Abstract
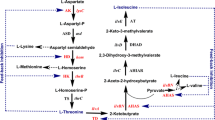
With the purpose of generating a microbial strain for l-ornithine production in Corynebacterium glutamicum, genes involved in the central carbon metabolism were inactivated so as to modulate the intracellular level of NADPH, and to evaluate their effects on l-ornithine production in C. glutamicum. Upon inactivation of the 6-phosphoglucoisomerase gene (pgi) in a C. glutamicum strain, the concomitant increase in intracellular NADPH concentrations from 2.55 to 5.75 mmol g−1 (dry cell weight) was accompanied by reduced growth rate and l-ornithine production, suggesting that l-ornithine production is not solely limited by NADPH availability. In contrast, inactivation of the gluconate kinase gene (gntK) led to a 51.8 % increase in intracellular NADPH concentration, which resulted in a 49.9 % increase in l-ornithine production. These results indicate that excess NADPH is not necessarily rate-limiting, but is required for increased l-ornithine production in C. glutamicum.


Similar content being viewed by others
References
Anderson WB, Nordlie RC (1968) Glucose dehydrogenase activity of yeast glucose 6-phosphate dehydrogenase. I. Selective stimulation by bicarbonate, phosphate, and sulfate. Biochemistry 7:1479–1485
Becker J, Klopproggr C, Herold A, Zelder O, Bolten CJ, Wittmann C (2007) Metabolic flux engineering of l-lysine production in Corynebacterium glutamicum over expression and modification of G6P dehydrogenase. J Biotechnol 132:99–109
Blombach B, Schreiner ME, Moch M, Oldiges M, Eikmanns BJ (2007) Effect of pyruvate dehydrogenase complex deficiency on l-lysine production with Corynebacterium glutamicum. Appl Microbiol Biotechnol 76:615–623
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Canonaco F, Hess TA, Heri S, Wang T, Szyperski T, Sauer U (2001) Metabolic flux response to phosphoglucose isomerase knock-out in Escherichia coli and impact of overexpression of the soluble transhydrogenase UdhA. FEMS Microbiol Lett 204:247–252
Cunin R, Glansdorff N, Pierard A, Stalon V (1986) Biosynthesis and metabolism of arginine in bacteria. Microbiol Rev 50:314–352
Fiaux J, Çakar P, Sonderegger M, Wüthrich K, Szyperski T, Sauer U (2003) Metabolic-flux profiling of the yeasts Saccharomyces cerevisiae and Pichia stipitis. Eukaryot Cell 2:170–180
Frunzke J, Engels V, Hasenbein S, Gätgens C, Bott M (2008) Co-ordinated regulation of gluconate catabolism and glucose uptake in Corynebacterium glutamicum by two functionally equivalent transcriptional regulators, GntR1 and GntR2. Mol Microbiol 67:305–322
Georgi T, Rittmann D, Wendisch VF (2005) Lysine and glutamate production by Corynebacterium glutamicum on glucose, fructose and sucrose: roles of malic enzyme and fructose-1,6-bisphosphate. Metab Eng 7:291–301
Hwang J-H, Hwang G-H, Cho J-Y (2008) Effect of increased glutamate availability on l-ornithine production in Corynebacterium glutamicum. J Microbiol Biotechnol 18:704–710
Hwang G-H, Cho J-Y (2010) Identification of a suppressor gene for the arginine-auxotrophic argJ mutation in Corynebacterium glutamicum. J Ind Microbiol Biotechnol 37:1131–1136
Lessie TG, Phibbs PV Jr (1984) Alternative pathways of carbohydrate utilization in pseudomonads. Ann Rev Microbiol 38:359–387
Marx A, Striegel K, de Graaf AA, Sahm H, Eggeling L (1997) Response of the central metabolism of Corynebacterium glutamicum to different flux burdens. Biotechnol Bioeng 56:168–180
Marx A, Eikmanns BJ, Sahm H, de Graaf AA, Eggeling L (1999) Response of the central metabolism in Corynebacterium glutamicum to the use of an NADH-dependent glutamate dehydrogenase. Metab Eng 1:35–48
Marx A, Hans S, Möckel B, Bathe B, de Graaf AA, McCormack AC, Stapleton C, Burke K, O’Donohue M, Dunican LK (2003) Metabolic phenotype of phosphoglucose isomerase mutants of Corynebacterium glutamicum. J Biotechnol 104:185–197
Mascarenhas D, Ashworth DJ, Chen CS (1991) Deletion of pgi alters tryptophan biosynthesis in a genetically engineered strain of Escherichia coli. Appl Environ Microbiol 57:2995–2999
Mortiz B, Striegel K, de Graaf AA, Sahm H (2000) Kinetic properties of the glucose 6-phosphate and 6-phosphogluconate dehydrogenases from Corynebacterium glutamicum and their application for predicting pentose phosphate pathway flux in vivo. Eur J Biochem 267:3442–3452
Ohnishi J, Katahira R, Mitsuhashi S, Kakita S, Ikeda M (2005) A novel gnd mutation leading to increased l-lysine production in Corynebacterium glutamicum. FEMS Microbiol Lett 242:265–274
Otani M, Ihara N, Umezawa C, Sano K (1986) Predominance of gluconate formation from glucose during germination of Bacillus megaterium QM B1551 spores. J Bacteriol 167:148–152
Sauer U, Hatzimanikatis V, Hohmann HP, Manneberg M, van Loon APGM, Bailey JE (1996) Physiology and metabolic fluxes of wild-type and riboflavin-producing Bacillus subtilis. Appl Environ Microbiol 62:3687–3696
Schäfer A, Tauch A, Jäger W, Kalinowski J, Thierbach G, Pühler A (1994) Small mobilizable multi-purpose cloning vectors derived from the Escherichia coli plasmids pK18 and pK19: selection of defined selections in the chromosome of Corynebacterium glutamicum. Gene 145:69–73
Vallino J, Stephanopoulos G (1994) Carbon flux distributions at the glucose 6-phosphate branch point in Corynebacterium glutamicum during lysine production. Biotechnol Prog 10:327–334
Wittmann C, Heinzle E (2002) Genealogy profiling through strain improvement by using metabolic network analysis: metabolic flux genealogy of several generations of lysine producing corynebacteria. Appl Environ Microbiol 68:5843–5849
Yoon K-H, Cho J-Y (2007) Transcriptional analysis of the gum gene cluster from Xanthomonas oryzae pathovar oryzae. Biotechnol Lett 29:95–103
Zamboni N, Fischer E, Laudert D, Aymerich S, Hohmann H-P, Sauer U (2004) The Bacillus subtilis yqjI gene encodes the NADP+-dependent 6-P-gluconate dehydrogenase in the pentose phosphate pathway. J Bacteriol 186:4528–4534
Acknowledgments
This study was supported by the Advanced R&D supporting business between industry and University funded by the Small and Medium Business Administration, Republic of Korea, a grant from the Next-Generation BioGreen 21 Program (no. PJ0080992011), Rural Development Administration, Republic of Korea, and, in part, by the Sangji University Research Fund 2011.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hwang, GH., Cho, JY. Implication of gluconate kinase activity in l-ornithine biosynthesis in Corynebacterium glutamicum . J Ind Microbiol Biotechnol 39, 1869–1874 (2012). https://doi.org/10.1007/s10295-012-1197-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-012-1197-7