Abstract
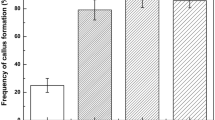
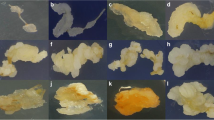
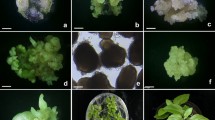
Improved suspension cell culture systems are needed to facilitate the application of recombinant DNA technology for wheat germplasm enhancement. This study evaluated three wheat (Triticum aestivum L.) cultivars, and the effects of medium basal salts, 2,4-D, sucrose, and L-proline concentrations on the establishment of rapidly growing and highly embryogenic callus and suspension cultures. Percent embryogenic calli was visually estimated and verified with light and scanning electron microscopy. The most highly embryogenic callus was produced by cultivar Bobwhite on medium with MS basal salts, 5.6 μ M 2,4-D, 58 mM sucrose, and zero proline. The suspension cultures that produced the greatest number of regenerated plants utilized callus tissue produced on solid medium with MS basal salts, 87 mM sucrose, 9 μM 2,4-D, and no proline.
Similar content being viewed by others
Abbreviations
- MS:
-
Murashige and Skoog medium (1962)
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
- IAA:
-
indole-3-acetic acid
- NAA:
-
napthaleneacetic acid; RG, relative growth
- %EC:
-
percent embryogenic calli
- RV:
-
Redway and Vasil medium (1990a)
- DPA:
-
days postanthesis
References
Ahmed KZ & Sagi F (1993) Plant Cell Rep. 12:175–179
Armstrong CL & Green CE (1985) Planta 164:207–214
Bapat SA, Joshi CP, & Mascarenhas AF (1988) Plant Cell Rep 7:538–541
Brown C, Brooks JF, Pearson D, & Mathias RJ (1989) J. Plant Physiol 133:727–733
Büter B, Schmid JE, & Stamp P (1991) Plant Cell Rep. 10:325–328
Carman JG & Campbell WF (1990) In: Bajaj YPS (ed) Biotechnology in Agriculture and Forestry, Vol 13. Wheat. Springer-Verlag Berlin pp. 68–87.
Carnes MG & Wright M (1988) Plant Sci. 57:195–203
Chang Y, Wang WC, Warfield CY, Nguyen HT, & Wong JR (1991) Plant Cell Rep. 9:611–614
Chu CC, Wang CC, & Sun CS (1975) Scientia Sinica 18:659–668
Galiba G & Erdai L (1986) Plant Sci. 45:65–70
Guo GQ, Xia GM, Li YM, & Chen HM (1991) Scientia Sinica Series B. 34:438–444
Harris R, Wright M, Byrne M, Varnum J, Brightwell B & Schubert K (1988) Plant Cell Rep. 7:337–340
He GY, Korbuly E & Barnabás B (1993) Plant Sci. 90:81–87
Heering DC, Guenzi AC, Peeper TP & Claypool PL (1992) Weed Sci. 40:174–179
Inagaki M, De Byser J, & Henry Y (1988) Japan J. Breed. 38:103–107
Kamo KK & Hodges TK (1986) Plant Sci. 45:111–117
Maddock SE (1987) Plant Cell Rep. 6:23–26
Murashige T & Skoog F (1962) Physiol. Plant 15:473–497
Ozias-Akins P & Vasil IK (1983) Protoplasma 117:40–44
Pauk J, Kertész Z, Jenes B, Purnhauser L, Manninen O Pulli S Barabás Z, & Dudits D (1994) Plant Cell Tiss. Org. Cult. 38:1–10
Redway FA, Vasil V, Lu D & Vasil IK (1990a) Theor. Appl Genet 79:609–617
Redway FA, Vasil V & Vasil IK (1990b) Plant Cell Rep. 8:714–717
Sears RG & Deckard EL (1982) Crop Sci. 22:546–550
Singer SR & McDaniel CN (1986) Can. J. Bot. 64:238–241
Vasil IK & Vasil V (1986) In: Vasil IK (ed) Cell Culture and Somatic Cell Genetics of Plants, Vol 3, Academic Press Orlando pp 121–150.
Vasil V, Redway F & Vasil IK (1990) Bio/Technology 8:429–434
Vasil V, Srivastava V, Castillo AM, Fromm ME, and Vasil IK (1993) Bio/Technology 11:1553–1558
Wang WC & Nguyen HT (1990) Plant Cell Rep. 8:639–642
Weeks JT, Anderson OD & Blechl AE (1993) Plant Physiol 102:1077–1084
Yang YM, He DG & Scott KJ (1991) Aust. J. Plant Physiol. 18:445–452
Zhou H, Zheng Y & Konzak CF (1991) Plant Cell Rep. 10:63–66
Author information
Authors and Affiliations
Additional information
Communicated by W. Parrott
Rights and permissions
About this article
Cite this article
Fellers, J.P., Guenzi, A.C. & Taliaferro, C.M. Factors affecting the establishment and maintenance of embryogenic callus and suspension cultures of wheat (Triticum aestivum L.). Plant Cell Reports 15, 232–237 (1995). https://doi.org/10.1007/BF00193726
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00193726