Abstract
The authors describe a method for the colorimetric determination of unamplified microRNA. It is based on the use of citrate-capped gold nanoparticles (AuNPs) and, alternatively, a microRNA-probe hybrid or a magnetically extracted microRNA that serve as stabilizers against the salt-induced aggregation of AuNPs. The absorbance ratios A525/A625 of the reacted AuNP solutions were used to quantify the amount of microRNA. The assay works in the range of 5–25 pmol microRNA. The lower limit of detection (LOD) is 10 pmol. The performance of the method was tested by detection of microRNA-210-3p in totally extracted urinary microRNA from normal, benign, and bladder cancer subjects. The sensitivity and specificity for qualitative detection of urinary microRNA-210-3p using the assay are 74% and 88% respectively, which is consistent with real time PCR based assays. The assay was applied to the determination of specific microRNA by using its specific oligo targeter or following magnetic isolation of the desired microRNA. The method is simple, cost-efficient, has a short turn-around time and requires minimal equipment and personnel.

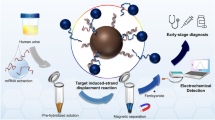
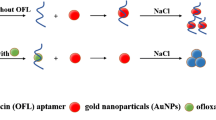
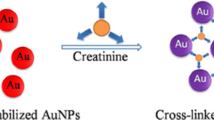
Schematic of the two detection schemes: In the first approach, matched microRNA hybridizes with its specific probe to stabilize gold nanoparticles (AuNPs) against salt induced aggregation and to leave the red color of the AuNPs unchanged. In the second one, microRNA extracted via magnetic nanoparticles (MNP) stabilizes AuNPs against aggregation.





Similar content being viewed by others
References
Ambros V (2004) The functions of animal microRNAs. Nature 431:350–355. https://doi.org/10.1038/nature02871
Krol J, Sobczak K, Wilczynska U, Drath M, Jasinska A, Kaczynska D, Krzyzosiak WJ (2004) Structural features of microRNA (miRNA) precursors and their relevance to miRNA biogenesis and small interfering RNA/short hairpin RNA design. J Biol Chem 279:42230–42239. https://doi.org/10.1074/jbc.M404931200
Iorio MV, Croce CM (2017) MicroRNA dysregulation in cancer: diagnostics, monitoring and therapeutics. A comprehensive review. EMBO Mol Med 9:582. https://doi.org/10.15252/emmm.201707779
Ben-Dov IZ, Tan YC, Morozov P, Wilson PD, Rennert H, Blumenfeld JD, Tuschl T (2014) Urine microRNA as potential biomarkers of autosomal dominant polycystic kidney disease progression: description of miRNA profiles at baseline. PLoS One 9:e86856. https://doi.org/10.1371/journal.pone.0086856
Verjans R, Van Bilsen M, Schroen B (2017) MiRNA Deregulation in Cardiac Aging and Associated Disorders. Int Rev Cell Mol Biol 334:207–263. https://doi.org/10.1016/bs.ircmb.2017.03.004
Cheng G (2015) Circulating miRNAs: roles in cancer diagnosis, prognosis and therapy. Adv Drug Deliv Rev 81:75–93. https://doi.org/10.1016/j.addr.2014.09.001
Bertoli G, Cava C, Castiglioni I (2015) MicroRNAs: New Biomarkers for Diagnosis, Prognosis, Therapy Prediction and Therapeutic Tools for Breast Cancer. Theranostics 5:1122–1143. https://doi.org/10.7150/thno.11543
Wang J, Chen J, Sen S (2016) MicroRNA as Biomarkers and Diagnostics. J Cell Physiol 231:25–30. https://doi.org/10.1002/jcp.25056
Leshkowitz D, Horn-Saban S, Parmet Y, Feldmesser E (2013) Differences in microRNA detection levels are technology and sequence dependent. RNA 19:527–538. https://doi.org/10.1261/rna.036475.112
Tian T, Wang J, Zhou X (2015) A review: microRNA detection methods. Org Biomol Chem 13:2226–2238. https://doi.org/10.1039/c4ob02104e
Hwang DW, Song IC, Lee DS, Kim S (2010) Smart magnetic fluorescent nanoparticle imaging probes to monitor micro RNAs. Small 6:81–88. https://doi.org/10.1002/smll.200901262
Li C, Li Z, Jia H, Yan J (2011) One-step ultrasensitive detection of microRNAs with loop-mediated isothermal amplification (LAMP). Chem Commun 47:2595–2597. https://doi.org/10.1039/c0cc03957h
Li F, Peng J, Wang J, Tang H, Tan L, Xie Q, Yao S (2014) Carbon nanotube-based label-free electrochemical biosensor for sensitive detection of miRNA-24. Biosens Bioelectron 54:158–164. https://doi.org/10.1016/j.bios.2013.10.061
Zhu X, Zhou X, Xing D (2013) Label-free detection of micro-RNA: two-step signal enhancement with a hairpin-probebased graphene fluorescence switch and isothermal amplification. Chemistry 19:5487–5494. https://doi.org/10.1002/chem.201204605
Mieszawska AJ, Mulder WJM, Fayad ZA, Cormode DP (2013) Multifunctional gold nanoparticles for diagnosis and therapy of disease. Mol Pharm 10:831–847. https://doi.org/10.1021/mp3005885
Rechberger W, Hohenau A, Leitner A, Krenn JR, Lamprecht B, Aussenegg FR (2003) Optical properties of two interacting gold nanoparticles. Opt Commun 220:137–141. https://doi.org/10.1016/S0030-4018(03)01357-9
Huang KS, Lin YC, Su KC, Chen HY (2007) An electroporation microchip system for the transfection of zebrafish embryos using quantum dots and GFP genes for evaluation. Biomed Microdevices 9:761–768. https://doi.org/10.1007/s10544-007-9087-x
Liandris E, Gazouli M, Andreadou M, Comor M, Abazovic N, Sechi LA, Ikonomopoulos J (2009) Direct detection of unamplified DNA from pathogenic mycobacteria using DNA-derivatized gold nanoparticles. J Microbiol Methods 78:260–264. https://doi.org/10.1016/j.mimet.2009.06.009
Nossier AI, Mohammed OS, Fakhr El-Deen RR, Zaghloul AS, Eissa S (2016) Gelatin-modified Gold Nanoparticles for Direct Detection of Urinary total Gelatinase activity: Diagnostic value in Bladder Cancer. Talanta 161:511–519. https://doi.org/10.1016/j.talanta.2016.09.015
Bonomi R, Cazzolaro A, Sansone A, Scrimin P, Prins LJ (2011) Detection of enzyme activity through catalytic signal amplification with functionalized gold nanoparticles. Angew Chem Int Ed Eng 50:2307–2312. https://doi.org/10.1002/anie.201007389
He S, Liu DB, Wang Z, Cai KY, Jiang XY (2011) Utilization of unmodified gold nanoparticles in colorimetric detection. Sci China Phys Mech Astron 54:1757–1765. https://doi.org/10.1007/s11433-011-4486-7
Li H, Rothberg LJ (2004) Colorimetric detection of DNA sequences based on electrostatic interactions with unmodified gold nanoparticles. Proc Natl Acad Sci U S A 101:14036–14039. https://doi.org/10.1073/pnas.0406115101
Li H, Rothberg LJ (2005) Detection of Specific Sequences in RNA Using Differential Adsorption of Single-Stranded Oligonucleotides on Gold Nanoparticles. Anal Chem 77:6229–6233. https://doi.org/10.1021/ac050921y
Farkhari N, Abbasian S, Moshaii A, Nikkhah M (2016) Mechanism of adsorption of single and double stranded DNA on gold and silver nanoparticles: Investigating some important parameters in bio-sensing applications. Colloids Surf B: Biointerfaces 148:657–664. https://doi.org/10.1016/j.colsurfb.2016.09.022
Hill HD, Mirkin CA (2006) The bio-barcode assay for the detection of protein and nucleic acid targets using DTT-induced ligand exchange. Nat Protoc 1:324–336. https://doi.org/10.1038/nprot.2006.51
Eissa S, Matboli M, Hegazy MG, Kotb YM, Essawy NO (2015) Evaluation of urinary microRNA panel in bladder cancer diagnosis: relation to bilharziasis. Transl Res 165:731–739. https://doi.org/10.1016/j.trsl.2014.12.008
Lorenzen JM, Volkmann I, Fiedler J, Schmidt M, Scheffner I, Haller H, Gwinner W, Thum T (2011) Urinary miR-210 as a mediator of acute T-cell mediated rejection in renal allograft recipients. Am J Transplant 11:2221–2227. https://doi.org/10.1111/j.1600-6143.2011.03679.x
Kilil GK, Tilton L, Karbiwnyk CM (2016) NaOH concentration and streptavidin bead type are key factors for optimal DNA aptamer strand separation and isolation. BioTechniques 61:114–116. https://doi.org/10.2144/000114449
Liu X, Wang Y, Chen P, Wang Y, Zhang J, Aili D, Liedberg B (2014) Biofunctionalized gold nanoparticles for colorimetric sensing of botulinum neurotoxin a light chain. Anal Chem 86:2345–2352. https://doi.org/10.1021/ac402626g
Li ZJ, Zheng XJ, Zhang L, Liang RP, Li ZM, Qiu JD (2015) Label-free colorimetric detection of biothiols utilizing SAM and un-modified Au nanoparticles. Biosens Bioelectron 68:668–674. https://doi.org/10.1016/j.bios.2015.01.062
Nelson EM, Rothberg LJ (2011) Kinetics and mechanism of single-stranded DNA adsorption onto citrate-stabilized gold nanoparticles in colloidal solution. Langmuir 27:1770–1777. https://doi.org/10.1021/la102613f
Li Y, Pu Q, Li J, Zhou L, Tao Y, Li Y, Xie G (2017) An “off-on” fluorescent switch assay for microRNA using nonenzymatic ligation-rolling circle amplification. Microchim Acta 184:4323–4330. https://doi.org/10.1007/s00604-017-2475-x
Zhou Y, Li B, Wang M, Wang J, Yin H, Ai S (2017) Fluorometric determination of microRNA based on strand displacement amplification and rolling circle amplification. Microchim Acta 184:4359–4365. https://doi.org/10.1007/s00604-017-2450-6
Shi HY, Yang L, Zhou XY, Bai J, Gao J, Jia HX, Li QG (2017) A gold nanoparticle-based colorimetric strategy coupled to duplex-specific nuclease signal amplification for the determination of microRNA. Microchim Acta 184:525–531. https://doi.org/10.1007/s00604-016-2030-1
Sang Y, Xu Y, Xu L, Cheng W, Li X, Wu J, Ding S (2017) Colorimetric and visual determination of microRNA via cycling signal amplification using T7 exonuclease. Microchim Acta 184(7):2465–2471. https://doi.org/10.1007/s00604-017-2238-8
Zeng K, Li H, Peng Y (2017) Gold nanoparticle enhanced surface plasmon resonance imaging of microRNA-155 using a functional nucleic acid-based amplification machine. Microchim Acta. https://doi.org/10.1007/s00604-017-2276-2
Borghei YS, Hosseini M, Ganjali MR (2017) Fluorescence based turn-on strategy for determination of microRNA-155 using DNA-templated copper nanoclusters. Microchim Acta 184:2671–2677. https://doi.org/10.1007/s00604-017-2272-6
Ji X, Lv H, Ma M, Lv B, Ding C (2017) An optical DNA logic gate based on strand displacement and magnetic separation, with response to multiple microRNAs in cancer cell lysates. Microchim Acta 184(8):2505–2513. https://doi.org/10.1007/s00604-017-2248-6
Paul A, Avci-Adali M, Ziemer G, Wendel HP (2009) Streptavidin-coated magnetic beads for DNA strand separation implicate a multitude of problems during cell-SELEX. Oligonucleotides 19:243–254. https://doi.org/10.1089/oli.2009.0194
Svobodová M, Pinto A, Nadal P, O’Sullivan CK (2012) Comparison of different methods for generation of single-stranded DNA for SELEX processes. Anal Bioanal Chem 404:835–842. https://doi.org/10.1007/s00216-012-6183-4
Liang C, Li D, Zhang G, Li H, Shao N, Liang Z, Zhang L, Lu A, Zhang G (2015) Comparison of the methods for generating single-stranded DNA in SELEX. Analyst 140:3439–3444. https://doi.org/10.1039/c5an00244c
Acknowledgements
This work was financially supported by the national grant: RSTDF 6635. Authors acknowledge Professor Mohamed Esmat, Prof of urology for provision of clinical samples and patient’s data. The National patent application number of this work is 1631/2017.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOCX 826 kb)
Rights and permissions
About this article
Cite this article
Nossier, A.I., Abdelzaher, H., Matboli, M. et al. Dual approach for the colorimetric determination of unamplified microRNAs by using citrate capped gold nanoparticles. Microchim Acta 185, 236 (2018). https://doi.org/10.1007/s00604-018-2767-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-018-2767-9