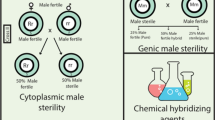
Abstract
Key message
The review outlines past failures, present status and future prospects of hybrid wheat, and includes information on CMS/CHA/transgenic approaches for male sterility, heterotic groups and cost-effective hybrid seed production.
Abstract
Hybrid varieties give increased yield and improved grain quality in both cross- and self-pollinated crops. However, hybrid varieties in self-pollinated crops (particularly cereals) have not been very successful, except for hybrid rice in China. In case of hybrid wheat, despite the earlier failures, renewed efforts in recent years have been made and hybrid varieties with desirable attributes have been produced and marketed in some European countries. This review builds upon previous reviews, with a new outlook and improved knowledge base, not covered in earlier reviews. New technologies have been described, which include the Hordeum chilense-based CMS–fertility restorer system, chromosomal XYZ-4E-ms system and the following transgenic technologies: (1) conditional male sterility involving use of tapetum-specific expression of a gene that converts a pro-toxin into a phytotoxin causing male sterility; (2) barnase-barstar SeedLink system of Bayer CropScience; (3) split-barnase system that obviates the need of a barstar-based male restorer line; and (4) seed production technology of DuPont-Pioneer that makes use of transgenes in production of male-sterile lines, but gives hybrid seed with no transgenes. This review also includes a brief account of studies for discovery of heterotic QTL, genomic prediction of hybrid vigour and the development of heterotic groups/patterns and their importance in hybrid wheat production. The problem of high cost of hybrid seed due to required high seed rate in wheat relative to hybrid rice has also been addressed. The review concludes with a brief account of the current efforts and future possibilities in making hybrid wheat a commercial success.



Similar content being viewed by others
References
Abdelkhalik AF, Shishido R, Nomura K, Ikehashi H (2005) QTL-based analysis of heterosis for grain shape traits and seedling characteristics in an indica–japonica hybrid in rice (Oryza sativa L.). Breed Sci 55:41–48
Adugna A, Nanda GS, Singh K, Bains NS (2004) A comparison of cytoplasmic and chemically-induced male sterility systems for hybrid seed production in wheat (Triticum aestivum). Euphytica 135:297–304
Aimin Z, Xiuling N, Dongchen L (2001) Advances of hybrid wheat breeding in China. Cereal Res Commun 9:343–350
Akel W, Thorwarth P, Mirdita V, Weissman EA, Liu G, Würschum T, Longin CFH (2018a) Can spelt wheat be used as heterotic group for hybrid wheat breeding? Theor Appl Genet 131(4):973–984
Akel W, Rapp M, Thorwarth Würschum T, Longin CFH (2018b) Hybrid durum wheat: heterosis of grain yield and quality traits and genetic architecture of anther extrusion. Theor Appl Genet. https://doi.org/10.1007/s00122-018-3248-6
Allen E, Gilbertson LA, Houmerd NM, Huarg S, Ivashuta SI, Robert JK (2007) Methods for producing hybrid seed. Patent WO/2007/047016 Monsanto Company, MO (US)
An G, Mitra A, Choi HK, Costa MA, An K, Thornburg RW, Ryan CA (1989) Functional analysis of the 3′ control region of the potato wound-inducible proteinase inhibitor II gene. Plant Cell 1:115–122
Baenziger PS, Rose D, Santra D, Xu L (2017) Improving small grains varieties for Nebraska. State breeding and quality evaluation report to the Nebraska Wheat Development, Utilization and Marketing Board, Nebraska, USA
Baenziger PS, Belamkar V, Easterly A, Garst N, Stoll H, Ibrahim AMH, Rudd JC, Adhikari A, Opena GB, Basent B, Longin F, Reif J, Sarazin Jean-Benoit, Poland J (2019) Developing the tools for hybrid wheat: American perspective. EWG breeding methods-workshop on hybrid wheat held on February 19, 2019 at University of Hoeinheim, Stuttgart, Germany
Bahl PN, Maan SS (1973) Chromosomal location of male fertility restoring genes in six lines of common wheat. Crop Sci 13:317–320
Basnet BR, Crossa J, Dreisigacker S, Perez-Rodriguez P, Manes Y, Singh RP, Camarillo-Castillo F, Murua M (2018) Hybrid wheat prediction using genomic, pedigree and environmental variables interaction models. Plant Gen. https://doi.org/10.3835/plantgenome2018.07.0051
Bernardo R (1994) Prediction of maize single-cross performance using RFLPs and information from related hybrids. Crop Sci 34:20–25
Bertin P, Gregoire D, Massart S, de Froidmont D (2001) Genetic diversity among European cultivated spelt revealed by microsatellites. Theor Appl Genet 102:148–156
Bertin P, Gregoire D, Massart S, de Froidmont D (2004) High level of genetic diversity among spelt germplasm revealed by microsatellite markers. Genome 47:1043–1052
Boeven PH, Longin CF, Wurschum T (2016a) A unified framework for hybrid breeding and establishment of heterotic groups in wheat. Theor Appl Genet 129:1231–1245
Boeven PHG, Longin CFH, Leiser WL, Kollers S, Ebmeyer E, Würschum T (2016b) Genetic architecture of male floral traits required for hybrid wheat breeding. Theor Appl Genet 129:2343–2357
Boeven PHG, Wurschum T, Rudloff J, Ebmeyer E, Longin CFH (2018) Hybrid seed set in wheat is a complex trait but can be improved indirectly by selection for male floral traits. Euphytica 214:110
Castillo A, Atienza SG, Martin AC (2014) Fertility of CMS wheat is restored by two Rf loci located on a recombined acrocentric chromosome. J Exp Bot 65:6667–6677
Changping Z (2013) Research and application of hybrid wheat in China. Eng Sci 11:19–21
Chu SH, Jiang W, Lee J, Chin JH, Koh HJ (2012) QTL analysis of heterosis for grain yield and yield related traits in indica-japonica crosses of rice (Oryza sativa L). Genes Genom 34:367–377
Cisar G, Cooper D (2002) Hybrid wheat. In: Curtis BC, Rajaram S, Macpherson HG (eds) Bread wheat: improvement and production. Food and Agriculture Organization of the United Nations, Rome, pp 157–174
Conner TW, Fabbri BJ, Huang J (2002) Methods for translational repression of gene expression in plants. Patent US0062499. Monsanto Company, MO (US)
Crow JF (1948) Alternative hypotheses of hybrid vigor. Genetics 33:477–487
Dotson SB, Lanahan MB, Smith AG, Kishore GM (1996) A phosphonate monoester hydrolase from Burkholderia caryophilli PG2982 is useful as a conditional lethal gene in plants. Plant J 10:383–392
Du HL, Maan SS, Hammond JJ (1991) Genetic analysis of male-fertility restoration in wheat. III: effects of neuploidy. Crop Sci 31:319–322
Duvick DN (1997) Heterosis: feeding people and protecting natural resources. In: Book of Abstracts. The Genetics and Exploitation of Heterosis in Crops. An International Symposium, Mexico City. 17–22 Aug 1997. CIMMYT, Mexico, DF, pp 6–9
East EM (1936) Heterosis. Genetics 21:375–397
Fischer S, Mohring J, Schön CC, Piepho H-P, Klein D, Schipprack W, Utz HF, Melchinger AE, Reif JC (2008) Trends in genetic variance components during 30 years of hybrid maize breeding at the University of Hohenheim. Plant Breed 127:446–451
Garcia AAF, Wang S, Melchinger AE, Zeng ZB (2008) Quantitative trait loci mapping and the genetic basis of heterosis in maize and rice. Genetics 180:1707–1724
Geiger HH, Miedaner T (2009) Rye breeding. In: Carena MJ (ed) Cereals. Springer, Berlin, pp 157–181
Geyer M, Bund A, Albrecht T, Hartl L, Mohler V (2016) Distribution of the fertility-restoring gene Rf3 in common and spelt wheat determined by an informative SNP marker. Mol Breed 36:167
Geyer M, Albrecht T, Hartl L, Mohlar V (2018) Exploring the genetics of fertility restoration controlled by Rf1 in common wheat (Triticum aestivum L.) using high-density linkage maps. Mol Genet Genom 293:451–462
Gowda M, Kling C, Würschum T, Reif JC (2010) Hybrid breeding in durum wheat: heterosis and combining ability. Crop Sci 50:2224–2230
Gowda M, Longin CFH, Lein V, Reif JC (2012) Relevance of specific versus general combining ability in winter wheat. Crop Sci 52:2494–2500
Gowda M, Zhao Y, Würschum T et al (2013) Relatedness severely impacts accuracy of marker assisted selection for disease resistance in hybrid wheat. Heredity 112:552–561
Guan RX, Liu DC, Zhang AM (2001) Genetic analysis and identification of RAPD markers of fertility restorer gene Rf6 for the T. timopheevii cytoplasmic male sterility of wheat. J Agric Biotechnol 9:159–162
Hawkes T, Pline-Srnic W, Dale R, Friend E, Hollinshead T, Howe P, Thompson P, Viner R, Greenland A (2011) D-glufosinate as a male sterility agent for hybrid seed production. Plant Biotech J 9:301–314
He ZH, Rajaram S, Huang GZ (1995) Wheat and wheat breeding in China: an overview. In: He ZH, Rajaram S, Xin ZY, Huang GZ (eds) 2001 A history of wheat breeding in China. CIMMYT, Mexico, DF, pp 1–14
Hermsen JGT (1965) Towards a more efficient utilization of genic male sterility in breeding hybrid barley and wheat. Euphytica 14:221–224
Hershey HP, Stoner TD (1991) Isolation and characterization of cDNA clones for RNA species induced by substituted benzenesulfonamides in corn. Plant Mol Biol 17:679–690
Hohn CE, Lukaszewski AJ (2016) Engineering the 1BS chromosome arm in wheat to remove the Rf multi locus restoring male fertility in cytoplasms of Aegilops kotschyi, Ae. uniaristata and Ae. mutica. Theor Appl Genet 129:1769–1774
Hull FH (1945) Recurrent selection for specific combining ability in corn. J Am Soc Agron 37:134–145
Jiang Y, Schmidt RH, Zhao Y, Reif JC (2017) A quantitative genetic framework highlights the role of epistatic effects for grain-yield heterosis in bread wheat. Nat Genet 49:1741–1746
Jinks JL, Jones RM (1958) Estimation of the components of heterosis. Genetics 43:223–234
Keil M, Sanches-Serrano J, Schell J, Willmitzer L (1986) Primary structure of a proteinase inhibitor II gene from potato. Nucleic Acids Res 14:5641–5650
Kempe K, Gils M (2011) Pollination control technologies for hybrid breeding. Mol Breed 27:417–437
Kempe K, Rubtsova M, Gils M (2014) Split-gene system for hybrid wheat seed production. Proc Natl Acad Sci 111:9097–9102
Kihara H, Tsunewaki K (1966) Basic studies on hybrid wheat breeding, carried out at the National Institute of Genetics. Seiken Ziho 18:55–63
Kihara H, Tsunewaki K (1967) Genetic principles applied to the breeding of crop plants. In: Brink RA (ed) Heritage from Mendel. University Wisconsin Press, Madison, pp 403–418
Koekemoer FP, Van Eeden E, Bonjean AP (2011) An overview of hybrid wheat production in South Africa and review of current worldwide wheat hybrid developments. In: Bonjean AP, Angus WJ, Van Ginkel M (eds) The world wheat book: a history of plant breeding. Lavoisier, Paris, pp 907–950
Koemel JE, Guenzi AC, Carver BF et al (2004) Hybrid and pure line hard winter wheat yield and stability. Crop Sci 44:107–113
Kojima T, Tsujimoto H, Ogihara Y (1997) High-resolution RFLP mapping of the fertility restoration (Rf3) gene against Triticum timopheevi cytoplasm located on chromosome 1BS of common wheat. Genes Genet Syst 72:353–359
Kriete G, Niehaus K, Perlick AM, Puhler A, Broer I (1996) Male sterility in transgenic tobacco plants induced by tapetum-specific deacetylation of the externally applied non-toxic compound N-acetyl-L-phosphinothricin. Plant J 9:809–818
Langer SM, Longin CFH, Würschum T (2014) Phenotypic evaluation of floral and flowering traits with relevance for hybrid breeding in wheat (Triticum aestivum L.). Plant Breed 133:433–441
Li XL, Liu LK, Hou N, Liu GQ, Liu CG (2005) SSR and SCAR markers linked to the fertility-restoring gene for a D2-type cytoplasmic male-sterile line in wheat. Plant Breed 124:413–415
Liable CA (1974) Chemical methods for pollen control. In: Proceedings of the 29th annual corn and sorghum research conference publication, vol 29, pp 174–182
Liu B, Deng J (1986) A dominant gene for male sterility in wheat. Plant Breed 97:204–209
Liu CG, Wu YW, Zhang CL, Ren SX, Zhang Y (1997) A preliminary study on the effects of Aegilops crassa cytoplasm on the characters of common wheat. J Genet Genom 24:241–247
Liu CG, Hou N, Liu GQ, Wu YW, Zhang CL, Zhang Y (2002) Studies on fertility genetic characters in D2-type CMS lines of common wheat. J Genet Genom 29:638–645
Liu CG, Hou N, Liu LK, Liu JC, Kang XS, Zhang AM (2006) A YA type cytoplasmic male sterile source in common wheat. Plant Breed 125:437–440
Liu G, Zhao Y, Gowda M, Longin CFH, Reif JC, Mette MF (2016) Predicting hybrid performance for quality traits through genomic-assisted approaches in Central European wheat. PLoS ONE 11:e0158635
Livers RW (1964) Fertility restoration and its inheritance in cytoplasmic male sterile wheat. Science 144:420
Longin CFH (2016) Future of wheat breeding is driven by hybrid wheat and efficient strategies for pre-breeding on quantitative traits. In: Zhang D (ed) Plant biotechnology and its applications, e-ISSN:2320-0189 and p-ISSN:2347-2380
Longin CF, Mühleisen J, Maurer HP, Zhang H, Gowda M, Reif JC (2012) Hybrid breeding in autogamous cereals. Theor Appl Genet 125:1087–1096
Longin CFH, Gowda M, Mühleisen J, Ebmeyer E, Kazman E, Schachschneider R, Schacht J, Kirchhoff M, Zhao Y, Reif JC (2013) Hybrid wheat: quantitative genetic parameters and consequences for the design of breeding programs. Theor Appl Genet 126:2791–2801
Longin CFH, Mi X, Wurschum T (2015) Genomic prediction in wheat: optimum allocation of test resources and comparison of breeding strategies for line and hybrid breeding. Thoer Appl Genet 128:12097–12306
Lucken KA (1987) Hybrid wheat. In: Heyne EG (ed) Wheat and wheat improvement. American Society of Agronomy, Madison, pp 444–452
Lukaszewski AJ (2017) Chromosomes 1BS and 1RS for control of male fertility in wheats and triticales with cytoplasms of Aegilops kotschyi, Ae. mutica and Ae. uniaristata. Theor Appl Genet 130:2521–2526
Ma ZQ, Sorrells ME (1995) Genetic analysis of fertility restoration in wheat using restriction fragment length polymorphisms. Crop Sci 35:1137–1143
Maan SS, McCracken EU (1968) Meiotic instability of common wheat strains derived from Triticum timopheevii zhuk crosses. Euphytica 17:445–450
Maan SS, Luchen KA, Bravo JM (1984) Genetic analyses of male-fertility restoration in wheat. I. Chromosomal location of Rf genes. Crop Sci 24:17–20
Mahajan V, Nagarjan S (1998) Opportunities in hybrid wheat-A review. PINSA B64:51–58
Mantle PG, Swan DJ (1995) Effect of male sterility on ergot disease spread in wheat. Plant Pathol 44:392–395
Mariani C, De Beuckeleer M, Truettner J, Leemans J, Goldberg RB (1990) Induction of male sterility in plants by a chimaeric ribonuclease gene. Nature 374:737–741
Mariani CV, Gossele V, de Beuckeleer M, de Block M, Goldberg RB, de Greef W, Leemans J (1992) A chimaeric ribonuclease-inhibitor gene restores fertility to male sterile plants. Nature 357:384–387
Martin AC, Atienza SG, Ramırez MC, Barro F, Martín A (2008) Male fertility restoration of wheat in Hordeum chilense cytoplasm is associated with 6HchS chromosome addition. Aust J Agri Res 59:206–213
Martin AC, Atienza SG, Ramírez MC, Barro F, Martín A (2010) Molecular and cytological characterization of an extra acrocentric chromosome that restores male fertility of wheat in the msH1 CMS system. Theor Appl Genet 121:1093–1101
Matuschke I, Qaim M (2006) Adoption and impact of hybrid wheat in India. Presented at ‘International Association of Agricultural Economists Conference’ held during August 12–18, 2006 at Gold Cost, Australia
McIntosh RA, Yamazaki Y, Dubcovsky J, Rogers J, Morris C, Appels R, Xia XC (2013) Catalogue of Gene Symbols for Wheat. In: The 12th International Wheat Genet Symposium September 8–13, 2013 Yokohama, Japan, pp 1–31
McRae DH (1985) Advances in chemical hybridisation. Plant Breed Rev 3:169–191
Melchinger AE, Gumber RK (1998) Overview of heterosis and heterotic groups in agronomic crops. In: CSSA special publication: concepts and breeding of heterosis in crop plants, pp 29–44
Melchinger AE, Utz HF, Piepho HP, Zeng ZB, Schön CC (2007) The role of epistasis in the manifestation of heterosis: a systems-oriented approach. Genetics 177:1815–1825
Mette MF, Gils M, Longin CFH, Reif JC (2015) Hybrid breeding in wheat. In: Ogihara Y, Takumi S, Handa H (eds) Advances in wheat Genetics: from genome to field. Springer, Tokyo, pp 225–232
Miedaner T, Laidig F (2019). Hybrid breeding in rye (Secale cereale L.). In: Al-Khayri et al JM (eds) Advances in plant breeding strategies. Vol 5 Cereals and legumes. Springer, Switzerland, pp 1–31
Mühleisen J, Piepho H-P, Maurer HP, Longin CFH, Reif JC (2014) Yield stability of hybrids versus lines in wheat, barley, and triticale. Theor Appl Genet 127:309–316
Mukai Y, Tsunewaki K (1979) Basic studies on hybrid wheat breeding.VIII. A new male sterility-fertility restoration system in common wheat utilizing the cytoplasms of Aegilops kotschyi and Ae. variabilis. Theor Appl Genet 54:153–160
Müller T, Schierscher-Viret B, Fossati D, Branant C, Schori A, Keller B (2018) Unlocking the diversity of genebanks: whole-genome marker analysis of Swiss bread wheat and spelt. Theor Appl Genet 131:407–416
Muqaddasi QH, Brassac J, Börner A, Pillen K, Roder MS (2017) Genetic architecture of anther extrusion in spring and winter wheat. Front Plant Sci 8:754
Murai K (1992). Basic studies on hybrid wheat breeding utilizing Aegilops crassa cytoplasm. Ph.D. dissertation. Kyoto University, Japan
Murai K (2002) Comparison of two fertility restoration systems against photoperiod-sensitive cytoplasmic male sterility in wheat. Plant Breed 121:363–365
Murai K, Tsunewaki K (1993) Photoperiod-sensitive cytoplasmic male sterility in wheat with Aegilops crassa cytoplasm. Euphytica 67:41–48
Murai K, Takumo S, Koga H, Ogihara Y (2002) Pistillody, homeotic transformation of stamens into pistil-like structures, caused by nuclear–cytoplasm interaction in wheat. Plant J 29:169–181
Murai K, Ohta H, Kurushima M, Ishikawa N (2016) Photoperiod-sensitive cytoplasmic male sterile elite lines for hybrid wheat breeding, showing high cross-pollination fertility under long-day conditions. Euphytica 212:313–322
Ni F, Qi J, Hao Q et al (2017) Wheat Ms2 encodes for an orphan protein that confers male sterility in grass species. Nat Commun 8:15121
Nielsen NH, Backes G, Stougaard J, Andersen SU, Jahoor A (2014) Genetic diversity and population structure analysis of European hexaploid bread wheat (Triticum aestivum L.) varieties. PLoS ONE 9:e94000
Niu N, Zhang GS, Liu HW, Wang JW, Li HX (2003) Inheritance on restoration performance of non-1BL/1RS male sterile line of Nian type in wheat. Acta Botanica Boreali-Occidentalia Sinica 23:608–614
O’Keefe DP, Tepperman JM, Dean C, Leto KJ, Erbes DL, Odell JT (1994) Plant expression of a bacterial cytochrome P450 that catalyzes activation of a sulfonylurea pro-herbicide. Plant Physiol 105:473–482
Parodi PC, de los Angeles Gaju M (2009) Male sterility induced by the chemical hybridizing agent clofencet on wheat, Triticum aestivum and T. turgidum var. durum. Cien Inv Agr 36:267–276
Pickett AA (1993) Hybrid wheat: results and problems. Adv Plant Breed Suppl J Plant Breed 15:1–259
Pickett AA, Galvey NV (1997) A further evaluation of hybrid wheat. Plant Var Seeds 10:15–32
Powers L (1944) An expansion of Jones’s theory for the explanation of heterosis. Am Nat 78:275–280
Qian CM, Xu A, Uiang GH (1986) Effects of low temperatures and genotypes on pollen development in wheat. Crop Sci 26:43–46
Qu Z, Li L, Luo J, Wang P, Yu S et al (2012) Qtl mapping of combining ability and heterosis of agronomic traits in rice backcross recombinant inbred lines and hybrid crosses. PLoS ONE 7:e28463
Reif JC, Hallauer AR, Melchinger AE (2005) Heterosis and heterotic patterns in maize. Maydica 50:215–223
Reif JC, Gumpert F, Fischer S, Melchinger AE (2007) Impact of genetic divergence on additive and dominance variance in hybrid populations. Genetics 176:1931–1934
Rembe M, Zhao Y, Jian Y, Reif JC (2018) Reciprocal recurrent genomic selection: an attractive tool to leverage hybrid wheat breeding. Theor Appl Genet. https://doi.org/10.1007/s00122-018-3244-x
Richey FD (1942) Mock-dominance and hybrid vigor. Science 96:280–281
Robertson LD, Curtis BC (1967) Monosomic analysis of fertility restoration in common wheat (T. aestivum L.). Crop Sci 9:493–495
Ru Z-G, Zhang L-P, Hu T-Z, Liu H-Y, Yang Q-K, Weng M-L, Wang B, Zhao C-P (2015) Genetic analysis and chromosome mapping of a thermo-sensitive genic male sterile gene in wheat. Euphytica 201:321–327
Schmidt W, Johnson VA, Mann SS (1962) Hybrid wheat. Neb Agric Exp Stn Q 9:9
Schulthess AW, Zhao Y, Longin CFH, Reif JC (2018) Advantage and limitations of multiple-trait genomic prediction for Fusarium head blight severity in hybrid wheat (Triticum aestivum L.). Theor Appl Genet 131:685–701. https://doi.org/10.1007/s00122-017-3029-7
Siedler H, Messmer MM, Schachermayr GM, Winzeler H, Winzeler M, Keller B (1994) Genetic diversity in European wheat and spelt breeding material based on RFLP data. Theor Appl Genet 88:994–1003
Singh SK, Chatrath R, Misra B (2010) Perspective of hybrid wheat research: a review. Indian J Agric Sci 80:1013–1027
Singh SP, Srivastava R, Kumar J (2015) Male sterility systems in wheat and opportunities for hybrid wheat development. Acta Physiol Plant 37:1713
Sinha P, Tomar SM, Vinod Singh VK, Balyan HS (2013) Genetic analysis and molecular mapping of a new fertility restorer gene Rf8 for Triticum timopheevi cytoplasm in wheat (Triticum aestivum L.) using SSR markers. Genetica 141:431–441
Sun B, Zhang A, Bonjean AP (2001) Chinese wheat pool (in) the world wheat book. In: Bonjean AP, Angus WJ (eds) A history of wheat breeding. Lavoisier Publishing Inc, Paris, pp 667–701
Tahir CM, Tsunewaki K (1969) Monosomic analysis of Triticum spelta var duhameliamum, a fertility-restorer for T. timopheevi cytoplasm. Jpn J Genet 44:1–9
Tsunewaki K (2015) Fine mapping of the first multi-fertility-restoring gene, Rf multi, of wheat for three Aegilops plasmons, using 1BS-1RS recombinant lines. Theor Appl Genet 128:723–732
Tucker EJ, Baumann U, Kouidri A et al (2017) Molecular identification of the wheat male fertility gene Ms1 and its prospects for hybrid breeding. Nat Commun 8:869
Virmani SS, Edwards JB (1983) Current status and future prospects for breeding hybrid rice and wheat. Adv Agron 36:145–214
Whitford R, Fleury D, Reif JC, Garcia M, Okada T, Korzun V, Langridge P (2013) Hybrid breeding in wheat: technologies to improve hybrid wheat seed production. J Exp Bot 64:5411–5428
Wilson JA (1984) Hybrid wheat breeding and commercial seed development. Plant Breeding Rev 2:303–319
Wilson P (1997) Hybrid wheat development in Australia and a proposal for hybrid wheat blends for developing countries. In: Book of abstracts. The genetics and exploitation of heterosis in crops. An international symposium, Mexico City. 17-22 Aug. 1997. CIMMYT, Mexico, DF, pp 216–217
Wilson JA, Ross WM (1962) Male sterility interaction of the Triticum aestivum nucleus and Triticum timopheevi cytoplasm. Wheat Inf Serv 14:29–30
Würschum T, Boeven PHG, Langer SM, Longin CFH, Leiser WL (2015) Multiply to conquer: copy number variations at Ppd-B1 and Vrn-A1 facilitate global adaptation in wheat. BMC Genet 16:96
Würschum T, Leiser WL, Longin CFH (2017a) Molecular genetic characterization and association mapping in spelt wheat. Plant Breed 136:214–223
Würschum T, Leiser WL, Weissmann S, Maurer HP (2017b) Genetic architecture of male fertility restoration of Triticum timopheevii cytoplasm and fine-mapping of the major restorer locus Rf3 on chromosome 1B. Theor Appl Genet 130:1253–1266
Xia XC, Reif JC, Hoisington D, Melchinger AE, Frisch M, Warburton ML (2004) Genetic diversity among CIMMYT maize inbred lines investigated with SSR markers: I. Lowland tropical maize. Crop Sci 44:2230–2237
Xiao J, Li J, Yuan L, Tanksley SD (1995) Dominance is the major genetic basis of heterosis in rice as revealed by QTL analysis using molecular markers. Genetics 140:745–754
Xie F, He Z, Esguerra MQ, Qiu F, Ramanathan V (2014) Determination of heterotic groups for tropical Indica hybrid rice germplasm. Theor Appl Genet 127:407–417
Xin XY, Wang WX, Yang JS, Luo XJ (2011) genetic analysis of heterotic loci detected in a cross between indica and japonica rice (Oryza sativa L.). Breed Sci 61:380–388
Xing QH, Ru ZG, Zhou CJ, Liang FS, Yang DE, Jin DM, Wang B (2003) Genic analysis and molecular mapping of thermosensitive genic male sterile gene (wtms1) in wheat. Theor Appl Genet 107:1500–1504
Xiyue S, Peng F, Lingjian M, Yajun X, Shudong L, Beiru H (2002) A comparison of wheat CMS lines of Ae. kotschyi cytoplasm of no 1B/1R type and 1B/1R type. Acta Univ Agric Boreali-occidentalis 30:1–4
Yang L, Liu B, Zhai H, Wang S, Liu H et al (2009) Dwarf male-sterile wheat: a revolutionary breeding approach to wheat. In: Shu QY (ed) Induced plant mutations in the genomics era. Food and Agriculture Organization of the United Nations, Rome, pp 370–372
Yen FS, Evans LE, Larter EH (1969) Monosomic analysis of fertility restoration in three restorer lines of wheat. Can J Genet Cytol 11:531–546
Yuan Q, Zhiying D, Peng T, Tian J (2012) QTL-based analysis of heterosis for number of grains per spike in wheat using DH and immortalized F2 populations. Euphytica 188:387–395
Zehr B (2001) Emerging biovision: Hybrid wheat. Paper presented at MSSRF-FAO expert meeting. Chennai, June 25–28
Zhang GS (1992) Breeding of wheat male sterile line having Aegilops uniaristata cytoplasm. Chin SciBull 7:641–645
Zhao Y, Zeng J, Fernando R, Reif JC (2013) Genomic prediction of hybrid wheat performance. Crop Sci 53:802–810
Zhao Y, Mette MF, Gowda M, Longin CFH, Reif JC (2014) Bridging the gap between marker-assisted and genomic selection of heading time and plant height in hybrid wheat. Heredity 112:638–645
Zhao Y, Liu G, Maurer HP et al (2015) Genome-based establishment of a high-yielding heterotic pattern for hybrid wheat breeding. Proc Natl Acad Sci 112:15624–15629
Zhou W, Frederic LK, Leslie LD, Wang S (2005) SSR markers associated with fertility restoration genes against Triticum timopheevii cytoplasm in Triticum aestivum. Euphytica 141:33–40
Zhou KJ, Wang SH, Feng YQ, Liu ZX, Wang GX (2006) The 4E-ms system of producing hybrid wheat. Crop Sci 46:250–255
Zhuo-Kun L, Kuan-Gang X, Zhan-Ling Z, Jin-Liang L, Shu-Xiao H, Bin T, Qian-Quian Y, Ji-Chun T (2010) Analysis of plant height heterosis based on QTL mapping in wheat. Acta Agron Sin 36:771–778
Acknowledgements
This review had a long incubation period, during which the authors received necessary facilities from Head, Department of Genetics and Plant Breeding, CCS University (Meerut, India); HSB continued to hold the position of INSA Senior Scientist, starting in 2015; PKG was initially awarded INSA Senior Scientist position during 2015–2017 and later continued to hold the position of Hony Senior Scientist, starting in June 2017; VG was awarded the position of INSPIRE Faculty by DST [DST/INSPIRE/04/2017/000413] and SG worked as SRF in an ICAR-NASF Project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Albrecht E. Melchinger.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Figure 1
XYZ 4E-ms system for production of hybrid wheat. ms1 recessive male-sterile gene located on chromosome 4BS of wheat; fertility can be restored to the homozygous male-sterile mutant (Z line) through the action of Ms1 dominant fertility restorer gene on alien chromosome 4E of Agropyron elongatum ssp. ruthenicum Beldie (Reproduced with permission from Whitford et al. 2013) (TIFF 111 kb)
Supplementary Figure 2a
Pollination control systems based on conditional male sterility. (a) The induction of male sterility through conversion of a non-phytotoxic substance, i.e. protoxin (N-acetyl-L-phosphinothricin) into a phytotoxin (L-phosphinothricin) in the anthers catalysed by the action of the transgene argE from Escherichia coli (encodes N-acetyl-L-ornithine deacetylase) having exclusive expression in male reproductive tissue (T, tapetum-specific promoter) (JPEG 50 kb)
Supplementary Figure 2b
Pollination control systems based on conditional male sterility. (b) Conditional male sterility through herbicide treatment. A transgene-impairing herbicide tolerance (HerbR) is constitutively expressed (C, constitutive promoter). Its inactivation in male tissue results in the selective destruction of cells necessary for pollen development after herbicide treatment. Transgene inactivation is achieved by male-specific expression (MS, male-specific promoter) of a protein that binds to a 50 UTL operator region (OP) of the HerbR-mRNA. Alternatively, a micro-RNA (miRNA) that binds specifically to an exogenous recognition site (RS) of the HerbR-mRNA may lead to suppression of the protein in the male reproductive tissue (modified after Kempe and Gils 2011) (JPEG 64 kb)
Supplementary Figure 3
Dual-component barnase-barstar system (SeedLink hybridization system) for production of hybrid wheat. Tapetal cell-specific expression (Ta) of barnase (bar) induces male sterility. Herbicide resistance gene (HR) allows selection of male-sterile individuals by herbicide spray. Barstar inhibitor (barstar) inactivates barnase resulting in restored fertility (Reproduced with permission from Whitford et al. 2013) (TIFF 92 kb)
Supplementary Figure 4a
(a) Vector constructs used in split-barnase technology (based on T-DNA; only T-DNA part of the vector is shown here); A is the barnase expression vector called provector; B (pICH13130) is the phiC31-integrase expression vector; (C) in F1 plants obtained by crossing two parents (one with the provector and the other with phiC31); (D) assembly of active barnase cytotoxin via the intein-mediated trans-splicing of two translated precursor protein molecules, Bar-N and Bar-C (with HPTII, hygromycin phosphotransferase; IntN and IntC). For details, see text. LB and RB, T-DNA left and right borders; ocs, octopine synthase terminator; NLS, SV40 T antigen nuclear localization signal (amino acids PKKKRKV); nos, nopaline synthase terminator; ocs, octopine synthase terminator; phiC31, phage phiC31 recombinase coding sequence; PSK, intron PSK7-i3 from Petunia hybrida; Ptap, tapetum-specific promoter from rice; Pubi, maize ubiquitin 1 promoter; S, (GGGGS)3 flexible peptide linker; UBQ, intron UBQ10-i1 from Arabidopsis thaliana (TIFF 69 kb)
Supplementary Figure 4b
(b) Experimental design for the production of hybrid seed using split-barnase approach. Genotypes that have an expected male-sterile phenotype are shown in green. (A) shows development of male-sterile line; (B) represents the strategy for maintaining the male-sterile line, and (C) shows hybrid seed production (reproduced from Kempe et al. 2014) (TIFF 95 kb)
Supplementary Figure 5
Pioneer-DuPont Seed Production Technology (SPT) for production of hybrid wheat. ms45, male sterility inducing gene; Ms45, a dominant fertility restorer gene; SC, seed colour marker; SC allows the visual separation of transgenic maintainer seed from non-transgenic male-sterile seed; PGI is the pollen germination inhibitor. F1 hybrid is non-transgenic (Reproduced with permission from Whitford et al. 2013) (TIFF 104 kb)
Rights and permissions
About this article
Cite this article
Gupta, P.K., Balyan, H.S., Gahlaut, V. et al. Hybrid wheat: past, present and future. Theor Appl Genet 132, 2463–2483 (2019). https://doi.org/10.1007/s00122-019-03397-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-019-03397-y