Abstract
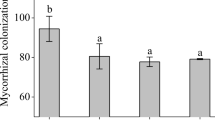
Gleditsia sinensis is a valuable tree species with important pharmaceutical uses. However, high soil NaCl concentration limits its growth in saline soil, including coastal areas. This study aimed to investigate the effects of arbuscular mycorrhizal fungi (AMF) on G. sinensis salinity tolerance and reveal its underlying physiological mechanism. A greenhouse experiment was performed. G. sinensis seedlings with and without AMF inoculation were subjected to four salinity levels: 0- (control), 50-, 100-, and 150-mM NaCl. After 2 months, the seedlings were harvested and analyzed for growth and biochemical parameters (antioxidant enzyme activity, superoxide anion content, malondialdehyde content, nutrient concentration, ion concentration, etc.). High AMF colonization rates (over 95%) and high mycorrhizal dependencies (over 75%) were observed across all NaCl levels. AMF effectively enhanced the salinity tolerance of G. sinensis seedlings by enhancing leaf stomatal conductance inducing higher net photosynthetic rates; improving peroxidase, catalase, and superoxide dismutase activities resulting in higher membrane stability indexes, lower superoxide anion and malondialdehyde contents in leaves and roots; increasing P/N ratio to mitigate P-limited biomass products; selectively absorbing less Na+ and more Ca2+ in their tissues to alleviate ion toxicity and maintain more favorable ion balances (e.g., K+/Na+) in their tissues. The results suggested AMF could effectively improve the salinity tolerance of G. sinensis, owning the great potential for afforestation and rehabilitation of G. sinensis in coastal areas.








Similar content being viewed by others
References
Abdel-Fattah GM, Asrar AWA (2012) Arbuscular mycorrhizal fungal application to improve growth and tolerance of wheat (Triticum aestivum L.) plants grown in saline soil. Acta Physiol Plant 34:267–277. https://doi.org/10.1007/s11738-011-0825-6
Alguacil MM, Hernández JA, Caravaca F, Portillo B, Roldán A (2003) Antioxidant enzyme activities in shoots from three mycorrhizal shrub species afforested in a degraded semi-arid soil. Physiol Plant 118:562–570. https://doi.org/10.1034/j.1399-3054.2003.00149.x
Al-Karaki GN (2006) Nursery inoculation of tomato with arbuscular mycorrhizal fungi and subsequent performance under irrigation with saline water. Sci Hortic 109:1–7. https://doi.org/10.1016/j.scienta.2006.02.019
Allen MF (1989) Mycorrhizae and rehabilitation of disturbed arid soils: processes and practices. Arid Soil Res Rehabil 3:229–241. https://doi.org/10.1080/15324988909381201
Alqarawi AA, Abd Allah EF, Hashem A (2014) Alleviation of salt induced adverse impact via mycorrhizal fungi in Ephedra aphylla Forssk. J Plant Interact 9:802–810. https://doi.org/10.1080/17429145.2014.949886
Augé RM (2001) Water relations, drought and VA mycorrhizal symbiosis. Mycorrhiza 11:3–42. https://doi.org/10.1007/s005720100
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Bolan NS (1991) A critical review on the role of mycorrhizal fungi in the uptake of phosphorus by plants. Plant Soil 134:189–207. https://doi.org/10.1007/BF00012037
Boyle E (2002) Oceanography. Oceanic salt switch. Science 298:1724–1725. https://doi.org/10.1126/science.1079238
Bulgarelli RG, Marcos FCC, Ribeiro RV, der Andrade SAL (2017) Mycorrhizae enhance nitrogen fixation and photosynthesis in phosphorus-starved soybean (Glycine max L. Merrill). Environ Exp Bot 140:26–33. https://doi.org/10.1016/j.envexpbot.2017.05.015
Cantrell IC, Linderman RG (2001) Preinoculation of lettuce and onion with VA mycorrhizal fungi reduces deleterious effects of soil salinity. Plant Soil 233:269–281. https://doi.org/10.1023/A:1010564013601
Cao K, Yu J, Xu DW, Ai KQ, Bao EC, Zou ZR (2018) Exposure to lower red to far-red light ratios improve tomato tolerance to salt stress. BMC Plant Biol 18:92. https://doi.org/10.1186/s12870-018-1310-9
Chance B, Maehly AC (1955) Assay of catalases and peroxidases. Method Enzymol 2:764–775. https://doi.org/10.1016/S0076-6879(55)02300-8
Chang W, Sui X, Fan XX, Jia TT, Song FQ (2018) Arbuscular mycorrhizal symbiosis modulates antioxidant response and ion distribution in salt-stressed Elaeagnus angustifolia seedlings. Front Microbiol 9:652. https://doi.org/10.3389/fmicb.2018.00652
Chow LMC, Tang JCO, Teo ITN, Chui CH, Lau FY, Leung TWT, Cheng G, Wong RSM, Wong ILK, Tsang KMS, Tan WQ, Zhao YZ, Lai KB, Lam WH, Guo DA, Chan ASC (2002) Antiproliferative activity of the extract of Gleditsia sinensis fruit on human solid tumour cell lines. Chemotherapy 48(6):303–308. https://doi.org/10.1159/000069713
Dasgan HY, Aktas H, Abak K, Cakmak I (2002) Determination of screening techniques to salinity tolerance in tomatoes and investigation of genotype responses. Plant Sci 163:695–703. https://doi.org/10.1016/S0168-9452(02)00091-2
Evelin H, Kapoor R, Giri B (2009) Arbuscular mycorrhizal fungi in alleviation of salt stress: a review. Ann Bot 104:1263–1280. https://doi.org/10.1093/aob/mcp251
Evelin H, Giri B, Kapoor R (2012) Contribution of Glomus intraradices inoculation to nutrient acquisition and mitigation of ionic imbalance in NaCl-stressed Trigonella foenum-graecum. Mycorrhiza 22:203–217. https://doi.org/10.1007/s00572-011-0392-0
Evelin H, Giri B, Kapoor R (2013) Ultrastructural evidence for AMF mediated salt stress mitigation in Trigonella foenum-graecum. Mycorrhiza 23:71–86. https://doi.org/10.1007/s00572-012-0449-8
Evelin H, Devi TS, Gupta S, Kapoor R (2019) Mitigation of salinity stress in plants by arbuscular mycorrhizal symbiosis: current understanding and new challenges. Front Plant Sci 10:470. https://doi.org/10.3389/fpls.2019.00470
Fernández N, Fontenla S, Messuti MI (2011) Co-occurrence of arbuscular mycorrhizas and dark septate endophytes in pteridophytes from a Valdivian Temperate Rainforest in Patagonia, Argentina. In: Pagano M (ed) Mycorrhiza: occurrence in natural and restored environments. Nova Science Publishers, New York, pp 99–126
Garg N, Bhandari P (2016) Silicon nutrition and mycorrhizal inoculations improve growth, nutrient status, K+/Na+ ratio and yield of Cicer arietinum L. genotypes under salinity stress. Plant Growth Regul 78:371–387. https://doi.org/10.1007/s10725-015-0099-x
Garg N, Chandel S (2014) Role of arbuscular mycorrhiza in arresting reactive oxygen species (ROS) and strengthening antioxidant defense in Cajanus cajan (L.) Millsp. Nodules under salinity (NaCl) and cadmium (Cd) stress. Plant Growth Regul 75:521–534. https://doi.org/10.1007/s10725-014-0016-8
Garg N, Singla P (2016) Stimulation of nitrogen fixation and trehalose biosynthesis by naringenin (Nar) and arbuacular mycorrhiza (AM) in chickpea under salinity stress. Plant Growth Regul 80:5–22. https://doi.org/10.1007/s10725-016-0146-2
Giannopolitis CN, Ries SK (1977) Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol 59:309–314. https://doi.org/10.1104/pp.59.2.309
Giovannetti M, Mosse B (1980) Evaluation of techniques for measuring vesicular arbuscular mycorrhizal infection in roots. New Phytol 84:489–500. https://doi.org/10.1111/j.1469-8137.1980.tb04556.x
Giri B, Kapoor R, Mukerji KG (2003) Influence of arbuscular mycorrhizal fungi and salinity on growth, biomass, and mineral nutrition of Acacia auriculiformis. Biol Fertil Soils 38:170–175. https://doi.org/10.1007/s00374-003-0636-z
Giri B, Kapoor R, Mukerji KG (2007) Improved tolerance of Acacia nilotica to salt stress by arbuscular mycorrhiza, Glomus fasciculatum may be partly related to elevated K/Na ratios in root and shoot tissues. Microb Ecol 54:753–760. https://doi.org/10.1007/s00248-007-9239-9
Goas G, Goas M, Larther F (1982) Accumulation of free proline and glycine betaine in Aster tripolium subjected to a saline shock: a kinetic study related to light period. Physiol Plant 55:383–388. https://doi.org/10.1111/j.1399-3054.1982.tb00309.x
Güsewel S (2004) N:P ratios in terrestrial plants: variation and functional significance. New Phytol 164:243–266. https://doi.org/10.1111/j.1469-8137.2004.01192.x
Hajiboland R, Aliasgharzadeh N, Laiegh SF, Poschenrieder C (2010) Colonization with arbuscular mycorrhizal fungi improves salinity tolerance of tomato (Solanum lycopersicum L.) plants. Plant Soil 331:313–327. https://doi.org/10.1007/s11104-009-0255-z
Hodges DM, DeLong JM, Forney CF, Prange RK (1999) Improving the thiobarbituric acid-reactive-substances assay for lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 207:604–611. https://doi.org/10.1007/s004250050524
Jarstfer AG, Farmer-Koppenol P, Sylvia DM (1998) Tissue magnesium and calcium affect mycorrhiza development and fungal reproduction. Mycorrhiza 7:237–242. https://doi.org/10.1007/s005720050186
Jiang MY, Zhang JH (2002) Water stress-induced abscisic acid accumulation triggers the increased generation of reactive oxygen species and up-regulates the activities of antioxidant enzymes in maize leaves. J Exp Bot 53:2401–2410. https://doi.org/10.1093/jxb/erf090
Jiang JX, Zhu LW, Wang L, Zhang WM (2007) Studies on modification of polysaccharide gum from Gleditsia sinensis Lam. seeds with α-galactosidase. Chem Ind for Prod 27(5):44–48 (in Chinese)
Jithesh MN, Prashanth SR, Sivaprakash KR, Parida AK (2006) Antioxidative response mechanisms in halophytes: their role in stress defiance. J Genet 85:237. https://doi.org/10.1007/BF02935340
Joslin JD, Henderson GS (1984) The determination of percentages of living tissue in woody fine root samples using triphenyltetrazolium chloride. For Sci 30:965–970. https://doi.org/10.1093/forestscience/30.4.965
Juniper S, Abbott L (1993) Vesicular-arbuscular mycorrhizas and soil salinity. Mycorrhiza 4:45–57. https://doi.org/10.1007/BF00204058
Khalil HA, Eissa AM, El-Shazly SM, Aboul Nasr AM (2011) Improved growth of salinity-stressed citrus after inoculation with mycorrhizal fungi. Sci Hortic 30:624–632. https://doi.org/10.1016/j.scienta.2011.08.019
Koltai H, Kapulnik Y (2010) Arbuscular mycorrhizas: physiology and function. Springer, Dordrecht, The Netherlands
Lei F, Bai ZY, Lu BS, Cai SW, Feng LN (2008) Effects of NaCl stress on Hovenia dulcis and Gleditsia sinensis seedlings growth, chlorophyll fluorescence, and active oxygen metabolism. Chin J Appl Ecol 19:2503–2508 (in Chinese)
Li JY, Zhao CY, Li J, Yan YY, Yu B, Han M (2013) Growth and leaf gas exchange in Populus euphratica across soil water and salinity gradients. Photosynthetica 51:321–329. https://doi.org/10.1007/s11099-013-0028-z
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Method Enzymol 148:350–382. https://doi.org/10.1016/0076-6879(87)48036-1
Lin JX, Wang YN, Sun SN, Mu CS, Yan XF (2017) Effects of arbuscular mycorrhizal fungi on the growth, photosynthesis and photosynthetic pigments of Leymus chinensis seedlings under salt-alkali stress and nitrogen deposition. Sci Total Environ 576:234–241. https://doi.org/10.1016/j.scitotenv.2016.10.091
Liu SL, Guo XL, Feng G, Maimaitiaili B, Fan JL, He XH (2016) Indigenous arbuscular mycorrhizal fungi can alleviate salt stress and promote growth of cotton and maize in saline fields. Plant Soil 398:195–206. https://doi.org/10.1007/s11104-015-2656-5
Lu RK (2000) Analytical methods for soil and agro-chemistry. China Agricultural Science and Technology Press, Beijing
Lu YW, Wang GQ, Meng QJ, Zhang WH, Duan BL (2014) Growth and physiological responses to arbuscular mycorrhizal fungi and salt stress in dioecious plant Populus tomentosa. Can J for Res 44:1020–1031. https://doi.org/10.1139/cjfr-2014-0009
Maathuis FJM (2009) Physiological functions of mineral macronutrients. Curr Opin Plant Biol 12:250–258. https://doi.org/10.1016/j.pbi.2009.04.003
Maathuis FJM, Amtmann A (1999) K+ nutrition and Na+ toxicity: the basis of cellular K+/Na+ ratios. Ann Bot 84:123–133. https://doi.org/10.1006/anbo.1999.0912
Marschner H (1995) Mineral nutrition of higher plant, 2nd edn. Academic, New York
Muchate NS, Nikalje GC, Rajurkar NS, Suprasanna P, Nikam TD (2016) Plant salt stress: adaptive responses, tolerance mechanism and bioengineering for salt tolerance. Bot Rev 82:371–406. https://doi.org/10.1007/s12229-016-9173-y
Munns R (2002) Comparative physiology of salt and water stress. Plant Cell Environ 25:239–250. https://doi.org/10.1046/j.0016-8025.2001.00808.x
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Parida AK, Das AB (2005) Salt tolerance and salinity effects on plants: a review. Ecotoxicol Environ Saf 60:324–349. https://doi.org/10.1016/j.ecoenv.2004.06.010
Phillips JM, Hayman DS (1970) Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans Br Mycol Soc 55:158–161. https://doi.org/10.1016/S0007-1536(70)80110-3
Pollastri S, Savvides A, Pesando M, Lumini E, Volpe MG, Ozudogru EA, Faccio A, Cunzo FD, Michelozzi M, Lambardi M, Fotopoulos V, Loreto F, Centritto M, Balestrini R (2018) Impact of two arbuscular mycorrhizal fungi on Arundo donax L. response to salt stress. Planta 247:573–585. https://doi.org/10.1007/s00425-017-2808-3
Porcel R, Aroca R, Ruiz-Lozano JM (2012) Salinity stress alleviation using arbuscular mycorrhizal fungi. A review. Agron Sustain Dev 32:181–200. https://doi.org/10.1007/s13593-011-0029-x
Porcel R, Redondo-Gómez S, Mateos-Naranjo E, Aroca R, Garcia R, Ruiz-Lozano JM (2015) Arbuscular mycorrhizal symbiosis ameliorates the optimum quantum yield of photosystem II and reduces non-photochemical quenching in rice plants subjected to salt stress. J Plant Physiol 185:75–83. https://doi.org/10.1016/j.jplph.2015.07.006
Rodríguez-Echeverría S, Hol WHG, Freitas H, Eason WR, Cook R (2008) Arbuscular mycorrhizal fungi of Ammophila arenaria (L.) Link: spore abundance and root colonisation in six locations of the European coast. Eur J Soil Biol 44:30–36. https://doi.org/10.1016/j.ejsobi.2007.01.003
Ruíz-lozano JM, Porcel R, Azcón C, Aroca R (2012) Regulation by arbuscular mycorrhizae of the integrated physiological response to salinity in plants: new challenges in physiological and molecular studies. J Exp Bot 63:4033–4044. https://doi.org/10.1093/jxb/ers126
Sarwat M, Hashem A, Ahanger MA, Abd-Allah EF, Alqarawl AA, Alyemeni MN, Ahmad P, Gucel S (2016) Mitigation of NaCl stress by arbuscular mycorrhizal fungi through the modulation of osmolytes, antioxidants and secondary metabolites in mustard (Brassica juncea L.) plants. Front Plant Sci 7:869. https://doi.org/10.3389/fpls.2016.00869
Sharifi M, Ghorbanli M, Ebrahimzadeh H (2007) Improved growth of salinity-stressed soybean after inoculation with pre-treated mycorrhizal fungi. J Plant Physiol 164:1144–1151. https://doi.org/10.1016/j.jplph.2006.06.016
Takai T, Kondo M, Yano M, Yamamoto T (2010) A quantitative trait locus for chlorophyll content and its association with leaf photosynthesis in rice. Rice 3:172–180. https://doi.org/10.1007/s12284-010-9047-6
Talaat NB, Shawky BT (2014) Protective effects of arbuscular mycorrhizal fungi on wheat (Triticum aestivum L.) plants exposed to salinity. Environ Exp Bot 98:20–31. https://doi.org/10.1016/j.envexpbot.2013.10.005
Thanaa E, Nawar A (1994) Salinity and mycorrhizal association in relation to carbohydrate status, leaf chlorophyll and activity of peroxidase and polyphenoloxidase enzymes in sour orange seedlings. Alex J Agric Res 29:342–351
Wang YH, Wang MQ, Li Y, Wu AP, Huang JY (2018) Effects of arbuscular mycorrhizal fungi on growth and nitrogen uptake of Chrysanthemum morifolium under salt stress. PLoS ONE 13(4):e0196408. https://doi.org/10.1371/journal.pone.0196408
Wilson JB, Sykes M (1999) Is zonation in coastal sand dunes determined primarily by sand burial or by salt spray? A test in New Zealand dunes. Ecol Lett 2:233–236. https://doi.org/10.1046/j.1461-0248.1999.00084.x
Yang HS, Zhang Q, Dai YJ, Liu Q, Tang JJ, Bian XM, Chen X (2015) Effects of arbuscular mycorrhizal fungi on plant growth depend on root system: a meta-analysis. Plant Soil 389:361–374. https://doi.org/10.1007/s11104-014-2370-8
Zhang JP, Tian XH, Yang YX, Liu QX, Wang Q, Chen LP, Li HL, Zhang WD (2016) Gleditsia species: an ethnomedical, phytochemical and pharmacological review. J Ethnopharmacol 178:155–171. https://doi.org/10.1016/j.jep.2015.11.044
Zhang T, Hu YJ, Zhang K, Tian CY, Guo JX (2018) Arbuscular mycorrhizal fungi improve plant growth of Ricinus communis by altering photosynthetic properties and increasing pigments under drought and salt stress. Ind Crop Prod 117:13–19. https://doi.org/10.1016/j.indcrop.2018.02.087
Zhou LG, Li D, Jiang WB, Qin ZZ, Zhao S, Qiu MH, Wu JY (2007) Two ellagic acid glycosides from Gleditsia sinensis Lam. with antifungal activity on Magnaporthe grisea. Nat Prod Res 21(4):303–309. https://doi.org/10.1080/14786410701192702
Zhu XC, Song FB, Xu HW (2010) Arbuscular mycorrhizae improves low temperature stress in maize via alterations in host water status and photosynthesis. Plant Soil 331:129–137. https://doi.org/10.1007/s11104-009-0239-z
Acknowledgements
We thank International Science Editing (http://www.internationalscienceediting.com) for editing this manuscript.
Funding
This work was financially supported by Jiangsu Agricultural Science and Technology Innovation Fund (Grant No. CX(17) 004), and the Greater Everglades Priority Ecosystem Science program and UC Davis Chancellors’ postdoc fellowship.
Author information
Authors and Affiliations
Contributions
The study conception and design were performed by JZ, GGW and JW; Material preparation and experiments were performed by JW, JY, BZ and QR; Data collection and analysis were performed by JW, JY and RH; Manuscript was written by JW; Funding were aquired by JZ and BZ.
Corresponding author
Ethics declarations
Conflict of interest
The author declared that they have no conflict of interest.
Additional information
Communicated by Pramod Kumar Nagar.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, J., Yuan, J., Ren, Q. et al. Arbuscular mycorrhizal fungi enhanced salt tolerance of Gleditsia sinensis by modulating antioxidant activity, ion balance and P/N ratio. Plant Growth Regul 97, 33–49 (2022). https://doi.org/10.1007/s10725-021-00792-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-021-00792-8