Abstract
The importance of addressing food safety is undeniable in today’s globalized food industry. Conventional thermal treatments negatively affect the nutritional and quality attributes of foods. Recently, non-thermal processing technologies have drawn much attention from the food industry and food research communities. Empirical data is available on the effectiveness of cold plasma, an emerging non-thermal technology, for eliminating chemical and biological hazards. This review aims to provide an overview of the impact of cold plasma on the major food hazards, including bacteria, bacterial spores, fungi, fungal spores, biofilms, viruses, mycotoxins, pesticides, and allergens. Cold plasma can effectively eliminate food hazards described above due to its numerous stress factors, including reactive oxygen/nitrogen species, charged particles, neutral or excited atoms and molecules, high energetic electrons, ultraviolet radiation, and electromagnetic fields. The oxidation, structural alteration, and destruction of cell components, dissociation of chemical bonds of mycotoxins as well as pesticides, and structural modification of allergens are the main inhibitory mechanisms of cold plasma. In several cases, nutritional and sensory attributes of cold plasma-treated foods were reported to be remained intact, and their shelf lives were extended. The factors affecting the decontamination efficacy of cold plasma are the treatment conditions, type of treated substrate, and the contaminants’ characteristics.



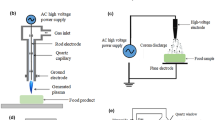
Copyright with permission from Elsevier.) c, d SEM images of S. Typhimurium cells; untreated (c) and treated by cold plasma (d). (Reprinted from Katsigiannis et al. (2021). Copyright with permission from Elsevier.) e, f TEM images of E. coli cells; untreated (e) and treated by cold plasma (f); g, h TEM images of S. cerevisiae cells; untreated (g) and treated by cold plasma (h); i, j SEM images of E. coli cells; untreated (i) and treated by cold plasma (j); k, l SEM images of S. cerevisiae cells; untreated (k) and treated by cold plasma (l). (Reprinted from Gan et al. [59]. Copyright with permission from Elsevier.) m, n SEM images of Z. rouxii cells; untreated (m) and treated by cold plasma (n). (Reprinted from Wang et al.[60, 61]. Copyright with permission from Elsevier.) o, p SEM images of A. flavus spores; untreated (o) and treated by cold plasma (p). (Reprinted from Ott et al. [64]. Copyright with permission from Elsevier)



Similar content being viewed by others
Abbreviations
- •OH:
-
Hydroxyl radical
- AFB1:
-
Aflatoxin B1
- AME:
-
Alternariol monomethyl ether
- AOH:
-
Alternariol
- ATR-FTIR:
-
Attenuated total reflectance–Fourier transform infrared spectroscopy
- DBD:
-
Dielectric barrier discharge
- DON:
-
Deoxynivalenol
- EPS:
-
Exopolysaccharide
- FB1:
-
Fumonisin B1
- H2O2 :
-
Hydrogen peroxide
- IgE:
-
Immunoglobulin E
- LPS:
-
Lipopolysaccharide
- O3 :
-
Ozone
- OH:
-
Hydroxyl
- PAW:
-
Plasma activated water
- PUFA:
-
Polyunsaturated fatty acid
- QS:
-
Quorum sensing
- RNS:
-
Reactive nitrogen species
- RONS:
-
Reactive oxygen and nitrogen species
- ROS:
-
Reactive oxygen species
- SDS-PAGE:
-
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis
- SEM:
-
Scanning electron microscopy
- TEM:
-
Transmission electron microscopy
- UV:
-
Ultraviolet
- ZEA:
-
Zearalenon
References
Ravash N, Hesari J (2021) A review on veterinary drug residues in foods of animal origin and the effect of different processes on their stability. Iranian J Biosyst Eng 52(1):147–168. https://doi.org/10.22059/ijbse.2021.307484.665326
Xin J, Wang X, Li N, Liu L, Lian Y, Wang M, Zhao RS (2020) Recent applications of covalent organic frameworks and their multifunctional composites for food contaminant analysis. Food Chem 330. https://doi.org/10.1016/j.foodchem.2020.127255
WHO (World Health Organization) (2022) Food safety. https://www.Who.Int/News-Room/Fact-Sheets/Detail/Food-Safety. Accessed 19 May 2022
Costa J, Bavaro SL, Benedé S, Diaz-Perales A, Bueno-Diaz C, Gelencser E, Klueber J, Larré C, Lozano-Ojalvo D, Lupi R, Mafra I, Mazzucchelli G, Molina E, Monaci L, Martín-Pedraza L, Piras C, Rodrigues PM, Roncada P, Schrama D, Holzhauser T (2022) Are physicochemical properties shaping the allergenic potency of plant allergens?. Clin Rev Allergy Immunol. https://doi.org/10.1007/s12016-020-08810-9
Costa J, Villa C, Verhoeckx K, Cirkovic-Velickovic T, Schrama D, Roncada P, Rodrigues PM, Piras C, Martín-Pedraza L, Monaci L, Molina E, Mazzucchelli G, Mafra I, Lupi R, Lozano-Ojalvo D, Larré C, Klueber J, Gelencser E, Bueno-Diaz C, Holzhauser T (2022) Are physicochemical properties shaping the allergenic potency of animal allergens?. Clin Rev Allergy Immunol. https://doi.org/10.1007/s12016-020-08826-1
Hooshyar L, Hesari J, Azadmard-Damirchi S (2020) Investigation of selected thermal and non-thermal preservative techniques to produce high quality and safe to drink sour cherry, red grape and pomegranate juices. J Food Sci Technol 57(5). https://doi.org/10.1007/s13197-019-04202-w
Hooshyar L, Jafarizadeh-Malmiri H, Berenjian A, Hesari J, Azadmard-Damirchi S, Şengül M (2020) Effects of thermosonication, sonication and mild heating on organoleptic attributes of three red fruit juices. Curr Nutr Food Sci 16(9). https://doi.org/10.2174/1573401316666200226105805
Xiang Q, Huangfu L, Dong S, Ma Y, Li K, Niu L, Bai Y (2021) Feasibility of atmospheric cold plasma for the elimination of food hazards: recent advances and future trends. Crit Rev Food Sci Nutr. https://doi.org/10.1080/10408398.2021.2002257
Ravash N, Hesari J (2022) The effect of high pressure processing (HPP) on milk and milk products. J Food Sci Technol (Iran) 18(121):131–146. https://doi.org/10.52547/FSCT.18.121.11
Ravash N, Peighambardoust SH, Soltanzadeh M, Pateiro M, Lorenzo JM (2020) Impact of high-pressure treatment on casein micelles, whey proteins, fat globules and enzymes activity in dairy products: a review. Crit Rev Food Sci Nutr. https://doi.org/10.1080/10408398.2020.1860899
Prasad A, Du L, Zubair M, Subedi S, Ullah A, Roopesh MS (2020) Applications of light-emitting diodes (LEDs) in food processing and water treatment. Food Eng Rev 12(3). https://doi.org/10.1007/s12393-020-09221-4
Prasad A, Gänzle M, Roopesh MS (2021). Food Res Int. https://doi.org/10.1016/j.foodres.2021.110601
Subedi S, Du L, Prasad A, Yadav B, Roopesh MS (2020) Inactivation of Salmonella and quality changes in wheat flour after pulsed light-emitting diode (LED) treatments. Food Bioprod Process 121. https://doi.org/10.1016/j.fbp.2020.02.004
Subedi S, Roopesh MS (2020) Simultaneous drying of pet food pellets and Salmonella inactivation by 395 nm light pulses in an LED reactor. J Food Eng 286. https://doi.org/10.1016/j.jfoodeng.2020.110110
Soltanzadeh M, Peighambardoust SH, Gullon P, Hesari J, Gullón B, Alirezalu K, Lorenzo J (2020) Quality aspects and safety of pulsed electric field (PEF) processing on dairy products: a comprehensive review. Food Rev Intl. https://doi.org/10.1080/87559129.2020.1849273
Sakooei-Vayghan R, Peighambardoust SH, Hesari J, Peressini D (2020) Effects of osmotic dehydration (with and without sonication) and pectin-based coating pretreatments on functional properties and color of hot-air dried apricot cubes. Food Chem. https://doi.org/10.1016/j.foodchem.2019.125978
Sakooei-Vayghan R, Peighambardoust SH, Hesari J, Soltanzadeh M, Peressini D (2020) Properties of dried apricots pretreated by ultrasound-assisted osmotic dehydration and application of active coatings. Food Technol Biotechnol 58(3). https://doi.org/10.17113/ftb.58.03.20.6471
Alirezalu K, Hesari J, Nemati Z, Munekata PES, Barba FJ, Lorenzo JM (2019) Combined effect of natural antioxidants and antimicrobial compounds during refrigerated storage of nitrite-free frankfurter-type sausage. Food Res Int. https://doi.org/10.1016/j.foodres.2018.11.048
Manafi Dizaj Yekan M, Manafi Dizaj Yekan M, Hedari J, Azadmard Damirchii S, Ravash N (2021) Effect of chitosan on the physico-chemical, microbiological and sensory properties of doogh. Food Sci Technol 18(112):169–178. https://fsct.modares.ac.ir/article-7-28096-en.html
Nottagh S, Hesari J, Peighambardoust SH, Rezaei-Mokarram R, Jafarizadeh-Malmiri H (2020) Effectiveness of edible coating based on chitosan and Natamycin on biological, physico-chemical and organoleptic attributes of Iranian ultra-filtrated cheese. Biologia 75(4). https://doi.org/10.2478/s11756-019-00378-w
Coutinho NM, Silveira MR, Rocha RS, Moraes J, Ferreira MVS, Pimentel TC, Freitas MQ, Silva MC, Raices RSL, Ranadheera CS, Borges FO, Mathias SP, Fernandes FAN, Rodrigues S, Cruz AG (2018) Cold plasma processing of milk and dairy products. Trends Food Sci Technol 74. https://doi.org/10.1016/j.tifs.2018.02.008
Dobeic M (2018) The potential of cold plasma applications in food safety. Meso 20(3). https://doi.org/10.31727/m.20.3.4
Misra NN, Roopesh MS (2019) Cold plasma for sustainable food production and processing. Green Food Processing Techniques. https://doi.org/10.1016/b978-0-12-815353-6.00016-1
Feizollahi E, Arshad M, Yadav B, Ullah A, Roopesh MS (2020) Degradation of deoxynivalenol by atmospheric-pressure cold plasma and sequential treatments with heat and UV light. Food Eng Rev. https://doi.org/10.1007/s12393-020-09241-0
Feizollahi E, Iqdiam B, Vasanthan T, Thilakarathna MS, Roopesh MS (2020) Effects of atmospheric-pressure cold plasma treatment on deoxynivalenol degradation, quality parameters, and germination of barley grains. Appl Sci (Switzerland). https://doi.org/10.3390/app10103530
Feizollahi E, Misra NN, Roopesh MS (2020) Factors influencing the antimicrobial efficacy of dielectric barrier discharge (DBD) atmospheric cold plasma (ACP) in food processing applications. Crit Rev Food Sci Nutr. https://doi.org/10.1080/10408398.2020.1743967
Sharma S, Singh RK (2020) Cold plasma treatment of dairy proteins in relation to functionality enhancement. Trends Food Sci Technol. https://doi.org/10.1016/j.tifs.2020.05.013
Yadav B, Spinelli AC, Govindan BN, Tsui YY, McMullen LM, Roopesh MS (2019) Cold plasma treatment of ready-to-eat ham: influence of process conditions and storage on inactivation of Listeria innocua. Food Res Int 123. https://doi.org/10.1016/j.foodres.2019.04.065
Yadav B, Spinelli AC, Misra NN, Tsui YY, McMullen LM, Roopesh MS (2020) Effect of in-package atmospheric cold plasma discharge on microbial safety and quality of ready-to-eat ham in modified atmospheric packaging during storage. J Food Sci 85(4). https://doi.org/10.1111/1750-3841.15072
Cheng JH, Lv X, Pan Y, Sun DW (2020) Foodborne bacterial stress responses to exogenous reactive oxygen species (ROS) induced by cold plasma treatments. Trends Food Sci Technol 103. https://doi.org/10.1016/j.tifs.2020.07.022
Liao X, Liu D, Xiang Q, Ahn J, Chen S, Ye X, Ding T (2017) Inactivation mechanisms of non-thermal plasma on microbes: a review. Food Control 75. https://doi.org/10.1016/j.foodcont.2016.12.021
Ucar Y, Ceylan Z, Durmus M, Tomar O, Cetinkaya T (2021) Application of cold plasma technology in the food industry and its combination with other emerging technologies. Trends Food Sci Technol. https://doi.org/10.1016/j.tifs.2021.06.004
Durek J, Fröhling A, Bußler S, Hase A, Ehlbeck J, Schlüter OK (2022) Pilot-scale generation of plasma processed air and its influence on microbial count, microbial diversity, and selected quality parameters of dried herbs. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2021.102890
Ziuzina D, Misra NN, Han L, Cullen PJ, Moiseev T, Mosnier JP, Keener K, Gaston E, Vilaró I, Bourke P (2020) Investigation of a large gap cold plasma reactor for continuous in-package decontamination of fresh strawberries and spinach. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2019.102229
Andrasch M, Stachowiak J, Schlüter O, Schnabel U, Ehlbeck J (2017) Scale-up to pilot plant dimensions of plasma processed water generation for fresh-cut lettuce treatment. Food Packag Shelf Life. https://doi.org/10.1016/j.fpsl.2017.08.007
Ziuzina D, Misra NN, Cullen PJ, Keener K, Mosnier JP, Vilaró I, Gaston E, Bourke P (2016) Demonstrating the potential of industrial scale in-package atmospheric cold plasma for decontamination of cherry tomatoes. Plasma Med. https://doi.org/10.1615/PlasmaMed.2017019498
Lee SY, In J, Chung MS, Min SC (2021) Microbial decontamination of particulate food using a pilot-scale atmospheric plasma jet treatment system. J Food Eng. https://doi.org/10.1016/j.jfoodeng.2020.110436
Charoux CMG, Free L, Hinds LM, Vijayaraghavan RK, Daniels S, O’Donnell CP, Tiwari BK (2020) Effect of non-thermal plasma technology on microbial inactivation and total phenolic content of a model liquid food system and black pepper grains. LWT. https://doi.org/10.1016/j.lwt.2019.108716
Misra NN, Jo C (2017) Applications of cold plasma technology for microbiological safety in meat industry. Trends Food Sci Technol. https://doi.org/10.1016/j.tifs.2017.04.005
Arjunan KP, Sharma VK, Ptasinska S (2015) Effects of atmospheric pressure plasmas on isolated and cellular DNA—a review. Int J Mol Sci. https://doi.org/10.3390/ijms16022971
Mai-Prochnow A, Clauson M, Hong J, Murphy AB (2016) Gram positive and Gram negative bacteria differ in their sensitivity to cold plasma. Sci Rep. https://doi.org/10.1038/srep38610
Huang M, Zhuang H, Zhao J, Wang J, Yan W, Zhang J (2020) Differences in cellular damages induced by dielectric barrier discharge plasma between Salmonella Typhimurium and Staphylococcus aureus. Bioelectrochemistry. https://doi.org/10.1016/j.bioelechem.2019.107445
Putker F, Bos MP, Tommassen J (2015) Transport of lipopolysaccharide to the Gram-negative bacterial cell surface. FEMS Microbiol Rev. https://doi.org/10.1093/femsre/fuv026
Yusupov M, Neyts EC, Verlackt CC, Khalilov U, Van Duin ACT, Bogaerts A (2015) Inactivation of the endotoxic biomolecule lipid a by oxygen plasma species: a reactive molecular dynamics study. Plasma Process Polym. https://doi.org/10.1002/ppap.201400064
Kartaschew K, Baldus S, Mischo M, Bründermann E, Awakowicz P, Havenith M (2016) Cold atmospheric-pressure plasma and bacteria: understanding the mode of action using vibrational microspectroscopy. J Phys D Appl Phys. https://doi.org/10.1088/0022-3727/49/37/374003
Misra NN, Tiwari BK, Raghavarao KSMS, Cullen PJ (2011) Nonthermal plasma inactivation of food-borne pathogens. Food Eng Rev. https://doi.org/10.1007/s12393-011-9041-9
Kiwi J, Nadtochenko V (2005) Evidence for the mechanism of photocatalytic degradation of the bacterial wall membrane at the TiO2 interface by ATR-FTIR and laser kinetic spectroscopy. Langmuir. https://doi.org/10.1021/la0469831
Koebnik R, Locher KP, Van Gelder P (2000) Structure and function of bacterial outer membrane proteins: barrels in a nutshell. Mol Microbiol. https://doi.org/10.1046/j.1365-2958.2000.01983.x
Laroussi M (2005) Low temperature plasma-based sterilization: overview and state-of-the-art. Plasma Process Polym. https://doi.org/10.1002/ppap.200400078
Katsigiannis AS, Bayliss DL, Walsh JL (2021) Cold plasma decontamination of stainless steel food processing surfaces assessed using an industrial disinfection protocol. Food Control. https://doi.org/10.1016/j.foodcont.2020.107543
Bai Y, Idris Muhammad A, Hu Y, Koseki S, Liao X, Chen S, Ye X, Liu D, Ding T (2020) Inactivation kinetics of Bacillus cereus spores by plasma activated water (PAW). Food Res Int. https://doi.org/10.1016/j.foodres.2020.109041
Bang IH, Kim YE, Lee SY, Min SC (2020) Microbial decontamination of black peppercorns by simultaneous treatment with cold plasma and ultraviolet C. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2020.102392
Beyrer M, Pina-Perez MC, Martinet D, Andlauer W (2020) Cold plasma processing of powdered Spirulina algae for spore inactivation and preservation of bioactive compounds. Food Control. https://doi.org/10.1016/j.foodcont.2020.107378
Deng X, Shi J, Kong MG (2006) Physical mechanisms of inactivation of Bacillus subtilis spores using cold atmospheric plasmas. IEEE Trans Plasma Sci. https://doi.org/10.1109/TPS.2006.877739
Pina-Perez MC, Martinet D, Palacios-Gorba C, Ellert C, Beyrer M (2020) Low-energy short-term cold atmospheric plasma: controlling the inactivation efficacy of bacterial spores in powders. Food Res Int. https://doi.org/10.1016/j.foodres.2019.108921
Tanino T, Matsui M, Uehara K, Ohshima T (2020) Inactivation of Bacillus subtilis spores on the surface of small spheres using low-pressure dielectric barrier discharge. Food Control. https://doi.org/10.1016/j.foodcont.2019.106890
Tseng S, Abramzon N, Jackson JO, Lin WJ (2012) Gas discharge plasmas are effective in inactivating Bacillus and Clostridium spores. Appl Microbiol Biotechnol. https://doi.org/10.1007/s00253-011-3661-0
Laroque DA, Seó ST, Valencia GA, Laurindo JB, Carciofi BAM (2022) Cold plasma in food processing: design, mechanisms, and application. J Food Eng. https://doi.org/10.1016/j.jfoodeng.2021.110748
Gan Z, Feng X, Hou Y, Sun A, Wang R (2021) Cold plasma jet with dielectric barrier configuration: investigating its effect on the cell membrane of E. coli and S. cerevisiae and its impact on the quality of chokeberry juice. LWT. https://doi.org/10.1016/j.lwt.2020.110223
Wang X, Wang S, Yan Y, Wang W, Zhang L, Zong W (2020) The degradation of Alternaria mycotoxins by dielectric barrier discharge cold plasma. Food Control. https://doi.org/10.1016/j.foodcont.2020.107333
Wang Y, Wang Z, Zhu X, Yuan Y, Gao Z, Yue T (2020) Application of electrical discharge plasma on the inactivation of Zygosaccharomyces rouxii in apple juice. LWT. https://doi.org/10.1016/j.lwt.2019.108974
Cao Y, Qu G, Li T, Jiang N, Wang T (2018) Review on reactive species in water treatment using electrical discharge plasma: formation, measurement, mechanisms and mass transfer. Plasma Sci Technol. https://doi.org/10.1088/2058-6272/aacff4
Hoppanová L, Dylíková J, Kováčik D, Medvecká V, Ďurina P, Kryštofová S, Zahoranová A, Kaliňáková B (2020) The effect of cold atmospheric pressure plasma on Aspergillus ochraceus and ochratoxin a production Antonie van Leeuwenhoek. Int J Gen Mol Microbiol. https://doi.org/10.1007/s10482-020-01457-8
Ott LC, Appleton HJ, Shi H, Keener K, Mellata M (2020) High voltage atmospheric cold plasma treatment inactivates Aspergillus flavus spores and deoxynivalenol toxin. Food Microbiol. https://doi.org/10.1016/j.fm.2020.103669
Nosanchuk JD, Casadevall A (2003) The contribution of melanin to microbial pathogenesis. Cell Microbiol. https://doi.org/10.1046/j.1462-5814.2003.00268.x
Paulussen C, Hallsworth JE, Álvarez-Pérez S, Nierman WC, Hamill PG, Blain D, Rediers H, Lievens B (2017) Ecology of aspergillosis: insights into the pathogenic potency of Aspergillus fumigatus and some other Aspergillus species. Microb Biotechnol. https://doi.org/10.1111/1751-7915.12367
Zhu Y, Li C, Cui H, Lin L (2020) Feasibility of cold plasma for the control of biofilms in food industry. Trends Food Sci Technol. https://doi.org/10.1016/j.tifs.2020.03.001
Hall-Stoodley L, Costerton JW, Stoodley P (2004) Bacterial biofilms: from the natural environment to infectious diseases. Nat Rev Microbiol. https://doi.org/10.1038/nrmicro821
Mai-Prochnow A (2020) Chapter 4 - Cold plasma to control biofilms on food and in the food-processing environment (D. B. T.-A. in C. P. A. for F. S. and P. Bermudez-Aguirre (ed.); pp. 109–143). Academic Press. https://doi.org/10.1016/B978-0-12-814921-8.00004-9
Stewart PS, Costerton JW (2001) Antibiotic resistance of bacteria in biofilms. Lancet. https://doi.org/10.1016/S0140-6736(01)05321-1
Ziuzina D, Patil S, Cullen PJ, Boehm D, Bourke P (2014) Dielectric barrier discharge atmospheric cold plasma for inactivation of Pseudomonas aeruginosa biofilms. Plasma Med. https://doi.org/10.1615/PlasmaMed.2014011996
Brun P, Bernabè G, Marchiori C, Scarpa M, Zuin M, Cavazzana R, Zaniol B, Martines E (2018) Antibacterial efficacy and mechanisms of action of low power atmospheric pressure cold plasma: membrane permeability, biofilm penetration and antimicrobial sensitization. J Appl Microbiol. https://doi.org/10.1111/jam.13780
Xiong Z, Du T, Lu X, Cao Y, Pan Y (2011) How deep can plasma penetrate into a biofilm?. Appl Phys Lett. https://doi.org/10.1063/1.3597622
Joaquin JC, Kwan C, Abramzon N, Vandervoort K, Brelles-Mariño G (2009) Is gas-discharge plasma a new solution to the old problem of biofilm inactivation? Microbiology. https://doi.org/10.1099/mic.0.021501-0
Vu B, Chen M, Crawford RJ, Ivanova EP (2009) Bacterial extracellular polysaccharides involved in biofilm formation. Molecules. https://doi.org/10.3390/molecules14072535
Stopforth JD, Samelis J, Sofos JN, Kendall PA, Smith GC (2002) Biofilm formation by acid-adapted and nonadapted Listeria monocytogenes in fresh beef decontamination washings and its subsequent inactivation with sanitizers. J Food Prot. https://doi.org/10.4315/0362-028X-65.11.1717
Vleugels M, Shama G, Deng XT, Greenacre E, Brocklehurst T, Kong MG (2005) Atmospheric plasma inactivation of biofilm-forming bacteria for food safety control. IEEE Trans Plasma Sci. https://doi.org/10.1109/TPS.2005.844524
Govaert M, Smet C, Baka M, Ećimović B, Walsh JL, Van Impe J (2018) Resistance of L. monocytogenes and S. Typhimurium towards cold atmospheric plasma as function of biofilm age. Appl Sci (Switzerland) 8(12). https://doi.org/10.3390/APP8122702
Gilmore BF, Flynn PB, O’Brien S, Hickok N, Freeman T, Bourke P (2018) Cold plasmas for biofilm control: opportunities and challenges. Trends Biotechnol. https://doi.org/10.1016/j.tibtech.2018.03.007
Flynn PB, Graham WG, Gilmore BF (2019) Acinetobacter baumannii biofilm biomass mediates tolerance to cold plasma. Lett Appl Microbiol. https://doi.org/10.1111/lam.13122
Abramzon N, Joaquin JC, Bray J, Brelles-Mariño G (2006) Biofilm destruction by RF high-pressure cold plasma jet. IEEE Trans Plasma Sci. https://doi.org/10.1109/TPS.2006.877515
Lin L, Liao X, Li C, Abdel-Samie MA, Cui H (2020) Inhibitory effect of cold nitrogen plasma on Salmonella Typhimurium biofilm and its application on poultry egg preservation. LWT. https://doi.org/10.1016/j.lwt.2020.109340
Bhardwaj A, Vinothkumar K, Rajpara N (2013) Bacterial quorum sensing inhibitors: attractive alternatives for control of infectious pathogens showing multiple drug resistance. Recent Pat Anti-infect Drug Discov 8(1)68–83. https://doi.org/10.2174/1574891x11308010012
Ziuzina D, Boehm D, Patil S, Cullen PJ, Bourke P (2015) Cold plasma inactivation of bacterial biofilms and reduction of quorum sensing regulated virulence factors. PLoS ONE. https://doi.org/10.1371/journal.pone.0138209
Gaballa A, Helmann JD (2002) A peroxide-induced zinc uptake system plays an important role in protection against oxidative stress in Bacillus subtilis. Mol Microbiol. https://doi.org/10.1046/j.1365-2958.2002.03068.x
Gupta TT, Ayan H (2019) Application of non-thermal plasma on biofilm: a review. Appl Sci (Switzerland) 9(17). https://doi.org/10.3390/app9173548
Mariño GB (2012) Challenges in biofilm inactivation: the use of cold plasma as a new approach. J Bioprocess Biotech. https://doi.org/10.4172/2155-9821.1000e107
Farber R, Dabush-Busheri I, Chaniel G, Rozenfeld S, Bormashenko E, Multanen V, Cahan R (2019) Biofilm grown on wood waste pretreated with cold low-pressure nitrogen plasma: utilization for toluene remediation. Int Biodeterior Biodegradation. https://doi.org/10.1016/j.ibiod.2019.03.003
Denes AR, Somers EB, Wong ACL, Denes F (2001) 12-crown-4-ether and tri(ethylene glycol) dimethyl-ether plasma-coated stainless steel surfaces and their ability to reduce bacterial biofilm deposition. J Appl Polym Sci. https://doi.org/10.1002/app.1799
Ma Y, Chen M, Jones JE, Ritts AC, Yu Q, Sun H (2012) Inhibition of Staphylococcus epidermidis biofilm by trimethylsilane plasma coating. Antimicrob Agents Chemother. https://doi.org/10.1128/AAC.01739-12
Mercier-Bonin M, Saulou C, Lebleu N, Schmitz P, Allion A, Zanna S, Marcus P, Raynaud P, Despax B (2011) Plasma-surface engineering for biofilm prevention: evaluation of anti-adhesive and antimicrobial properties of asilvernanocomposite thin film. Biofilms: Formation, Development and Properties.
Muro-Fraguas I, Sainz-García A, Fernández Gómez P, López M, Múgica-Vidal R, Sainz-García E, Toledano P, Sáenz Y, López M, González-Raurich M, Prieto M, Alvarez-Ordóñez A, González-Marcos A, Alba-Elías F (2020) Atmospheric pressure cold plasma anti-biofilm coatings for 3D printed food tools. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2020.102404
Stewart-Wade SM (2011) Plant pathogens in recycled irrigation water in commercial plant nurseries and greenhouses: their detection and management. Irrig Sci. https://doi.org/10.1007/s00271-011-0285-1
Zhang CM, Xu LM, Xu PC, Wang XC (2016) Elimination of viruses from domestic wastewater: requirements and technologies. World J Microbiol Biotechnol. https://doi.org/10.1007/s11274-016-2018-3
M Moisan JP Moreau 1993 Low-temperature sterilization using gas plasmas: a review of the experiments and an analysis of the inactivation mechanisms Med Device Technol
Min SC, Roh SH, Niemira BA, Sites JE, Boyd G, Lacombe A (2016) Dielectric barrier discharge atmospheric cold plasma inhibits Escherichia coli O157:H7, Salmonella, Listeria monocytogenes, and Tulane virus in Romaine lettuce. Int J Food Microbiol. https://doi.org/10.1016/j.ijfoodmicro.2016.08.025
Filipić A, Gutierrez-Aguirre I, Primc G, Mozetič M, Dobnik D (2020) Cold plasma, a new hope in the field of virus inactivation. Trends Biotechnol. https://doi.org/10.1016/j.tibtech.2020.04.003
Hanbal SE, Takashima K, Miyashita S, Ando S, Ito K, Elsharkawy MM, Kaneko T, Takahashi H (2018) Atmospheric-pressure plasma irradiation can disrupt tobacco mosaic virus particles and RNAs to inactivate their infectivity. Adv Virol. https://doi.org/10.1007/s00705-018-3909-4
Aboubakr HA, Gangal U, Youssef MM, Goyal SM, Bruggeman PJ (2016) Inactivation of virus in solution by cold atmospheric pressure plasma: identification of chemical inactivation pathways. J Phys D Appl Phys. https://doi.org/10.1088/0022-3727/49/20/204001
Amiran MR, Akhavan Sepahi A, Zabiollahi R, Ghomi H, Momen SB, Aghasadeghi MR (2016) In vitro assessment of antiviral activity of cold atmospheric pressure plasma jet against the human immunodeficiency virus (HIV). Journal of Medical Microbiology and Infectious Diseases 4(3):62–67
Sakudo A, Shimizu N, Imanishi Y, Ikuta K (2013) N2 gas plasma inactivates influenza virus by inducing changes in viral surface morphology, protein, and genomic RNA. Biomed Res Int. https://doi.org/10.1155/2013/694269
Wu Y, Liang Y, Wei K, Li W, Yao M, Zhang J, Grinshpun SA (2015) MS2 virus inactivation by atmospheric-pressure cold plasma using different gas carriers and power levels. Appl Environ Microbiol. https://doi.org/10.1128/AEM.03322-14
Gracanin M, Hawkins CL, Pattison DI, Davies MJ (2009) Singlet-oxygen-mediated amino acid and protein oxidation: formation of tryptophan peroxides and decomposition products. Free Radical Biol Med. https://doi.org/10.1016/j.freeradbiomed.2009.04.015
Li G, Ruobing X, Lu G, Zhichao L, Yiming Z, Dingxin L, Lei Z, Hailan C, Michael G (2018) Mechanism of virus inactivation by cold atmospheric-pressure plasma and plasma-activated water. Appl Environ Microbiol 84(17):1–10
Aboubakr HA, Mor SK, Higgins LA, Armien A, Youssef MM, Bruggeman PJ, Goyal SM (2018) Cold argon-oxygen plasma species oxidize and disintegrate capsid protein of feline calicivirus. PLoS ONE. https://doi.org/10.1371/journal.pone.0194618
Filipić A, Primc G, Zaplotnik R, Mehle N, Gutierrez-Aguirre I, Ravnikar M, Mozetič M, Žel J, Dobnik D (2019) Cold atmospheric plasma as a novel method for inactivation of potato virus Y in water samples. Food and Environmental Virology. https://doi.org/10.1007/s12560-019-09388-y
Puligundla P, Lee T, Mok C (2020) Effect of corona discharge plasma jet treatment on the degradation of aflatoxin B1 on glass slides and in spiked food commodities. LWT. https://doi.org/10.1016/j.lwt.2019.108333
Feizollahi E, Roopesh MS (2021) Mechanisms of deoxynivalenol (DON) degradation during different treatments: a review. Crit Rev Food Sci Nutr. https://doi.org/10.1080/10408398.2021.1895056
Gavahian M, Cullen PJ (2020) Cold plasma as an emerging technique for mycotoxin-free food: efficacy, mechanisms, and trends. Food Rev Intl. https://doi.org/10.1080/87559129.2019.1630638
Hojnik N, Cvelbar U, Tavčar-Kalcher G, Walsh JL, Križaj I (2017) Mycotoxin decontamination of food: cold atmospheric pressure plasma versus “classic” decontamination. Toxins 9(5). https://doi.org/10.3390/toxins9050151
Gavahian M, Pallares N, Al Khawli F, Ferrer E, Barba FJ (2020) Recent advances in the application of innovative food processing technologies for mycotoxins and pesticide reduction in foods. Trends Food Sci Technol. https://doi.org/10.1016/j.tifs.2020.09.018
Misra NN, Yadav B, Roopesh MS, Jo C (2019) Cold plasma for effective fungal and mycotoxin control in foods: mechanisms, inactivation effects, and applications. Compr Rev Food Sci Food Saf. https://doi.org/10.1111/1541-4337.12398
Shi H, Ileleji K, Stroshine RL, Keener K, Jensen JL (2017) Reduction of aflatoxin in corn by high voltage atmospheric cold plasma. Food Bioprocess Technol. https://doi.org/10.1007/s11947-017-1873-8
Wu Y, Cheng JH, Sun DW (2021) Blocking and degradation of aflatoxins by cold plasma treatments: applications and mechanisms. Trends Food Sci Technol. https://doi.org/10.1016/j.tifs.2021.01.053
Muthukumar M, Sargunamani D, Selvakumar N (2005) Statistical analysis of the effect of aromatic, azo and sulphonic acid groups on decolouration of acid dye effluents using advanced oxidation processes. Dyes Pigm. https://doi.org/10.1016/j.dyepig.2004.07.012
Shi H, Cooper B, Stroshine RL, Ileleji KE, Keener KM (2017) Structures of degradation products and degradation pathways of aflatoxin B1 by high-voltage atmospheric cold plasma (HVACP) treatment. J Agric Food Chem. https://doi.org/10.1021/acs.jafc.7b01604
Iqdiam BM, Feizollahi E, Arif MF, Jeganathan B, Vasanthan T, Thilakarathna MS, Roopesh MS (2021) Reduction of T-2 and HT-2 mycotoxins by atmospheric cold plasma and its impact on quality changes and germination of wheat grains. J Food Sci. https://doi.org/10.1111/1750-3841.15658
Sen Y, Onal-Ulusoy B, Mutlu M (2019) Detoxification of hazelnuts by different cold plasmas and gamma irradiation treatments. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2019.05.002
Siciliano I, Spadaro D, Prelle A, Vallauri D, Cavallero MC, Garibaldi A, Gullino ML (2016) Use of cold atmospheric plasma to detoxify hazelnuts from aflatoxins. Toxins. https://doi.org/10.3390/toxins8050125
Pham TH, Bui HM, Bui TX (2020) Advanced oxidation processes for the removal of pesticides. Curr Dev Biotechnol Bioeng. https://doi.org/10.1016/b978-0-12-819594-9.00013-9
Zhou R, Zhou R, Yu F, Xi D, Wang P, Li J, Wang X, Zhang X, Bazaka K, Ostrikov K, (Ken), (2018) Removal of organophosphorus pesticide residues from Lycium barbarum by gas phase surface discharge plasma. Chem Eng J. https://doi.org/10.1016/j.cej.2018.02.107
Sarangapani C, Scally L, Gulan M, Cullen PJ (2020) Dissipation of pesticide residues on grapes and strawberries using plasma-activated water. Food Bioprocess Technol. https://doi.org/10.1007/s11947-020-02515-9
Cong L, Huang M, Zhang J, Yan W (2020) Effect of dielectric barrier discharge plasma on the degradation of malathion and chlorpyrifos on lettuce. J Sci Food Agric. https://doi.org/10.1002/jsfa.10651
Gavahian M, Khaneghah AM (2020) Cold plasma as a tool for the elimination of food contaminants: recent advances and future trends. Crit Rev Food Sci Nutr. https://doi.org/10.1080/10408398.2019.1584600
Moutiq R, Pankaj SK, Wan Z, Mendonca A, Keener K, Misra NN (2020) Atmospheric pressure cold plasma as a potential technology to degrade carbamate residues in water. Plasma Chem Plasma Process. https://doi.org/10.1007/s11090-020-10093-z
Gavahian M, Meng-Jen T, Khaneghah AM (2020) Emerging techniques in food science: the resistance of chlorpyrifos pesticide pollution against arc and dielectric barrier discharge plasma. Quality Assurance and Safety of Crops and Foods 12(1). https://doi.org/10.15586/qas.v12iSP1.807
Bai Y, Chen J, Yang Y, Guo L, Zhang C (2010) Degradation of organophosphorus pesticide induced by oxygen plasma: Effects of operating parameters and reaction mechanisms. Chemosphere. https://doi.org/10.1016/j.chemosphere.2010.06.071
Lozano-Ojalvo D, Benedé S, Van Bilsen JBT (2021) Food allergy: etiology, allergens, and analytical strategies. Elsevier. https://doi.org/10.1016/B978-0-08-100596-5.22845-X
Morais S, Tortajada-Genaro LA, Maquieira Á, Gonzalez Martinez MÁ (2020) Biosensors for food allergy detection according to specific IgE levels in serum. Trends Anal Chem. https://doi.org/10.1016/j.trac.2020.115904
Zhang Q, Cheng Z, Zhang J, Nasiru MM, Wang Y, Fu L (2021) Atmospheric cold plasma treatment of soybean protein isolate: insights into the structural, physicochemical, and allergenic characteristics. J Food Sci. https://doi.org/10.1111/1750-3841.15556
Zhang S, Huang W, Feizollahi E, Roopesh MS, Chen L (2021) Improvement of pea protein gelation at reduced temperature by atmospheric cold plasma and the gelling mechanism study. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2020.102567
Chizoba Ekezie FG, Cheng JH, Sun DW (2018) Effects of nonthermal food processing technologies on food allergens: a review of recent research advances. Trends Food Sci Technol 74:12–25. https://doi.org/10.1016/j.tifs.2018.01.007
Ojha S, Fröhling A, Durek J, Ehlbeck J, Tiwari BK, Schlüter OK, Bußler S (2021) Principles and application of cold plasma in food processing. Innov Food Process Technol. https://doi.org/10.1016/b978-0-08-100596-5.23033-3
Sarangapani C, Patange A, Bourke P, Keener K, Cullen PJ (2018) Recent advances in the application of cold plasma technology in foods. Annu Rev Food Sci Technol. https://doi.org/10.1146/annurev-food-030117-012517
Meinlschmidt P, Ueberham E, Lehmann J, Reineke K, Schlüter O, Schweiggert-Weisz U, Eisner P (2016) The effects of pulsed ultraviolet light, cold atmospheric pressure plasma, and gamma-irradiation on the immunoreactivity of soy protein isolate. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2016.06.007
Ekezie FGC, Sun DW, Cheng JH (2019) Altering the IgE binding capacity of king prawn (Litopenaeus Vannamei) tropomyosin through conformational changes induced by cold argon-plasma jet. Food Chem. https://doi.org/10.1016/j.foodchem.2019.125143
Li J, Xiang Q, Liu X, Ding T, Zhang X, Zhai Y, Bai Y (2017) Inactivation of soybean trypsin inhibitor by dielectric-barrier discharge (DBD) plasma. Food Chem. https://doi.org/10.1016/j.foodchem.2017.03.167
Wu Y, Liang Y, Wei K, Li W, Yao M, Zhang J (2014) Rapid allergen inactivation using atmospheric pressure cold plasma. Environ Sci Technol. https://doi.org/10.1021/es5003988
Wei Ng S, Lu P, Rulikowska A, Boehm D, O’Neill G, Bourke P (2020) The effect of atmospheric cold plasma treatment on the antigenic properties of bovine milk casein and whey proteins. Food Chem. https://doi.org/10.1016/j.foodchem.2020.128283
Meinlschmidt P, Brode V, Sevenich R, Ueberham E, Schweiggert-Weisz U, Lehmann J, Rauh C, Knorr D, Eisner P (2017) High pressure processing assisted enzymatic hydrolysis – an innovative approach for the reduction of soy immunoreactivity. Innov Food Sci Emerg Technol. https://doi.org/10.1016/J.IFSET.2016.06.022
Cabanillas B, Cuadrado C, Rodriguez J, Hart J, Burbano C, Crespo JF, Novak N (2015) Potential changes in the allergenicity of three forms of peanut after thermal processing. Food Chem. https://doi.org/10.1016/j.foodchem.2015.03.023
Izquierdo FJ, Peñas E, Baeza ML, Gomez R (2008) Effects of combined microwave and enzymatic treatments on the hydrolysis and immunoreactivity of dairy whey proteins. Int Dairy J. https://doi.org/10.1016/J.IDAIRYJ.2008.01.005
Li H, Yu J, Ahmedna M, Goktepe I (2013) Reduction of major peanut allergens Ara h 1 and Ara h 2, in roasted peanuts by ultrasound assisted enzymatic treatment. Food Chem. https://doi.org/10.1016/J.FOODCHEM.2013.03.049
Chen D, Peng P, Zhou N, Cheng Y, Min M, Ma Y, Mao Q, Chen P, Chen C, Ruan R (2019) Evaluation of Cronobacter sakazakii inactivation and physicochemical property changes of non-fat dry milk powder by cold atmospheric plasma. Food Chem. https://doi.org/10.1016/j.foodchem.2019.03.149
Choi MS, Jeon EB, Kim JY, Choi EH, Lim JS, Choi J, Park SY (2020) Impact of non-thermal dielectric barrier discharge plasma on Staphylococcus aureus and Bacillus cereus and quality of dried blackmouth angler (Lophiomus setigerus). J Food Eng. https://doi.org/10.1016/j.jfoodeng.2020.109952
Costello KM, Smet C, Gutierrez-Merino J, Bussemaker M, Van Impe JF, Velliou EG (2021) The impact of food model system structure on the inactivation of Listeria innocua by cold atmospheric plasma and nisin combined treatments. Int J Food Microbiol. https://doi.org/10.1016/j.ijfoodmicro.2020.108948
Giannoglou M, Stergiou P, Dimitrakellis P, Gogolides E, Stoforos NG, Katsaros G (2020) Effect of cold atmospheric plasma processing on quality and shelf-life of ready-to-eat rocket leafy salad. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2020.102502
Wang J, Han R, Liao X, Ding T (2021) Application of plasma-activated water (PAW) for mitigating methicillin-resistant Staphylococcus aureus (MRSA) on cooked chicken surface. LWT. https://doi.org/10.1016/j.lwt.2020.110465
Yadav B, Roopesh MS (2020) In-package atmospheric cold plasma inactivation of Salmonella in freeze-dried pet foods: effect of inoculum population, water activity, and storage. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2020.102543
Hojnik N, Modic M, Žigon D, Kovač J, Jurov A, Dickenson A, Walsh JL, Cvelbar U (2020) Cold atmospheric pressure plasma-assisted removal of aflatoxin B1 from contaminated corn kernels. Plasma Processes Polym. https://doi.org/10.1002/ppap.202000163
Feng X, Ma X, Liu H, Xie J, He C, Fan R (2019) Argon plasma effects on maize: pesticide degradation and quality changes. J Sci Food Agric. https://doi.org/10.1002/jsfa.9810
Ranjitha Gracy TK, Gupta V, Mahendran R (2019) Influence of low-pressure nonthermal dielectric barrier discharge plasma on chlorpyrifos reduction in tomatoes. J Food Process Eng. https://doi.org/10.1111/jfpe.13242
Sarangapani C, Misra NN, Milosavljevic V, Bourke P, O’Regan F, Cullen PJ (2016) Pesticide degradation in water using atmospheric air cold plasma. J Water Process Eng. https://doi.org/10.1016/j.jwpe.2016.01.003
Venkataratnam H, Sarangapani C, Cahill O, Ryan CB (2019) Effect of cold plasma treatment on the antigenicity of peanut allergen Ara h 1. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2019.02.001
Sainz-García A, González-Marcos A, Múgica-Vidal R, Muro-Fraguas I, Escribano-Viana R, González-Arenzana L, López-Alfaro I, Alba-Elías F, Sainz-García E (2021) Application of atmospheric pressure cold plasma to sanitize oak wine barrels. LWT. https://doi.org/10.1016/j.lwt.2020.110509
Farr SB, Kogoma T (1991) Oxidative stress responses in Escherichia coli and Salmonella typhimurium. Microbiol Rev. https://doi.org/10.1128/mmbr.55.4.561-585.1991
Govaert M, Smet C, Vergauwen L, Ećimović B, Walsh JL, Baka M, Van Impe J (2019) Influence of plasma characteristics on the efficacy of Cold Atmospheric Plasma (CAP) for inactivation of Listeria monocytogenes and Salmonella Typhimurium biofilms. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2019.01.013
Duan J, Lu X, He G (2017) On the penetration depth of reactive oxygen and nitrogen species generated by a plasma jet through real biological tissue. Phys Plasmas. https://doi.org/10.1063/1.4990554
Kim J, Rodriguez ME, Guo M, Kenney ME, Oleinick NL, Anderson VE (2008) Oxidative modification of cytochrome c by singlet oxygen. Free Radical Biol Med. https://doi.org/10.1016/j.freeradbiomed.2007.12.031
Kiš M, Milošević S, Vulić A, Herceg Z, Vukušić T, Pleadin J (2020) Efficacy of low pressure DBD plasma in the reduction of T-2 and HT-2 toxin in oat flour. Food Chem. https://doi.org/10.1016/j.foodchem.2020.126372
Feizollahi E, Roopesh MS (2021) Degradation of zearalenone by atmospheric cold plasma: effect of selected process and product factors. Food Bioprocess Technol. https://doi.org/10.1007/s11947-021-02692-1
Attri P, Kumar N, Park JH, Yadav DK, Choi S, Uhm HS, Kim IT, Choi EH, Lee W (2015) Influence of reactive species on the modification of biomolecules generated from the soft plasma. Sci Rep. https://doi.org/10.1038/srep08221
Zhang H, Ma D, Qiu R, Tang Y, Du C (2017) Non-thermal plasma technology for organic contaminated soil remediation: a review. Chem Eng J. https://doi.org/10.1016/j.cej.2016.12.067
Laroussi M, Leipold F (2004) Evaluation of the roles of reactive species, heat, and UV radiation in the inactivation of bacterial cells by air plasmas at atmospheric pressure. Int J Mass Spectrom. https://doi.org/10.1016/j.ijms.2003.11.016
Patil S, Moiseev T, Misra NN, Cullen PJ, Mosnier JP, Keener KM, Bourke P (2014) Influence of high voltage atmospheric cold plasma process parameters and role of relative humidity on inactivation of Bacillus atrophaeus spores inside a sealed package. J Hosp Infect. https://doi.org/10.1016/j.jhin.2014.08.009
Mousavi SM, Imani S, Dorranian D, Larijani K, Shojaee M (2017) Effect of cold plasma on degradation of organophosphorus pesticides used on some agricultural products. Journal of Plant Protection Research. https://doi.org/10.1515/jppr-2017-0004
Surowsky B, Bußler S, Schlüter OK (2016) Cold plasma interactions with food constituents in liquid and solid food matrices. Cold Plasma in Food and Agriculture: Fundamentals and Applications. https://doi.org/10.1016/B978-0-12-801365-6.00007-X
Thirumdas R, Kothakota A, Annapure U, Siliveru K, Blundell R, Gatt R, Valdramidis VP (2018) Plasma activated water (PAW): Chemistry, physico-chemical properties, applications in food and agriculture. Trends Food Sci Technol. https://doi.org/10.1016/j.tifs.2018.05.007
Khan MSI, Lee NR, Ahn J, Kim JY, Kim JH, Kwon KH, Kim YJ (2020) Degradation of different pesticides in water by microplasma: the roles of individual radicals and degradation pathways. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-020-11127-x
Sawangrat C, Leksakul K, Bonyawan D, Anantana T, Jomjunyong S (2019) Decontamination of pesticide residues on tangerine fruit using non-thermal plasma technology. IOP Conference Series: Earth and Environmental Science. https://doi.org/10.1088/1755-1315/347/1/012048
Chizoba Ekezie FG, Sun DW, Cheng JH (2017) A review on recent advances in cold plasma technology for the food industry: current applications and future trends. Trends Food Sci Technol. https://doi.org/10.1016/j.tifs.2017.08.007
Liao X, Cullen PJ, Muhammad AI, Jiang Z, Ye X, Liu D, Ding T (2020) Cold plasma–based hurdle interventions: new strategies for improving food safety. Food Engineering Reviews. https://doi.org/10.1007/s12393-020-09222-3
Kayes MM, Critzer FJ, Kelly-Wintenberg K, Roth JR, Montie TC, Golden DA (2007) Inactivation of foodborne pathogens using a one atmosphere uniform glow discharge plasma. Foodborne Pathog Dis. https://doi.org/10.1089/fpd.2006.62
Yadav B, Roopesh MS (2022) Synergistically enhanced Salmonella Typhimurium reduction by sequential treatment of organic acids and atmospheric cold plasma and the mechanism study. Food Microbiol. https://doi.org/10.1016/J.FM.2021.103976
Adam AM, Jeganathan B, Vasanthan T, Roopesh MS (2021) Dipping fresh-cut apples in citric acid before plasma-integrated low-pressure cooling improves Salmonella and polyphenol oxidase inactivation. J Sci Food Agric. https://doi.org/10.1002/jsfa.11690
Patange A, Boehm D, Ziuzina D, Cullen PJ, Gilmore B, Bourke P (2019) High voltage atmospheric cold air plasma control of bacterial biofilms on fresh produce. Int J Food Microbiol. https://doi.org/10.1016/j.ijfoodmicro.2019.01.005
Cui H, Bai M, Yuan L, Surendhiran D, Lin L (2018) Sequential effect of phages and cold nitrogen plasma against Escherichia coli O157:H7 biofilms on different vegetables. Int J Food Microbiol. https://doi.org/10.1016/j.ijfoodmicro.2018.01.004
Tschang CYT, Thoma M (2019) Biofilm inactivation by synergistic treatment of atmospheric pressure plasma and chelating agents. Clin Plasma Med. https://doi.org/10.1016/j.cpme.2019.100091
Chaijan M, Chaijan S, Panya A, Nisoa M, Cheong LZ, Panpipat W (2022) Combined effects of prior plasma-activated water soaking and whey protein isolate-ginger extract coating on the cold storage stability of Asian sea bass (Lates calcarifer) steak. Food Control. https://doi.org/10.1016/J.FOODCONT.2021.108787
Shiekh KA, Zhou P, Benjakul S (2021) Combined effects of pulsed electric field, Chamuang leaf extract and cold plasma on quality and shelf-life of Litopenaeus vannamei. Food Biosci https://doi.org/10.1016/J.FBIO.2021.100975
Liao X, Li J, Suo Y, Ahn J, Liu D, Chen S, Hu Y, Ye X, Ding T (2018) Effect of preliminary stresses on the resistance of Escherichia coli and Staphylococcus aureus toward non-thermal plasma (NTP) challenge. Food Res Int. https://doi.org/10.1016/j.foodres.2017.11.010
Kim JY, Jeon EB, Song MG, Choi EH, Lim JS, Choi J, Park SY (2022) Inhibitory effects of atmospheric dielectric barrier discharge plasma against Bacillus cereus and Staphylococcus aureus on Gwamegi (semi-dried Cololabis saira). J Appl Microbiol. https://doi.org/10.1111/jam.15445
Kim YH, Lee C, Lee SJ, Yoon KS (2022) Reduction of E. coli O157: H7 and Bacillus cereus levels in red pepper powder using dielectric barrier discharge (DBD) plasma for enhanced quality. Innovative Food Sci Emerg Technol https://doi.org/10.1016/J.IFSET.2022.102916
Wang Y, Liu Y, Li M, Ma M, Sun Q (2022). Evaluation of the storage stability and quality properties of fresh noodles mixed with plasma-activated water. https://doi.org/10.3390/foods11010133
Molina-Hernandez JB, Laika J, Peralta-Ruiz Y, Palivala VK, Tappi S, Cappelli F, Ricci A, Neri L, Chaves-López C (2022) Influence of atmospheric cold plasma exposure on naturally present fungal spores and physicochemical characteristics of sundried tomatoes (Solanum lycopersicum L.). Foods. https://doi.org/10.3390/foods11020210
Machado-Moreira B, Tiwari BK, Richards KG, Abram F, Burgess CM (2021) Application of plasma activated water for decontamination of alfalfa and mung bean seeds. Food Microbiol. https://doi.org/10.1016/j.fm.2020.103708
Taheri S, Brodie GI, Gupta D, Jacob MV (2020) Afterglow of atmospheric non-thermal plasma for disinfection of lentil seeds from Botrytis Grey Mould. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2020.102488
Shirani K, Shahidi F, Mortazavi SA (2020) Investigation of decontamination effect of argon cold plasma on physicochemical and sensory properties of almond slices. Int J Food Microbiol. https://doi.org/10.1016/j.ijfoodmicro.2020.108892
Park H, Puligundla P, Mok C (2020) Cold plasma decontamination of brown rice grains: impact on biochemical and sensory qualities of their corresponding seedlings and aqueous tea infusions. LWT. https://doi.org/10.1016/j.lwt.2020.109508
Liu C, Chen C, Jiang A, Sun X, Guan Q, Hu W (2020) Effects of plasma-activated water on microbial growth and storage quality of fresh-cut apple. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2019.102256
Zhao N, Ge L, Huang Y, Wang Y, Wang Y, Lai H, Wang Y, Zhu Y, Zhang J (2020) Impact of cold plasma processing on quality parameters of packaged fermented vegetable (radish paocai) in comparison with pasteurization processing: insight into safety and storage stability of products. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2020.102300
Cui H, Li H, Abdel-Samie MA, Surendhiran D, Lin L (2021) Anti-Listeria monocytogenes biofilm mechanism of cold nitrogen plasma. Innov Food Sci Emerg Technol. https://doi.org/10.1016/j.ifset.2020.102571
Aboubakr HA, Sampedro Parra F, Collins J, Bruggeman P, Goyal SM (2020) Ìn situ inactivation of human norovirus GII.4 by cold plasma: ethidium monoazide (EMA)-coupled RT-qPCR underestimates virus reduction and fecal material suppresses inactivation. Food Microbiol. https://doi.org/10.1016/j.fm.2019.103307
Chen Z, Garcia G, Arumugaswami V, Wirz RE (2020) Cold atmospheric plasma for SARS-CoV-2 inactivation. Phys Fluids. https://doi.org/10.1063/5.0031332
Roh SH, Oh YJ, Lee SY, Kang JH, Min SC (2020) Inactivation of Escherichia coli O157:H7, Salmonella, Listeria monocytogenes, and Tulane virus in processed chicken breast via atmospheric in-package cold plasma treatment. LWT. https://doi.org/10.1016/j.lwt.2020.109429
Xia T, Kleinheksel A, Lee EM, Qiao Z, Wigginton KR, Clack HL (2019) Inactivation of airborne viruses using a packed bed non-thermal plasma reactor. J Phys D Appl Phys. https://doi.org/10.1088/1361-6463/ab1466
Park SY, Ha SD (2018) Assessment of cold oxygen plasma technology for the inactivation of major foodborne viruses on stainless steel. J Food Eng. https://doi.org/10.1016/j.jfoodeng.2017.11.041
Bae SC, Park SY, Choe W, Ha SD (2015) Inactivation of murine norovirus-1 and hepatitis A virus on fresh meats by atmospheric pressure plasma jets. Food Res Int. https://doi.org/10.1016/j.foodres.2015.06.039
Hojnik N, Modic M, Walsh JL, Žigon D, Javornik U, Plavec J, Žegura B, Filipič M, Cvelbar U (2021) Unravelling the pathways of air plasma induced aflatoxin B1 degradation and detoxification. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2020.123593
Wielogorska E, Ahmed Y, Meneely J, Graham WG, Elliott CT, Gilmore BF (2019) A holistic study to understand the detoxification of mycotoxins in maize and impact on its molecular integrity using cold atmospheric plasma treatment. Food Chem. https://doi.org/10.1016/j.foodchem.2019.125281
Ali M, Cheng JH, Sun DW (2021) Effect of plasma activated water and buffer solution on fungicide degradation from tomato (Solanum lycopersicum) fruit. Food Chem. https://doi.org/10.1016/j.foodchem.2021.129195
Alves Filho EG, Silva LMA, Oiram Filho F, Rodrigues S, Fernandes FAN, Gallão MI, Mattison CP, De Brito ES (2019) Cold plasma processing effect on cashew nuts composition and allergenicity. Food Res Int. https://doi.org/10.1016/j.foodres.2019.108621
Liu ZW, Liu LJ, Zhou YX, Tan YC, Cheng JH, Bekhit AED, Inam-Ur-Raheem M, Aadil RM (2021) Dielectric-barrier discharge (DBD) plasma treatment reduces IgG binding capacity of β-lactoglobulin by inducing structural changes. Food Chem. https://doi.org/10.1016/j.foodchem.2021.129821
Author information
Authors and Affiliations
Contributions
Negar Ravash had the idea for the article. Negar Ravash wrote the original draft and prepared figures. Negar Ravash, Ehsan Feizollahi, and Harleen Kaur Dhaliwal performed the literature search and data analysis. Negar Ravash, Javad Hesari, Ehsan Feizollahi, Harleen Kaur Dhaliwal, and M. S. Roopesh wrote, reviewed, and edited the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ravash, N., Hesari, J., Feizollahi, E. et al. Valorization of Cold Plasma Technologies for Eliminating Biological and Chemical Food Hazards. Food Eng Rev 16, 22–58 (2024). https://doi.org/10.1007/s12393-023-09348-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12393-023-09348-0