Abstract
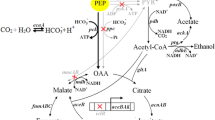
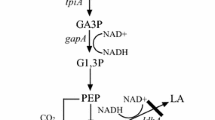
Corynebacterium glutamicum is particularly known for its potentiality in succinate production. We engineered C. glutamicum for the production of succinate. To enhance C3–C4 carboxylation efficiency, chromosomal integration of the pyruvate carboxylase gene pyc resulted in strain NC-4. To increase intracellular NADH pools, the pntAB gene from Escherichia coli, encoding for transhydrogenase, was chromosomally integrated into NC-4, leading to strain NC-5. Furthermore, we deleted pgi gene in strain NC-5 to redirect carbon flux to the pentose phosphate pathway (PPP). To solve the drastic reduction of PTS-mediated glucose uptake, the ptsG gene from C. glutamicum, encoding for the glucose-specific transporter, was chromosomally integrated into pgi-deficient strain resulted in strain NC-6. In anaerobic batch fermentation, the production of succinate in pntAB-overexpressing strain NC-5 increased by 14% and a product yield of 1.22 mol/mol was obtained. In anaerobic fed-batch process, succinic acid concentration reached 856 mM by NC-6. The yields of succinate from glucose were 1.37 mol/mol accompanied by a very low level of by-products. Activating PPP and transhydrogenase in combination led to a succinate yield of 1.37 mol/mol, suggesting that they exhibited a synergistic effect for improving succinate yield.





Similar content being viewed by others
References
Bartek T, Blombach B, Zonnchen E, Makus P, Lang S, Eikmanns BJ, Oldiges M (2010) Importance of NADPH supply for improved l-valine formation in Corynebacterium glutamicum. Biotechnol Prog 26:361–371
Blombach B, Schreiner ME, Bartek T, Oldiges M, Eikmanns BJ (2008) Corynebacterium glutamicum tailored for high-yield l-valine production. Appl Microbiol Biotechnol 79:471–479
Blombach B, Riester T, Wieschalka S, Ziert C, Youn JW, Wendisch VF, Eikmanns BJ (2011) Corynebacterium glutamicum tailored for efficient isobutanol production. Appl Environ Microbiol 77:3300–3310
Boonstra B, French CE, Wainwright I, Bruce NC (1999) The udhA gene of Escherichia coli encodes a soluble pyridine nucleotide transhydrogenase. J Bacteriol 181:1030–1034
Bozell JJ, Petersen GR (2010) Technology development for the production of biobased products from biorefinery carbohydrates-the US Department of Energyʼs “Top 10” revisited. Green Chem 12:539–554
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Canonaco F, Hess TA, Heri S, Wang T, Szyperski T, Sauer U (2001) Metabolic flux response to phosphoglucose isomerase knock-out in Escherichia coli and impact of overexpression of the soluble transhydrogenase UdhA. FEMS Microbiol Lett 204:247–252
Chen XJ, Jiang ST, Zheng Z, Pan LJ, Luo SZ (2012) Effects of culture redox potential on succinic acid production by Corynebacterium crenatum under anaerobic conditions. Process Biochem 47:1250–1255
Chin JW, Khankal R, Monroe CA, Maranas CD, Cirino PC (2009) Analysis of NADPH supply during xylitol production by engineered Escherichia coli. Biotechnol Bioeng 102:209–220
Clarke DM, Loo TW, Gillam S, Bragg PD (1986) Nucleotide sequence of the pntA and pntB genes encoding the pyridine nucleotide transhydrogenase of Escherichia coli. Eur J Biochem 158:647–653
Eggeling L, Bott M (2005) Handbook of Corynebacterium glutamicum. CRC Press, Boca Raton, pp 13–16
Hwang GH, Cho JY (2012) Implication of gluconate kinase activity in l-ornithine biosynthesis in Corynebacterium glutamicum. J Ind Microbiol Biotechnol 39:1869–1874
Ikeda M, Nakagawa S (2003) The Corynebacterium glutamicum genome: features and impacts on biotechnological processes. Appl Microbiol Biotechnol 62:99–109
Ikeda M, Mitsuhashi S, Tanaka K, Hayashi M (2009) Reengineering of a Corynebacterium glutamicum l-arginine and l-citrulline producer. Appl Environ Microbiol 75:1635–1641
Inui M, Murakami S, Okino S, Kawaguchi H, Vertes AA, Yukawa H (2004) Metabolic analysis of Corynebacterium glutamicum during lactate and succinate productions under oxygen deprivation conditions. Appl Microbiol Biotechnol 7:182–196
Jensen JVK, Wendisch VF (2013) Ornithine cyclodeaminase-based proline production by Corynebacterium glutamicum. Microb Cell Fact 12:63
Jo JH, Seol HY, Lee YB, Kim MH, Hyun HH, Lee HH (2012) Disruption of genes for enhanced biosynthesis of a-ketoglutarate in Corynebacterium glutamicum. Can J Microbiol 58:278–286
Kabus A, Georgi T, Wendisch VF, Bott M (2007) Expression of the Escherichia coli pntAB genes encoding a membrane-bound transhydrogenase in Corynebacterium glutamicum improves l-lysine formation. Appl Microbiol Biotechnol 75:47–53
Kalinowski J, Bathe B, Bartels D, Bischoff N, Bott M, Burkovski A, Dusch N, Eggeling L, Eikmanns BJ, Gaigalat L, Goesmann A, Hartmann M, Huthmacher K, Kramer R, Linke B, McHardy AC, Meyer F, Mockel B, Pfefferle W, Puhler A, Rey DA, Ruckert C, Rupp O, Sahm H, Wendisch VF, Wiegrabe I, Tauch A (2003) The complete Corynebacterium glutamicum ATCC 13032 genome sequence and its impact on the production of l-aspartate-derived amino acids and vitamins. J Biotechnol 104:5–25
Krause FS, Blombach B, Eikmanns BJ (2010) Metabolic engineering of Corynebacterium glutamicum for 2-ketoisovalerate production. Appl Environ Microbiol 76:8053–8061
Lindner SN, Petrov DP, Hagmann CT, Henrich A, Kramer R, Eikmanns BJ, Wendisch VF, Seibold GM (2013) Phosphotransferase system-mediated glucose uptake is repressed in phosphoglucoisomerase-deficient Corynebacterium glutamicum strains. Appl Environ Microbiol 79:2588–2595
Litsanov B, Brocker M, Bott M (2012) Towards homosuccinate fermentation: metabolic engineering of Corynebacterium glutamicum for anaerobic succinate production from glucose and formate. Appl Environ Microbiol 78:3325–3337
Litsanov B, Kabus A, Brocker M, Bott M (2012) Efficient aerobic succinate production from glucose in minimal medium with Corynebacterium glutamicum. Microb Biotechnol 5:116–128
Lilius EM, Multanen VM, Toivonen V (1979) Quantitative extraction and estimation of intracellular nicotinamide nucleotides in Escherichia coli. Anal Biochem 99:22–27
Lv YY, Wu ZH, Han SY (2011) Genome sequence of Corynebacterium glutamicum S9114, a strain for industrial production of glutamate. J Bacteriol 193:6096–6097
Lv YY, Liao JJ, Wu ZH, Han SY, Lin Y, Zheng SP (2012) Genome sequence of Corynebacterium glutamicum ATCC 14067, which provides insight into amino acid biosynthesis in Coryneform bacteria. J Bacteriol 194:742–743
Maki K, Uno K, Morita T, Aiba H (2008) RNA but not protein partners, is directly responsible for translational silencing by a bacterial Hfq-binding small RNA. Proc Natl Acad Sci USA 105:10332–10337
Marx A, Hans S, Mockel B, Bathe B, de Graaf AA, McCormack AC, Stapleton C, Burke K, O’Donohue M, Dunican LK (2003) Metabolic phenotype of phosphoglucose isomerase mutants of Corynebacterium glutamicum. J Biotechnol 104:185–197
Mckinlay JB, Vieille C, Zeikus JG (2007) Prospects for a bio-based succinate industry. Appl Microbiol Biothechnol 76:727–740
Morita T, Maki K, Aiba H (2005) RNase E-based ribonucleoprotein complexes: mechanical basis of mRNA destabilization mediated by bacterial noncoding RNAs. Genes Dev 19:2176–2186
Okino S, Inui M, Yukawa H (2005) Production of organic acids by Corynebacterium glutamicum under oxygen deprivation. Appl Microbiol Biotechnol 68:465–480
Okino S, Noburyu R, Suda M, Jojima T, Inui M, Yukawa H (2008) An efficient succinic acid production process in a metabolically engineered Corynebacterium glutamicum strain. Appl Microbiol Biotechnol 81:459–464
Okino S, SudaM FujikuraK, InuiM Yukawa H (2008) Production of d-lactic acid by Corynebacterium glutamicum under oxygen deprivation. Appl Microbiol Biotechnol 78:449–454
Payne J, Morri JG (1969) Pyruvate carboxylase in Rhodopseudomonas spheroids. J Gen Microbiol 59:97–101
Peters-Wendisch PG, Schiel B, Wendisch VF, Katsoulidis E, Mockel B, Sahm H, Eikmanns BJ (2001) Pyruvate carboxylase is a major bottleneck for glutamate and lysine production by Corynebacterium glutamicum. J Mol Microbiol Biotechnol 3:295–300
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual. Cold Spring Harbor, Cold Spring Harbor Laboratory
Sauer U, Canonaco F, Heri S, Perrenoud A, Fischer E (2004) The soluble and membrane-bound transhydrogenases UdhA and PntAB have divergent functions in NADPH metabolism of Escherichia coli. J Biol Chem 279:6613–6619
Schäfer A, Tauch A, Jäger W, Kalinowski J, Thierbach G, Pühler A (1994) Small mobilizable multi-purpose cloning vectors derived from the Escherichia coli plasmids pK18 and pK19: selection of defined deletions in the chromosome of Corynebacterium glutamicum. Gene 145:69–73
Stols L, Donnely MI (1997) Production of succinic acid through overexpression of NAD-dependent malic enzyme in an Escherichia coli mutant. Appl Environ Microbiol 63:2695–2701
Takors R, Bathe B, Rieping M, Hans S, Kelle R, Huthmacher K (2007) Systems biology for industrial strains and fermentation processes-example: amino acids. J Biotechnol 129:181–190
Van der Rest ME, Lange C, Molenaar D (1999) A heat shock following electroporation induces highly efficient transformation of Corynebacterium glutamicum with xenogeneic plasmid DNA. Appl Microbiol Biotechnol 52(4):541–545
Wang C, Zhang HL, Cai H, Zhou ZH, Chen YL, Chen YL, Ouyang PK (2014) Succinic acid production from corn cob hydrolysates by genetically engineered Corynebacterium glutamicum. Appl Biochem Biotechnol 172:340–350
Wang C, Cai H, Zhou ZH, Zhang K, Chen ZJ, Chen YL, Wan HG, Ouyang PK (2014) Investigation of ptsG gene in response to xylose utilization in Corynebacterium glutamicum. J Ind Microbiol Biotechnol 41:1249–1258
Werpy T, Petersen G (2004) Top value added chemicals from biomass. US Department of Energy, Office of Energy Efficiency and Renewable Energy, Office of the Biomass Program, Washington
Wieschalka S, Blombach B, Eikmanns BJ (2012) Engineering Corynebacterium glutamicum for the production of pyruvate. Appl Microbiol Biotechnol 94:449–459
Wieschalka S, Blombach B, Bott M, Eikmanns BJ (2013) Bio-based production of organic acids with Corynebacterium glutamicum. Microb Biotechnol 6:87–102
Yasuda K, Jojima T, Suda M, Okino S, Inui M, Yukawa H (2007) Analyses of the acetate-producing pathways in Corynebacterium glutamicum under oxygen-deprived conditions. Appl Microbiol Biotechnol 77:853–860
Yukawa H, Omumasaba CA, Nonaka H, Kos P, Okai N, Suzuki N, Suda N, Tsuge Y, Watanabe J, Ikeda Y, Vertès AA, Inui M (2007) Comparative analysis of the Corynebacterium glutamicum group and complete genome sequence of strain R. Microbiol-SGM 153:1042–1058
Zhou ZH, Wang C, Chen ZJ, Chen YL, Zhang K, Xu HT, Cai H (2015) Increasing available NADH supply during succinic acid production by Corynebacterium glutamicum. Biotechnol Progr 31(1):12–19
Zhu NQ, Xia HH, Yang JG, Zhao XM, Chen T (2014) Improved succinate production in Corynebacterium glutamicum by engineering glyoxylate pathway and succinate export system. Biotechnol Lett 36:553–560
Acknowledgements
This work was supported by the 973 Program of China (Grant No. 2011CB707405).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declares that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, C., Zhou, Z., Cai, H. et al. Redirecting carbon flux through pgi-deficient and heterologous transhydrogenase toward efficient succinate production in Corynebacterium glutamicum . J Ind Microbiol Biotechnol 44, 1115–1126 (2017). https://doi.org/10.1007/s10295-017-1933-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-017-1933-0