Abstract
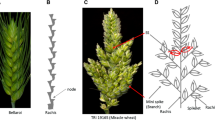
The development and morphology of the wheat spike is important because the spike is where reproduction occurs and it holds the grains until harvest. Therefore, genes that influence spike morphology are of interest from both theoretical and practical stand points. When substituted for the native chromosome 2A in the tetraploid Langdon (LDN) durum wheat background, the Triticum turgidum ssp. dicoccoides chromosome 2A from accession IsraelA confers a short, compact spike with fewer spikelets per spike compared to LDN. Molecular mapping and quantitative trait loci (QTL) analysis of these traits in a homozygous recombinant population derived from LDN × the chromosome 2A substitution line (LDNIsA-2A) indicated that the number of spikelets per spike and spike length were controlled by linked, but different, loci on the long arm of 2A. A QTL explaining most of the variation for spike compactness coincided with the QTL for spike length. Comparative mapping indicated that the QTL for number of spikelets per spike overlapped with a previously mapped QTL for Fusarium head blight susceptibility. The genes governing spike length and compactness were not orthologous to either sog or C, genes known to confer compact spikes in diploid and hexaploid wheat, respectively. Mapping and sequence analysis indicated that the gene governing spike length and compactness derived from wild emmer could be an ortholog of the barley Cly1/Zeo gene, which research indicates is an AP2-like gene pleiotropically affecting cleistogamy, flowering time, and rachis internode length. This work provides researchers with knowledge of new genetic loci and associated markers that may be useful for manipulating spike morphology in durum wheat.





Similar content being viewed by others
References
Cantrell RG, Joppa LR (1991) Genetic analysis of quantitative traits in wild emmer (Triticum turgidum L. var. dicoccoides). Crop Sci 31:645–649
Chen A, Baumann U, Fincher GB, Collins NC (2009a) Flt-2L, a locus in barley controlling flowering time, spike density, and plant height. Funct Integr Genomics 9:243–254
Chen A, Reinheimer J, Brule-Babel A, Baumann U, Pallotta M, Fincher GB, Collins NC (2009b) Genes and traits associated with chromosome 2H and 5H regions controlling sensitivity of reproductive tissues to frost in barley. Theor Appl Genet 118:1465–1476
Druka A, Franckowiak J, Lundqvist U, Bonar N, Alexander J, Houston K, Radovic S, Shahinnia F, Vendramin V, Morgante M, Stein N, Waugh R (2011) Genetic dissection of barley morphology and development. Plant Physiol 155:617–627
Endo TR, Gill BS (1996) The deletion stocks of common wheat. J Hered 87:295–307
Faris JD, Friesen TL (2009) Reevaluation of a tetraploid wheat population indicates that the Tsn1-ToxA interaction is the only factor governing susceptibility to Stagonospora nodorum blotch. Phytopathology 99:906–912
Faris JD, Gill BS (2002) Genomic targeting and high-resolution mapping of the domestication gene Q in wheat. Genome 45:706–718
Faris JD, Haen KM, Gill BS (2000) Saturation mapping of a gene-rich recombination hot spot region in wheat. Genetics 154:823–835
Faris JD, Fellers JP, Brooks SA, Gill BS (2003) A bacterial artificial chromosome contig spanning the major domestication locus Q in wheat and identification of a candidate gene. Genetics 164:311–321
Garvin DF, Stack RW, Hansen JM (2009) Quantitative trait locus mapping of increased Fusarium head blight susceptibility associated with a wild emmer wheat chromosome. Phytopathology 99:447–452
Gilsinger J, Kong L, Shen X, Ohm H (2005) DNA markers associated with low Fusarium head blight incidence and narrow flower opening in wheat. Theor Appl Genet 110:1218–1225
Gonzalez-Hernadez JL, Elias EM, Kianian SF (2004) Mapping genes for grain protein concentration and grain yield on chromosome 5B of Triticum turgidum (L.) var. dicoccoides. Euphytica 139:217–225
Gonzalez-Hernandez JL, Singh PK, Mergoum M, Adhikari TB, Kianian SF, Simsek S, Elias EM (2009) A quantitative trait locus on chromosome 5B controls resistance of Triticum turgidum (L.) var. dicoccoides to Stagonospora nodorum blotch. Euphytica 166:199–206
Houston K, McKim SM, Comadran J, Bonar N, Druka I, Uzrek N, Cirillo E, Guzy-Wrobelska J, Collins NC, Halpin C, Hansson M, Dockter C, Druka A, Waugh R (2013) Variation in the interaction between alleles of HvAPETALA2 and microRNA172 determines the density of grains on the barley inflorescence. Proc Natl Acad Sci USA 110:16675–16680
Jantasuriyarat C, Vales MI, Watson CJW, Riera-Lizarazu O (2004) Identification and mapping of genetic loci affecting the free-threshing habit and spike compactness in wheat (Triticum aestivum L.). Theor Appl Genet 108:261–273
Joehanes R, Nelson JC (2008) QGene 4.0, an extensive Java QTL-analysis platform. Bioinformatics 24:2788–2789
Johnson EB, Nalam VJ, Zemetra RS, Riera-Lizarazu O (2008) Mapping the compactum locus in wheat (Triticum aestivum L.) and its relationship to other spike morphology genes of the Triticeae. Euphytica 163:193–201
Joppa LR (1993) Chromosome engineering in tetraploid wheat. Crop Sci 33:908–913
Joppa LR, Cantrell RG (1990) Chromosomal location of genes for grain protein content of wild tetraploid wheat. Crop Sci 30:1059–1064
Joppa LR, Williams ND (1988) Langdon durum disomic substitution lines and aneuploidy analysis in tetraploid wheat. Genome 30:222–228
Joppa LR, Hareland GA, Cantrell RG (1991) Quality characteristics of the Langdon durum-dicoccoides chromosome substitution lines. Crop Sci 31:1513–1517
Joppa LR, Du C, Hart GE, Hareland GA (1997) Mapping gene(s) for grain protein in tetraploid wheat (Triticum turgidum L.) using a population of recombinant inbred chromosome lines. Crop Sci 37:1586–1589
Kato K, Miura H, Sawada S (1999) QTL mapping of genes controlling ear emergence time and plant height on chromosome 5A of wheat. Theor Appl Genet 98:472–476
Kato K, Sonokawa R, Miura H, Sawada S (2003) Dwarfing effect associated with the threshability gene Q on wheat chromosome 5A. Plant Breed 122:489–492
Klindworth DL, Hareland GA, Elias EM, Faris JD, Chao S, Xu SS (2009) Agronomic and quality characteristics of two new sets of Langdon durum-wild emmer wheat chromosome substitution lines. J Cereal Sci 50:29–35
Kosambi DD (1944) The estimation of map distances from recombination values. Ann Eugen 12:172–175
Kumar S, Gill BS, Faris JD (2007) Identification and characterization of segregation distortion loci along chromosome 5B in tetraploid wheat. Mol Genet Genomics 278:1870196
Kuspira J, Unrau J (1957) Genetic analysis of certain characters in common wheat using whole chromosome substitution lines. Can J Plant Sci 37:300–326
Lorieux M (2012) MapDisto: fast and efficient computation of genetic linkage maps. Mol Breed 30:1231–1235
Mackey J (1954) Neutron and X-ray experiments in wheat and revision of the speltoid problem. Hereditas 40:65–180
Muramatsu M (1963) Dosage effect of the spelta gene q of hexaploid wheat. Genetics 48:469–482
Muramatsu M (1979) Presence of the vulgare gene, Q, in a dense-spike variety of Triticum dicoccum Schübl. Report of the Plant Germ-Plasm Institute, Kyoto University, No. 4 pp 39–41
Muramatsu M (1985) Spike type in two cultivars of Triticum dicoccum with the spelta gene q compared with the Q-bearing variety liguliforme. Jpn J Breed 35:255–267
Muramatsu M (1986) The vulgare super gene, Q: its universality in durum wheat and its phenotypic effects in tetraploid and hexaploid wheats. Can J Genet Cytol 28:30–41
Nair SK, Wang N, Turuspekov Y, Pourkheirandish M, Sinsuwongwat S, Chen G, Sameri M, Tagiri A, Honda I, Watanabe Y, Kanamori H, Wicker T, Stein N, Nagamura Y, Matsumoto T, Komatsuda T (2010) Cleistogamous flowering in barley arises from the suppression of microRNA-guided HvAP2 mRNA cleavage. Proc Natl Acad Sci USA 107:490–495
Nalam VJ, Vales MI, Watson CJW, Kianian SF, Riera-Lizarazu O (2006) Map-based analysis of genes affecting the brittle rachis character in tetraploid wheat (Triticum turgidum L.). Theor Appl Genet 112:373–381
Ning S, Wang N, Sakuma S, Pourkheirandish M, Wu J, Matsumoto T, Koba T, Komatsuda T (2013) Structure, transcription and post-translational regulation of the bread wheat orthologs of the barley cleistogamy gene Cly1. Theor Appl Genet 126:1273–1283
Otto CD, Kianian SF, Elias EM, Stack RW, Joppa LR (2002) Genetic dissection of a major Fusarium head blight QTL in tetraploid wheat. Plant Mol Biol 48:625–632
Peng J, Ronin Y, Fahima T, Roder MS, Li Y, Nevo E, Korol A (2003) Domestication quantitative trait loci in Triticum dicoccoides, the progenitor of wheat. Proc Natl Acad Sci USA 100:2489–2494
Röder MS, Korzun V, Wendehake K, Plaschke J, Tixier M-H, Leroy P, Ganal MW (1998) A microsatellite map of wheat. Genetics 149:2007–2023
SAS Institute (2011) SAS/STAT 9.3 User’s Guide. SAS Institute Inc, Cary
Sears ER (1947) The sphaerococcum gene in wheat. Genetics 32:102–103
Sears ER (1952) Misdivision of univalents in common wheat. Chromosoma 4:535–550
Sears ER (1954) The aneuploids of common wheat. Missouri Agric Exp Stn Res Bull 572:59
Sears ER, Loegering WQ, Rodenhiser HA (1957) Identification of chromosomes carrying genes for stem rust resistance in four varieties of wheat. Agron J 49:208–212
Simons KJ, Fellers JP, Trick HN, Zhang Z, Tai Y-S, Gill BS, Faris JD (2006) Molecular characterization of the major wheat domestication gene Q. Genetics 172:547–555
Singh PK, Mergoum M, Adhikari TB, Kianian SF, Elias EM (2007) Chromosomal location of genes for seeding resistance to tan spot and Stagonospora nodorum blotch in tetraploid wheat. Euphytica 155:27–34
Somers DJ, Isaac P, Edwards K (2004) A high-density microsatellite consensus map for bread wheat (Trticum aestivum L.). Theor Appl Genet 109:1105–1114
Song QJ, Shi JR, Singh S, Fickus EW, Costa JM, Lewis J, Gill BS, Ward R, Cregan PB (2005) Development and mapping of microsatellite (SSR) markers in wheat. Theor Appl Genet 110:550–560
Sood S, Kuraparthy V, Bai GH, Gill BS (2009) The major threshability genes soft glume (sog) and tenacious glume (Tg), of diploid and polyploid wheat, trace their origin to independent mutations at non-orthologous loci. Theor Appl Genet 119:341–351
Sourdille P, Tixier MH, Charmet G, Gay G, Cadalen T, Bernard S, Bernard M (2000) Location of genes involved in ear compactness in wheat (Triticum aestivum) by means of molecular markers. Mol Breed 6:247–255
Sourdille P, Singh S, Cadalen T, Brown-Guedira GL, Gay G, Qi L, Gill BS, Dufour P, Murigneux A, Bernard M (2004) Microsatellite-based deletion bin system for the establishment of genetic-physical map relationships in wheat (Triticum aestivum L.). Funct Integr Genomics 4:12–25
Stack RW, Elias EM, Mitchell Fetch J, Miller JD, Joppa LR (2002) Fusarium head blight reaction of Langdon durum-Triticum dicoccoides chromosome substitution lines. Crop Sci 42:637–642
Torada A, Koike M, Mochida K, Ogihara Y (2006) SSR-based linkage map with new markers using an intraspecific population of common wheat. Theor Appl Genet 112:1042–1051
Tsunewaki K (1966) Comparative gene analysis of common wheat and its ancestral species. II. Waxiness, growth habit and awnedness. Jpn J Bot 19:175–254
Tsunewaki K, Ebona K (1999) Production of near-isogenic lines of common wheat for glaucousness and genetic basis of this trait clarified by their use. Genes Genet Syst 74:33–41
Unrau J, Smith WE, McGinnis RC (1950) Spike density, speltoidy and compactoidy in hexaploid wheat. Can J Res Com 28:273–276
Yu J-K, Dake TM, Singh S, Benscher D, Li W, Gill BS, Sorrells ME (2004) Development and mapping of EST-derived simple sequence repeat markers for hexaploid wheat. Genome 47:805–818
Zhang ZC, Friesen TL, Simons KJ, Xu SS, Faris JD (2009) Development, identification, and validation of markers for marker-assisted selection against Stagonospora nodorum toxin sensitivity genes Tsn1 and Snn2 in wheat. Mol Breed 23:35–49
Zhang ZC, Belcram H, Gornicki P, Charles M, Just J, Huneau C, Magdelenat G, Couloux A, Samain S, Gill BS, Rasmussen JB, Barbe V, Faris JD, Chalhoub B (2011) Duplication and partitioning in evolution and function of homoeologous Q loci governing domestication characters in polyploid wheat. Proc Natl Acad Sci USA 108:18737–18742
Acknowledgments
The authors thank Breanna Oldenburg and Megan Stoley for technical assistance and Dr. Steven S. Xu for assistance with statistical analyses. This research was supported by USDA-ARS CRIS project 5442-22000-037-00D.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Hohmann.
Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture. USDA is an equal opportunity provider and employer.
Rights and permissions
About this article
Cite this article
Faris, J.D., Zhang, Z., Garvin, D.F. et al. Molecular and comparative mapping of genes governing spike compactness from wild emmer wheat. Mol Genet Genomics 289, 641–651 (2014). https://doi.org/10.1007/s00438-014-0836-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-014-0836-2