Abstract
Low-moisture foods such as spices, grains, and seeds constitute an important part of the human diet. Increased consumer concern for food safety and food quality has focused on the decontamination technologies required for low-moisture foods. Cold plasma treatment has been a promising novel technology in the food processing industry due to its advantages in safety, efficiency, versatility, and environmentally friendly nature. It has shown various capabilities on safety and quality control in low-moisture foods. This paper comprehensively reviewed the application of cold plasma in low-moisture foods, including inactivation of microorganisms, degradation of mycotoxins, influences on the quality of low-moisture foods, and promotion of seed germination. Cold plasma can inactivate the pathogenic microorganisms on the surface of low-moisture foods, by generating active species, ultraviolet radiation, and electric fields, which helps to extend the shelf life of foods while having minimal impact on food quality. Cold plasma technology is also an effective approach to detoxify mycotoxin-contaminated low-moisture foods by degrading various mycotoxins. With the manipulation of parameters for cold plasma generation, target functional properties of food products may be obtained. In addition, the application of cold plasma in seed germination is promising and could be of great significance to the global food crisis. This review also suggests that more systematic studies are needed to employ cold plasma in the low-moisture foods industry for selected applications.
Similar content being viewed by others
Introduction
Low-moisture foods (LMFs) refer to food or food ingredients with a water activity (aw) less than 0.85 [1]. They can be broadly categorized as natural LMFs and processed LMFs. LMFs such as nuts, milk powder, seasonings, peanut butter, and flour were originally considered microbiologically safe. However, their frequent contamination with pathogenic microorganisms has drawn serious attention worldwide [2]. In 2014, more than 150 people in China were infected by consuming milk powder contaminated with Staphylococcus aureus [3]. In 2019, 21 people in the USA contracted E. coli O26 from eating contaminated flour [4]. Studies have reported the long-term prevalence of foodborne pathogens in LMFs or dry food processing and manufacturing environments [5]. Moreover, foodborne pathogens in LMFs usually have stronger resistance to high temperature and harsh environments than pathogens that existed in high water activity environments [6]. For instance, molds can grow in low-moisture environments, and produce mycotoxins that are carcinogenic and teratogenic, and thus pose a challenge to food safety.
Different physical and chemical treatment methods are used to minimize the safety problems associated with LMFs. For instance, preservatives such as benzoic acid and sorbic acid are often used in the food industry [7]. However, some chemical agents have carcinogenic and mutagenic effects and can cause environmental safety problems [8]. The most common physical treatment method is heat treatment,however, it can very likely lead to a reduction in the sensory properties and nutritional value of food. Also, some microorganisms are prone to heat resistance in a hot environment [9]. Due to the limitations of conventional heat treatments, the potential of new non-thermal processing techniques to solve food safety issues has been explored [10]. Among the non-thermal techniques, UV, radiation, pulsed light, and gas treatments, i.e., ethylene oxide, have limitations such as lower efficiency, overheating problems, higher cost, and chemical residue issues [11]. While ensuring the safety of LMFs, food industry often uses different treatments to enhance the functional performance of LMFs in order to improve their applicability (e.g., rheological properties, foaming ability) [12]. Seeds are an important part of LMFs. Seed germination is easily affected by environmental conditions such as temperature, humidity, and light [13, 14]. Under the pressure of global warming and the increasing world population, improving the seed germination rate and seedling survival rate is of great significance to solve the world food crisis [15]. In addition to external factors, seed germination is also affected by internal factors such as seed quality and its dormancy period [16]. To shorten the dormant period of seeds, various physical and chemical methods have been adopted.
CP is a novel non-thermal technology widely used in medicine, electronics, material science, and agricultural science. Plasma, also referred to as the fourth form of matter, can be generated either under atmospheric pressure or vacuum. Recent reviews on CP applications are focused on microbial inactivation and its degradation of mycotoxins. CP has been investigated for its potential to modify the nutritional and functional properties of LMFs [17]. CP has also been proved to have great potential to improve seed germination by enhancing seed germination rate and seedling survival rate [18]. Based on the above information, this review provides a short introduction to CP technology with its detailed application in LMFs. The four main areas discussed are shown in Fig. 1.
Cold plasma Technology
Plasma Chemistry
In nature, matter generally exists in three states: solid, gas, and liquid [19]. With the change of temperature or pressure, these three states can be transformed into each other. In a gaseous substance, if the temperature continues to rise to several thousand degrees Celsius, the gas will be ionized. The electrons around the nucleus will get rid of the bondage, and the electrons and positive ions will be separated. During this motion, the collision occurs and the neutral atoms are ionized to generate electrons, positive ions, free radicals, and neutral particles, which constitutes the fourth state of matter—the plasma state [19].
Plasma can be divided into two categories: high-temperature plasma and low-temperature plasma [20]. In high-temperature plasma, the gas is completely ionized and the particle temperature is as high as 106–108 K, with no heat transfer [21]. The low-temperature plasma is classified into thermal plasma and cold plasma. Thermal plasma is usually generated by the ionization of dense gas under high pressure and is in a state of thermodynamic equilibrium. CP is generated by the ionization of gas at atmospheric pressure or low pressure, with relatively high electron temperature but low ion temperature, which is in a non-equilibrium state [22]. This review mainly focuses on CP technology.
Plasma Source
CP can be generated in different ways, and each of these plasma generation methods has its own advantages and disadvantages [23]. The commonly used methods include dielectric barrier discharge, corona discharge, microwave discharge, and radiofrequency discharge. These methods have been described in detail in previous literature [24], therefore we only discussed them briefly in this review.
Dielectric barrier discharge (DBD) is the most common method to generate CP. It consists of a high voltage electrode and a ground electrode. Insulation material covers one or both electrodes or floats between the two electrodes, which can prevent the generation of thermal plasma [25]. DBD usually works at a 50 Hz–10 MHz frequency and 104–106 Pa pressure [26]. Its advantages include a large discharge area, room temperature operation, low maintenance cost, and strong sterilization effect, and can be applied to food in various states [27]. The corona discharge consists of a power supply, a transformer, a curved electrode, and a blower [28]. The application of high voltage will cause a strong electric field to concentrate on the curved electrode surface, and the gas near the curved electrode surface is induced to ionize due to the concentrated electric field, resulting in corona discharge plasma through localized gas breakdown [23]. The corona discharge is relatively easy to operate, but it has limited discharge area [29]. The microwave discharge does not require electrodes. The magnetron transmits the microwaves into the process chamber, and the gas absorbs the microwaves to generate heat, thus inelastic collisions [30]. It can work in a low-pressure environment and is able to generate a large amount of active material with less gas. However, the microwave discharge plasma is limited in space, and the application of a large area requires a discharge matrix [23]. The radiofrequency discharge consists of two coaxial electrodes, one is grounded, and the other is excited. The electrons are accelerated and ionized by collision with gas atoms [23]. It can work under low pressure to generate high-density plasma. However, radiofrequency discharge is expensive due to the usage of helium and argon. It is therefore usually used in the medical industry [31].
Application
CP has been widely used in many fields such as medical, machinery, environmental protection, and the food industry. In the medical area, it is reported that CP could induce stem cell proliferation and treat cancer [32]. CP will not only combat the current coronavirus disease (COVID-19) crisis but will also help with any future viral pandemic outbreak [33]. In machinery, plasma is used for surface activation, functionalization, etching, and polymerization of nanocomposites to improve the structure and properties of composites [34]. In terms of environmental protection, the treatment of CP helps to reduce the number of microorganisms and bacterial spores, as well as the toxicity of industrial wastewater [35].
To date, CP has been closely integrated with the food industry to inactivate microorganisms such as bacteria, molds, and yeasts in plant and animal foods, ensuring food safety and extending the shelf life of food [36]. For example, it is used for inactivating Salmonella, Listeria monocytogenes, and Duran virus [37], and removing aflatoxin B-1 from contaminated corn [38]. The application of CP technology has also been expanded to dissipation of agrochemical residues [39], such as the removal of malathion and chlorpyrifos on vegetables [40, 41], and the reduction of azoxystrobin residues on strawberry fruit [42]. CP can modify the physicochemical properties of food materials. It has been used in food processing to improve food functionalities, such as the foaming ability and hydration properties of wheat flour [43]. It can also be used to promote seed germination and growth [44].
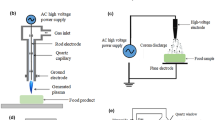
Although most of these studies have been carried out at the laboratory level, plasma equipment that can be applied to the food industry has already taken shape. Misra and Jo [15] proposed a plasma device model for food processing based on the European Union’s SAFE-PAG project, which consists of multiple plasma units assembled into a tunnel-like structure through which food passes and pathogens are eliminated (Fig. 2). Ziuzina et al. [45] designed a DBD reactor that purifies the fruits and vegetables in the package, which includes two 1 m long electrodes and an adjustable discharge gap to process multiple flexible packages at the same time. Based on the above research, we propose a plasma device that may be applied in the food industry, as shown in Fig. 3. The CP system is designed by adding a conveyor belt between the two electrodes of the DBD reactor through which the plasma contacts the food surface to kill pathogens. The number of patents for cold plasma devices exceeds one million, but most of them are only based on the design of plasma devices and have very few practical applications in the food processing industry. In recent years, the application of plasma in food processing and preservation has increased, and the number of patents has also increased [46].
Cold Plasma Application in Low-Moisture Foods
Inactivation of Microorganisms
With the popularization of health concepts, the safety of LMFs has gradually become a focus of attention. The contamination of LMFs by pathogens and spoilage microorganisms not only causes waste of resources but also affects the economic value of food. It is a common practice in the food industry to ensure food quality by inhibiting the growth and reproduction of pathogens by using various physicochemical sterilization methods. In many cases, preservatives used by the food industry contain controversial ingredients, which often make consumers feel disturbed [47]. Although heat treatment is effective in deactivating pathogens and spoilage microorganisms in food, pigments and nutrients are lost along with water, which reduces the nutritional and sensory quality of food [48]. Several non-thermal processing techniques are alternatives to traditional heat treatment, such as high-pressure processing and ozonation. These techniques are not dependent on temperature and can maximize food quality, but they have certain limitations. High-pressure processing is not suitable for high-protein foods because high pressure can lead to protein denaturation and gel formation [49]. Ozone is a toxic gas and may form various toxic by-products in food [50]. Among these new non-thermal technologies, CP technology has the unique advantage of being safe, efficient, non-toxic, and residue-free [51]. Therefore, it is chosen by an increasing number of professionals.
Mechanisms of Microbial Inactivation
CP is effective in the field of microbial purification. Studies have shown that the contents of Escherichia coli, Salmonella, and Listeria are significantly reduced after CP treatment for a short time [52]. The effect of plasma treatment depends on the product type, the type of bacteria, the surface properties of food, and the diffusivity of the plasma species [53]. So far, the mechanisms of plasma sterilization have not been fully elucidated due to the complexity of the plasma species’ interaction with microorganisms.
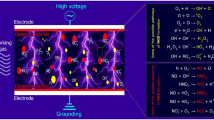
Several studies have shown that when gases are ionized, the resulting UV radiation and charged particles work as active species to play a major role in microbial inactivation. The mechanisms of microbial inactivation by CP are shown in Fig. 4. In general, plasma-generated reactive species oxidize lipids and sugars on microbial cell membranes, thus causing the cell membranes to rupture [54]. Reactive oxygen (ROS) and nitrogen species (RNS) also interact with peptidoglycan and important chemical bonds in the cell wall. C-O and C-N bonds are broken and the bacterial cell loses the protection as the entire cell structure is damaged [55]. In addition to basic cellular structures, the active species also react with biomolecules such as proteins, DNA, and lipids within bacterial cells, leading to amino acid oxidation, lipid peroxide formation, and DNA rupture [56]. During plasma generation, the intensity of initiated UV radiation is dependent on the plasma devices and gas types. The microbial inactivation mechanisms are that the ultraviolet radiation is irradiated to the biological cells where the nucleic acid absorbs the photon energy forming a thymine dimer to inhibit bacteria proliferation [57].
Fridman et al. [58] found that the electrostatic force is thought to destroy the cell membrane, which leads to cell membrane damage and even cell death. Other studies have shown that when the electric field strength is high enough, it can change the three-dimensional structure of the protein, causing it to separate from the cell membrane, and resulting in membrane perforation and leakage of cell contents [59]. Cui et al. [60] observed that cold nitrogen plasma (CNP) treatment can destroy the structure and affect the metabolism of L. monocytogenes biofilms. The main antibacterial mechanism of CNP is that large numbers of active free radicals generated during discharge react with biological macromolecules such as phospholipids, DNA, and proteins, destroying the integrity of biofilms. At the same time, ROS can further damage the exposed bacterial cell membrane, resulting in increased oxidative stress of various substances in the cell. This inhibits the expression of quorum sensing-related genes and virulence genes and finally leads to bacterial cell death. The physical impact of CP on microorganisms is mainly the electrostatic force generated by some collection of charged particles. The study by Qian et al. [61] also obtained similar results. After plasma treatment, reactive species ROS and RNS attack the cell membrane to change its permeability, resulting in the leakage of inorganic ions (K, Mg+2+, Na), which eventually inactivates the cell. The study also pointed out that the effect of plasma treatment on Gram-negative bacteria was significantly higher than that of Gram-positive bacteria under the same conditions, which may be resulted from the differences in the peptidoglycan layer.+
Effects of Cold Plasma on Microbial Inactivation in Low-Moisture Foods
Factors affecting the efficiency of plasma treatment include process, product, and microbial factors [25]. This review focuses on the effect of processing parameters on microbial inactivation. An overview of the recent studies on the inactivation of microorganisms in LMFs by CP is shown in Table 1.
The electrical input parameters (power, time, frequency, voltage, etc.) of plasma generation affect the efficiency of microbial inactivation. Deng et al. [62] reported that the microbial inactivation efficiency of CP increased with the increase of voltage and frequency. Similar results were obtained in food-grade rice starch and spices [63, 64]. Beyrer et al. [63] observed that among the three power levels of 4.1, 5.7, and 7.1 W, the inactivation rate of Bacillus coagulans reached the highest in 5 s under 7.1 W. Charoux et al. [65] observed a significant reduction of 5.64 log CFU/mL in Bacillus subtilis vegetative cell population in black pepper grains after 5 min of treatment at 30 kV and 1.16 ± 0.98 log (CFU/g) after 3 min of treatment at 15 kV. Ahangari et al. [66] investigated walnut kernels treated with radiofrequency low-pressure cold plasma (LPCP) at different powers and observed that the efficiency of microbial inactivation was increased with the increase of power and time. The decrease of the microbial population was the highest at 50 W power and 20 min treatment time. The total viable count, coliform, and mold log reduction were 1.09, 0.97, and 0.89 log CFU/g, respectively. Appropriate increase in CP treatment time will also increase microbial inactivation efficiency, but longer treatment time is not better. Hemmati et al. [67] explored the effect of cold atmospheric-pressure plasma on the microbial load of turmeric powder. The number of aerobic viable cell count was observed to decrease by about 1.5 log CFU g−1 at 7 min after plasma treatment. However, the decreasing effect of plasma treatment was only significant in the first 3 min of the process. Other studies have also yielded the same results [76]. It is necessary to understand the optimized plasma treatment parameters for maximum plasma treatment efficiency [77].
Moreover, the selection of different plasma generation systems affects the microbial inactivation efficiency. Hertwig et al. [68] observed that after using microwave-driven plasma to treat pepper for 30 min, S. enterica, Bacillus subtilis spores, and Bacillus atrophaeus spores were reduced by 4.1, 2.4, and 2.8 log, while radiofrequency plasma treatment did not achieve similar inactivation. The microbial inactivation effect is influenced by the type of ionized gas in the plasma system and the distance from the plasma generation source. Sen et al. [69] used atmospheric-pressure (AP) and low-pressure (LP) plasmas to treat A. flavus and A. parasiticus in hazelnuts, respectively. Researchers observed that higher plasma power, frequency, and voltage resulted in greater inactivation in a short period. And regardless of plasma type (LP or AP), when the air was used as a reactive gas, the inactivation efficiency was much greater than using nitrogen. Different treatment methods also affect the microbial inactivation effect. Taking dried peppermint as an example, the total viable count (TVC) decreased significantly by up to 1.5 log after 7.5 min of continuous plasma treatment, while the total viable count (TVC) decreased only by about 0.8 log after 3 × 2.5 min of pulsed plasma treatment. Spore-forming bacteria, Enterobacteriaceae, yeasts, and molds were significantly reduced after plasma treatment for 7.5 min compared to 3 × 2.5 min treatment [70].
Different gas ionizations produce different types of complex compounds. The chemical properties of the process gas determine the nature of reactions that occur during food processing. Basaran et al. [71] treated hazelnuts, peanuts, and pistachios with low-temperature radiofrequency plasma by changing the processing gas. Researchers observed that the use of sulfur hexafluoride was more efficient than the use of air as the reaction gas for the same length of time. Similarly, Niemira [72] studied Salmonella and E. coli on almonds, and also changed the distance of the food from the plasma generation source. By changing the distances to 2, 4, and 6 cm, the process gases were air and nitrogen, respectively. Researchers observed that for E. coli O157:H7C9490, regardless of the reaction gas used, the microbial inactivation efficiency was the strongest when it was 6 cm away from the emitter, but for E. coli O157:H735150, the distance effect was not obvious, and the change in the distance did not cause the change in bactericidal efficiency. Salmonella PT30 had the highest inactivation efficiency at 6 cm and 4 cm under the action of air and nitrogen, respectively. Therefore, the microbial inactivation efficiency is restrained by a variety of factors, and appropriate microbial inactivation parameters should be selected correspondingly to the appropriate strains.
The microbial inactivation efficiency is also affected by different food matrices. The study found that when different cultivars of walnut kernels are treated, the bactericidal effect may be different due to the different thickness of walnut kernels [73]. After 3, 5, and 7 min of treatment, A. flavus cells reduction was highest in the Mazandaran cultivar and lowest in the Taleghan cultivar. After 10 min treatments, the reduction rate of A. flavus was the highest in the Taleghan cultivar and lowest in the Shahmirzad cultivar. In addition to the aforementioned foods, plasma can significantly reduce microorganisms in spices [78] and dried fruits [75].
Reduction of Mycotoxins on Low-Moisture Food Surfaces
LMFs lack the water necessary for microbial growth and reproduction, limiting microbial growth. However, some xerophilic spoilage fungi can grow at aw < 0.6 [79]. For instance, LMFs such as corn, rice, wheat, and spices are susceptible to contamination by Aspergillus, Fusarium, and Penicillium [80]. According to the Food and Agriculture Organization (FAO) of the United Nations, 25% of the food in the world is contaminated by fungal pathogens every year [81], causing food waste and economic losses. Certain fungal pathogens can produce toxic metabolite mycotoxins, which are highly detrimental to human health. The common effects are liver toxicity, kidney toxicity, carcinogenicity, teratogenicity, and immunosuppression [82]. Among the mycotoxins, the most distributed groups are aflatoxin (AF), fumonisin (FB), ochratoxin (OTA), T-2 toxin, zearalenone (ZEN), deoxynivalenol (DON), nivalenol (NIV), etc. [82]. There are different ways to inhibit the growth of fungal pathogens and decompose mycotoxins in contaminated food [83,84,85,86]. However, given the specificity of the food industry, we should constantly seek new approaches to make up for the shortcomings of existing technologies.
Conventional Methods for Mycotoxin Degradation
Common methods to prevent fungal contamination from food include the application of pesticides, preventing pests and diseases, cultivating new varieties with fungal resistance, and low-temperature dry storage, etc., but it is unrealistic to ensure that food is completely free from fungal contamination [87]. Mycotoxin removal from contaminated food is crucial, and a range of physicochemical purification techniques are employed in the food industry.
Physical degradation methods of mycotoxins include removal of contaminated fractions, ultraviolet (UV), gamma rays, pulsed light (PL), and more. UV irradiation has been shown to effectively degrade Aflatoxin B1 (AFB1). The absorption of UV light at 222, 265, and 362 nm by AFB1 destroys the structure of the terminal furan ring and eventually reduces its toxicity [88]. Ultraviolet light is an economical and environmentally friendly fungal inactivation technology characterized by low heat generation and no toxic waste [89]. UV radiation is limited to the low UV penetration, which can only interact with contaminants on the food surface. It also takes a long time to achieve mycotoxin reduction, which may lead to the deterioration of food quality [90]. Pulsed light is the use of gas flash lamps to generate short and high-intensity flashes in the frequency range of ultraviolet, visible, and infrared (200–1100 nm). Pulsed light treatment can degrade mycotoxins [91], but its degradation pathway and degradation products are still unclear [92]. Moreover, the limited penetration of pulsed light can only remove mycotoxins on the food surface, and can eventually overheat the food [93]. Gamma rays can promote water splitting to generate a variety of reactive free radicals that can destroy mycotoxins [94]. Gamma rays have the advantages of high reactivity and strong penetration, but the free radicals can also cause oxidative deterioration of nutrients such as lipids and vitamins in food [95].
Two methods for the chemical degradation of mycotoxins are ozone degradation and electrolytic oxidized water (EOW) degradation. Ozone is a strong oxidant, which can generate free radicals and change the functional group structure of mycotoxins to produce low-toxic products with fewer double bonds [96]. Studies have shown that ozone can degrade mycotoxins in LMFs [97]. However, if the ozone concentration is too low or the treatment time is too short, mycotoxins cannot be effectively degraded, and long-term ozone treatment will adversely affect food quality. For example, after ozone treatment at 5 ppm for 6 h, the appearance and flavor of red peppers were significantly reduced [98]. Electrolyzed oxidized water is produced by electrolysis of salt solution, which can oxidatively denature mycotoxins through OH radicals and active chlorine [99]. Studies have shown that electrolyzed oxidized water has good potential in the degradation of deoxynivalenol (DON) and aflatoxin [100].
CP can kill mold and mold spores on food and inhibit the formation of mycotoxins, while the active species contained in CP can react with some structures of mycotoxins and degrade mycotoxins [24]. Numerous studies have demonstrated the use of CP to reduce mycotoxins on the surface of LMFs [101,102,103], such as aflatoxin in corn [104, 105] and ochratoxin A in coffee [106].
Mechanisms of Mycotoxin Degradation by Cold Plasma Technology
The degradation pathways and degradation products of mycotoxins after plasma treatment have not been extensively studied, and the specific mechanisms are still unclear [107]. Currently, only AFB1, ZEN, and patulin have been reported to explain mycotoxin degradation. The degradation of AFB1 starts from the C (8)-C (9) bond of the terminal furan ring and goes through various degradation pathways to obtain the final degradation product [38].
Wang et al. [108] treated AFB1 with low-temperature radio-frequency plasma and analyzed the quantity and structure of degradation products. They observed that five previously unreported degradation products were produced. A possible degradation pathway was proposed based on the structures of these five degradation products. Low-temperature radio-frequency plasma treatment can induce the addition reaction of the C (8)-C (9) bond of AFB1 to form the intermediate m/z 331(C17H15O7), this compound is also the main degradation product of AFB1 after UV and Co60 gamma radiation treatment. Researchers also observed that these five degradation products were less toxic than AFB1 due to the loss of the double bond on the terminal furan ring.
Shi et al. [104] treated aflatoxin B1 with high-pressure atmospheric cold plasma (HVACP) and used liquid-chromatography time-of-flight mass spectrometry to analyze the degradation products of AFB1. AFB1 produced six major degradation products after HVACP treatment, two of which were ozone products of AFB1 and four were new products that had not been reported before. Shi et al. [104] also reported that reactive oxygen species are the main species for the degradation of AFB1, and the temperature and UV intensity during HVACP treatment did not meet the conditions required for AFB1 degradation. Based on the structure of the degradation products, they proposed two possible pathways. The first pathway was the addition reaction of water molecules, hydrogen molecules, and aldehyde groups with AFB1. The second pathway involved an epoxidation reaction by HO2• radicals and oxidation reactions through the combined effects of the oxidative species OH•, H2O2, and O3. All the six degradation products lost double bonds, and the structures of the furan ring, cyclopentenone, and methoxy group were changed accordingly. Their toxicity was significantly lower than that of AFB1. Wielogorska et al. [109] treated six common mycotoxins with CP, degradation products were only found in AFB1 and ZEN. They speculated that the degradation products of AFB1 were generated by the modification of the terminal furan ring and the lactone ring. The methoxy group was not changed during the treatment. Xue et al. [110] explored the degradation pathways of patulin under low-temperature plasma treatment, and they observed that the lactone and hemiacetal structures in the patulin molecule were disrupted and the toxicity of patulin was significantly reduced. Although researchers have put forward many speculations about the pathway of plasma degradation of mycotoxins, there is no definite evidence to clarify the specific pathway of plasma degradation of mycotoxins. More research should be carried out in the future to fill this gap.
Research Progress of Cold Plasma Degradation of Mycotoxins
CP processing is a promising mycotoxin decontamination method for various types of LMFs (Table 2). Park et al. [111] successfully degraded three mycotoxins using CP, which opened a new chapter in the application of plasma technology. They treated AFB1, DON, and NIV with CP generated by a microwave-induced argon plasma system at atmospheric pressure. The results showed that these mycotoxins were completely degraded within 5 s of treatment. Since then, many researchers have used CP to degrade mycotoxins in LMFs.
Aflatoxin
Aflatoxin B1 is a Group 1 carcinogen that induces cancer cell formation through an additive reaction with guanine [124]. LMFs such as nuts, peanuts, corn, wheat, and rice are generally susceptible to aflatoxin contamination. Most studies have focused on pistachios and hazelnuts, while other types of nuts have been less frequently reported. Basaran et al. [71] used low-pressure CP to treat aflatoxin on hazelnuts and observed that after 20 min treatment with air plasma and sulfur hexafluoride (SF6) plasma, total aflatoxin (B1, B2, G1, G2) was reduced by 50 and 20%, respectively. A few years later, to explore the optimal degradation conditions of aflatoxin in hazelnuts, Siciliano et al. [112] optimized the process parameters, i.e. gas composition and power, of cold atmospheric pressure plasma. Irrespective of the treatment time, the 21% oxygen content was insufficient in degrading aflatoxin B1. The higher the processing power, the better the degradation of aflatoxin on hazelnuts. Besides, the highest degradation efficiency was obtained when pure nitrogen or nitrogen–oxygen mixture (0.1% O2) was used. Makari et al. [113] compared the aflatoxin B1 degradation on glass slides and pistachios, using dielectric barrier discharge CP and observed that CP treatment for 60, 120, and 180 s, degraded aflatoxin by 35.41%, 60.97%, and 64.63% on the glass slides, and by 32.31%, 44.77%, and 52.42% on pistachios, respectively. Because of the complexity of the food matrix, the degradation rates of AFB1 on food were significantly lower than that on glass slides.
Devi et al. [114] investigated the effects of CP on aflatoxin production of A. flavus and A. parasiticus inoculated on peanuts. They observed that after treatment with 40 W for 15 min and 60 W for 12 min, the amount of aflatoxin B1 was reduced by more than 70% and 90%. Iqdiam et al. [115] used an atmospheric pressure plasma jet to treat aflatoxins on peanuts in both constant and agitation states. The content of AFTs was reduced by 23% after 2 min of constant treatment and by 38% after 5 min of stirring treatment, having no significant difference in the degradation rate.
Corn is an important source of feed and is highly susceptible to aflatoxin contamination. Many studies have shown the effectiveness of CP technology for degrading aflatoxin in corn. Shi et al. [105] evaluated the effects of gas type (air and modified atmosphere gas (MA65)) and relative humidity on the degradation effect of aflatoxin in corn. The results showed that MA65 degraded aflatoxin better than air at the same treatment time and relative humidity, which was due to the active substances (ozone and NOx) in MA65 at higher concentrations. Researchers also observed that humid air (40% and 80%) had a higher rate of aflatoxin degradation than dry air (5%), possibly due to the higher content of OH radicals produced by humid air. In addition, it was also observed that when the treatment time was longer than 10 min, the degradation rate increased slowly, and the aflatoxin could not be completely degraded. Researchers speculated that this may be either due to insufficient exposure of the corn to the active substances or due to the lesser penetration and subsequent interaction of ROS with the aflatoxins. Treatment power was equally important for aflatoxin degradation. However, they did not mention the effect of power on the degradation of AFB1 on corn in their study. Hojnik et al. [116] treated aflatoxin B1 on corn kernels using cold atmospheric pressure plasma generated by both low discharge power (0.18 W/cm) and high discharge power (0.31 W/cm). The results showed that both methods were able to significantly reduce AFB1, and the effect was better at high power. After 240 s of cold atmospheric pressure plasma treatment, AFB1 was completely degraded.
The magnitude of the current and the sample-electrode distance also affected the degradation of AFB1. Puligundla et al. [117] investigated the effect of corona discharge plasma on the degradation of aflatoxin in rice and wheat. They set different current (1.00, 1.25, and 1.50 A) and sample-electrode distances (15,25, and 35 mm) groups. The results showed that the treatment effect of AFB1 was the best when the current was 1.5 A with a sample-electrode distance of 15 mm.
Ochratoxin (OTA)
Ochratoxin A, originally found in corn, is classified as a group 2B carcinogen by the International Agency for Research on Cancer and the World Health Organization and is cytotoxic and genotoxic, inhibiting protein synthesis, interfering with the phenylalanine metabolic system, and damaging DNA [125, 126]. Durek et al. [119] observed that when barley was treated with CO2 plasma generated by diffuse coplanar surface barrier discharge (DCSBD), the production of OTA increased from 49 ± 13.8 ng/g (control group) to 72.9 ± 45.8 ng/g (3 min). Hoppanova et al. [126] observed that OTA production increased significantly in the early stages of incubation (4 days) after 60 and 90 s of treatment of OTA with plasma, but was lower than the control after seven days of incubation. The final yield of OTA was lower in plasma-treated samples compared to untreated samples. In actual processing, process conditions such as gas composition and storage time should be considered. However, there are also studies showing that plasma treatment can reduce the OTA content in food. Patricia Casas-Junco et al. [106] used helium plasma to treat ochratoxin A in coffee. They observed that after 30 min plasma treatment, OTA was reduced by 50% and toxicity was reduced from “toxic” to “slightly toxic.”
Alternaria Toxins
Food and feed are also highly susceptible to contamination of Alternaria alternate which produces Alternaria toxins with different structures such as alternariol (AOH) and alternariol monomethyl ether (AME). Hajnal et al. [118] used surface dielectric barrier discharge cold atmospheric plasma to treat AOH, AME, and tentoxin (TEN) toxins on wheat flour using different treatment times and plasma source distances. It was found that CP was able to reduce the content of these three mycotoxins. A better degradation effect was obtained with a closer distance to the plasma source and a longer treatment time. In the following year, Wang et al. [127] used dielectric barrier discharge CP to degrade solid and liquid AOH and AME and reported their 100% degradation after 180 s and 300 s, respectively. These two studies indicate that the degradation of AOH and AME on food substrates may not be as effective as that using pure mycotoxins, which may be due to insufficient contact or incomplete entry of the active species generated by the CP into the food. The effects of processing parameters such as voltage, pH, initial concentration, direct and indirect CP treatment, and catalyst on the degradation of AOH and AME were also optimized by Wang et al. [127]. The results showed that a higher voltage alkaline environment, high substrate concentration, and using catalysts such as FeSO4 and H2O2 can promote the degradation of AOH and AME.
T-2 and HT-2 Toxins
T-2 and HT-2 toxins are natural contaminants present in cereals, with the highest levels found in oats and oat-based products. Kis et al. [120] treated T-2 toxin and HT-2 toxin in oat flour with low-pressure DBD plasma generated by the ionization of four gases: oxygen, argon, air, and nitrogen. The results showed that the four plasmas had significant effects on the degradation of T-2 toxin, with a reduction of 43.25% after nitrogen treatment for 30 min, and the average degradation rate of HT-2 toxin (20.98%) was lower than that of T-2 toxin (25.01%). Iqdiam et al. [121] used dielectric barrier discharge air atmospheric cold plasma (DBD-ACP) to treat T-2 and HT-2 toxins on glass slides and wheat and showed that after 10 min of air-ACP treatment, pure T-2 and HT-2 toxins were decreased by 63.63% and 51.5%, while T-2 toxin and HT-2 toxin on wheat kernels were decreased by 79.8% and 70.4%. The degradation rate of T-2 and HT-2 toxins in wheat was higher, probably due to the limited amount of solvent and short vortexing time, which was not sufficient to extract all pure toxins.
In addition to the four mycotoxins mentioned above, cold plasma has also shown good degradation of other mycotoxins, such as Citrinin, ZEN, and DON. Vaseghi et al. [128] studied the feasibility of degrading Citrinin by cold plasma, and the results showed that cold plasma could effectively degrade Citrinin, and the degradation rate was different depending on the source of Citrinin. Feizollahi and Roopesh [122] explored substrate properties (canola grain, canola meal, and barley grains) on ZEA degradation. They observed that the characteristics of the substrate affected the degradation effect of CP on ZEN. Feizollahi et al. [123] explored the effect of atmospheric cold plasma on DON-contaminated barley grains. They observed that at different treatment times (0–10 min), DON was significantly reduced in the first 6 min, and thereafter the reduction rate was not significant. Although CP applies to a wide range of low-moisture foods and is effective in degrading mycotoxins, further research is needed on process optimization, the reaction between cold plasma and food components, and the safety of the process.
Effects of Cold Plasma Treatment on Low-Moisture Food Quality
Microbial inhibition and modification methods commonly used for LMFs have certain limitations, including sensory changes, ingredient oxidation, and nutrient loss [129]. For example, the common household spices such as turmeric and red pepper have a loss of 27–53% curcumin and 18–36% capsaicin, respectively, after heat treatment [130]. CP has attracted interest as an alternative to conventional food processing methods in improving the nutritional and functional properties of foods [131]. Studies have investigated the efficacy of plasma technology in the modification of raw materials to improve the overall product characteristics (Table 3, Fig. 5).
Effects on Sensory Quality
The sensory properties of food (color, aroma, taste, etc.) are important attributes of food, which will directly affect consumers’ choices. The results of Basaran et al. [71] observed that after treating nuts with air and sulfur hexafluoride plasma for 20 min, the surface morphology and sensory attributes were not significantly different compared to the control group. Iqdiam et al. [115] treated peanuts with an atmospheric pressure plasma jet and observed that the overall appearance, likeability, taste, and texture of peanuts under both constant and agitated treatments were not significantly different from those in the control group. The peanuts treated for constant 2 min had even better quality attributes than the control group. Another study by the same group used DBD-ACP to treat the T-2 and HT-2 toxins in wheat and observed that after 5 min and 10 min of wheat treatment, the L*, a*, b*, chroma, hue angle, and total color difference were not significantly different from the control group [121]. Hojnik et al. [116] observed that plasma did not cause surface damage to the corn, while Wielogorska et al. [109] observed that after CP treated corn, more than half of the molecular compound composition in maize was affected. In conclusion, the effect of CP on the sensory quality of food is negligible.
Effects on Water and Dry Matter Content
Water is an important component of food and is a solvent for many substances. Changes in food temperature introduced by CP treatment can affect the moisture content of food, thus affecting the shelf life, sensory properties, and the content of other components of foods. Selcuk et al. [132] investigated the effect of CP treatment on the quality characteristics of grains and legumes and observed no change in food quality except for a slight change in moisture content. CP treatment can also produce some desirable effects on the color of certain foods. Lee et al. [129] evaluated the changes in the quality of brown rice and found that the brightness of brown rice increased, and the whiteness index increased from 56.9 to 58.3, which may also cause by changes in moisture. The study also pointed out that the amylase activity was significantly enhanced when brown rice was plasma was treated for 5 min, 10 min, and 20 min, which together with the change in water absorption, was considered to be the main reason for the change in the hardness of brown rice. Wiktor et al. [133] investigated the effect of plasma treatment on the physicochemical properties of spices and observed that water activity and dry matter changed significantly in all spice types, with increments of 2 and 3.5% in the dry matter content of juniper berries and black pepper seeds, respectively. The change in dry matter content may be related to water evaporation due to higher temperatures after plasma treatment.
Effects on Lipids
Lipid oxidation, which produces a variety of primary and secondary products that negatively affect food quality, is one of the main parameters in the analysis of food shelf life. Bahrami et al. [134] evaluated the effect of plasma treatment on the quality of wheat flour and observed no significant differences in the non-polar and glycolipid fractions when determining total extractable lipids (non-polar, FFA, glycolipids, and phospholipids). But the free fatty acids and phospholipid complement in wheat flour were significantly affected by CP. Choi et al. [135] used a corona discharge plasma jet to treat dried squid shreds and found that the oxidation of unsaturated fatty acids in the samples increased with increasing treatment time and TBARS values increased significantly. Lipid oxidation deteriorates the sensory and organoleptic properties of the foods. Lee et al. [136] also observed that CP treatment caused adverse oxidation of brown and white cooked rice affecting their quality, and the 2-thiobarbituric acid reactive substance (TBARS) values of brown and white cooked rice increased from 0.49 and 0.04 to 0.59 and 0.25 after 20 min of plasma treatment. The oxidation of lipids by plasma increased with increasing treatment time. The effect of CP on food lipids is not always negative either. Foligni et al. [137] evaluated the degree of oxidation of pistachio kernels after surface medium blocking discharge plasma treatment. They observed that hydroperoxides and TBARS in the plasma processed group were not significantly different from the control group in either high or low ozone concentration environments. The rate of lipid oxidation can be delayed by optimizing various plasma process parameters such as modified gas mixtures with reduced oxygen levels, low input power, reduced treatment temperatures, and shorter processing times [144].
Effects on Proteins
The rising popularity of alternative proteins has propelled the exploration of sustainable and environmentally friendly methods for protein production. The alternative protein market is dominated by plant proteins. CP treatment provides a green approach to improving the physical and chemical properties of plant proteins. The mechanism of the interaction of the plasma-generated ROS with proteins is through amino acid derivatization. Bombardment of ROS on the protein moieties creates active sites and results in an increased addition of the hydrophilic groups [145]. Moreover, CP treatment can modify the primary, secondary, tertiary, and quaternary structures of the protein aggregates, thereby resulting in their improved functional and nutritional properties [131]. It was found that in CP-modified wheat protein, the oxidation of amino acids directly affected the functional properties of wheat [146]. The total protein level of plasma-treated wheat flour did not change significantly. However, the ratio of different protein components and their molecular weight were affected, which may further alter the water-binding capacity. Misra et al. [138] observed that ACP treatment changed the protein secondary structure of wheat flour, which was mainly manifested by differences in rheological properties, and an improved dough strength by promoting the formation of disulfide bonds. The same result was observed by Zare et al. [139]. ACP treatment altered the rheological properties of quinoa flour, probably due to the polymerization of proteins and the formation of disulfide bonds by plasma treatment. Bahrami et al. [134] observed that the total protein of wheat flour was not affected by the CP treatment, but the protein profile shifted to higher molecular weight and the dough strength increased. The CP process variables such as input voltage, working gas, and treatment time can be optimized to improve the solubility, gelation, and foaming properties of the plant proteins [145, 147].
Effects on Carbohydrates
Carbohydrates are biological molecules composed of carbon, hydrogen, and oxygen atoms. Starch is a very common type of carbohydrate, which is one of the widely used biopolymers in different fields such as the food industry, paper industry, textile, and pharmaceutical. The use of cold plasma modified starch is an effective alternative to chemical modification [17]. Several studies have summarized the changes in the properties of various types of starch (red adzuki bean, potato, rice, and mango seed kernels) after plasma treatment [148,149,150,151]. Wu et al. [140] investigated the effects of CP generated by different powers on the physicochemical properties of corn starch. The results showed that plasma of different intensities triggered the depolymerization of corn starch, increased the solubility of starch and gelatinization degree, and decreased peak viscosity and final viscosity significantly. Scanning electron microscope images showed that plasma treatment caused slight cracks on the surface of starch granules, but the overall morphology did not change significantly. Chaple et al. [43] treated wheat flour with a dielectric barrier discharge plasma of 80 kV for 5–20 min and observed an increase in the viscosity, hydration, and gelatinization properties of wheat flour. The endothermic enthalpy and crystallinity of wheat flour decreased due to plasma-induced depolymerization of starch. The L* and Whiteness Index values of flour increased with the treatment time, and conversely, the a* and b* values decreased with the treatment time. The authors proposed that the function of food composition can be selectively regulated by adjusting the plasma process parameters.
The change of molecular weight by plasma treatment was mentioned in the study of Bie et al. [141], the average molecular weight of cornstarch decreased after 10 min dielectric barrier discharge (DBD) air plasma treatment (1.934 × 107 to 0.098 × 107 g/mol). The study also concluded that the viscosity of cornstarch decreased gradually with increasing treatment time. Changes in its rheological properties tend to transition from pseudoplastic (non-Newtonian) fluids to Newtonian fluids. Okyere et al. [142] studied the effects of carbon dioxide-argon RF CP treatment on the in vitro digestion and structural characteristics of granular and non-granulated waxy maize, potato, and rice starch, and observed that plasma treatment could also change the damaged starch content, making the relative number of short chains decrease, resulting in a slight increase in long chains. The treated starch is beneficial to slow the release of glucose in the blood and affects the in vitro digestibility of starch, which certainly improves the quality of starch.
In addition to proteins, lipids, and carbohydrates, plasma treatment also affects the content of other nutrients in LMFs. Alves Filho et al. [143] studied the effect on the composition of cashew nuts. The results showed that plasma treatment caused little sensitization, reduced sucrose content, increased fatty acid, and glycerol content, and increased aromatic acid content. CP has both beneficial and adverse effects on food quality, and attention should be paid to the selection of parameters during food processing.
Effects of Cold Plasma on Seed Germination
Seed germination is a key link in the plant life cycle and is affected by various environmental factors [14], such as sunlight, air, moisture, and temperature. In addition, seed germination is also affected by internal factors such as seed coat hardness, phytohormones, and seed maturity [152]. In recent years, the pressure of population growth and climate change has led to a rise in the food demand, and it is thus of great significance to find new technologies to improve seed germination rate, accelerate seedling growth, and subsequently increase the production yield [18]. Researchers have explored the feasibility of various physicochemical techniques such as scratching, delamination, ultrasound, electromagnetic fields, and gamma rays, to improve seed germination and seedling growth [153]. Many studies have demonstrated the direct or indirect effect of CP in significantly improving the germination rate of seeds and the growth of seedlings [154,155,156,157].
Action Mechanisms of Plasma Promoting Seed Germination
The efficacy of plasma treatment in promoting seed germination has been discussed in many reports, but the specific mechanisms underlying the remain poorly understood and may be related to the physical, chemical, and biochemical factors derived from plasma treatment [158]. Studies have reported an increased association between the seed germination rate and the hydrophilicity of the seed surface [159, 160]. The bombardment of plasma-generated ions and radicals causes etching of the seed coat, thus improving the hydrophilicity and the water uptake capacity of the seed (Fig. 6). Plasma improves the seed surface wettability and enhances the seed germination characteristics [161, 162]. The results are in agreement with the finding of Bormashenko et al. [163] in 15 s cold air plasma treated lentils and beans, where the water contact angles (WCA) decreased from 127 ± 2° and 98 ± 2° to 20 ± 1° and 53 ± 1.5°, respectively. Changes in the water contact angle may be due to chemical changes occurring on the seed coat surface. Gomez-Ramierez et al. [164] reported after a dielectric barrier discharge and a radiofrequency plasma treatment, the surface oxygen and nitrogen content of quinoa seeds significantly increased, while the carbon content decreased significantly. The results showed that after plasma treatment the outer layer of quinoa seeds was highly oxidized and enriched with potassium ions and nitrates which eventually disappeared on the surface of the seeds. This was probably due to the diffusion of potassium ions and nitrates inside the seeds with water vapor, thus eventually promoting seed germination. Recek et al. [165] reported that after treating common bean (Phaseolus vulgaris L.) with low-pressure oxygen plasma for only 0.5 s, the WCA of the sample decreased from the initial 85° to 10°, when treated for 3 s, the WCA reduced to unmeasurable. The results of X-ray photoelectron spectroscopy showed that the carbon content on the surface of the sample was significantly reduced and the oxygen content was significantly increased, while the nitrogen and silicon content remained within a reasonable range. Other elements such as calcium and magnesium appeared on the surface. This indicates that oxygen-containing functional groups were introduced on the surface of seeds, resulting in the rise of oxygen and the fall of carbon.
Another possible mechanism could be due to the interaction and penetration of plasma-generated RONS into the seeds, followed by subsequent activation of various biochemical reactions required for seed germination [166]. The interaction of plasma with seeds can affect the activities of various enzymes, such as dehydrogenase, and superoxide dismutase peroxidase, and eventually alter the physiological metabolism of seeds [167]. Ji et al. [168] observed that after spinach seeds were treated with high voltage pulsed plasma for 1 d, the gene expression levels of gibberellic acid and amylase increased in the seeds, and they speculated activation of biochemical reactions in the seeds. Mildaziene et al. [169] reported that after the treatment of sunflower seeds with radio-frequency CP, the content of gibberellic acid in seeds increased, while the content of zeatin decreased. The increase of gibberellic acid promotes the synthesis of amylase, causes starch hydrolysis, and contributes to seed germination. Ussenov et al. [170] investigated the effect of SDBD plasma on wheat seed germination parameters and α-amylase enzyme activity, and they observed a significant increase in α-amylase enzyme activity after 15 s of plasma treatment and a significantly higher seed germination rate than the group without plasma treatment.
Research Progress of Cold Plasma Application on Seed Germination
Seeds is a type of LMFs and it has been proved that CP is able to promote the germination of many plant seeds, improve seedling survival, and accelerate seedling growth [44, 171, 172]. CP is generally considered a safe and inexpensive method for seed processing [173].
Seed germination rate is influenced by the nature of chemically reactive species. Sarinont et al. [174] observed that oxygen and nitrogen active species were closely associated with seed germination. Upon using air, O2, NO (10%), and N2 as the processing gas, the rate of seed germination was enhanced, researchers speculated hydroxyl and oxygen radicals as key factors in promoting seed germination. Ji et al. [168] observed that the germination rate of spinach seeds treated with air DBD plasma was slightly higher compared to N2 plasma treatment, and the presence of an appropriate dose of elemental nitrogen was important.
An appropriate increase in treatment power can promote seed germination, but excessive treatment power is not better. [175] treated wheat seeds with cold nitrogen plasma treatment of different power and found that 80 W treatment could significantly improve seed germination potential (6.0%) and germination rate (6.7%), while the 100 W treatment was not significantly different compared to the control. Li et al. [176] investigated the effect of low vacuum helium plasma treatment on oilseeds and found that treatment with a treatment power of 80 and 100 W significantly improved the germination of rapeseed. The 100 W treatment showed the highest germination rate, germination index, and vigor index, which were 8.71%, 9.52%, and 21.86% higher than the control group, respectively. Li et al. [171] studied the effect of different doses (0–210 W) of CP on the germination of centipedegrass seeds and they observed that CP treatment at 120 W significantly increased the germination rate of seeds, while there was no significant difference in the germination rate of CP treatment at 210 W compared to that of non-CP treated seeds.
The effect of treatment time on seed germination was similar to that of the effect of power. Shashikanthalu et al. [172] used dielectric barrier discharge plasma to treat cumin seeds. At the same voltage (2 kV), 2 min exposure time showed 25.34% of germination and 3 min showed the highest germination percentage of 28.67%, but when the treatment time was increased to 4 min, the germination percentage decreased to 20%. The germination rate of seeds increased significantly at all power levels for the appropriate treatment duration, but the increasing trend was not consistently maintained, and longer treatment durations showed a negative effect on the seeds. Mravlje et al. [177] also observed a negative effect of CP treatment above 60 s on the germination of buckwheat seeds. This may be due to the higher density of electrons, stronger UV radiation, and more heat generated by the prolonged CP processing. Therefore, the duration of the plasma treatment can be decreased to increase the seed germination rate at high power.
Future Prospects
Although there are a lot of laboratory studies about CP application on LMFs, there is a lack of application reports in the agricultural field and food processing plants. Applying plasma technology to the food industry presents many challenges. First, enlarging the discharge area for continuous processing without compromising plasma uniformity is the main challenge [178]. Devices such as multiple plasma sources or microplasma arrays have been shown to perform scale-up for large-volume food processing. However, issues such as system volume, sample surface interaction, power and current supply, and cost-effectiveness have yet to be addressed. In addition, precise control of the CP generation process is required, which is affected by several variables, including processing gas, gas flow rate, current size, and process time which alter the outcome of treatment. The most likely methods of process control are electrical and optical measurements, but studies of electrical and optical signatures of plasma species have been underreported. Another consideration during processing is treatment heterogeneity, where there may be uneven contact between plasma and food, which can lead to different results in the same batch of samples. Many studies have reported the use of plasma-treated liquids, such as plasma-activated water, in contact with food matrices to compensate for these deficiencies [179]. However, this method does not apply to LMFs, and the introduction of excess moisture may affect the shelf life of the products and add additional processing steps. A proper vibrating unit for containing LMFs during treatment may be an option to solve the heterogeneity issue. In order to apply CP in practice, issues such as safety evaluation, regulatory policy, and consumer acceptance also need to be considered. More research on the feasibility of CP is needed in the future to pave the way for its application in the industry.
Conclusions
CP can significantly reduce harmful microorganisms on the surface of various LMFs such as walnuts, hazelnuts, peppers, starch, and spices, by generating active species, ultraviolet radiation, and electric fields. This helps to extend the shelf life of foods while having minimal impact on food quality. At the same time, many studies have shown that CP technology can change the functional properties of food. By adjusting the parameters of the cold plasma generation process, it is possible to obtain target products that meet people’s expectations. CP technology is also an effective approach to detoxify mycotoxin-contaminated LMFs by degrading various mycotoxins such as AF, ZEN, OTA, T-2, and HT-2. In addition, the application of CP in seed germination is promising and could be of great significance to the global food crisis. Even though CP technology has numerous advantages, it is still in the early stages of commercialization, and there are still many problems that have not yet been solved. For instance, due to the diversity of species contained in CP and the complexity of food matrices, the mechanisms by which CP inactivates microorganisms, degrades mycotoxins, and promotes seed germination are still unclear and future studies are still in need for a comprehensive understanding. The transition of plasma technology from the laboratory to the food industry also has problems such as unstable work efficiency, lack of process compatible technology design, and imperfect process control technology, and the scientific community is connecting plasma technology with other disciplines to solve these issues.
References
Liu S, Wei X, Tang J, Qin W, Wu Q (2021) Recent developments in low-moisture foods: microbial validation studies of thermal pasteurization processes. Crit Rev Food Sci Nutr 343:Article 128398. https://doi.org/10.1080/10408398.2021.2016601
Liu S, Roopesh MS, Tang J, Wu Q, Qin W (2022) Recent development in low-moisture foods: Microbial safety and thermal process. Food Res Int 155:111072. https://doi.org/10.1016/j.foodres.2022.111072
Huang Z, Yu X, Yang Q, Zhao Y, Wu W (2021) Aptasensors for Staphylococcus aureus risk assessment in food. Front Microbiol 12:Article 714265. https://doi.org/10.3389/fmicb.2021.714265
Wason S, Verma T, Subbiah J (2021) Validation of process technologies for enhancing the safety of low-moisture foods: A review. Compr Rev Food Sci Food Safety 20(5):4950–4992. https://doi.org/10.1111/1541-4337.12800
Chitrakar B, Zhang M, Adhikari B (2018) Dehydrated foods: Are they microbiologically safe? Crit Rev Food Sci Nutr 59(17):1–43. https://doi.org/10.1080/10408398.2018.1466265
Denes T, Wiedmann M (2014) Environmental responses and phage susceptibility in foodborne pathogens: implications for improving applications in food safety. Curr Opin Biotechnol 26:45–49. https://doi.org/10.1016/j.copbio.2013.09.001
Dastyar H, Ghoreishi SM, Ghani M (2022) Shaker-Assisted Liquid-Liquid Microextraction Followed by Solidification of Floating Organic Droplet and Back-Extraction Procedure besides Partial Least Squares Regression for Simultaneous Spectrophotometric Determination of Benzoic Acid and Sorbic Acid. Polycyclic Aromat Compd. https://doi.org/10.1080/10406638.2022.2039226
Dey S, Nagababu BH (2022) Applications of food color and bio-preservatives in the food and its effect on the human health. Food Chem Adv 1:Article 100019. https://doi.org/10.1016/j.focha.2022.100019
Kim TD, Begyn K, Khanal S, Taghlaoui F, Heyndrickx M, Rajkovic A, ... Aertsen A (2021) Bacillus weihenstephanensis can readily evolve for increased endospore heat resistance without compromising its thermotype. Int J Food Microbiol 341:109072. https://doi.org/10.1016/j.ijfoodmicro.2021.109072
Khouryieh HA (2021) Novel and emerging technologies used by the US food processing industry. Innov Food Sci Emerg Technol 67:Article 102559. https://doi.org/10.1016/j.ifset.2020.102559
Adebo OA, Molelekoa T, Makhuvele R, Adebiyi JA, Oyedeji AB, Gbashi S, Njobeh PB (2021) A review on novel non-thermal food processing techniques for mycotoxin reduction. Int J Food Sci Technol 56(1):13–27. https://doi.org/10.1111/ijfs.14734
Abedi E, Pourmohammadi K (2021) Chemical modifications and their effects on gluten protein: An extensive review. Food Chem 343:Article 128398. https://doi.org/10.1016/j.foodchem.2020.128398
Ali F, Qanmber G, Li F, Wang Z (2022) Updated role of ABA in seed maturation, dormancy, and germination. J Adv Res 35:199–214. https://doi.org/10.1016/j.jare.2021.03.011
Klupczynska EA, Pawlowski TA (2021) Regulation of seed dormancy and germination mechanisms in a changing environment. Int J Mol Sc 22(3):Article 1357. https://doi.org/10.3390/ijms22031357
Waskow A, Avino F, Howling A, Furno I (2022) Entering the plasma agriculture field: An attempt to standardize protocols for plasma treatment of seeds. Plasma Process Polym 19(1):Article e2100152. https://doi.org/10.1002/ppap.202100152
Iwasaki M, Penfield S, Lopez-Molina L (2022) Parental and environmental control of seed dormancy in arabidopsis thaliana. Annu Rev Plant Biol 73:355–378. https://doi.org/10.1146/annurev-arplant-102820-090750
Okyere AY, Rajendran S, Annor GA (2022) Cold plasma technologies: Their effect on starch properties and industrial scale-up for starch modification. Curr Res Food Sci 5:451–463. https://doi.org/10.1016/j.crfs.2022.02.007
Shelar A, Singh AV, Dietrich P, Maharjan RS, Thissen A, Didwal PN, Patil R (2022) Emerging cold plasma treatment and machine learning prospects for seed priming: a step towards sustainable food production. RSC Adv 12(17):10467–10488. https://doi.org/10.1039/d2ra00809b
Kletschkus K, Haralambiev L, Mustea A, Bekeschus S, Stope MB (2020) Review of innovative physical therapy methods: Introduction to the principles of cold physical plasma. In Vivo 34(6):3103–3107. https://doi.org/10.21873/invivo.12143
Mandal R, Singh A, Singh AP (2018) Recent developments in cold plasma decontamination technology in the food industry. Trends Food Sci Technol 80:93–103. https://doi.org/10.1016/j.tifs.2018.07.014
Mollakhalili-Meybodi N, Yousefi M, Nematollahi A, Khorshidian N (2021) Effect of atmospheric cold plasma treatment on technological and nutrition functionality of protein in foods. Eur Food Res Technol 247(7):1579–1594. https://doi.org/10.1007/s00217-021-03750-w
Xiang Q, Liu X, Li J, Ding T, Zhang H, Zhang X, Bai Y (2018) Influences of cold atmospheric plasma on microbial safety, physicochemical and sensorial qualities of meat products. J Food Sci Technol-Mysore 55(3):846–857. https://doi.org/10.1007/s13197-017-3020-y
Sakudo A, Misawa T, Yagyu Y (2020) Equipment design for cold plasma disinfection of food products. Adv Cold Plasma Appl Food Safety Preserv 289–307. https://doi.org/10.1016/b978-0-12-814921-8.00010-4
Xiang Q, Huangfu L, Dong S, Ma Y, Li K, Niu L, Bai Y (2021) Feasibility of atmospheric cold plasma for the elimination of food hazards: Recent advances and future trends. Crit Rev Food Sci Nutr. https://doi.org/10.1080/10408398.2021.2002257
Feizollahi E, Misra NN, Roopesh MS (2021) Factors influencing the antimicrobial efficacy of dielectric barrier discharge (DBD) atmospheric cold plasma (ACP) in food processing applications. Crit Rev Food Sci Nutr 61(4):666–689. https://doi.org/10.1080/10408398.2020.1743967
Zhang H, Ma D, Qiu R, Tang Y, Du C (2017) Non-thermal plasma technology for organic contaminated soil remediation: A review. Chem Eng J 313:157–170. https://doi.org/10.1016/j.cej.2016.12.067
Nasiru MM, Frimpong EB, Muhammad U, Qian J, Mustapha AT, Yan W, Zhang J (2021) Dielectric barrier discharge cold atmospheric plasma: Influence of processing parameters on microbial inactivation in meat and meat products. Compr Rev Food Sci Food Safety 20(3):2626–2659. https://doi.org/10.1111/1541-4337.12740
Mok C, Lee T, Puligundla P (2015) Afterglow corona discharge air plasma (ACDAP) for inactivation of common food-borne pathogens. Food Res Int 69:418–423. https://doi.org/10.1016/j.foodres.2014.11.034
Scholtz V, Julak J, Kriha V (2010) The microbicidal effect of low-temperature plasma generated by corona discharge: Comparison of various microorganisms on an agar surface or in aqueous suspension. Plasma Processes Polym 7(3–4):237–243. https://doi.org/10.1002/ppap.200900072
Tolouie H, Mohammadifar MA, Ghomi H, Hashemi M (2018) Cold atmospheric plasma manipulation of proteins in food systems. Crit Rev Food Sci Nutr 58(15):2583–2597. https://doi.org/10.1080/10408398.2017.1335689
Coutinho NM, Silveira MR, Rocha RS, Moraes J, Ferreira MVS, Pimentel TC, Cruz AG (2018) Cold plasma processing of milk and dairy products. Trends Food Sci Technol 74:56–68. https://doi.org/10.1016/j.tifs.2018.02.008
Tanaka H, Bekeschus S, Yan D, Hori M, Keidar M, Laroussi M (2021) Plasma-treated solutions (PTS) in cancer therapy. Cancers 13(7):Article 1737. https://doi.org/10.3390/cancers13071737
Misra N, Bhatt S, Khonsari FA, Kumar V (2021) State of the art in nonthermal plasma processing for biomedical applications: Can it help fight viral pandemics like COVID-19? Plasma Process Polym 18(7):Article e2000215. https://doi.org/10.1002/ppap.202000215
Wu M, Jia L, Lu S, Qin Z, Wei S, Yan R (2021) Interfacial performance of high-performance fiber-reinforced composites improved by cold plasma treatment: A review. Surf Interfaces 24:Article 101077. https://doi.org/10.1016/j.surfin.2021.101077
Patange A, Boehm D, Giltrap M, Lu P, Cullen PJ, Bourke P (2018) Assessment of the disinfection capacity and eco-toxicological impact of atmospheric cold plasma for treatment of food industry effluents. Sci Total Environ 631–632:298–307. https://doi.org/10.1016/j.scitotenv.2018.02.269
Laroque DA, Seo ST, Valencia GA, Laurindo JB, Mattar Carciofi BA (2022) Cold plasma in food processing: Design, mechanisms, and application. J Food Eng 312:Article 110748. https://doi.org/10.1016/j.jfoodeng.2021.110748
Roh SH, Oh YJ, Lee SY, Kang JH, Min SC (2020) Inactivation of Escherichia coli O157:H7, Salmonella, Listeria monocytogenes, and Tulane virus in processed chicken breast via atmospheric in-package cold plasma treatment. LWT 127:109429. https://doi.org/10.1016/j.lwt.2020.109429
Hojnik N, Modic M, Walsh JL, Zigon D, Javornik U, Plavec J, ... Cvelbar U (2021) Unravelling the pathways of air plasma induced aflatoxin B-1 degradation and detoxification. J Hazard Mater 403:123593. https://doi.org/10.1016/j.jhazmat.2020.123593
Gavahian M, Sarangapani C, Misra NN (2021) Cold plasma for mitigating agrochemical and pesticide residue in food and water: Similarities with ozone and ultraviolet technologies. Food Res Int 141:110138. https://doi.org/10.1016/j.foodres.2021.110138
Anbarasan R, Jaspin S, Bhavadharini B, Pare A, Pandiselvam R, Mahendran R (2022) Chlorpyrifos pesticide reduction in soybean using cold plasma and ozone treatments. LWT 159:113193. https://doi.org/10.1016/j.lwt.2022.113193
Cong L, Huang M, Zhang J, Yan W (2021) Effect of dielectric barrier discharge plasma on the degradation of malathion and chlorpyrifos on lettuce. J Sci Food Agric 101(2):424–432. https://doi.org/10.1002/jsfa.10651
Mir SA, Siddiqui MW, Dar BN, Shah MA, Wani MH, Roohinejad S, Ali A (2020) Promising applications of cold plasma for microbial safety, chemical decontamination and quality enhancement in fruits. J Appl Microbiol 129(3):474–485. https://doi.org/10.1111/jam.14541
Chaple S, Sarangapani C, Jones J, Carey E, Causeret L, Genson A, ... Bourke P (2020) Effect of atmospheric cold plasma on the functional properties of whole wheat (Triticum aestivum L.) grain and wheat flour. Innov Food Sci Emerg Technol 66:Article 102529. https://doi.org/10.1016/j.ifset.2020.102529
Terebun P, Kwiatkowski M, Starek A, Reuter S, Mok YS, Pawlat J (2021) Impact of short time atmospheric plasma treatment on onion seeds. Plasma Chem Plasma Process 41(2):559–571. https://doi.org/10.1007/s11090-020-10146-3
Ziuzina D, Misra N, Han L (2019) Investigation of a large gap cold plasma reactor for continuous in-package decontamination of fresh strawberries and spinach. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2019.102229
Hernandez-Torres CJ, Reyes-Acosta YK, Chavez-Gonzalez ML, Davila-Medina MD, Verma DK, Martinez-Hernandez JL, Aguilar CN (2022) Recent trends and technological development in plasma as an emerging and promising technology for food biosystems. Saudi J Biol Sci 29(4):1957–1980. https://doi.org/10.1016/j.sjbs.2021.12.023
Vandenberg LN, Bugos J (2021) Assessing the public health implications of the food preservative propylparaben: Has this chemical been safely used for decades. Curr Environ Health Rep 8(1):54–70. https://doi.org/10.1007/s40572-020-00300-6
Hernandez-Hernandez HM, Moreno-Vilet L, Villanueva-Rodriguez SJ (2019) Current status of emerging food processing technologies in Latin America: Novel non-thermal processing. Innov Food Sci Emerg Technol 58:Article 102233. https://doi.org/10.1016/j.ifset.2019.102233
Ozkan G, Guldiken B, Capanoglu E (2019) Effect of novel food processing technologies on beverage antioxidants. Process Sustain Beverages 413–449. https://doi.org/10.1016/b978-0-12-815259-1.00012-4
Temba BA, Sultanbawa Y, Kriticos DJ, Fox GP, Harvey JJW, Fletcher MT (2016) Tools for defusing a major global food and feed safety risk: Nonbiological postharvest procedures to decontaminate mycotoxins in foods and feeds. J Agric Food Chem 64(47):8959–8972. https://doi.org/10.1021/acs.jafc.6b03777
Ganesan AR, Tiwari U, Ezhilarasi PN, Rajauria G (2021) Application of cold plasma on food matrices: A review on current and future prospects. J Food Process Preserv 45(1):Article e15070. https://doi.org/10.1111/jfpp.15070
Umair M, Jabbar S, Ayub Z, Aadil RM, Abid M, Zhang J, Zhao L (2021) Recent advances in plasma technology: Influence of atmospheric cold plasma on spore inactivation. Food Rev Intl. https://doi.org/10.1080/87559129.2021.1888972
Ziuzina D, Petil S, Cullen PJ, Keener KM, Bourke P (2014) Atmospheric cold plasma inactivation of Escherichia coli, Salmonella enterica serovar Typhimurium and Listeria monocytogenes inoculated on fresh produce. Food Microbiol 42:109–116. https://doi.org/10.1016/j.fm.2014.02.007
Gaunt LF, Beggs CB, Georghiou GE (2006) Bactericidal action of the reactive species produced by gas-discharge nonthermal plasma at atmospheric pressure: A review. IEEE Trans Plasma Sci 34(4):1257–1269. https://doi.org/10.1109/tps.2006.878381
Yusupov M, Bogaerts A, Huygh S, Snoeckx R, van Duin ACT, Neyts EC (2013) Plasma-induced destruction of bacterial cell wall components: A reactive molecular dynamics simulation. J Phys Chem C 117(11):5993–5998. https://doi.org/10.1021/jp3128516
Misra NN, Jo C (2017) Applications of cold plasma technology for microbiological safety in meat industry. Trends Food Sci Technol 64:74–86. https://doi.org/10.1016/j.tifs.2017.04.005
Eto H, Ono Y, Ogino A, Nagatsu M (2008) Low-temperature sterilization of wrapped materials using flexible sheet-type dielectric barrier discharge. Appl Phys Lett 93(22):Article 221502. https://doi.org/10.1063/1.3039808
Fridman G, Brooks AD, Balasubramanian M, Fridman A, Gutsol A, Vasilets VN, Friedman G (2007) Comparison of direct and indirect effects of non-thermal atmospheric-pressure plasma on bacteria. Plasma Processes Polym 4(4):370–375. https://doi.org/10.1002/ppap.200600217
Han L, Patil S, Boehm D, Milosavljevic V, Cullen PJ, Bourke P (2016) Mechanisms of inactivation by high-voltage atmospheric cold plasma differ for Escherichia coli and Staphylococcus aureus. Appl Environ Microbiol 82(2):450–458. https://doi.org/10.1128/aem.02660-15
Cui H, Li H, Abdel-Samie MA, Surendhiran D, Lin L (2021) Anti-Listeria monocytogenes biofilm mechanism of cold nitrogen plasma. Innov Food Sci Emerg Technol 67:Article 102571. https://doi.org/10.1016/j.ifset.2020.102571
Qian J, Ma L, Yan W, Zhuang H, Huang M, Zhang J, Wang J (2022) Inactivation kinetics and cell envelope damages of foodborne pathogens Listeria monocytogenes and Salmonella Enteritidis treated with cold plasma. Food Microbiol 101:Article 103891. https://doi.org/10.1016/j.fm.2021.103891
Deng S, Ruan R, Mok CK, Huang G, Lin X, Chen P (2007) Inactivation of Escherichia coli on almonds using nonthermal plasma. J Food Sci 72(2):M62–M66. https://doi.org/10.1111/j.1750-3841.2007.00275.x
Beyrer M, Smeu I, Martinet D, Howling A, Pina-Perez MC, Ellert C (2020) Cold atmospheric plasma inactivation of microbial spores compared on reference surfaces and powder particles. Food Bioprocess Technol 13(5):827–837. https://doi.org/10.1007/s11947-020-02438-5
Darvish H, Ramezan Y, Khani MR, Kamkari A (2022) Effect of low-pressure cold plasma processing on decontamination and quality attributes of Saffron (Crocus sativus L.). Food Sci Nutr. https://doi.org/10.1002/fsn3.2824
Charoux CMG, Free L, Hinds LM, Vijayaraghavan RK, Daniels S, O’Donnell CP, Tiwari BK (2020) Effect of non-thermal plasma technology on microbial inactivation and total phenolic content of a model liquid food system and black pepper grains. LWT 118:108716. https://doi.org/10.1016/j.lwt.2019.108716
Ahangari M, Ramezan Y, Khani MR (2021) Effect of low pressure cold plasma treatment on microbial decontamination and physicochemical properties of dried walnut kernels (Juglans regia L.). J Food Process Eng 44(1):Article e13593. https://doi.org/10.1111/jfpe.13593
Hemmati V, Garavand F, Goudarzi M, Sarlak Z, Cacciotti I, Tiwari BK (2021) Cold atmospheric-pressure plasma treatment of turmeric powder: microbial load, essential oil profile, bioactivity and microstructure analyses. Int J Food Sci Technol 56(5):2224–2232. https://doi.org/10.1111/ijfs.14838
Hertwig C, Reineke K, Ehlbeck J, Knorr D, Schlueter O (2015) Decontamination of whole black pepper using different cold atmospheric pressure plasma applications. Food Control 55:221–229. https://doi.org/10.1016/j.foodcont.2015.03.003
Sen Y, Onal-Ulusoy B, Mutlu M (2019) Aspergillus decontamination in hazelnuts: Evaluation of atmospheric and low-pressure plasma technology. Innov Food Sci Emerg Technol 54:235–242. https://doi.org/10.1016/j.ifset.2019.04.014
Durek J, Froehling A, Bussler S, Hase A, Ehlbeck J, Schlueter OK (2022) Pilot-scale generation of plasma processed air and its influence on microbial count, microbial diversity, and selected quality parameters of dried herbs. Innov Food Sci Emerg Technol 75:Article 102890. https://doi.org/10.1016/j.ifset.2021.102890
Basaran P, Basaran-Akgul N, Oksuz L (2008) Elimination of Aspergillus parasiticus from nut surface with low pressure cold plasma (LPCP) treatment. Food Microbiol 25(4):626–632. https://doi.org/10.1016/j.fm.2007.12.005
Niemira BA (2012) Cold plasma reduction of Salmonella and Escherichia coli O157:H7 on almonds using ambient pressure gases. J Food Sci 77(3):M171–M175. https://doi.org/10.1111/j.1750-3841.2011.02594.x
Amini M, Ghoranneviss M (2016) Effects of cold plasma treatment on antioxidants activity, phenolic contents and shelf life of fresh and dried walnut (Juglans regia L.) cultivars during storage. LWT 73:178–184. https://doi.org/10.1016/j.lwt.2016.06.014
Hertwig C, Reineke K, Ehlbeck J, Erdogdu B, Rauh C, Schlueter O (2015) Impact of remote plasma treatment on natural microbial load and quality parameters of selected herbs and spices. J Food Eng 167:12–17. https://doi.org/10.1016/j.jfoodeng.2014.12.017
Molina-Hernandez JB, Laika J, Peralta-Ruiz Y, Palivala VK, Tappi S, Cappelli F, ... Chaves-Lopez C (2022) Influence of atmospheric cold plasma exposure on naturally present fungal spores and physicochemical characteristics of sundried tomatoes (Solanum lycopersicum L.). Foods 11(2):Article 210. https://doi.org/10.3390/foods11020210
Lee SY, In J, Chung M-S, Min SC (2021) Microbial decontamination of particulate food using a pilot-scale atmospheric plasma jet treatment system. J Food Eng 294:110436. https://doi.org/10.1016/j.jfoodeng.2020.110436
Kang M-H, Veerana M, Eom S, Uhm H-S, Ryu S, Park G (2020) Plasma mediated disinfection of rice seeds in water and air. J Phys D-Appl Phys 53(21):Article 214001. https://doi.org/10.1088/1361-6463/ab79de
Charoux CMG, Patange A, Lamba S, O’Donnell CP, Tiwari BK, Scannell AGM (2021) Applications of nonthermal plasma technology on safety and quality of dried food ingredients. J Appl Microbiol 130(2):325–340. https://doi.org/10.1111/jam.14823
Alp D, Bulantekin O (2021) The microbiological quality of various foods dried by applying different drying methods: a review. Eur Food Res Technol 247(6):1333–1343. https://doi.org/10.1007/s00217-021-03731-z
Reddy KRN, Salleh B, Saad B, Abbas HK, Abel CA, Shier WT (2010) An overview of mycotoxin contamination in foods and its implications for human health. Toxin Rev 29(1):3–26. https://doi.org/10.3109/15569541003598553
Sun Z, Xu J, Wang G, Song A, Li C, Zheng S (2020) Hydrothermal fabrication of rectorite based biocomposite modified by chitosan derived carbon nanoparticles as efficient mycotoxins adsorbents. Appl Clay Sci 184:Article 105373. https://doi.org/10.1016/j.clay.2019.105373
Janik E, Niemcewicz M, Ceremuga M, Stela M, Saluk-Bijak J, Siadkowski A, Bijak M (2020) Molecular aspects of mycotoxins-A serious problem for human health. Int J Mol Sci 21(21):Article 8187. https://doi.org/10.3390/ijms21218187
Alizadeh AM, Hashempour-Baltork F, Khaneghah AM, Hosseini H (2021) New perspective approaches in controlling fungi and mycotoxins in food using emerging and green technologies. Curr Opin Food Sci 39:7–15. https://doi.org/10.1016/j.cofs.2020.12.006
Gavahian M, Mathad GN, Oliveira CAF, Khaneghah AM (2021) Combinations of emerging technologies with fermentation: Interaction effects for detoxification of mycotoxins? Food Res Int 141:110104. https://doi.org/10.1016/j.foodres.2021.110104
Sipos P, Peles F, Brasso DL, Beri B, Pusztahelyi T, Pocsi I, Gyori Z (2021) Physical and chemical methods for reduction in aflatoxin content of feed and food. Toxins 13(3):Article 204. https://doi.org/10.3390/toxins13030204
Yousefi M, Mohammadi MA, Khajavi MZ, Ehsani A, Scholtz V (2021) Application of novel non-thermal physical technologies to degrade mycotoxins. J Fungi 7(5):Article 395. https://doi.org/10.3390/jof7050395
Liu Y, Galani Yamdeu JH, Gong YY, Orfila C (2020) A review of postharvest approaches to reduce fungal and mycotoxin contamination of foods. Compr Rev Food Sci Food Safety 19(4):1521–1560. https://doi.org/10.1111/1541-4337.12562
Pankaj SK, Shi H, Keener KM (2018) A review of novel physical and chemical decontamination technologies for aflatoxin in food. Trends Food Sci Technol 71:73–83. https://doi.org/10.1016/j.tifs.2017.11.007
Gayan E, Condon S, Alvarez I (2014) Biological aspects in food preservation by ultraviolet light: A review. Food Bioprocess Technol 7(1):1–20. https://doi.org/10.1007/s11947-013-1168-7
Byun K-H, Park SY, Lee DU, Chun HS, Ha S-D (2020) Effect of UV-C irradiation on inactivation of Aspergillus flavus and Aspergillus parasiticus and quality parameters of roasted coffee bean (Coffea arabica L.). Food Addit Contam Chem Anal Control Expo Risk Assess 37(3), 507–518. https://doi.org/10.1080/19440049.2020.1711971
Oms-Oliu G, Martin-Belloso O, Soliva-Fortuny R (2010) Pulsed light treatments for food preservation. A review. Food Bioprocess Technol 3(1):13–23. https://doi.org/10.1007/s11947-008-0147-x
Guo Y, Zhao L, Ma Q, Ji C (2021) Novel strategies for degradation of aflatoxins in food and feed: A review. Food Res Int 140:Article 109878. https://doi.org/10.1016/j.foodres.2020.109878
Mir SA, Dar BN, Shah MA, Sofi SA, Hamdani AM, Oliveira CAF, ... Sant’Ana AS (2021) Application of new technologies in decontamination of mycotoxins in cereal grains: Challenges, and perspectives. Food Chem Toxicol 148:Article 111976. https://doi.org/10.1016/j.fct.2021.111976
Rustom IYS (1997) Aflatoxin in food and feed: Occurrence, legislation and inactivation by physical methods. Food Chem 59(1):57–67. https://doi.org/10.1016/s0308-8146(96)00096-9
Luo X, Qi L, Liu Y, Wang R, Yang D, Li K, ... Chen Z (2017) Effects of electron beam lrradiation on zearalenone and ochratoxin A in naturally contaminated corn and corn quality parameters. Toxins 9(3):84. https://doi.org/10.3390/toxins9030084
Sujayasree OJ, Chaitanya AK, Bhoite R, Pandiselvam R, Kothakota A, Gavahian M, Khaneghah AM (2022) Ozone: An advanced oxidation technology to enhance sustainable food consumption through mycotoxin degradation. Ozone Sci Eng 44(1):17–37. https://doi.org/10.1080/01919512.2021.1948388
Qi L, Li Y, Luo X, Wang R, Zheng R, Wang L, Chen Z (2016) Detoxification of zearalenone and ochratoxin A by ozone and quality evaluation of ozonised corn. Food Addit Contam Chem Anal Control Expo Risk Assess 33(11):1700–1710. https://doi.org/10.1080/19440049.2016.1232863
Akbas MY, Ozdemir M (2008) Effect of gaseous ozone on microbial inactivation and sensory of flaked red peppers. Int J Food Sci Technol 43(9):1657–1662. https://doi.org/10.1111/j.1365-2621.2008.01722.x
Drishya C, Yoha KS, Perumal AB, Moses JA, Anandharamakrishnan C, Balasubramaniam VM (2022) Impact of nonthermal food processing techniques on mycotoxins and their producing fungi. Int J Food Sci Technol 57(4):2140–2148. https://doi.org/10.1111/ijfs.15444
Villarreal-Barajas T, Vazquez-Duran A, Mendez-Albores A (2022) Effectiveness of electrolyzed oxidizing water on fungi and mycotoxins in food. Food Control 131:108454. https://doi.org/10.1016/j.foodcont.2021.108454
Feizollahi E, Roopesh MS (2021) Mechanisms of deoxynivalenol (DON) degradation during different treatments: A review. Crit Rev Food Sci Nutr. https://doi.org/10.1080/10408398.2021.1895056
Kaur M, Hueberli D, Bayliss KL (2020) Cold plasma: exploring a new option for management of postharvest fungal pathogens, mycotoxins and insect pests in Australian stored cereal grain. Crop Pasture Sci 71(8):715–724. https://doi.org/10.1071/cp20078
Nishimwe K, Agbemafle I, Reddy MB, Keener K, Maier DE (2021) Cytotoxicity assessment of Aflatoxin B1 after high voltage atmospheric cold plasma treatment. Toxicon 194:17–22. https://doi.org/10.1016/j.toxicon.2021.02.008
Shi H, Cooper B, Stroshine RL, Ileleji KE, Keener KM (2017) Structures of degradation products and degradation pathways of aflatoxin B-1 by high-voltage atmospheric cold plasma (HVACP) treatment. J Agric Food Chem 65(30):6222–6230. https://doi.org/10.1021/acs.jafc.7b01604
Shi H, Ileleji K, Stroshine RL, Keener K, Jensen JL (2017) Reduction of aflatoxin in corn by high voltage atmospheric cold plasma. Food Bioprocess Technol 10(6):1042–1052. https://doi.org/10.1007/s11947-017-1873-8
Patricia Casas-Junco P, Raymundo Solis-Pacheco J, Arturo Ragazzo-Sanchez J, Rosa Aguilar-Uscanga B, Ulises Bautista-Rosales P, Calderon-Santoyo M (2019) Cold plasma treatment as an alternative for ochratoxin A detoxification and inhibition of mycotoxigenic fungi in roasted coffee. Toxins 11(6). https://doi.org/10.3390/toxins11060337
Misra NN, Yadav B, Roopesh MS, Jo C (2019) Cold plasma for effective fungal and mycotoxin control in foods: Mechanisms, inactivation effects, and applications. Compr Rev Food Sci Food Safety 18(1):106–120. https://doi.org/10.1111/1541-4337.12398
Wang S-Q, Huang G-Q, Li Y-P, Xiao J-X, Zhang Y, Jiang W-L (2015) Degradation of aflatoxin B-1 by low-temperature radio frequency plasma and degradation product elucidation. Eur Food Res Technol 241(1):103–113. https://doi.org/10.1007/s00217-015-2439-5
Wielogorska E, Ahmed Y, Meneely J, Graham WG, Elliott CT, Gilmore BF (2019) A holistic study to understand the detoxification of mycotoxins in maize and impact on its molecular integrity using cold atmospheric plasma treatment. Food Chem 301:125281. https://doi.org/10.1016/j.foodchem.2019.125281
Xue M, Wang T, Sun Q, Qu G, Jia H, Zhu L (2021) Insights into the highly efficient detoxification of the biotoxin patulin in water by discharge plasma oxidation. Chem Eng J 411:128432. https://doi.org/10.1016/j.cej.2021.128432
Park BJ, Takatori K, Sugita-Konishi Y, Kim I-H, Lee M-H, Han D-W, Park J-C (2007) Degradation of mycotoxins using microwave-induced argon plasma at atmospheric pressure. Surf Coat Technol 201(9–11):5733–5737. https://doi.org/10.1016/j.surfcoat.2006.07.092
Siciliano I, Spadaro D, Prelle A, Vallauri D, Cavallero MC, Garibaldi A, Gullino ML (2016) Use of cold atmospheric plasma to detoxify hazelnuts from aflatoxins. Toxins 8(5):125. https://doi.org/10.3390/toxins8050125
Makari M, Hojjati M, Shahbazi S, Askari H (2021) Elimination of Aspergillus flavus from pistachio nuts with dielectric barrier discharge (DBD) cold plasma and its impacts on biochemical indices. J Food Qual 2021:9968711. https://doi.org/10.1155/2021/9968711
Devi Y, Thirumdas R, Sarangapani C, Deshmukh RR, Annapure US (2017) Influence of cold plasma on fungal growth and aflatoxins production on groundnuts. Food Control 77:187–191. https://doi.org/10.1016/j.foodcont.2017.02.019
Iqdiam BM, Abuagela MO, Boz Z, Marshall SM, Goodrich-Schneider R, Sims CA, ... Welt BA (2020) Effects of atmospheric pressure plasma jet treatment on aflatoxin level, physiochemical quality, and sensory attributes of peanuts. J Food Process Preser 44(1):e14305. https://doi.org/10.1111/jfpp.14305
Hojnik N, Modic M, Zigon D, Kovac J, Jurov A, Dickenson A, ... Cvelbar U (2021) Cold atmospheric pressure plasma-assisted removal of aflatoxin B-1 from contaminated corn kernels. Plasma Process Polym 18(1):e2000163. https://doi.org/10.1002/ppap.202000163
Puligundla P, Lee T, Mok C (2020) Effect of corona discharge plasma jet treatment on the degradation of aflatoxin B1 on glass slides and in spiked food commodities. LWT 124:108333. https://doi.org/10.1016/j.lwt.2019.108333
Hajnal EJ, Vukic M, Pezo L, Orcic D, Puac N, Skoro N, ... Simovic DS (2019) Effect of atmospheric cold plasma treatments on reduction of Alternaria toxins content in wheat flour. Toxins 11(12):704. https://doi.org/10.3390/toxins11120704
Durek J, Schlueter O, Roscher A, Durek P, Froehling A (2018) Inhibition or stimulation of ochratoxin A synthesis on inoculated barley triggered by diffuse coplanar surface barrier discharge plasma. Front Microbiol 9:2782. https://doi.org/10.3389/fmicb.2018.02782
Kis M, Milosevic S, Vulic A, Herceg Z, Vukusic T, Pleadin J (2020) Efficacy of low pressure DBD plasma in the reduction of T-2 and HT-2 toxin in oat flour. Food Chem 316:126372. https://doi.org/10.1016/j.foodchem.2020.126372
Iqdiam BM, Feizollahi E, Arif MF, Jeganathan B, Vasanthan T, Thilakarathna MS, Roopesh MS (2021) Reduction of T-2 and HT-2 mycotoxins by atmospheric cold plasma and its impact on quality changes and germination of wheat grains. J Food Sci 86(4):1354–1371. https://doi.org/10.1111/1750-3841.15658
Feizollahi E, Roopesh MS (2021) Degradation of zearalenone by atmospheric cold plasma: Effect of selected process and product factors. Food Bioprocess Technol 14(11):2107–2119. https://doi.org/10.1007/s11947-021-02692-1
Feizollahi E, Iqdiam B, Vasanthan T, Thilakarathna MS, Roopesh MS (2020) Effects of atmospheric-pressure cold plasma treatment on deoxynivalenol degradation, Quality Parameters, and Germination of Barley Grains. Appl Sci-Basel 10(10):3530. https://doi.org/10.3390/app10103530
Marchese S, Polo A, Ariano A, Velotto S, Costantini S, Severino L (2018) Aflatoxin B1 and M1: Biological properties and their involvement in cancer development. Toxins 10(6):214. https://doi.org/10.3390/toxins10060214
Ganesan AR, Mohan K, Rajan DK, Pillay AA, Palanisami T, Sathishkumar P, Conterno L (2022) Distribution, toxicity, interactive effects, and detection of ochratoxin and deoxynivalenol in food: A review. Food Chem. https://doi.org/10.1016/j.foodchem.2021.131978
Hoppanova L, Dylikova J, Kovacik D, Medvecka V, Durina P, Krystofova S, Kalinakova B (2020) The effect of cold atmospheric pressure plasma on Aspergillus ochraceus and ochratoxin A production. Antonie Van Leeuwenhoek Int J Gen Mol Microbiol 113(10):1479–1488. https://doi.org/10.1007/s10482-020-01457-8
Wang X, Wang S, Yan Y, Wang W, Zhang L, Zong W (2020) The degradation of Alternaria mycotoxins by dielectric barrier discharge cold plasma. Food Control 117:107333. https://doi.org/10.1016/j.foodcont.2020.107333
Vaseghi N, Bayat M, Chaichi Nosrati A, Ghorannevis M, Hashemi JJIJ, o. M., & Microbiology, C. (2017) Plasma jet impacts on Citrinin production in isolates belonging to Penicillium Spp. Int J Mol Clin Microbiol 7(2):875–889
Lee KH, Kim H-J, Woo KS, Jo C, Kim J-K, Kim SH, ... Kim WH (2016) Evaluation of cold plasma treatments for improved microbial and physicochemical qualities of brown rice. LWT 73:442–447. https://doi.org/10.1016/j.lwt.2016.06.055
Suresh D, Manjunatha H, Srinivasan K (2007) Effect of heat processing of spices on the concentrations of their bioactive principles: Turmeric (Curcuma longa), red pepper (Capsicum annuum) and black pepper (Piper nigrum). J Food Compos Anal 20(3–4):346–351. https://doi.org/10.1016/j.jfca.2006.10.002
Kopuk B, Gunes R, Palabiyik I (2022) Cold plasma modification of food macromolecules and effects on related products. Food Chem 382:132356. https://doi.org/10.1016/j.foodchem.2022.132356
Selcuk M, Oksuz L, Basaran P (2008) Decontamination of grains and legumes infected with Aspergillus spp. and Penicillum spp. by cold plasma treatment. Bioresource Technol 99(11):5104–5109. https://doi.org/10.1016/j.biortech.2007.09.076
Wiktor A, Hrycak B, Jasinski M, Rybak K, Kieliszek M, Krasniewska K, Witrowa-Rajchert D (2020) Impact of atmospheric pressure microwave plasma treatment on quality of selected spices. Appl Sci-Basel 10(19):6815. https://doi.org/10.3390/app10196815
Bahrami N, Bayliss D, Chope G, Penson S, Perehinec T, Fisk ID (2016) Cold plasma: A new technology to modify wheat flour functionality. Food Chem 202:247–253. https://doi.org/10.1016/j.foodchem.2016.01.113
Choi S, Puligundla P, Mok C (2017) Effect of corona discharge plasma on microbial decontamination of dried squid shreds including physico-chemical and sensory evaluation. LWT 75:323–328. https://doi.org/10.1016/j.lwt.2016.08.063
Lee KH, Woo KS, Yong HI, Jo C, Lee SK, Lee BW, Kim H-J (2018) Assessment of microbial safety and quality changes of brown and white cooked rice treated with atmospheric pressure plasma. Food Sci Biotechnol 27(3):661–667. https://doi.org/10.1007/s10068-017-0297-6
Foligni R, Mannozzi C, Ismaiel L, Capelli F, Laurita R, Tappi S, ... Mozzon M (2022) Impact of cold atmospheric plasma (CAP) treatments on the oxidation of pistachio kernel lipids. Foods 11(3):419. https://doi.org/10.3390/foods11030419
Misra NN, Kaur S, Tiwari BK, Kaur A, Singh N, Cullen PJ (2015) Atmospheric pressure cold plasma (ACP) treatment of wheat flour. Food Hydrocolloids 44:115–121. https://doi.org/10.1016/j.foodhyd.2014.08.019
Zare L, Mollakhalili-Meybodi N, Fallahzadeh H, Arab M (2022) Effect of atmospheric pressure cold plasma (ACP) treatment on the technological characteristics of quinoa flour. LWT-Food Sci Technol 155:112898. https://doi.org/10.1016/j.lwt.2021.112898
Wu T-Y, Chang C-R, Chang T-J, Chang Y-J, Liew Y, Chau C-F (2019) Changes in physicochemical properties of corn starch upon modifications by atmospheric pressure plasma jet. Food Chem 283:46–51. https://doi.org/10.1016/j.foodchem.2019.01.043
Bie P, Pu H, Zhang B, Su J, Chen L, Li X (2016) Structural characteristics and rheological properties of plasma-treated starch. Innov Food Sci Emerg Technol 34:196–204. https://doi.org/10.1016/j.ifset.2015.11.019
Okyere AY, Boakye PG, Bertoft E, Annor GA (2022) Structural characterization and enzymatic hydrolysis of radio frequency cold plasma treated starches. J Food Sci 87(2):686–698. https://doi.org/10.1111/1750-3841.16037
Alves Filho EG, Silva LMA, Oiram Filho F, Rodrigues S, Fernandes FAN, Gallao MI, ... de Brito ES (2019) Cold plasma processing effect on cashew nuts composition and allergenicity. Food Res Int 125:108621. https://doi.org/10.1016/j.foodres.2019.108621
Gavahian M, Chu Y-H, Mousavi Khaneghah A, Barba FJ, Misra NN (2018) A critical analysis of the cold plasma induced lipid oxidation in foods. Trends Food Sci Technol 77:32–41. https://doi.org/10.1016/j.tifs.2018.04.009
Basak S, Annapure US (2022) Recent trends in the application of cold plasma for the modification of plant proteins - A review. Future Foods 5:100119. https://doi.org/10.1016/j.fufo.2022.100119
Rosell CM, Wang J, Aja S, Bean S, Lookhart G (2003) Wheat flour proteins as affected by transglutaminase and glucose oxidase. Cereal Chem 80(1):52–55. https://doi.org/10.1094/cchem.2003.80.1.52
Zhang S, Huang W, Feizollahi E, Roopesh MS, Chen L (2021) Improvement of pea protein gelation at reduced temperature by atmospheric cold plasma and the gelling mechanism study. Innov Food Sci Emerg Technol 67:102567. https://doi.org/10.1016/j.ifset.2020.102567
Ge X, Shen H, Su C, Zhang B, Zhang Q, Jiang H, Li W (2021) The improving effects of cold plasma on multi-scale structure, physicochemical and digestive properties of dry heated red adzuki bean starch. Food Chem 349:129159. https://doi.org/10.1016/j.foodchem.2021.129159
Kalaivendan RGT, Mishra A, Eazhumalai G, Annapure US (2022) Effect of atmospheric pressure non-thermal pin to plate plasma on the functional, rheological, thermal, and morphological properties of mango seed kernel starch. Int J Biol Macromol 196:63–71. https://doi.org/10.1016/j.ijbiomac.2021.12.013
Sun X, Saleh ASM, Sun Z, Ge X, Shen H, Zhang Q, ... Li W (2022). Modification of multi-scale structure, physicochemical properties, and digestibility of rice starch via microwave and cold plasma treatments. LWT-Food Sci Technol 153:112483. https://doi.org/10.1016/j.lwt.2021.112483
Sun X, Sun Z, Guo Y, Zhao J, Zhao J, Ge X, ... Yan W (2021) Effect of twin-xuscrew extrusion combined with cold plasma on multi-scale structure, physicochemical properties, and digestibility of potato starches. Innov Food Sci Emerg Technol 74:102855. https://doi.org/10.1016/j.ifset.2021.102855
Adhikari B, Adhikari M, Park G (2020) The effects of plasma on plant growth, development, and sustainability. Appl Sci-Basel 10(17):6045. https://doi.org/10.3390/app10176045
Holonec R, Viman O, Morar IM, Singeorzan S, Scheau C, Vlasin HD, ... Truta AM (2021) Non-chemical treatments to improve the seeds germination and plantlets growth of sessile oak. Not Bot Horti Agrobot Cluj-Napoca 49(3):12401. https://doi.org/10.15835/nbha49312401
Guragain RP, Baniya HB, Pradhan SP, Pandey BP, Subedi DP (2021) Influence of plasma-activated water (PAW) on the germination of radish, fenugreek, and pea seeds. AIP Adv 11(12):125304. https://doi.org/10.1063/5.0070800
Lotfy K, Al-Harbi NA, Abd El-Raheem H (2019) Cold atmospheric pressure nitrogen plasma jet for enhancement germination of wheat seeds. Plasma Chem Plasma Process 39(4):897–912. https://doi.org/10.1007/s11090-019-09969-6
Šerá B, Scholtz V, Jirešová J, Khun J, Julák J, Šerý M (2021) Effects of non-thermal plasma treatment on seed germination and early growth of leguminous plants—A Review. 10(8):1616. https://www.mdpi.com/2223-7747/10/8/1616
Sera B, Sery M, Zahoranova A, Tomekova J (2021) Germination improvement of three pine species (Pinus) after diffuse coplanar surface barrier discharge plasma treatment. Plasma Chem Plasma Process 41(1):211–226. https://doi.org/10.1007/s11090-020-10128-5
Waskow A, Howling A, Furno I (2021) Mechanisms of plasma-seed treatments as a potential seed processing technology. Front Phys 9:617345. https://doi.org/10.3389/fphy.2021.617345
Ahmed N, Shahid M, Siow KS, Wee MFMR, Haron FF, Patra A, Fazry S (2022) Germination and growth improvement of papaya utilizing oxygen (O-2) plasma treatment. J Phys D-Appl Phys 55(25):255205. https://doi.org/10.1088/1361-6463/ac6068
Perez Piza MC, Prevosto L, Zilli C, Cejas E, Kelly H, Balestrasse K (2018) Effects of non-thermal plasmas on seed-borne Diaporthe/Phomopsis complex and germination parameters of soybean seeds. Innov Food Sci Emerg Technol 49:82–91. https://doi.org/10.1016/j.ifset.2018.07.009
Holc M, Mozetic M, Recek N, Primc G, Vesel A, Zaplotnik R, Gselman P (2021) Wettability increase in plasma-treated agricultural seeds and its relation to germination improvement. Agronomy-Basel 11(8):1467. https://doi.org/10.3390/agronomy11081467
Los A, Ziuzina D, Akkermans S, Boehm D, Cullen PJ, Van Impe J, Bourke P (2018) Improving microbiological safety and quality characteristics of wheat and barley by high voltage atmospheric cold plasma closed processing. Food Res Int 106:509–521. https://doi.org/10.1016/j.foodres.2018.01.009
Bormashenko E, Grynyov R, Bormashenko Y, Drori E (2012) Cold radiofrequency plasma treatment modifies wettability and germination speed of plant seeds. Sci Rep 2:741. https://doi.org/10.1038/srep00741
Gomez-Ramierez A, Lopez-Santos C, Cantos M, Garcia JL, Molina R, Cotrino J, ... Gonzalez-Elipe AR (2017) Surface chemistry and germination improvement of Quinoa seeds subjected to plasma activation. Sci Rep 7:5924. https://doi.org/10.1038/s41598-017-06164-5
Recek N, Holc M, Vesel A, Zaplotnik R, Gselman P, Mozetic M, Primc G (2021) Germination of Phaseolus vulgaris L. seeds after a short treatment with a powerful RF plasma. Int J Mol Sci 22(13):6672. https://doi.org/10.3390/ijms22136672
de Groot GJJB, Hundt A, Murphy AB, Bange MP, Mai-Prochnow A (2018) Cold plasma treatment for cotton seed germination improvement. Sci Rep 8:14372. https://doi.org/10.1038/s41598-018-32692-9
Jiang J, Lu Y, Li J, Li L, He X, Shao H, Dong Y (2014) Effect of seed treatment by cold plasma on the resistance of tomato to ralstonia solanacearum (Bacterial Wilt). Plos One 9(5):Article e97753. https://doi.org/10.1371/journal.pone.0097753
Ji S-H, Choi K-H, Pengkit A, Im JS, Kim JS, Kim YH, Park G (2016) Effects of high voltage nanosecond pulsed plasma and micro DBD plasma on seed germination, growth development and physiological activities in spinach. Arch Biochem Biophys 605:117–128. https://doi.org/10.1016/j.abb.2016.02.028
Mildaziene V, Aleknaviciute V, Zukiene R, Pauzaite G, Nauciene Z, Filatova I, ... Baniulis D (2019) Treatment of Common Sunflower (Helianthus annus L.) Seeds with radio-frequency electromagnetic field and cold plasma induces changes in seed phytohormone balance, seedling development and leaf protein expression. Sci Rep 9:6437. https://doi.org/10.1038/s41598-019-42893-5
Ussenov YA, Akildinova A, Kuanbaevich BA, Serikovna KA, Gabdullin M, Dosbolayev M, Ramazanov T (2022) The Effect of Non-Thermal Atmospheric pressure plasma treatment of wheat seeds on germination parameters and α-amylase enzyme activity. IEEE Trans Plasma Sci 50(2):330–340. https://doi.org/10.1109/TPS.2022.3145831
Li L, Chen J, Wang H, Guo H, Li D, Li J, ... Zong J (2021) Cold plasma treatment improves seed germination and accelerates the establishment of centipedegrass. Crop Sci 61(4):2827–2836, Article 1–10. https://doi.org/10.1002/csc2.20513
Shashikanthalu SP, Ramireddy L, Radhakrishnan M (2020) Stimulation of the germination and seedling growth of Cuminum cyminum L. seeds by cold plasma. J Appl Res Med Aroma Plants 18:Article 100259. https://doi.org/10.1016/j.jarmap.2020.100259
Kitazaki S, Sarinont T, Koga K, Hayashi N, Shiratani M (2014) Plasma induced long-term growth enhancement of Raphanus sativus L. using combinatorial atmospheric air dielectric barrier discharge plasmas. Curr Appl Phys 14:S149–S153. https://doi.org/10.1016/j.cap.2013.11.056
Sarinont T, Amano T, Attri P, Koga K, Hayashi N, Shiratani M (2016) Effects of plasma irradiation using various feeding gases on growth of Raphanus sativus L. Arch Biochem Biophys 605:129–140. https://doi.org/10.1016/j.abb.2016.03.024
Jiang J, He X, Li L, Li J, Shao H, Xu Q, ... Dong Y (2014) Effect of cold plasma treatment on seed germination and growth of wheat. Plasma Sci Technol 16(1). https://doi.org/10.1088/1009-0630/16/1/12
Li L, Li J, Shao H, Dong Y (2018) Effects of low-vacuum helium cold plasma treatment on seed germination, plant growth and yield of oilseed rape. Plasma Sci Technol 20(9):Article 095502. https://doi.org/10.1088/2058-6272/aac3d0
Mravlje J, Regvar M, Staric P, Mozetic M, Vogel-Mikus K (2021) Cold plasma affects germination and fungal community structure of buckwheat seeds. Plants-Basel 10(5):Article 851. https://doi.org/10.3390/plants10050851
Cullen PJ, Lalor J, Scally L, Boehm D, Milosavljevic V, Bourke P, Keener K (2018) Translation of plasma technology from the lab to the food industry. Plasma Process Polym 15(2):Article e1700085. https://doi.org/10.1002/ppap.201700085
Gao Y, Francis K, Zhang X (2022) Review on formation of cold plasma activated water (PAW) and the applications in food and agriculture. Food Res Int 157:Article 111246. https://doi.org/10.1016/j.foodres.2022.111246
Funding
We gratefully acknowledge the financial support of Yongjiang Talent Introduction Programme (Young talent sub-project, 2021).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rao, W., Li, Y., Dhaliwal, H. et al. The Application of Cold Plasma Technology in Low-Moisture Foods. Food Eng Rev 15, 86–112 (2023). https://doi.org/10.1007/s12393-022-09329-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12393-022-09329-9