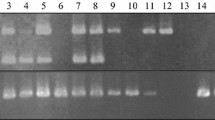
Abstract
Retrotransposon-based molecular markers are a powerful tool for mapping and diversity studies. The scarcity of retrotransposon long terminal repeat (LTR) sequences limits the application of retrotransposon-based molecular marker systems. Here, we isolated two novel complete Ty1-copia retrotransposons (CTcrm1 and CTcrm2) in apple using a genome walking strategy. The CTcrm retrotransposons are nearly 5 kb long, and they have all the features of Ty1-copia retrotransposons. The differences in gene organization and nucleotide sequence length between the CTcrm retrotransposons and other reported complete retrotransposons in apple showed that CTcrm1 and CTcrm2 are the first two distinct complete Ty1-copia retrotransposons in the apple genome. To investigate the potential utility of the two retrotransposons as molecular markers, primers complementary to the CTcrm LTRs were designed to develop sequence-specific amplification polymorphism markers for discriminating bud sports of Fuji apple. Multiple polymorphisms corresponding to CTcrm1 and CTcrm2 were detected and could easily be used to discriminate bud sports from their Fuji progenitor, as well as from each other.



Similar content being viewed by others
References
Acquadro A, Portis E, Moglia A, Lanteri S (2006) Retrotransposon-based S-SAP as a platform for the analysis of genetic variation and linkage in globe artichoke. Genome 49:1149–1159
Antonius-Klemola K, Kalendar R, Schulman AH (2006) TRIM retrotransposons occur in apple and are polymorphic between varieties but not sports. Theor Appl Genet 112:999–1008
Berenyi M, Gichuki ST, Schmidt J, Burg K (2002) Ty1-copia retrotransposon-based S-SAP (sequence-specific amplified polymorphism) for genetic analysis of sweetpotato. Theor Appl Genet 105:862–869
Boeke JD, Corces VG (1989) Transcription and reverse transcription of retrotransposons. Annu Rev Microbiol 43:403–434
Bowman E, Pathak V, Hu W (1996) Efficient initiation and strand transfer of polypurine tract-primed plus-strand DNA prevent strand transfer of internally initiated plus-strand DNAs. J Virol 70:1687–1694
Breto MP, Ruiz C, Pina JA, Asins MJ (2001) The diversification of Citrus clementina Hort. ex Tan., a vegetatively propagated crop species. Mol Phylogenet Evol 21:285–293
Casacuberta JM, Santiago N (2003) Plant LTR-retrotransposons and MITEs: control of transposition and impact on the evolution of plant genes and genomes. Gene 311:1–11
Cornman RS, Arnold ML (2009) Characterization and comparative analysis of sequence-specific amplified polymorphisms based on two subfamilies of IRRE retrotransposons in Iris missouriensis (Iridaceae). Genetica 135:25–38
Dice LR (1945) Measures of the amount of ecologic association between species. Ecology 26:297–302
Ellis THN, Poyser MR, Knox MR, Versinin AV, Ambrose MJ (1998) Polymorphism of insertion sites of Ty1-copia class retrotransposons and its use for linkage and diversity analysis in pea. Mol Gen Genet 260:9–19
Feschotte C, Jiang N, Wessler SR (2002) Plant transposable elements: where genetics meets genomics. Nat Rev Genet 3:329–341
Flavell AJ, Dunbar E, Anderson PSR, Hartley R, Kumar A (1992) Ty1-copia group retrotransposons are ubiquitous and heterogeneous in higher plants. Nucleic Acids Res 20:3639–3644
Flavell AJ, Knox MR, Pearce SR, Ellis THN (1998) Retrotransposon-based insertion polymorphisms (RBIP) for high throughput marker analysis. Plant J 16:643–650
Grandbastien MA (1992) Retroelements in higher plants. Trends Genet 8:103–108
Grandbastien MA, Spielmann A, Caboche M (1989) Tnt1, a mobile retroviral-like transposable element of tobacco isolated by plant cell genetics. Nature 337:376–380
Han YP, Kseni G, Schuyler SK (2007) Multiple-copy cluster-type organization and evolution of genes encoding O-methyltransferases in the apple. Genetics 176:2625–2635
Hirochika H, Hirochika R (1993) Ty1-copia group retrotransposons as ubiquitous components of plant genomes. Jpn J Genet 68:35–46
Hirochika H, Otsuki H, Yoshikawa M, Otsuki Y, Sugimoto K, Takeda S (1996) Autonomous transposition of the tobacco retrotransposon Tto1 in rice. The Plant Cell 8:725–734
Hoying SA, Rosenberger DA, Lamont G (2006) The apple industry in China. New York Fruit Quarterly 14:13–20
Hui EKW, Wang PC, Lo SJ (1998) Strategies for cloning unknown cellular flanking DNA sequences from foreign integrants. Cell Mol Life Sci 54:1403–1411
Jin YK, Bennetzen JL (1989) Structure and coding properties of Bsl, a maize retrovirus-like transposon (transposable element/retrotransposon/reverse transcription/translation frameshift). Proc Nati Acad Sci 86:6235–6239
Kalendar R, Grob T, Regina M, Suoniemi A, Schulman AH (1999) IRAP and REMAP: two new retrotransposon-based DNA fingerprinting techniques. Theor Appl Genet 98:704–711
Komatsu H (1998) Red Fuji in Japan—choosing the best strain for your business strategy. IDFTA, Compact Fruit Tree 31:No 2
Kumar A, Bennetzen JL (1999) Plant retrotransposons. Annu Rev Genet 33:479–532
Kumar A, Hirochika H (2001) Applications of retrotransposons as genetic tools in plant biology. Trends Plant Sci 6:127–134
Kunze R, Saedler H, Lonnig W (1997) Plant transposable elements. Adv Bot Res 27:331–470
Lou QF, Chen JF (2007) Ty1-copia retrotransposon-based SSAP marker development and its potential in the genetic study of cucurbits. Genome 50:802–810
Lowe TM, Eddy SR (1997) TRNAscan-SE: a program for improved detection of transfer RNA genes in genomic sequence. Nucleic Acids Res 25:955–964
Macas J, Neumann P (2007) Ogre elements—a distinct group of plant Ty3/gypsy-like retrotransposons. Gene 390:108–116
Mount SM, Rubin GM (1985) Complete nucleotide sequence of the Drosophila transposable element Copia: homology between copia and retroviral proteins. Mol Cell Biol 5:1630–1638
Nagy ED, Molnar I, Schneider A, Kovacs G, Molnar-Lang M (2006) Characterization of chromosome-specific S-SAP markers and their use in studying genetic diversity in Aegilops species. Genome 49:289–296
Nicholas KB, Nicholas HB, Deerfield DW (1997) GeneDoc: analysis and visualization of genetic variation EMBnet. News 4:1–4
Oosterhout CV, Hutchinson WF, Wills DPM, Shipley P (2004) micro-checker: software for identifying and correcting genotyping errors in microsatellite data. Mol Ecol Notes 4:535–538
Pearce SR, Stuart-Rogers C, Knox MG, Kumar A, Ellis THN, Flavell AJ (1999) Rapid isolation of plant Ty1-copia group retrotransposons LTR sequences for molecular marker studies. Plant J 19:711–717
Pelsy F (2007) Untranslated leader region polymorphism of Tvv1, a retrotransposon family, is a novel marker useful for analyzing genetic diversity and relatedness in the genus Vitis. Theor Appl Genet 116:15–27
Pelsy F, Merdinoglu D (2002) Complete sequence of Tvv1, a family of Ty1 copia-like retrotransposons of Vitis vinifera L., reconstituted by chromosome walking. Theor Appl Genet 105:614–621
Perlman PS, Boeke-Science JD (2004) Molecular biology: ring around the retroelement. Science 303:182–184
Porceddu A, Albertini E, Barcaccia G, Marconi G, Bertoli FB, Veronesi F (2002) Development of S-SAP markers based on an LTR-like sequence from Medicago sativa L. Mol Genet Genomics 267:107–114
Powell M, Levin J (1996) Sequence and structural determinant required for priming of plus-strand DNA synthesis by the human immunodeficiency virus type 1 polypurine tract. J Virol 70:5288–5296
Queen RA, Gribbon BM, James C (2004) Retrotransposon-based molecular markers for linkage and genetic diversity analysis in wheat. Mol Genet Genomics 271:91–97
Rico-Cabanas L, Martinez-Izquierdo JA (2007) CIRE1, a novel transcriptionally active Ty1-copia retrotransposon from Citrus sinensis. Mol Genet Genomics 277:365–377
Schulman AH, Flavell AJ, Ellis THN (2004) The application of LTR retrotransposons as genetic markers in plants. In: Miller W, Capy P (eds) Mobile genetic elements: protocols and genomic applications. Humana, Totowa, pp 145–173
Sharbel TF (1999) Amplified fragment length polymorphism (AFLP): a non-random PCR-based technique for multilocus sampling. In: Epplen JT, Lubjuhn T (eds) A laboratory guide to DNA fingerprinting/profiling. Birkhauser, Basel, pp 177–194
Shirasu K, Schulman A, Lahaye T, Shulze L (2000) A contiguous 66-kb barley DNA sequence provides evidence for reversible genome expansion. Genome Res 10:908–915
Soleimani VD, Baum BR, Johnson DA (2005) Genetic diversity among barley cultivars assessed by sequence-specific amplification polymorphism. Theor Appl Genet 110:1290–1300
Sun HY, Dai HY, Zhao GL, Ma Y, Ou CQ, Li H, Li LG, Zhang ZH (2008) Genome-wide characterization of long terminal repeat-retrotransposons in apple reveals the differences in heterogeneity and copy number between Ty1-copia and Ty3-gypsy retrotransposons. J Integr Plant Biol 50:1130–1139
Suoniemi A, Tanskanen J, Schulman AH (1998) Gypsy-like retrotransposons are widespread in the plant kingdom. Plant J 13:699–705
Syed NH, Flavell AJ (2007) Sequence-specific amplification polymorphisms (SSAPs): a multi-locus approach for analyzing transposon insertions. Nat Protoc 1:2746–2752
Syed NH, Sørensen AP, Antonise R, Wiel CVD, Linden GVD, Westende WV, Hooftman DAP, Nijs HCMD, Flavell AJ (2005) A detailed linkage map of lettuce based on SSAP, AFLP and NBS markers. Theor Appl Genet 112:517–527
Tahara M, Aoki T, Suzuka S, Yamashita H, Tanaka M, Matsunaga S, Kokumai S (2004) Isolation of an active element from a high-copy-number family of retrotransposons in the sweetpotato genome. Mol Genet Genomics 272:116–127
Tam SM, Mhiri C, Vogelaar A, Kerkveld M, Pearce SR, Grandbastien M-A (2005) Comparative analyses of genetic diversities within tomato and pepper collection detected by retrotransposon-based SSAP, AFLP and SSR. Theor Appl Genet 110:819–831
Tan GH, Gao Y, Shi M, Zhang XY, He SH, Chen ZL, An CC (2005) SiteFinding-PCR: a simple and efficient PCR method for chromosome walking. Nucleic Acids Res 33:e122
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) The CLUSTAL-X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res 25:4876–4882
Tignon M, Watillon B, Kettman R (2001) Identification of copia like retrotransposable element by apple. Acta Hort 546:515–520
Varmus H (1982) Form and function of retroviral proviruses. Science 216:812–820
Venturi S, Dondini L, Donini P, Sansavini S (2006) Retrotransposon characterisation and fingerprinting of apple clones by S-SAP markers. Theor Appl Genet 112:440–444
Venturi S, Franceschi PD, Dondini L, Sansavini S (2009) Retrotransposon based markers to discriminate sports in pear. Acta Hort 814:701–704
Waugh R, McLean K, Flavell AJ, Pearce SR, Kumar A, Thomas BBT, Powell W (1997) Genetic distribution of BARE-1-like retrotransposable elements in the barley genome revealed by sequence-specific amplification polymorphisms (S-SAP). Mol Gen Genet 253:687–694
White SE, Habera LF, Wessler SR (1994) Retrotransposons in the flanking regions of normal plant genes: a role for copia-like elements in the evolution of gene structure and expression. Proc Natl Acad Sci USA 91:11792–11796
Wünsch A, Hormaza JI (2002) Cultivar identification and genetic fingerprinting of temperate fruit tree species using DNA markers. Euphytica 125:59–67
Yanez M, Verdugo I, Rodriguez PS, Ruiz-Lara S (1998) Highly heterogeneous families of Ty1/copia retrotransposons in the Lycopersicon chilense genome. Gene 222:223–228
Yao J, Dong Y, Morris BA (2001) Parthenocarpic apple fruit production conferred by transposon insertion mutations in a MADS-box transcription factor. Proc Natl Acad Sci USA 98:1306–1311
Zhao GL, Zhang ZH, Sun HY, Li H, Dai HY (2007) Isolation of Ty1-copia-like retrotransposon sequences from the apple genome by chromosome walking based on modified SiteFinding-polymerase chain reaction. Acta Bioch Bioph Sin 39:675–683
Acknowledgments
This work was supported by National Natural Science Foundation of China (No. 30500348) and Science Foundation of Department of Education from Liaoning Province (No. 2008T177).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by A. Abbott
Guiling Zhao and Hongyan Dai contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Fig. 1
S-SAP spectrograms obtained with the primer combination LTRP1 and MseI(ggt) in selective amplification. The horizontal scale indicates fragment size (in base pairs), and the vertical scale indicates relative fluorescence intensity (in RFU). The number in the pane specifies the fragment size (in base pairs) of the corresponding S-SAP peak. Spectrograms of each sample from ZGL-01 to ZGL-20 are as follows: FJ, NFN2, NFN3, NFN6, NFN7, AF, AF1, YF, IL1, L1, MHF1, HMS, SF, ES, MS, K, IF10, G, CH, 2001. (PDF 407 kb)
Supplemental Fig. 2
S-SAP spectrograms obtained with the primer combination LTRP2 and PstI(aat) in selective amplification. The horizontal scale indicates fragment size (in base pairs), and the vertical scale indicates relative fluorescence intensity (in RFU). The number in the pane specifies the fragment size (in base pairs) of the corresponding S-SAP peak. Spectrograms of each sample from ZGL-01 to ZGL-20 are as follows: FJ, NFN2, NFN3, NFN6, NFN7, AF, AF1, YF, IL1, L1, MHF1, HMS, SF, ES, MS, K, IF10, G, CH, 2001. (PDF 371 kb)
Rights and permissions
About this article
Cite this article
Zhao, G., Dai, H., Chang, L. et al. Isolation of two novel complete Ty1-copia retrotransposons from apple and demonstration of use of derived S-SAP markers for distinguishing bud sports of Malus domestica cv. Fuji. Tree Genetics & Genomes 6, 149–159 (2010). https://doi.org/10.1007/s11295-009-0236-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11295-009-0236-7