou JS, Yao LG, Jiao ZJ, Kan YC (2016) Genome mining and motif truncation of glycoside hydrolase family 5 endo-β-1,4-mannanase encoded by aspergillus oryzae RIB40 for potential konjac flour hydrolysis or feed additive. Enzyme Microb Technol 93–94:99–104. https://doi.org/10.1016/j.enzmictec.2016.08.003
von Freiesleben P, Spodsberg N, Blicher TH, Anderson L, Jørgensen H, Stålbrand H, Meyer AS, Krogh KB (2016) An Aspergillus nidulans GH26 endo-β-mannanase with a novel degradation pattern on highly substituted galactomannans. Enzyme Microb Technol 83:68–77. https://doi.org/10.1016/j.enzmictec.2015.10.011
Vu TT, Quyen DT, Dao TT, Nguyen Sle T (2012) Cloning, high-level expression, purification, and properties of a novel endo-beta-1,4-mannanase from Bacillus subtilis G1 in Pichia pastoris. J Microbiol Biotechnol 22:331–338. https://doi.org/10.4014/jmb.1106.06052
Wan J, Shahid MS, Yuan J (2023) The comparative effects of supplementing protease combined with carbohydrase enzymes on the performance and Egg n-3 Deposition of Laying Hens Fed with Corn-Flaxseed or Wheat-Flaxseed diets. Anim (Basel) 13:3510. https://doi.org/10.3390/ani13223510
Wang C, Luo H, Niu C, Shi P, Huang H, Meng K, Bai Y, Wang K, Hua H, Yao B (2015) Biochemical characterization of a thermophilic β-mannanase from Talaromyces Leycettanus JCM12802 with high specific activity. Appl Microbiol Biotechnol 99:1217–1228. https://doi.org/10.1007/s00253-014-5979-x
Wang YP, Rao B, Zhang L, Ma LX (2017) High-level expression of two thermophilic β-mannanases in Yarrowialipolytica. Protein Expr Purif 133:1–7. https://doi.org/10.1016/j.pep.2017.02.008
White D, Adhikari R, Wang J, Chen C, Lee JH, Kim WK (2021) Effects of dietary protein, energy and β-mannanase on laying performance, egg quality, and ileal amino acid digestibility in laying hens. Poult Sci 100:101312. https://doi.org/10.1016/j.psj.2021.101312
Wu M, Tang C, Li J, Zhang H, Guo J (2011) Bimutation breeding of Aspergillus Niger strain for enhancing β-mannanase production by solid-state fermentation. Carbohydr Res 346:2149–2155. https://doi.org/10.1016/j.carres.2011.06.035
Xie J, He Z, Wang Z, Wang B, Pan L (2019) Efficient expression of a novel thermophilic fungal β-mannosidase from Lichtheimia ramosa with broad-range pH stability and its synergistic hydrolysis of Locust bean gum. J Biosci Bioeng 128:416–423. https://doi.org/10.1016/j.jbiosc.2019.04.007
Xu X, Zhang Y, Meng Q, Meng K, Zhang W, Zhou X, Luo H, Chen R, Yang P, Yao B (2013) Overexpression of a fungal β-mannanase from Bispora sp. MEY-1 in maize seeds and enzyme characterization. PLoS ONE 8:e56146. https://doi.org/10.1371/journal.pone.0056146
Xu L, Zhang Y, Dong Y, Qin G, Zhao X, Shen Y (2022) Enhanced extracellular β-mannanase production by overexpressing PrsA lipoprotein in Bacillus subtilis and optimizing culture conditions. J Basic Microbiol 62:815–823. https://doi.org/10.1002/jobm.202200080
Yamabhai M, Sak-Ubol S, Srila W, Haltrich D (2016) Mannan biotechnology: from biofuels to health. Crit Rev Biotechnol 36:32–42. https://doi.org/10.3109/07388551.2014.923372
Yan S, Duan B, Liu C, Liu G, Kang L, Sun L, Yi L, Zhang Z, Liu Z, Yuan S (2023) Heterologous Expression, Purification and Characterization of an Alkalic Thermophilic β-Mannanase CcMan5C from Coprinopsis cinerea. J Fungi (Basel) 9:378. https://doi.org/10.3390/jof9030378. PMID: 36983546
Yang XS, Jiang ZB, Song HT, Jiang SJ, Madzak C, Ma LX (2009) Cell-surface display of the active mannanase in Yarrowia Lipolytica with a novel surface-display system. Biotechnol Appl Biochem 54:171–176. https://doi.org/10.1042/ba20090222
Yang F, Yang X, Li Z, Du C, Wang J, Li S (2015) Overexpression and characterization of a glucose-tolerant β-glucosidase from T. aotearoense with high specific activity for cellobiose. Appl Microbiol Biotechnol 99:8903–8915. https://doi.org/10.1007/s00253-015-6619-9
Yang W, Qiao X, Li K, Fan J, Bo T, Guo DA, Ye M (2016) Identification and differentiation of Panax ginseng, Panax Quinquefolium, and Panax notoginseng by monitoring multiple diagnostic chemical markers. Acta Pharm Sin B 6:568–575. https://doi.org/10.1016/j.apsb.2016.05.005
Yoo HY, Pradeep GC, Kim SW, Park DH, Choi YH, Suh JW, Yoo JC (2015) A novel low-molecular weight alkaline mannanase from Streptomyces tendae. Biotechnol Bioproc Eng 20:453–461. https://doi.org/10.1007/s12257-014-0885-8
You J, Liu JF, Yang SZ, Mu BZ (2016) Low-temperature-active and salt-tolerant β-mannanase from a newly isolated Enterobacter sp. strain N18. J Biosci Bioeng 121:140–146. https://doi.org/10.1016/j.jbiosc.2015.06.001
You X, Qin Z, Li YX, Yan QJ, Li B, Jiang ZQ (2018) Structural and biochemical insights into the substrate-binding mechanism of a novel glycoside hydrolase family 134 β-mannanase. Biochim Biophys Acta Gen Subj 1862:1376–1388. https://doi.org/10.1016/j.bbagen.2018.03.016
Zang H, Xie S, Wu H, Wang W, Shao X, Wu L, Rajer FU, Gao X (2015) A novel thermostable GH5_7 β-mannanase from Bacillus pumilus GBSW19 and its application in manno-oligosaccharides (MOS) production. Enzyme Microb Technol 78:1–9. https://doi.org/10.1016/j.enzmictec.2015.06.007
Zhang M, Chen XL, Zhang ZH, Sun CY, Chen LL, He HL, Zhou BC, Zhang YZ (2009) Purification and functional characterization of endo-beta-mannanase MAN5 and its application in oligosaccharide production from konjac flour. Appl Microbiol Biotechnol 83:865–873. https://doi.org/10.1007/s00253-009-1920-0
Zhang W, Liu Z, Zhou S, Mou H, Zhang R (2019) Cloning and expression of a β-mannanase gene from Bacillus sp. MK-2 and its directed evolution by random mutagenesis. Enzyme Microb Technol 124:70–78. https://doi.org/10.1016/j.enzmictec.2019.02.003
Zhang X, Chen Y, Lv Z, Zhou L, Guo Y (2024) Analysis of the effects of β-mannanase on immune function and intestinal flora in broilers fed the low energy diet based on 16S rRNA sequencing and metagenomic sequencing. Poult Sci 103:103581. https://doi.org/10.1016/j.psj.2024.103581
Zhao J, Shi P, Luo H, Yang P, Zhao H, Bai Y, Huang H, Wang H, Yao B (2010) An acidophilic and acid-stable beta-mannanase from phialophora sp. p13 with high mannan hydrolysis activity under simulated gastric conditions. J Agric Food Chem 58:3184–3190. https://doi.org/10.1021/jf904367r
Zhao D, Wang Y, Na J, Ping W, Ge J (2019) The response surface optimization of β-mannanase produced by Lactobacillus casei HDS-01 and its potential in juice clarification. Prep Biochem Biotechnol 49:202–207. https://doi.org/10.1080/10826068.2019.1566151
Zhao D, Zhang X, Wang Y, Na J, Ping W, Ge J (2020) Purification, biochemical and secondary structural characterisation of β-mannanase from Lactobacillus casei HDS-01 and juice clarification potential. Int J Biol Macromol 154:826–834. https://doi.org/10.1016/j.ijbiomac.2020.03.157
Zheng F, Basit A, Zhang Z, Zhuang H, Chen J, Zhang J (2023) Improved production of recombinant β-mannanase (TaMan5) in Pichia pastoris and its synergistic degradation of lignocellulosic biomass. Front Bioeng Biotechnol 11:1244772. https://doi.org/10.3389/fbioe.2023.1244772
Zhou J, Zhang R, Gao Y, Li J, Tang X, Mu Y, Wang F, Li C, Dong Y, Huang Z (2012) Novel low-temperature-active, salt-tolerant and proteases-resistant endo-1,4-β-mannanase from a new Sphingomonas strain. J Biosci Bioeng 113:568–574. https://doi.org/10.1016/j.jbiosc.2011.12.011
Zhou C, Xue Y, Ma Y (2018) Characterization and high-efficiency secreted expression in Bacillus subtilis of a thermo-alkaline β-mannanase from an alkaliphilic Bacillus clausii strain S10. Microb Cell Fact 17:124. https://doi.org/10.1186/s12934-018-0973-0
Acknowledgements
This work was financially supported by Key R&D Program of Shandong Province, China (2022SFGC0102), the Innovation Capability Improvement Project of technology-based medium and small enterprises in Shandong Province (2021TSGC1247).
Author information
Authors and Affiliations
Contributions
All the authors were involved in the conception and design of this review. P.W., X.P., Y.Z., P.G., Y.L and J.G. wrote the article; W.Z. prepared figures; P.G., W.Z., Y.L. and J.G. read and corrected the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, P., Pei, X., Zhou, W. et al. Research and application progress of microbial β-mannanases: a mini-review. World J Microbiol Biotechnol 40, 169 (2024). https://doi.org/10.1007/s11274-024-03985-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-024-03985-1