Abstract
Adsorption can be efficiently employed for the removal of various toxic dyes from water and wastewater. In this article, the authors reviewed variety of adsorbents used by various researchers for the removal of malachite green (MG) dye from an aqueous environment. The main motto of this review article was to assemble the scattered available information of adsorbents used for the removal of MG to enlighten their wide potential. In addition to this, various optimal experimental conditions (solution pH, equilibrium contact time, amount of adsorbent and temperature) as well as adsorption isotherms, kinetics and thermodynamics data of different adsorbents towards MG were also analyzed and tabulated. Finally, it was concluded that the agricultural solid wastes and biosorbents such as biopolymers and biomass adsorbents have demonstrated outstanding adsorption capabilities for removal of MG dye.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Till the late nineteenth century, all the colorants were extracted from shells, flowers, roots, insects and molluscs. But, with the historic discovery of first synthetic dye, Mauveine, most of the uses of natural dyes have been replaced with synthetic ones as they can be manufactured on a large scale (Gordon and Gregory 1987). At present, more than 1,00,000 synthetic dyes exist with an annual production of over 7 × 105 tonnes/year and are mostly associated with water pollution (Mittal et al. 2009; Gupta et al. 2011; Saravanan et al. 2016).
Malachite green (MG) is water soluble cationic dye that appears as green crystalline powder and belongs to triphenylmethane category (Raval et al. 2016b). The important physicochemical characteristics of MG were presented in Tables 1 and Table 2 narrated its applications and toxic effects. It was revealed from the Table 2 that the extensive usage of MG dye has caused several health hazards and hence, proper treatment of effluent containing MG dye is extremely necessary.
Many treatment technologies have been applied to decolorize MG from aqueous medium, such as biodegradation and decolorization (An et al. 2002; Levin et al. 2004; Ren et al. 2006; Eichlerová et al. 2006; Daneshvar et al. 2007a, b; Ayed et al. 2008; Ali et al. 2009; Wu et al. 2009; Cheriaa and Bakhrouf 2009; Ayed et al. 2010; Du et al. 2011; Khataee et al. 2011a, b; Shedbalkar and Jadhav 2011; Hu et al. 2011; Abd-El-Kareem and Taha 2012); photocatalytic degradation (Hasnat et al. 2003; Kominami et al. 2003; Lv et al. 2005; Yang et al. 2005; Chen et al. 2007; Sayılkan et al. 2007a, b, 2008; Tayade et al. 2007; Wang et al. 2008a; Ju et al. 2008; Asiltürk et al. 2009; Prado and Costa 2009; Bansal et al. 2009; Kaneva et al. 2010; Liu et al. 2010; Bojinova and Dushkin 2011; Chen et al. 2011; Tolia et al. 2012; Saha et al. 2012; Aliyan et al. 2013); photooxidative degradation (Modirshahla and Behnajady 2006); Solar degradation (Pirsaheb et al. 2015); coagulation–flocculation (Oladoja and Aliu 2009; Man et al. 2012); ozonation (Zhou et al. 2013); fenton reagent (Chen et al. 2002; Karimi et al. 2012; Bai et al. 2013; Nidheesh et al. 2013); Solvent extraction (Pandit and Basu 2004); sonochemical and sonophotocatalytic degradation (Berberidou et al. 2007; Bejarano-Pérez and Suárez-Herrera 2008; Moumeni and Hamdaoui 2012). However, all these techniques have certain limitations in terms of design, dye separation efficiency, cost and effectiveness. Advantages and disadvantages of various techniques used for the removal of MG dye have been summarized in Table 3 (Dawood and Sen 2014; Yang et al. 2014). More than 250 research articles have been published related to the adsorption of MG which confirmed that amongst all the available treatment techniques, adsorption has more frequently used one.
In addition, though there was a number of review articles such as Microbial decolorization of textile-dye containing effluents: a review by Banat et al. (1996); Fungal decolorization of dye wastewaters: a review by Fu and Viraraghavan (2001); Remediation of dyes in textile effluent: a critical review on current treatment technologies with a proposed alternative Robinson et al. (2001); The removal of color from textile wastewater using whole bacterial cells: a review by Pearce et al. (2003); Removal of synthetic dyes from wastewaters: a review by Forgacs et al. (2004); Decolourization of industrial effluents—available methods and emerging technologies—a review by Anjaneyulu et al. (2005); Adsorption–desorption characteristics of phenol and reactive dyes from aqueous solution on mesoporous activated carbon prepared from waste tires by Tanthapanichakoon et al. (2005); Non-conventional low-cost adsorbents for dye removal: a review by Crini (2006); Methods of dye removal from dye house effluent—an overview by Mondal (2008); Agricultural based activated carbons for the removal of dyes from aqueous solutions: a review by Demirbas (2009); Application of low-cost adsorbents for dye removal—a review by Gupta and Suhas (2009); Biodegradation of synthetic dyes—a review by Ali (2010); Decolorization of dye wastewaters by biosorbents: A review by Srinivasan and Viraraghavan (2010); Cationic and anionic dye adsorption by agricultural solid wastes: a comprehensive review by Salleh et al. (2011); A review on applicability of naturally available adsorbents for the removal of hazardous dyes from aqueous waste by Sharma et al. (2011); Adsorption of dyes and heavy metal ions by chitosan composites: a review by Wan Ngah et al. (2011); Microbial decolouration of azo dyes: A review by Solís et al. (2012); Adsorption of dyes using different types of sand: a review by Bello et al. (2013); Adsorptive removal of dyes from aqueous solution onto carbon nanotubes: a review by Gupta et al. (2013); Agricultural peels for dye adsorption: a review of recent literature by Anastopoulos and Kyzas (2014); Magnetic composite an environmental super adsorbent for dye sequestration—a review by Sivashankar et al. (2014); Dye and its removal from aqueous solution by adsorption: a review by Yagub et al. (2014), none of them has given comprehensive knowledge on the adsorptive removal of MG from aqueous media.
Therefore, in this review article we tried to congregate the available information on various adsorbents used for the removal of MG, a cationic dye, from water and wastewater. The main objective of this review article was to compile an extensive list of adsorbents which will help the future generation to furnish comprehensive up-to-date research summary on adsorbents used for removal of MG dye. Also the reported optimal experimental conditions (solution pH, contact time and amount of adsorbent) as well as adsorption isotherms, kinetics and thermodynamics data of different adsorbents towards MG were also mentioned. The authors embolden the readers to refer to the original research articles for information regarding the experimental conditions.
Dyes and their classification
Dyes (a natural or synthetic substance) are mainly chemical compounds that can affix themselves on the surface of fabrics to impart color (Yagub et al. 2014). It refers as a substance, usually organic, which is designed to be absorbed or adsorbed by, made to react with, or deposited within a substrate to impart color to that substrate with some degree of permanence (Burkinshaw 2016). Dyes may be classified in several ways (Adegoke and Bello 2015). Figure 1 summarizes the complete classification of dyes.
Based on the source of materials: Dyes can be classified mainly into two types based on their origin
Natural dyes
Natural dyes can be derived from the three primary sources—plants, animals and minerals. They are mostly negatively charged. Positively charged natural dyes can also exist, but they are not very common. Irrespective of the source of origin, natural dyes can be categorized as:
-
1.
Substantive dyes: Those dyes which become chemically fixed to the fiber without the aid of any other chemicals or additives are referred to as substantive dyes or direct dyes.
-
2.
Adjective dyes: Those natural dyes which require an added substance or mordant to make the dyes colorfast are known as adjective dyes or mordant dyes. Most of the natural dyes belong to this category (Cardon 2007).
Synthetic dyes
Dyes derived from organic or inorganic compounds are known as synthetic dyes. Because of their easy use, cost effectiveness and wide range of colors these dyes became popular.
Based on their method of application to the substrate
Acid dyes
Acid dyes are highly water soluble anionic dyes which contain one or more sulfonic groups/substituents and other acidic groups. They have better light fastness compare to basic dyes. The existence of sulfonic acid groups upsurge their solubility in water and give the dye molecules a negative charge. Acid dyes can be further divided into two sub-groups:
-
1.
Acid-leveling dyes: These dyes are smaller or medium sized, and show a moderate affinity for wool fibers. Hence, due to average affinity these dyes are not very resistant to washing.
-
2.
Acid-milling dyes: These dyes are larger than acid-leveling dyes, and show a much stronger inter-molecular attractions for wool fibers. Due to this strong affinity these dyes are very resistant to washing.
Basic dyes
These water soluble dyes possess cationic functional groups such as –NR3+ or =NR2+. They are generally amino and substituted amino compounds soluble in acid. They become attached to the fibers by formation of ionic bonds with fiber’s anionic groups.
Direct dyes
Although these dyes are water soluble anionic dyes, they cannot be classified as acid dyes because the acid groups are not the means of attachment to the fiber. Since, these dyes do not require any kind of fixing, they are called direct dyes.
Mordant dyes
A latin word mordant means ‘to bite’. It acts as ‘fixing agent’ to improve the color fastness of some acid dyes, because they have the ability to form insoluble colored complexes with metal ions. They are usually metal salts. Besides, alum there is a large range of other metallic salt mordants available. Aluminum, chromium and iron salts are frequently used for synthetic mordant dyes.
Reactive dyes
Fiber reactive dyes will form a covalent bond while they react with the cellulosic fiber. As a result they are very difficult to remove, once attached with the fiber. Initially, these dyes were designed for cellulose fibers but now some fiber-reactive dyes for protein and polyamide fibers are also commercially available.
Disperse dyes
These dyes have substantially low solubility in water. Their structure is small, planner and non-ionic with attached polar functional groups, such as –NO2 and –CN. They are mainly used for the dyeing of polyesters because they can interact with the polyester chains by forming dispersed particles.
Solvent dyes
These dyes are insoluble in water but soluble in alcohols, chlorinated hydrocarbons and liquid ammonia. They are mainly used for coloring plastics, synthetic fibers, gasoline, oils and waxes.
Sulfur dyes
Sulfur dyes are applied to cotton from an alkaline reducing bath with sodium sulfide as the reducing agent. These dyes are water insoluble but they are soluble in their reduced form and exhibit affinity for cellulose. They are low cost and have good fastness to light, washings and acids. The actual structures of sulfur dyes are largely unknown although it is considered that they possess sulfur-containing heterocyclic rings.
Based on their chemical structure: (Gordon and Gregory 1987; Waring and Hallas 1990)
Azo dyes
Approximately half of all dyes are azo dyes, making them the largest group of synthetic colorants used in textile industries. This type of dyes contain minimum one azo group but can also contain two or three, but very rarely, four azo groups. Azo dyes are complex aromatic compounds with significant structural diversity and are of great environmental concern because the reductive cleavage of azo linkages is responsible for the formation of amines, which are classified as toxic and carcinogenic (Raval et al. 2016a).
Anthraquinone dyes
Anthraquinone dyes are the second most important class after azo dyes. They are also one of the oldest types of dyes since they have been found in the covering of mummies dating back over 4000 years. In contrast to the azo dyes, which have no natural counterparts, all the natural red dyes were anthraquinones. The lower tinctorial strength and reduced flexibility increase the production cost of anthraquinone dyes and hence, they are not as widely used as azo dyes.
Benzodifuranone dyes
Benzodifuranone (BDF) dyes are one such type which attacking anthraquinone dyes. The BDF chromogen is one of the very few novel chromogens to have been accidently discovered this century
Polymethine and related dyes
Dyes containing a conjugated chain of carbon atoms terminated by an ammonium group and, in addition, a nitrogen, sulfur, or oxygen atom, or an equivalent unsaturated group is termed as polymethine dyes. This type of dyes may be neutral, cationic or anionic. The best known cationic polymethine dyes are the cyanine dyes. Cyanine dyes cannot be used as textile dyes because they have poor light fastness. They are used in photography.
Polycyclic aromatic carbonyl dyes
Polycyclic aromatic carbonyl dyes have one or more carbonyl groups. These carbonyl groups are linked by a quinonoid system. These dyes are relatively very large molecules which are made up from smaller units of anthraquinones. These dyes are often termed as anthraquinonoid vat dyes because they are applied to the substrate by a vatting process.
Indigoid dyes
Indigoid dyes also contain carbonyl groups, similar to the anthraquinone, benzodifuranone and polycyclic aromatic carbonyl dyes. They are vat dyes and represent one of the oldest known classes of dyes.
Phthalocyanines
Phthalocyanines are one of the novel chromophores which have been discovered since the nineteenth century. They are structurally similar to the natural porphyrin pigments haemin and chlorophyll. These natural pigments have poor stability, but phthalocyanines have exceptional stability and tinctorially stronger. All the phthalocyanine compounds are blue to green in color because substituents in their fused benzene rings exert only a minor sway on the color of phthalocyanines. They are widely used in printing inks and paints.
Di- and tri-aryl carbonium and related dyes
Di- and triarylcarbonium dyes and their heterocyclic derivatives comprise the oldest class of synthetic dyes—the majority of which were discovered in the 19th and early 20th century. Diphenylmethane and Triphenylmethane dyes are the most important aryl carbonium dyes. Structurally and property wise they are similar to the cyanine dyes i.e. exceptional brightness, high tinctorial strength and low light fastness.
Nitro and nitroso dyes
Nitro and nitroso dyes are minor commercial importance and constitute only a fraction of the total dyes market. Nitro dyes have simple chemical structures. They are small aromatic molecules which contain minimum one nitro group and one or more amino or hydroxy groups. Initially, the nitro dyes were acid dyes which were used for dyeing the natural animal fibers i.e. wool and silk. Picric acid was the first synthetic nitro dye which imparted a greenish-yellow color to silk but could not be used for longer period of time because of its toxicity and poor fastness properties.
Nitroso dyes are prepared by nitrosation of the appropriate phenol or naphthol. They are generally metal complex derivatives of o-nitrosophenols or naphthols. Nitroso dyes are polygenetic because they form differently colored complexes with different metals. They have good light fastness.
Based upon the electronic origins of color
Griffith (1976, 1981) has classified dyes based on the electronic mechanism by which color is generated. According to this, purely on theoretical basis, dyes can be arranged into four classes.
-
(II)
Donor–acceptor chromogens
-
(III)
Polyene chromogens
-
(IIII)
n → п2 chromogens
-
(IIV)
Cyanine type chromogens
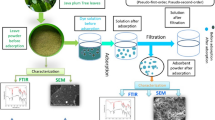
Literature on various adsorbents used for the removal of MG
See (Fig. 2)
Activated carbon adsorbents
Activated carbons (AC) are known as very efficacious adsorbents due to their large surface area, a micro-porous structure and a high degree of surface reactivity. However, due to their high production costs, these materials tend to be more expensive than other adsorbents. Four classes of AC include: powder (PAC), granular (GAC), fibrous (ACF) and cloth (ACC). Also two more frequently used ACs are: (1) commercial activated carbons and (2) activated carbons prepared from agricultural waste materials (Raval et al. 2016c).
Commercial activated carbons
A number of commercially available activated carbons such as powdered activated carbon (Kumar 2006); coal-based activated carbon (Aitcheson et al. 2000); commercially available powdered activated carbon (CPAC) (Malik et al. 2007); activated carbon prepared from lignite (Önal et al. 2007); activated Charcoal (Iqbal and Ashiq 2007); activated carbon prepared from Tuncbileklignite (Önal et al. 2006); oxyhumolite (Young brown coal) (Janos et al. 2005); Ordered mesoporous carbons (OMCs) (Tian et al. 2011); etc. have been extensively studied for the adsorption of MG.
Mesoporous carbon was synthesized by Anbia and Ghaffari (2012) for the removal of a cationic dye malachite green (MG) from aqueous solution. They carried out study under various experimental conditions and concluded that the equilibrium was attained within 30 min of contact with adsorbent and more than 99% removal of MG was reached at the optimum pH value of 8.5.
Aitcheson et al. (2000) used coal-based activated carbon 207EA (Sutcliffe Speakman) for the removal of aquaculture therapeutants (Malachite Green, formalin, Chloramine-T and Oxytetracycline) which were widely used in aquaculture to control fish parasites and disease. The adsorption process was well fitted to the Freundlich isotherm and confirmed that adsorption was heterogeneous in nature.
Adsorption of MG was studied by Mall et al. (2005) using commercial grade (ACC) and laboratory grade (ACL) activated carbons. Authors conducted batch adsorption studies to evaluate the effect of various parameters such as pH, adsorbent dose, contact time and initial MG concentration on the removal of MG and concluded that the effective pH was 7.0 for adsorption of MG. Equilibrium reached in about 4 h contact time and optimum ACC and ACL dosages were 20 and 4.0 g/L, respectively.
To overcome deficiencies of CACs the use of activated carbon adsorbents derived from various agricultural waste and industrial solid waste materials have received increasing consideration in the purification of wastewater over the last several years because they are generated in excess, inexpensive, locally available and their use will further solve the problem of their disposal (Pehlivan and Arslan 2007).
Activated carbons prepared from agricultural and industrial solid wastes
As biomass and other waste materials have little or no economic value and often present a disposal problem, they offer an inexpensive and additional renewable source of activated carbon. Thus, conversion of these low cost by products into activated carbon would provide an inexpensive substitute to the existing CACs, add monetary value and help in dropping the expenditure of waste disposal (Raval et al. 2016c).
Polygonum orientale Linn is an annual plant of the genus Polygonum in the family Polygonaceae, which is native to eastern Asia and widely distributed in China. This plant has a developed caudex system and generally produces large and continuous caudex masses. Therefore, this plant offers a good basis for the development of adsorbent materials. Looking upon this property of plant (Wang et al. 2010) prepared P. orientale Linn activated carbon (PLAC) from waste P. orientale Linn by chemical activation with H3PO4 and used as an adsorbent to remove malachite green (MG) and rhodamine B (RB) from aqueous solution.
Durian (Durio zibethinus Murray) is the most popular seasonal fruit in Southeast Asia, particularly Malaysia, Indonesia, Thailand, and the Philippines, and belongs to the family Bombacaceae. A significant percentage of the planted durian fruit crop is wasted each year. Only one third of durian fruit is edible, whereas the seeds (20–25%) and the shells are usually discarded. The wasted durian seed represents a significant potential for the development of value-added products. Thus, Ahmad et al. (2014) studied the removal of Malachite green dye using durian seed-based activated carbon.
Rambutan (Nephelium lappaceum L.) is a popular tropical fruit which belongs to Sapindaceae family. However, due to the high consumption of rambutan’s edible part, massive amount of the peel is disposed, causing a severe problem in the community as they gradually ferment and release off odors. Therefore, Ahmad and Alrozi (2011) converted rambutan peel into activated carbon by physiochemical activation for its potential to remove MG dye from wastewater.
Akar et al. (2013) produced active carbon from spent tea leaves (STAC) by activation with 4% NaOH solution for the removal of malachite green dye because tea is basically dried and processed leaves of plant called Camellia sinensis. It is consumed by the most of people in the world and is the second most popular beverage in the world. It is estimated that between 18 and 20 billion cups of tea are drunk daily in world and disposal of spent leaves become a major problem.
Banana (Musa paradisiaca) is native to Southeast Asia, most of the bananas produced are consumed locally and ∼10% are exported. Massive amounts of stalk (as waste products) are disposed, resulting in a serious environmental problem through the emission of foul odors when it is left to decompose. Hence, Bello et al. (2012) attempted to use banana stalk, an abundantly available lignocellulosic agricultural waste, as a cheap and renewable precursor for activated carbon preparation for MG dye removal and helped in placing value on this agricultural waste and provide a potentially cheap alternative to existing commercial activated carbons.
In addition to above described materials, a wide variety of other agricultural and industrial waste materials have also been used for the preparation of activated carbon and applied for the adsorptive removal of MG dye from aqueous solution. It includes Tamarind fruit shells carbon (Vasu 2008); Juglans regia shells carbon (Nethaji et al. 2012); Annona squmosa seed (Santhi et al. 2010a); Groundnut shell carbon (Malik et al. 2007); Pine sawdust (Akmil-Başar et al. 2005); Tea leaves (Singh and Rastogi 2004); Bamboo (Hameed and El-Khaiary 2008a); Jute fiber (Porkodi and Vasanth Kumar 2007) and waste printed circuit boards (Kan et al. 2015), etc.
In addition, Table 4 presented various commercially available and agricultural waste based activated carbon adsorbents used for the removal of MG and their optimum conditions.
Non-conventional low-cost adsorbents
Activated carbon adsorption has been cited by the US Environmental Protection Agency as one of the best available control technologies. However, although activated carbon is a preferred sorbent, its widespread use is restricted due to high cost. To decrease the cost of treatment, attempts have been made to find inexpensive alternative adsorbents. Recently, numerous approaches have been studied for the development of cheaper and effective adsorbents. Thus, there is a need to search for new and economical adsorbent that could remove metal. In this context, agricultural by-products and industrial waste can be seen as having a great potential to be developed as a low cost sorbent. A low-cost adsorbent is defined as the one which is plentiful in nature, or is a by-product or waste from industry and requires little or no processing (Raval et al. 2016c). Literature survey reveals that multifarious materials have been utilized as adsorbents under this category.
Waste materials from agriculture and industry
Agricultural solid wastes
Agricultural solid wastes are relatively cheap and are available in huge quantities. They can be used as an adsorbent due to their physic-chemical properties. The utilization of these wastes can play a significant role in national economy (Crini 2006).
Orange peel is discarded in the orange-juice and soft-drink industries all over the world. It has been used as an adsorbent for the removal of MG from wastewater by Kumar and Porkodi (2007). The results revealed that the adsorption process followed both Langmuir and Redlich–Peterson isotherms, the adsorption capacity was 483.63 mg/g.
A study of Rahman et al. (2005) reported the removal of MG from aqueous solutions onto Phosphoric acid (H3PO4) and sodium hydroxide (NaOH) treated rice husks.
Rice straw is a lignocellulosic agricultural stalk containing cellulose (37.4%), hemi-cellulose (44.9%), lignin (4.9%) and silica ash (13.1%). Gong et al. (2006) compared MG adsorption efficiency of native and thermos chemically modified rice straw and found their adsorption efficiency 94.34 and 256.41 mg/g, respectively.
Chieng et al. (2015) investigated the use of breadnut (Artocarpus camansi) as a low-cost adsorbent for the removal of MG toxic dye. In Brunei Darussalam, breadnut was locally known as ‘Kemangsi’, whose seeds were consumed either cooked or roasted. The skin and core of Artocorpus fruits, the major parts of the fruits, were inedible and have no economical value, and discarded as waste. Authors concluded that the adsorption characteristics of breadnut skin towards MG can be significantly enhanced by modification through treatment with dilute NaOH solution.
Other solid wastes from cheap and readily available resources have also been successfully employed for the removal of MG from aqueous solution and were given in Table 5.
Industrial by-products/industrial solid wastes
Industrial by products, such as metal hydroxide sludge, fly ash, red mud and bio-solids, can be used as low-cost adsorbents for MG removal. Table 6 represented the maximum adsorption capacities together with the isotherm, kinetics and thermodynamic results of various industrial solid wastes adsorbents for MG.
Fly ash: Witek-Krowiak et al. (2012) investigated the use of fly ash for the adsorption MG from solution with different contact times, initial dye concentrations, pHs and temperatures. The adsorption equilibrium has been described in terms of Langmuir model. The enthalpy values obtained in the experiment were not high, which suggested that physical sorption dominates in the process of cationic dyes bonding on microspheres of fly ash.
Bagasse fly ash (BFA) is a waste collected from the particulate separation equipment attached to the flue gas line of the sugarcane bagasse-fired boilers and has been used as an effective adsorbent for the removal of dyes. This is available in plenty, and almost free of cost from the sugar mills. In addition, bagasse fly ash generated in the sugar industry does not contain large amounts of toxic metals and hence, Mall et al. (2005) used bagasse fly ash (BFA) as adsorbent for the removal of MG from aqueous solutions. Maximum adsorption capacity for MG was reported as 170.33 mg/g (at pH 7.0 and adsorbent dosage of 1.0 g/L) and within 240 min of contact time.
Waste red mud: Zhang et al. (2014b) used the acid activated (Acid treatment neutralize the alkalinity of red mud, which could also be recognized as a method for activating the material) sintering process red mud (ASRM) for the adsorption of MG. The optimum process variables were found to be pH > 3.2, 180 min of contact time and 0.25 g/250 mL of adsorbent dosage. The adsorption process followed the Langmuir isotherm with 336.40 mg/g of adsorption capacity.
Activated slag: Steel plants produce granular blast furnace slag as a byproduct, and this material also causes a disposal problem. Hence, Gupta et al. (1997) utilized this waste product for the adsorption of MG from aqueous medium both in batch and column modes. Further, they concluded that the column capacity was found to be higher than the batch capacity. The removal takes place through a film diffusion mechanism at lower concentrations and by particle diffusion at higher concentrations.
Natural materials
Clays
Clay is mineral comprised of alumina and silica that usually includes bound water. Clays have a sheet-like structure and are composed of tetrahedrally arranged silicates and octahedrally arranged aluminates. Due to their low-cost, abundance in nature, high sorption capacity, chemical and mechanical stability, layered structure and high cation-exchange capacity (CEC), clay minerals are frequently used as an adsorbent (Krauskopf 1956; da Fonseca et al. 2006).The clays always contain exchangeable ions on their surface and play vital role in the environment by acting as a natural scavengers of contaminants by taking up cations and/or anions through either ion exchange or adsorption (Babel and Kurniawan 2003).
A low-cost inorganic powder (Persian Kaolin) was used as adsorbent for the sequestration of cationic dye from aqueous solution by Tehrani-Bagha et al. (2011). The sorption of the dye malachite green on kaolin confirmed to linear form of Langmuir adsorption equation. The maximum adsorption capacity was 52.91 mg/g. In addition, kinetic and thermodynamic parameters, such as enthalpy (∆H) and free energy (∆G) were calculated. The negative values of ΔG and positive values of ΔH indicated that the dye adsorption process was spontaneous and endothermic in nature
The efficacy of montmorillonite clay had been investigated by Tahir et al. (2010) for the adsorptive removal of malachite green dye from aqueous solution. Authors concluded that the MG dye was homogenously adsorbed on the surface of montmorillonite clay adsorbent.
Bulut et al. (2008) investigated the sorption of MG from aqueous solution onto bentonite as a function of contact time, pH and initial dye concentration. The adsorption equilibriums have been described in terms of Langmuir, Freundlich, Temkin and Dubinin–Radushkevich models. Authors reported that the adsorption process obeyed the Langmuir isotherm and pseudo-second-order kinetic model. The results of the thermodynamic study indicated that the sorption process was endothermic and spontaneous.
Tahir and Rauf (2006) utilized the natural bentonite clay for the adsorption of MG dye from aqueous solution. The authors finally concluded that the values of sorption free energy (E a) of 1.00–1.12 kJ/mol indicated physical adsorption of dye on clay mineral.
Siliceous materials
Siliceous rocks are sedimentary rocks that have silica (SiO2) as the principal constituent. The most common siliceous rock is chert other types include diatomite. They commonly form silica-secreting organisms, such as radiolarians, diatoms, or some types of sponges. The natural siliceous materials used for MG adsorption were perlite, silica, glass fibers and alunite because of their high abundance, easy availability and low cost.
Govindasamy et al. (2009) studied the removal of MG from aqueous solution by perlite. Authors reported 40 min of contact time, 0.1 g/50 mL of adsorbent dosage and 8.0–9.0 pH value were optimum conditions for the removal of dye. It was suggested that the adsorption of MG onto perlite was spontaneous, chemical and exothermic in nature and followed Freundlich isotherm as well as pseudo-first-order kinetic rate model.
Silica gel is a hydrophilic porous adsorbent. Aromatic compounds are found to involve the pi-cloud in hydrogen bonding with silanol (SiOH) group during adsorption. Cationic and nonionic surfactants adsorb on silica surface involving hydrogen bonding (Parida et al. 2006).
Kannan et al. (2008) utilized tetrahedral silica for the adsorption of MG. The adsorbent was characterized by Fourier transformed infrared spectroscopy (FT-IR) to confirm the tetrahedral framework of silica. The thermodynamic studies proved that the adsorption was endothermic and chemisorptions (ΔH 0 > 40 kJ/mol) on silica. Recovery of dye on silica was studied from 30 to 90 °C and observed that only 3.5% was recovered from silica. The less recovery on silica proved the strong adsorption of dye on silica.
Talc is the most common silicate material, with an idealized chemical formula of Mg3Si4O10(OH)2. Its ability to remove aqueous MG from solution had been studied by Lee et al. (2013).
Zeolites
Zeolites are highly porous crystalline aluminosilicates with tetrahedrally connected three dimensional frameworks and extra-framework charge balancing cations. Zeolite structures are commonly described in terms of the size, geometry and connectivity of the pore space. The use of natural zeolite as an adsorbent has gained interest among researchers; mainly because of its sorption properties provide a combination of ion exchange and molecular sieve properties which can also be easily modified (Goursot et al. 1997; Sánchez and Pariente 2011).
Zeolites have been used as chemical sieves, as water softeners, as removers of ammonia from urban, agricultural and industrial waste waters and of toxic gases from gaseous emissions, as filters for odor control, as chemical fertilizer, herbicide and pesticide carriers and in animal nutrition (Sand and Mumpton 1978; Breck 1984; Karge and Weitkamp 1989).
Han et al. (2010) investigated the use of natural zeolite, obtained from Xinyang city in China, for the removal of MG from aqueous solution. The rate constants of sorption for different kinetic models were calculated. Good correlation coefficients were obtained for the pseudo-second order kinetic model. In addition, authors were treated spent zeolite by microwave irradiation and it was found that yield of regeneration was 85.8% in the case of microwave irradiated time 10 min at 160 W.
Table 7 represented the maximum adsorption capacities together with the isotherm, kinetics and thermodynamic results of various natural materials used for the removal of MG
Biosorbents
Biosorption is defined as the accumulation and concentration of pollutants from aqueous solutions by the use of biological materials. Biological materials, such as yeast, fungi or bacterial biomass and biopolymers are used as adsorbents for the removal of MG from aqueous media (Raval et al. 2016d; Gupta et al. 2015).
Biomass
Biomass is biological material derived from living, or recently living organisms, which can be applied to both animal and vegetable derived material. The major advantages of biosorption over conventional treatment methods include, low cost; high efficiency; minimization of chemical or biological sludge; no additional nutrient requirement and possibility of regeneration of biosorbent (Volesky 1990).
Decolorization/bioadsorption of MG dye from wastewater by (dead/living) biomass of submerged aquatic plant (Rajesh Kannan et al. 2010); white-rot fungi (Kuhar and Papinutti 2013); fresh water algae (Kumar et al. 2005); brown marine algae (Kannan et al. 2009) and immobilized baker’s yeast cells (Godbole and Sawant 2006) were studied by many researchers (shown in Table 8)
Wang (2009) used alligator weed (a freshwater macrophyte) as biosorbent for the removal of basic dye malachite green from aqueous solution mainly because alligator weed, originated from South America, occupies agricultural areas and blocks drainage and irrigation channels causing problems on agricultural land. In addition, it causes water pollution from plant decomposition and an increase in mosquito breeding areas. Finally, author concluded that the alligator weed could serve as low-cost adsorbents for removing MG from aqueous medium.
Parthenium (Parthenium hysterophorus L.), also known as white top or carrot weed, an annual herbaceous weed was pretreated with H3PO4 and H2SO4 by Lata et al. (2008) and employed for the adsorption of MG dye.
Biopolymers
Biopolymers are high molecular weight compounds synthesized by living organisms. The performance of chitin hydrogel as an adsorbent to remove MG has been demonstrated by Tang et al. (2012). They found that the maximum adsorption capacity of hydrogel for MG was 26.88 mg/g. Authors concluded from the calculated activation energy (37.18 kJ/mol) and from the results of SEM and FTIR that the adsorption of malachite green on the hydrogel was physical process.
Bekçi et al. (2008) synthesized chitosan beads and examined its adsorption capacity towards MG. In contrast to the chitin hydrogels the authors concluded from the calculated activation energy value (85.6 kJ/mol) that the adsorption of MG onto chitosan beads was chemical in nature.
Although chitin and chitosan are abundantly available biopolymers and are biocompatible and biodegradable material, they have less chemical versatility due to their strong crystal structure, and hence their chemical structures have been modified to overcome this undesirable characteristic. In an effort to increase the chemical versatility and adsorption efficiency, chitin and chitosan biopolymers have been modified or crosslinked by various surfactants and crosslinking agents.
Huang et al. (2012) explored the use of Chitin grafted poly (acrylic acid) (chi-g-PAA) for the removal of MG from wastewater and concluded that the carboxylic groups that are grafted to the surface of chitin can be hydrolyzed to a sodium salt using dilute NaOH(aq) and can be readily utilized to adsorb malachite green from aqueous solution. The monolayer adsorption capacity obtained was 285.70 mg/g which was 10 times higher than the chitin hydrogels (26.88 mg/g).
Wan Ngah et al. (2010) studied the removal of MG from aqueous solutions through adsorption on to chitosan coated bentonite beads. The characterization of adsorbents was performed by Fourier Transform Infrared (FTIR) spectroscopy, scanning electron microscopy (SEM) coupled with energy dispersive spectroscopy (EDS) and X-ray diffraction (XRD) analysis. Authors mentioned that the monolayer adsorption capacity obtained was 435.00 mg/g which was five times greater than the chitosan beads (93.55 mg/g).
Peat
Peat is a porous and complex natural material, widely available and studied as an alternative adsorbent for different pollutants as well as dyes (Raval et al. 2016c).
Coco-peat was used by Vijayaraghavan et al. (2015) as an adsorbent to study the adsorption of malachite green and crystal violet from wastewater. It was found that a contact time of 60 min was sufficient for the adsorption to reach equilibrium. The optimum pH was found to be 7.0. The data for the adsorption of MG was described well by both the Sips adsorption isotherm model. The adsorption process was found to be endothermic.
Nanomaterials
Typical nanomaterials include nanotubes, nanoparticles, nanofibers, nanorods, fullerenes, and nanowires (Schmidt et al. 2002; Gupta and Saleh 2013; Gupta and Nayak 2012; Saravanan et al. 2013, 2014).
Carbon nanotubes were discovered accidentally by Iijima (1991), while studying the surfaces of graphite electrodes used in an electric arc discharge. CNTs are divided into two types based on the arrangement of their graphene cylinders: (1) single-walled CNTs (SWCNTs) and (2) multi-walled CNTs (MWCNTs) (Khani et al. 2010; Saleh and Gupta 2011).
MWNTs are collections of several concentric graphene cylinders and are larger structures compared to SWNTs which are individual cylinders of 1–2 nm diameter. The former can be considered as a mesoscale graphite system, whereas the latter is truly a single large molecule (Odom et al. 1998). The mechanisms by which the dye molecules are sorbed onto CNTs are very complicated and appear attributable to electrostatic attraction, sorption, precipitation and chemical interaction between the reactive groups of dye molecules and the surface functional groups of CNTs.
Recently, Rajabi et al. (2016) functionalized MWCNTs with the carboxylate group and used as an adsorbent for the rapid and fast removal of MG dye from the aqueous solutions. The optimized contact time and pH were 10 min and 9, respectively. The effect of temperature revealed that the adsorption capacity of MG dye increased with increasing contact time, temperature and pH. The adsorption equilibrium and kinetic data was well fitted and found to be in good agreement with the Langmuir isotherm model and pseudo second order kinetic model, respectively.
Shirmardi et al. (2013) prepared functionalized multi-walled carbon nanotubes (f-MWCNTs) by acid treatment and used for the adsorption of MG dye from aqueous solution in batch mode. The results indicated that by increasing contact time, pH and adsorbent dosage, the removal percentage increased, but by increasing the initial MG concentration, the removal percentage decreased. Based on correlation coefficient (R 2), the experimental data follows the Langmuir isotherm with maximum adsorption capacity of 142.85 mg/g.
Halloysite, a mineral of the kaolin group [forms hollow tubular crystals (halloysite nanotubes—HNTs)], can be used as MG dye adsorbent by Kiani et al. (2011) and found that the equilibrium adsorption was reached within 30 min. The adsorption of MG by HNTs obeyed pseudo-second-order kinetics with activation energy 18.28 kJ/mol, suggested that the process was physisorption. The best-fit adsorption isotherm was achieved with the Langmuir model, indicating that homogeneous adsorption occurred. The negative values of ΔG° and positive value of ΔH° showed that the adsorption was a spontaneous and endothermic.
The particles of at least one dimension smaller than 1 µm, and potentially as small as atomic and molecular length scales (0.2 nm) are called nanoparticles. Nanoparticles can have amorphous or crystalline form (Buzea et al. 2007). Due to some of the unique characteristics such as small size, catalytic potential, large surface area, ease of separation, and large number of active sites for interaction with different contaminants the nanoparticles have established themselves as excellent adsorbents (Raval et al. 2016d).
Composites and nano-composites
The nano-scale inclusions that are imbedded within matrix of a material are referred as composites (Chen et al. 2013).
Poly(vinyl alcohol) (PVA) is a water-soluble material containing many reactive hydroxyl groups. It has been extensively applied in biomedical and pharmaceutical fields due to its low cost, non-toxicity, biocompatibility, good mechanical strength and chemical stability. However, the PVA foam illustrated low adsorption for organic and inorganic pollutants. Chitosan (CS) is one of the most abundant biopolymers in nature. It exhibits good adsorption capacity for organic dyes and heavy metals because it has a large number of hydroxyl and amine groups. However, the use of CS in the form of flake or powder cannot be separated easily. Therefore, Li et al. (2012) prepared a novel foam adsorbent based on PVA/CS composites by the interpenetrating polymer network (IPN), foaming process and freeze-thaw treatment and found that the composite material had higher adsorption efficiency (380.65 mg/g) compared with the single component.
Hussain et al. (2006) defined nanocomposites as materials consisting of at least two phases with one dispersed in another that is called matrix, and thus forms a three dimensional network.
Wang et al. (2015) prepared Magnetic β-cyclodextrin-graphene oxide nanocomposites (Fe3O4/β–CD/GO) as an adsorbent for MG dye because graphene oxide have abundant hydrophilic groups on its surface, and thus it has good adsorption capacity still its separation was difficult task from sample solution and hence, to simplify the retrieval procedure, magnetic adsorbent (Fe3O4) has been incorporated with GO. In addition, β-CD was also added onto composite (Magnetic-graphene oxide) to provide hydrophobic inner cavity and a hydrophilic exterior which can enhance the adsorption capacity of composite adsorbent for MG dye.
Table 9 presented the maximum adsorption capacities of various nanomaterials, composite and nanocomposite adsorbents used for MG.
Miscellaneous adsorbents
See Table 10.
Conclusions and future perspectives
Adsorption process being very easy, economical, effective and versatile had become the most favored methods for amputation of toxic and hazardous dyes from wastewater. This review article revealed about various adsorbents, such as activated carbons, low-cost materials, nanomaterials, composites and nanocomposites, used for the adsorption and removal of MG from water and wastewater. It was concluded that amongst all the other adsorbents, agricultural solid wastes and biosorbents, such as biopolymers and biomass, have been extensively used for the removal of MG and are acknowledged as effective and economic adsorbents for the removal of MG dye. Various experimental conditions (pH, temperature, contact time and amount of adsorbent) have been also mentioned for related adsorbents in the present article.
This review article also disclosed that the Langmuir and Freundlich adsorption isotherm models were frequently applied to evaluate the adsorption capacity of various adsorbents towards MG. In kinetic study, it was observed that the pseudo-second-order rate model was been followed for the adsorption of MG and according to thermodynamic parameters the adsorption process was found to be spontaneous in nature.
Since, most of the reported studies had been performed using batch and small-scale column adsorption tests, further research is required for the development of more effective adsorbents, modeling of adsorption mechanism, regeneration of spent adsorbents and treatment of real industrial wastewater.
References
Abd-El-Kareem MS, Taha HM (2012) Decolorization of malachite green and methylene blue by two microalgal species. Int J Chem Environ Eng 3:297–302
Adegoke KA, Bello OS (2015) Dye sequestration using agricultural wastes as adsorbents. Water Resour Ind 12:8–24. doi:10.1016/j.wri.2015.09.002
Afkhami A, Moosavi R, Madrakian T (2010) Preconcentration and spectrophotometric determination of low concentrations of malachite green and leuco-malachite green in water samples by high performance solid phase extraction using maghemite nanoparticles. Talanta 82:785–789. doi:10.1016/j.talanta.2010.05.054
Agarwal S, Tyagi I, Gupta VK et al (2016) Kinetics and thermodynamics of Malachite Green dye removal from aqueous phase using iron nanoparticles loaded on ash. J Mol Liq. doi:10.1016/j.molliq.2016.04.039
Ahmad MA, Ahmad N, Bello OS (2014) Adsorptive removal of malachite green dye using durian seed-based activated carbon. Water Air Soil Pollut 225:1–18. doi:10.1007/s11270-014-2057-z
Ahmad MA, Alrozi R (2011) Removal of malachite green dye from aqueous solution using rambutan peel-based activated carbon: equilibrium, kinetic and thermodynamic studies. Chem Eng J 171:510–516. doi:10.1016/j.cej.2011.04.018
Ahmad R, Kumar R (2010) Adsorption studies of hazardous malachite green onto treated ginger waste. J Environ Manag 91:1032–1038. doi:10.1016/j.jenvman.2009.12.016
Ahmad R, Mondal PK (2010) Application of modified water nut carbon as a sorbent in congo red and malachite green dye contaminated wastewater remediation. Sep Sci Technol 45:394–403. doi:10.1080/01496390903484875
Ai L, Huang H, Chen Z et al (2010) Activated carbon/CoFe2O4 composites: facile synthesis, magnetic performance and their potential application for the removal of malachite green from water. Chem Eng J 156:243–249. doi:10.1016/j.cej.2009.08.028
Aitcheson SJ, Arnett J, Murray KR, Zhang J (2000) Removal of aquaculture therapeutants by carbon adsorption: 1. Equilibrium adsorption behaviour of single components. Aquaculture 183:269–284. doi:10.1016/S0044-8486(99)00304-X
Akar E, Altinişik A, Seki Y (2013) Using of activated carbon produced from spent tea leaves for the removal of malachite green from aqueous solution. Ecol Eng 52:19–27. doi:10.1016/j.ecoleng.2012.12.032
Akmil-Başar C, Onal Y, Kiliçer T, Eren D (2005) Adsorptions of high concentration malachite green by two activated carbons having different porous structures. J Hazard Mater 127:73–80. doi:10.1016/j.jhazmat.2005.06.025
Aksakal O, Ucun H, Kaya Y (2009) Application of Eriobotrya japonica (Thunb.) Lindley (Loquat) seed biomass as a new biosorbent for the removal of malachite green from aqueous solution. Water Sci Technol J Int Assoc Water Pollut Res 59:1631–1639. doi:10.2166/wst.2009.130
Ali H (2010) Biodegradation of synthetic dyes—a review. Water Air Soil Pollut 213:251–273. doi:10.1007/s11270-010-0382-4
Ali H, Ahmad W, Haq T (2009) Decolorization and degradation of malachite green by Aspergillus flavus and Alternaria solani. Afr J Biotechnol. doi:10.4314/ajb.v8i8.60239
Aliyan H, Fazaeli R, Jalilian R (2013) Fe3O4@mesoporous SBA-15: a magnetically recoverable catalyst for photodegradation of malachite green. Appl Surf Sci 276:147–153. doi:10.1016/j.apsusc.2013.03.049
Altınışık A, Gür E, Seki Y (2010) A natural sorbent, Luffa cylindrica for the removal of a model basic dye. J Hazard Mater 179:658–664. doi:10.1016/j.jhazmat.2010.03.053
An S-Y, Min S-K, Cha I-H et al (2002) Decolorization of triphenylmethane and azo dyes by Citrobacter sp. Biotechnol Lett 24:1037–1040. doi:10.1023/A:1015610018103
Anastopoulos I, Kyzas GZ (2014) Agricultural peels for dye adsorption: a review of recent literature. J Mol Liq 200(Part B):381–389. doi:10.1016/j.molliq.2014.11.006
Anbia M, Ghaffari A (2012) Removal of malachite green from dye wastewater using mesoporous carbon adsorbent. J Iran Chem Soc 8:S67–S76. doi:10.1007/BF03254283
Anirudhan TS, Suchithra PS (2009) Adsorption characteristics of humic acid-immobilized amine modified polyacrylamide/bentonite composite for cationic dyes in aqueous solutions. J Environ Sci China 21:884–891
Anirudhan TS, Suchithra PS, Radhakrishnan PG (2009) Synthesis and characterization of humic acid immobilized-polymer/bentonite composites and their ability to adsorb basic dyes from aqueous solutions. Appl Clay Sci 43:336–342. doi:10.1016/j.clay.2008.09.015
Anjaneyulu Y, Chary NS, Raj DSS (2005) Decolourization of industrial effluents—available methods and emerging technologies—a review. Rev Environ Sci Biotechnol 4:245–273. doi:10.1007/s11157-005-1246-z
Arellano-Cárdenas S, López-Cortez S, Cornejo-Mazón M, Mares-Gutiérrez JC (2013) Study of malachite green adsorption by organically modified clay using a batch method. Appl Surf Sci 280:74–78. doi:10.1016/j.apsusc.2013.04.097
Arivoli S, Sundaravadivelu M, Elango KP (2008) Removal of basic and acidic dyes from aqueous solution by adsorption on a low cost activated carbon: kinetic and thermodynamic study. Indian J Chem Technol 15:130
Asiltürk M, Sayılkan F, Arpaç E (2009) Effect of Fe3+ ion doping to TiO2 on the photocatalytic degradation of malachite green dye under UV and vis-irradiation. J Photochem Photobiol Chem 203:64–71. doi:10.1016/j.jphotochem.2008.12.021
Ayed L, Chaieb K, Cheref A, Bakhrouf A (2008) Biodegradation of triphenylmethane dye Malachite Green by Sphingomonas paucimobilis. World J Microbiol Biotechnol 25:705–711. doi:10.1007/s11274-008-9941-x
Ayed L, Chaieb K, Cheref A, Bakhrouf A (2010) Biodegradation and decolorization of triphenylmethane dyes by Staphylococcus epidermidis. Desalination 260:137–146. doi:10.1016/j.desal.2010.04.052
Babel S, Kurniawan TA (2003) Low-cost adsorbents for heavy metals uptake from contaminated water: a review. J Hazard Mater 97:219–243. doi:10.1016/S0304-3894(02)00263-7
Baek M-H, Ijagbemi CO, Se-Jin O, Kim D-S (2010) Removal of malachite green from aqueous solution using degreased coffee bean. J Hazard Mater 176:820–828. doi:10.1016/j.jhazmat.2009.11.110
Bai C, Xiao W, Feng D et al (2013) Efficient decolorization of Malachite Green in the Fenton reaction catalyzed by [Fe(III)-salen]Cl complex. Chem Eng J 215–216:227–234. doi:10.1016/j.cej.2012.09.124
Banat IM, Nigam P, Singh D, Marchant R (1996) Microbial decolorization of textile-dye containing effluents: a review. Bioresour Technol 58:217–227. doi:10.1016/S0960-8524(96)00113-7
Bansal P, Bhullar N, Sud D (2009) Studies on photodegradation of malachite green using TiO2/ZnO photocatalyst. Desalin Water Treat 12:108–113. doi:10.5004/dwt.2009.944
Başar CA (2006) Applicability of the various adsorption models of three dyes adsorption onto activated carbon prepared waste apricot. J Hazard Mater 135:232–241. doi:10.1016/j.jhazmat.2005.11.055
Beak MH, Ijagbemi CO, Kim DS (2009) Treatment of malachite green-containing wastewater using poultry feathers as adsorbent. J Environ Sci Health Part A Tox Hazard Subst Environ Eng 44:536–542. doi:10.1080/10934520902720132
Bejarano-Pérez NJ, Suárez-Herrera MF (2008) Sonochemical and sonophotocatalytic degradation of malachite green: the effect of carbon tetrachloride on reaction rates. Ultrason Sonochem 15:612–617. doi:10.1016/j.ultsonch.2007.09.009
Bekçi Z, Özveri C, Seki Y, Yurdakoç K (2008) Sorption of malachite green on chitosan bead. J Hazard Mater 154:254–261. doi:10.1016/j.jhazmat.2007.10.021
Bekçi Z, Seki Y, Cavas L (2009) Removal of malachite green by using an invasive marine alga Caulerpa racemosa var. cylindracea. J Hazard Mater 161:1454–1460. doi:10.1016/j.jhazmat.2008.04.125
Bello OS, Ahmad MA, Ahmad N (2012) Adsorptive features of banana (Musa paradisiaca) stalk-based activated carbon for malachite green dye removal. Chem Ecol 28:153–167. doi:10.1080/02757540.2011.628318
Bello OS, Ahmad MA, Semire B (2014) Scavenging malachite green dye from aqueous solutions using pomelo (Citrus grandis) peels: kinetic, equilibrium and thermodynamic studies. Desalin Water Treat 56:521–535. doi:10.1080/19443994.2014.940387
Bello OS, Bello IA, Adegoke KA (2013) Adsorption of dyes using different types of sand: a review. South Afr J Chem 66:117–129
Berberidou C, Poulios I, Xekoukoulotakis NP, Mantzavinos D (2007) Sonolytic, photocatalytic and sonophotocatalytic degradation of malachite green in aqueous solutions. Appl Catal B Environ 74:63–72. doi:10.1016/j.apcatb.2007.01.013
Bhagavathi Pushpa T, Vijayaraghavan J, Sardhar Basha SJ et al (2015) Investigation on removal of malachite green using EM based compost as adsorbent. Ecotoxicol Environ Saf 118:177–182. doi:10.1016/j.ecoenv.2015.04.033
Bhattacharyya KG, Sarma A (2003) Adsorption characteristics of the dye, brilliant green, on neem leaf powder. Dyes Pigments 57:211–222. doi:10.1016/S0143-7208(03)00009-3
Bojinova A, Dushkin C (2011) Photodegradation of malachite green in water solutions by means of thin films of TiO2/WO3 under visible light. React Kinet Mech Catal 103:239–250. doi:10.1007/s11144-011-0295-2
Boutemedjet S, Hamdaoui O (2009) Sorption of malachite green by eucalyptus bark as a non-conventional low-cost biosorbent. Desalin Water Treat 8:201–210. doi:10.5004/dwt.2009.684
Bradder P, Ling SK, Wang S, Liu S (2011) Dye adsorption on layered graphite oxide. J Chem Eng Data 56:138–141. doi:10.1021/je101049g
Breck DW (1984) Zeolite molecular sieves. Krieger, Chicago
Bulut E, Özacar M, Şengil İA (2008) Equilibrium and kinetic data and process design for adsorption of Congo Red onto bentonite. J Hazard Mater 154:613–622. doi:10.1016/j.jhazmat.2007.10.071
Burkinshaw SM (2016) Physico-chemical aspects of textile coloration, 1st edn. Wiley, New York
Buvaneswari N, Kannan C (2011) Plant toxic and non-toxic nature of organic dyes through adsorption mechanism on cellulose surface. J Hazard Mater 189:294–300. doi:10.1016/j.jhazmat.2011.02.036
Buzea C, Pacheco II, Robbie K (2007) Nanomaterials and nanoparticles: sources and toxicity. Biointerphases 2:MR17–MR71. doi:10.1116/1.2815690
Cardon D (2007) Natural dyes: sources, tradition, technology and science. Archetype, London
Chanzu HA, Onyari JM, Shiundu PM (2012) Biosorption of malachite green from aqueous solutions onto polylactide/spent brewery grains films: kinetic and equilibrium studies. J Polym Environ 20:665–672. doi:10.1007/s10924-012-0479-5
Cheknane B, Baudu M, Basly J-P, Bouras O (2010) Adsorption of basic dyes in single and mixture systems on granular inorganic–organic pillared clays. Environ Technol 31:815–822. doi:10.1080/09593331003667758
Chen CC, Lu CS, Chung YC, Jan JL (2007) UV light induced photodegradation of malachite green on TiO2 nanoparticles. J Hazard Mater 141:520–528. doi:10.1016/j.jhazmat.2006.07.011
Chen F, Ma W, He J, Zhao J (2002) Fenton degradation of malachite green catalyzed by aromatic additives. J Phys Chem A 106:9485–9490. doi:10.1021/jp0144350
Chen J, Mao J, Mo X et al (2009) Study of adsorption behavior of malachite green on polyethylene glycol micelles in cloud point extraction procedure. Colloids Surf Physicochem Eng Asp 345:231–236. doi:10.1016/j.colsurfa.2009.05.015
Chen Y, Zhang Y, Liu C et al (2011) Photodegradation of malachite green by nanostructured Bi2WO6 visible light-induced photocatalyst. Int J Photoenergy 2012:e510158. doi:10.1155/2012/510158
Chen Z, Deng H, Chen C et al (2014) Biosorption of malachite green from aqueous solutions by Pleurotus ostreatus using Taguchi method. J Environ Health Sci Eng 12:63. doi:10.1186/2052-336X-12-63
Chen Z-Y, Gao H-W, He Y-Y (2013) Selective photodegradation and backfilling for regeneration of the inorganic–organic hybrid composite Fe3O4@C18ADB@Zn2SiO4 which captures organic pollutants from aqueous solution. RSC Adv 3:5815–5818. doi:10.1039/C3RA22324H
Cheng W, Wang S-G, Lu L et al (2008) Removal of malachite green (MG) from aqueous solutions by native and heat-treated anaerobic granular sludge. Biochem Eng J 39:538–546. doi:10.1016/j.bej.2007.10.016
Cheriaa J, Bakhrouf A (2009) Triphenylmethanes, malachite green and crystal violet dyes decolourisation by Sphingomonas paucimobilis. Ann Microbiol 59:57–61. doi:10.1007/BF03175599
Chieng HI, Lim LBL, Priyantha N (2015) Enhancing adsorption capacity of toxic malachite green dye through chemically modified breadnut peel: equilibrium, thermodynamics, kinetics and regeneration studies. Environ Technol 36:86–97. doi:10.1080/09593330.2014.938124
Chieng HI, Zehra T, Lim LBL et al (2014) Sorption characteristics of peat of Brunei Darussalam IV: equilibrium, thermodynamics and kinetics of adsorption of methylene blue and malachite green dyes from aqueous solution. Environ Earth Sci 72:2263–2277. doi:10.1007/s12665-014-3135-7
Chowdhury S, Chakraborty S, Saha P (2011a) Biosorption of Basic Green 4 from aqueous solution by Ananas comosus (pineapple) leaf powder. Colloids Surf B Biointerfaces 84:520–527. doi:10.1016/j.colsurfb.2011.02.009
Chowdhury S, Das P (2012) Utilization of a domestic waste—eggshells for removal of hazardous malachite green from aqueous solutions. Environ Prog Sustain Energy 31:415–425. doi:10.1002/ep.10564
Chowdhury S, Mishra R, Saha P, Kushwaha P (2011b) Adsorption thermodynamics, kinetics and isosteric heat of adsorption of malachite green onto chemically modified rice husk. Desalination 265:159–168. doi:10.1016/j.desal.2010.07.047
Chowdhury S, Saha P (2010a) Sea shell powder as a new adsorbent to remove Basic Green 4 (malachite green) from aqueous solutions: equilibrium, kinetic and thermodynamic studies. Chem Eng J 164:168–177. doi:10.1016/j.cej.2010.08.050
Chowdhury S, Saha P (2010b) Pseudo-second-order kinetic model for sorption of malachite green onto sea shell: comparison of linear and non-linear methods. IIOAB J 1:3–7
Chowdhury S, Saha P (2011a) Adsorption thermodynamics and kinetics of malachite green onto Ca(OH)2-treated fly ash. J Environ Eng 137:388–397. doi:10.1061/(ASCE)EE.1943-7870.0000334
Chowdhury S, Saha PD (2011) Mechanistic, Kinetic, and Thermodynamic Evaluation of Adsorption of Hazardous Malachite Green onto Conch Shell Powder. Sep Sci Technol 46:1966–1976. doi:10.1080/01496395.2011.584930
Chowdhury S, Saha PD (2012) Fixed-bed adsorption of malachite green onto binary solid mixture of adsorbents: seashells and eggshells. Toxicol Environ Chem 94:1272–1282. doi:10.1080/02772248.2012.703205
Chowdhury S, Saha PD (2013) Adsorption of malachite green from aqueous solution by naoh-modified rice husk: fixed-bed column studies. Environ Prog Sustain Energy 32:633–639. doi:10.1002/ep.11674
Chowdhury S, Saha PD (2011b) Comparative analysis of linear and nonlinear methods of estimating the pseudo-second-order kinetic parameters for sorption of malachite green onto pretreated rice husk. Bioremediat J 15:181–188. doi:10.1080/10889868.2011.624140
Chowdhury S, Saha PD, Ghosh U (2012) Fish (Labeo rohita) scales as potential low-cost biosorbent for removal of malachite green from aqueous solutions. Bioremediat J 16:235–242. doi:10.1080/10889868.2012.731444
Crini G (2006) Non-conventional low-cost adsorbents for dye removal: a review. Bioresour Technol 97:1061–1085. doi:10.1016/j.biortech.2005.05.001
Crini G, Peindy HN, Gimbert F, Robert C (2007) Removal of C.I. Basic Green 4 (malachite green) from aqueous solutions by adsorption using cyclodextrin-based adsorbent: kinetic and equilibrium studies. Sep Purif Technol 53:97–110. doi:10.1016/j.seppur.2006.06.018
da Fonseca MG, de Oliveira MM, Arakaki LNH (2006) Removal of cadmium, zinc, manganese and chromium cations from aqueous solution by a clay mineral. J Hazard Mater 137:288–292. doi:10.1016/j.jhazmat.2006.02.001
Dadhaniya PV, Patel MP, Patel RG (2006) Swelling and dye adsorption study of novel superswelling [acrylamide/N-vinylpyrrolidone/3(2-hydroxyethyl carbamoyl) acrylic acid] hydrogels. Polym Bull 57:21–31. doi:10.1007/s00289-006-0531-5
Dahri MK, Kooh MRR, Lim LBL (2014) Water remediation using low cost adsorbent walnut shell for removal of malachite green: equilibrium, kinetics, thermodynamic and regeneration studies. J Environ Chem Eng 2:1434–1444. doi:10.1016/j.jece.2014.07.008
Daneshvar N, Ayazloo M, Khataee AR, Pourhassan M (2007a) Biological decolorization of dye solution containing malachite green by microalgae Cosmarium sp. Bioresour Technol 98:1176–1182. doi:10.1016/j.biortech.2006.05.025
Daneshvar N, Khataee AR, Rasoulifard MH, Pourhassan M (2007b) Biodegradation of dye solution containing malachite green: optimization of effective parameters using Taguchi method. J Hazard Mater 143:214–219. doi:10.1016/j.jhazmat.2006.09.016
Das A, Pal A, Saha S, Maji SK (2009a) Behaviour of fixed-bed column for the adsorption of malachite green on surfactant-modified alumina. J Environ Sci Health Part A Tox Hazard Subst Environ Eng 44:265–272. doi:10.1080/10934520802597929
Das AK, Saha S, Pal A, Maji SK (2009b) Surfactant-modified alumina: an efficient adsorbent for malachite green removal from water environment. J Environ Sci Health Part A 44:896–905. doi:10.1080/10934520902958708
Dawood S, Sen TK (2014) Review on dye removal from Its aqueous solution into alternative cost effective and non-conventional adsorbents. J Chem Process Eng 1:1–7
Debrassi A, Corrêa AF, Baccarin T et al (2012) Removal of cationic dyes from aqueous solutions using N-benzyl-O-carboxymethylchitosan magnetic nanoparticles. Chem Eng J 183:284–293. doi:10.1016/j.cej.2011.12.078
Demirbas A (2009) Agricultural based activated carbons for the removal of dyes from aqueous solutions: a review. J Hazard Mater 167:1–9. doi:10.1016/j.jhazmat.2008.12.114
Dhodapkar R, Rao NN, Pande SP et al (2007) Adsorption of cationic dyes on Jalshakti®, super absorbent polymer and photocatalytic regeneration of the adsorbent. React Funct Polym 67:540–548. doi:10.1016/j.reactfunctpolym.2007.03.007
Dos Reis LGT, Robaina NF, Pacheco WF, Cassella RJ (2011) Separation of malachite green and methyl green cationic dyes from aqueous medium by adsorption on Amberlite XAD-2 and XAD-4 resins using sodium dodecylsulfate as carrier. Chem Eng J 171:532–540. doi:10.1016/j.cej.2011.04.024
Du L-N, Wang S, Li G et al (2011) Biodegradation of malachite green by Pseudomonas sp. strain DY1 under aerobic condition: characteristics, degradation products, enzyme analysis and phytotoxicity. Ecotoxicology 20:438–446. doi:10.1007/s10646-011-0595-3
Eckenrode HM, Jen S-H, Han J et al (2005) Adsorption of a cationic dye molecule on polystyrene microspheres in colloids: effect of surface charge and composition probed by second harmonic generation. J Phys Chem B 109:4646–4653. doi:10.1021/jp045610q
Eichlerová I, Homolka L, Nerud F (2006) Synthetic dye decolorization capacity of white rot fungus Dichomitus squalens. Bioresour Technol 97:2153–2159. doi:10.1016/j.biortech.2005.09.014
Eskizeybek V, Sarı F, Gülce H et al (2012) Preparation of the new polyaniline/ZnO nanocomposite and its photocatalytic activity for degradation of methylene blue and malachite green dyes under UV and natural sun lights irradiations. Appl Catal B Environ 119–120:197–206. doi:10.1016/j.apcatb.2012.02.034
Farhadi K, Maleki R, Nezhad NM, Samadi N (2010) Spectrophotometric determination of malachite green residue in water samples after preconcentration on surfactant-coated alumina. Spectrosc Lett 43:101–107. doi:10.1080/00387010903278309
Forgacs E, Cserháti T, Oros G (2004) Removal of synthetic dyes from wastewaters: a review. Environ Int 30:953–971. doi:10.1016/j.envint.2004.02.001
Franca AS, Oliveira LS, Nunes AA (2010a) Malachite green adsorption by a residue-based microwave-activated adsorbent. CLEAN—Soil Air Water 38:843–849. doi:10.1002/clen.201000153
Franca AS, Oliveira LS, Saldanha SA et al (2010b) Malachite green adsorption by mango (Mangifera indica L.) seed husks: kinetic, equilibrium and thermodynamic studies. Desalin Water Treat 19:241–248. doi:10.5004/dwt.2010.1105
Fu Y, Viraraghavan T (2001) Fungal decolorization of dye wastewaters: a review. Bioresour Technol 79:251–262. doi:10.1016/S0960-8524(01)00028-1
Gao J-F, Zhang Q, Wang J-H et al (2011) Contributions of functional groups and extracellular polymeric substances on the biosorption of dyes by aerobic granules. Bioresour Technol 102:805–813. doi:10.1016/j.biortech.2010.08.119
Garg VK, Gupta R, Bala Yadav A, Kumar R (2003) Dye removal from aqueous solution by adsorption on treated sawdust. Bioresour Technol 89:121–124. doi:10.1016/S0960-8524(03)00058-0
Garg VK, Kumar R, Gupta R (2004) Removal of malachite green dye from aqueous solution by adsorption using agro-industry waste: a case study of Prosopis cineraria. Dyes Pigments 62:1–10. doi:10.1016/j.dyepig.2003.10.016
Geng Z, Lin Y, Yu X et al (2012) Highly efficient dye adsorption and removal: a functional hybrid of reduced graphene oxide–Fe3O4 nanoparticles as an easily regenerative adsorbent. J Mater Chem 22:3527. doi:10.1039/c2jm15544c
Ghaedi M, Ansari A, Habibi MH, Asghari AR (2014) Removal of malachite green from aqueous solution by zinc oxide nanoparticle loaded on activated carbon: kinetics and isotherm study. J Ind Eng Chem 20:17–28. doi:10.1016/j.jiec.2013.04.031
Ghaedi M, Mosallanejad N (2014) Study of competitive adsorption of malachite green and sunset yellow dyes on cadmium hydroxide nanowires loaded on activated carbon. J Ind Eng Chem 20:1085–1096. doi:10.1016/j.jiec.2013.06.046
Ghaedi M, Shojaeipour E, Ghaedi AM, Sahraei R (2015) Isotherm and kinetics study of malachite green adsorption onto copper nanowires loaded on activated carbon: artificial neural network modeling and genetic algorithm optimization. Spectrochim Acta A Mol Biomol Spectrosc 142:135–149. doi:10.1016/j.saa.2015.01.086
Godbole PT, Sawant AD (2006) Removal of malachite green from aqueous solutions using immobilised Saccharomyces cerevisiae. J Sci Ind Res 65:440–443
Gong R, Feng M, Zhao J et al (2009) Functionalization of sawdust with monosodium glutamate for enhancing its malachite green removal capacity. Bioresour Technol 100:975–978. doi:10.1016/j.biortech.2008.06.031
Gong R, Jin Y, Chen F et al (2006) Enhanced malachite green removal from aqueous solution by citric acid modified rice straw. J Hazard Mater 137:865–870. doi:10.1016/j.jhazmat.2006.03.010
Gong R, Sun J, Zhang D et al (2008) Kinetics and thermodynamics of basic dye sorption on phosphoric acid esterifying soybean hull with solid phase preparation technique. Bioresour Technol 99:4510–4514. doi:10.1016/j.biortech.2007.08.061
Gordon PF, Gregory P (1987) Organic Chemistry in Colour. Springer, Berlin, Heidelberg
Goursot A, Vasilyev V, Arbuznikov A (1997) Modeling of adsorption properties of zeolites: correlation with the structure. J Phys Chem B 101:6420–6428. doi:10.1021/jp971230b
Govindasamy V, Sahadevan R, Subramanian S, Mahendradas DK (2009) Removal of malachite green from aqueous solutions by perlite. Int J Chem React Eng 7:1–22. doi:10.2202/1542-6580.1889
Griffiths J (1976) Colour and constitution of organic molecules. Academic Press, London
Griffiths J (1981) Recent developments in the colour and constitution of organic dyes. Rev Prog Color Relat Top 11:37–57. doi:10.1111/j.1478-4408.1981.tb03714.x
Guo Y, Yang S, Fu W et al (2003a) Adsorption of malachite green on micro- and mesoporous rice husk-based active carbon. Dyes Pigments 56:219–229. doi:10.1016/S0143-7208(02)00160-2
Guo Y, Zhang H, Tao N et al (2003b) Adsorption of malachite green and iodine on rice husk-based porous carbon. Mater Chem Phys 82:107–115. doi:10.1016/S0254-0584(03)00191-3
Gupta N, Kushwaha AK, Chattopadhyaya MC (2014) Application of potato (Solanum tuberosum) plant wastes for the removal of methylene blue and malachite green dye from aqueous solution. Arab J Chem. doi:10.1016/j.arabjc.2011.07.021
Gupta N, Kushwaha AK, Chattopadhyaya MC (2012) Adsorption studies of cationic dyes onto Ashoka (Saraca asoca) leaf powder. J Taiwan Inst Chem Eng 43:604–613. doi:10.1016/j.jtice.2012.01.008
Gupta VK, Jain R, Nayak A et al (2011) Removal of the hazardous dye—tartrazine by photodegradation on titanium dioxide surface. Mater Sci Eng C 31:1062–1067. doi:10.1016/j.msec.2011.03.006
Gupta VK, Kumar R, Nayak A et al (2013) Adsorptive removal of dyes from aqueous solution onto carbon nanotubes: a review. Adv Colloid Interface Sci 193–194:24–34. doi:10.1016/j.cis.2013.03.003
Gupta VK, Mittal A, Krishnan L, Gajbe V (2004) Adsorption kinetics and column operations for the removal and recovery of malachite green from wastewater using bottom ash. Sep Purif Technol 40:87–96. doi:10.1016/j.seppur.2004.01.008
Gupta VK, Nayak A (2012) Cadmium removal and recovery from aqueous solutions by novel adsorbents prepared from orange peel and Fe2O3 nanoparticles. Chem Eng J 180:81–90. doi:10.1016/j.cej.2011.11.006
Gupta VK, Nayak A, Agarwal S (2015) Bioadsorbents for remediation of heavy metals: current status and their future prospects. Environ Eng Res 20:1–18
Gupta VK, Saleh TA (2013) Sorption of pollutants by porous carbon, carbon nanotubes and fullerene—an overview. Environ Sci Pollut Res 20:2828–2843. doi:10.1007/s11356-013-1524-1
Gupta VK, Srivastava SK, Mohan D (1997) Equilibrium uptake, sorption dynamics, process optimization, and column operations for the removal and recovery of malachite green from wastewater using activated carbon and activated slag. Ind Eng Chem Res 36:2207–2218. doi:10.1021/ie960442c
Gupta VK, Suhas (2009) Application of low-cost adsorbents for dye removal—a review. J Environ Manage 90:2313–2342. doi:10.1016/j.jenvman.2008.11.017
Hamdaoui O, Chiha M, Naffrechoux E (2008a) Ultrasound-assisted removal of malachite green from aqueous solution by dead pine needles. Ultrason Sonochem 15:799–807. doi:10.1016/j.ultsonch.2008.01.003
Hamdaoui O, Saoudi F, Chiha M, Naffrechoux E (2008b) Sorption of malachite green by a novel sorbent, dead leaves of plane tree: equilibrium and kinetic modeling. Chem Eng J 143:73–84. doi:10.1016/j.cej.2007.12.018
Hameed BH, El-Khaiary MI (2008a) Equilibrium, kinetics and mechanism of malachite green adsorption on activated carbon prepared from bamboo by K2CO3 activation and subsequent gasification with CO2. J Hazard Mater 157:344–351. doi:10.1016/j.jhazmat.2007.12.105
Hameed BH, El-Khaiary MI (2008b) Batch removal of malachite green from aqueous solutions by adsorption on oil palm trunk fibre: equilibrium isotherms and kinetic studies. J Hazard Mater 154:237–244. doi:10.1016/j.jhazmat.2007.10.017
Hameed BH, El-Khaiary MI (2008c) Kinetics and equilibrium studies of malachite green adsorption on rice straw-derived char. J Hazard Mater 153:701–708. doi:10.1016/j.jhazmat.2007.09.019
Hameed BH, El-Khaiary MI (2008d) Malachite green adsorption by rattan sawdust: isotherm, kinetic and mechanism modeling. J Hazard Mater 159:574–579. doi:10.1016/j.jhazmat.2008.02.054
Hammud HH, Shmait A, Hourani N (2015) Removal of malachite green from water using hydrothermally carbonized pine needles. RSC Adv 5:7909–7920. doi:10.1039/C4RA15505J
Han R, Wang Y, Sun Q et al (2010) Malachite green adsorption onto natural zeolite and reuse by microwave irradiation. J Hazard Mater 175:1056–1061. doi:10.1016/j.jhazmat.2009.10.118
Hasnat MA, Siddiquey IA, Saiful IS (2003) Photodegradation of malachite green in the aqueous medium. Indian J Chem Sect A 42:1865–1867
Hema M, Arivoli S (2007) Comparative study on the adsorption kinetics and thermodynamics of dyes onto acid activated low cost carbon. Int J Phys Sci 2:10–17
Heydartaemeh MR, Ardejani FD, Badii K et al (2014) FeCl2/FeCl3 perlite nanoparticles as a novel magnetic material for adsorption of green malachite dye. Arab J Sci Eng 39:3383–3392. doi:10.1007/s13369-014-0978-x
Hu R, Huang J-B, Yang Z-P et al (2011) Biosorption of crystal violet and malachite green by Rhodotorula graminis Y-5. Ying Yong Sheng Tai Xue Bao J Appl Ecol Zhongguo Sheng Tai Xue Xue Hui Zhongguo Ke Xue Yuan Shenyang Ying Yong Sheng Tai Yan Jiu Suo Zhu Ban 22:3293–3299
Huang C-M, Chen L-C, Yang H-C et al (2012) Preparation of acrylic acid-modified chitin improved by an experimental design and its application in absorbing toxic organic compounds. J Hazard Mater 241–242:190–196. doi:10.1016/j.jhazmat.2012.09.032
Huang L, Weng X, Chen Z et al (2014) Green synthesis of iron nanoparticles by various tea extracts: comparative study of the reactivity. Spectrochim Acta A Mol Biomol Spectrosc 130:295–301. doi:10.1016/j.saa.2014.04.037
Huo S-H, Yan X-P (2012) Metal–organic framework MIL-100(Fe) for the adsorption of malachite green from aqueous solution. J Mater Chem 22:7449. doi:10.1039/c2jm16513a
Hussain F, Hojjati M, Okamoto M, Gorga RE (2006) Review article: polymer-matrix nanocomposites, processing, manufacturing, and application: an overview. J Compos Mater 40:1511–1575. doi:10.1177/0021998306067321
Idris MN, Ahmad ZA, Ahmad MA (2011) Adsorption equilibrium of malachite green dye onto rubber seed coat based activated carbon. Int J Basic Appl Sci 11:32–37
Iijima S (1991) Helical microtubules of graphitic carbon. Nature 354:56–58
Inbaraj BS, Sulochana N (2002) Basic dye adsorption on a low cost carbonaceous sorbent—kinetic and equilibrium studies. Indian J Chem Technol 9:201–208
Iqbal MJ, Ashiq MN (2007) Adsorption of dyes from aqueous solutions on activated charcoal. J Hazard Mater 139:57–66. doi:10.1016/j.jhazmat.2006.06.007
Issa AA, Al-Degs YS, Al-Ghouti MA, Olimat AAM (2014) Studying competitive sorption behavior of methylene blue and malachite green using multivariate calibration. Chem Eng J 240:554–564. doi:10.1016/j.cej.2013.10.084
Jalil AA, Triwahyono S, Yaakob MR et al (2012) Utilization of bivalve shell-treated Zea mays L. (maize) husk leaf as a low-cost biosorbent for enhanced adsorption of malachite green. Bioresour Technol 120:218–224. doi:10.1016/j.biortech.2012.06.066
Janoš P (2003) Sorption of basic dyes onto iron humate. Environ Sci Technol 37:5792–5798. doi:10.1021/es020142o
Janoš P, Michálek P, Turek L (2007) Sorption of ionic dyes onto untreated low-rank coal–oxihumolite: a kinetic study. Dyes Pigments 74:363–370. doi:10.1016/j.dyepig.2006.02.017
Janos P, Sedivý P, Rýznarová M, Grötschelová S (2005) Sorption of basic and acid dyes from aqueous solutions onto oxihumolite. Chemosphere 59:881–886. doi:10.1016/j.chemosphere.2004.11.018
Janoš P, Šmídová V (2005) Effects of surfactants on the adsorptive removal of basic dyes from water using an organomineral sorbent—iron humate. J Colloid Interface Sci 291:19–27. doi:10.1016/j.jcis.2005.04.065
Jasińska A, Bernat P, Paraszkiewicz K (2013) Malachite green removal from aqueous solution using the system rapeseed press cake and fungus Myrothecium roridum. Desalin Water Treat 51:7663–7671. doi:10.1080/19443994.2013.779939
Jayaraj R, Mohan MC, Prasath PMD, Khan TH (2011) Malachite green dye removal using the seaweed enteromorpha. J Chem 8:649–656. doi:10.1155/2011/141305
Jeon YS, Lei J, Kim J-H (2008) Dye adsorption characteristics of alginate/polyaspartate hydrogels. J Ind Eng Chem 14:726–731. doi:10.1016/j.jiec.2008.07.007
Jeyagowri B, Yamuna RT (2015) Potential efficacy of a mesoporous biosorbent Simarouba glauca seed shell powder for the removal of malachite green from aqueous solutions. Desalin Water Treat 57:11326–11336. doi:10.1080/19443994.2015.1042060
Ju Y, Yang S, Ding Y et al (2008) Microwave-assisted rapid photocatalytic degradation of malachite green in TiO2 suspensions: mechanism and pathways. J Phys Chem A 112:11172–11177. doi:10.1021/jp804439z
Kadirvelu K, Kavipriya M, Karthika C et al (2003) Utilization of various agricultural wastes for activated carbon preparation and application for the removal of dyes and metal ions from aqueous solutions. Bioresour Technol 87:129–132. doi:10.1016/S0960-8524(02)00201-8
Kan Y, Yue Q, Kong J et al (2015) The application of activated carbon produced from waste printed circuit boards (PCBs) by H3PO4 and steam activation for the removal of malachite green. Chem Eng J 260:541–549. doi:10.1016/j.cej.2014.09.047
Kaneva N, Stambolova I, Blaskov V et al (2010) Photocatalytic activity of nanostructured ZnO films prepared by two different methods for the photoinitiated decolorization of malachite green. J Alloys Compd 500:252–258. doi:10.1016/j.jallcom.2010.04.020
Kannan C, Muthuraja K, Devi MR (2013) Hazardous dyes removal from aqueous solution over mesoporous aluminophosphate with textural porosity by adsorption. J Hazard Mater 244–245:10–20. doi:10.1016/j.jhazmat.2012.11.016
Kannan C, Sundaram T, Palvannan T (2008) Environmentally stable adsorbent of tetrahedral silica and non-tetrahedral alumina for removal and recovery of malachite green dye from aqueous solution. J Hazard Mater 157:137–145. doi:10.1016/j.jhazmat.2007.12.116
Kannan RR, Rajasimman M, Rajamohan N, Sivaprakash B (2009) Brown marine algae Turbinaria conoides as biosorbent for malachite green removal: equilibrium and kinetic modeling. Front Environ Sci Eng China 4:116–122. doi:10.1007/s11783-010-0006-7
Kant S, Pathania D, Singh P et al (2014) Removal of malachite green and methylene blue by Fe0.01Ni0.01Zn0.98O/polyacrylamide nanocomposite using coupled adsorption and photocatalysis. Appl Catal B Environ 147:340–352. doi:10.1016/j.apcatb.2013.09.001
Karge HG, Weitkamp J (1989) Zeolites as catalysts, sorbents and detergent builders: applications and innovations. Elsevier, Amsterdam
Karimi A, Aghbolaghy M, Khataee A, Shoa Bargh S (2012) Use of enzymatic bio-fenton as a new approach in decolorization of malachite green. Sci World J. doi:10.1100/2012/691569
Kattri SD, Singh MK (1999) Adsorption of basic dyes from aqueous solution by natural adsorbent. Indian J Chem Technol 6:112–116
Khan AA, Ahmad R, Khan A, Mondal PK (2013) Preparation of unsaturated polyester Ce(IV) phosphate by plastic waste bottles and its application for removal of Malachite green dye from water samples. Arab J Chem 6:361–368. doi:10.1016/j.arabjc.2010.10.012
Khan TA, Rahman R, Ali I et al (2014) Removal of malachite green from aqueous solution using waste pea shells as low-cost adsorbent—adsorption isotherms and dynamics. Toxicol Environ Chem 96:569–578. doi:10.1080/02772248.2014.969268
Khani H, Rofouei MK, Arab P et al (2010) Multi-walled carbon nanotubes-ionic liquid-carbon paste electrode as a super selectivity sensor: application to potentiometric monitoring of mercury ion(II). J Hazard Mater 183:402–409. doi:10.1016/j.jhazmat.2010.07.039
Khataee AR, Dehghan G, Zarei M et al (2011a) Neural network modeling of biotreatment of triphenylmethane dye solution by a green macroalgae. Chem Eng Res Des 89:172–178. doi:10.1016/j.cherd.2010.05.009
Khataee AR, Zarei M, Dehghan G et al (2011b) Biotreatment of a triphenylmethane dye solution using a Xanthophyta alga: modeling of key factors by neural network. J Taiwan Inst Chem Eng 42:380–386. doi:10.1016/j.jtice.2010.08.006
Khattri S, Singh M (1999) Colour removal from dye wastewater using sugar cane dust as an adsorbent. Adsorpt Sci Technol 17:269–282
Khattri SD, Singh MK (2009) Removal of malachite green from dye wastewater using neem sawdust by adsorption. J Hazard Mater 167:1089–1094. doi:10.1016/j.jhazmat.2009.01.101
Khattri SD, Singh MK (2000) Colour removal from synthetic dye wastewater using a bioadsorbent. Water Air Soil Pollut 120:283–294. doi:10.1023/A:1005207803041
Kiani G, Dostali M, Rostami A, Khataee AR (2011) Adsorption studies on the removal of malachite green from aqueous solutions onto halloysite nanotubes. Appl Clay Sci 54:34–39. doi:10.1016/j.clay.2011.07.008
Kominami H, Kumamoto H, Kera Y, Ohtani B (2003) Photocatalytic decolorization and mineralization of malachite green in an aqueous suspension of titanium(IV) oxide nano-particles under aerated conditions: correlation between some physical properties and their photocatalytic activity. J Photochem Photobiol Chem 160:99–104. doi:10.1016/S1010-6030(03)00227-2
Krauskopf KB (1956) Factors controlling the concentrations of thirteen rare metals in sea-water. Geochim Cosmochim Acta 9:1-B32. doi:10.1016/0016-7037(56)90055-2
Kuhar F, Papinutti L (2013) Protective effect of vanilloids against chemical stress on the white-rot fungus Ganoderma lucidum. J Environ Manage 124:1–7. doi:10.1016/j.jenvman.2013.03.040
Kumar KV (2006) Comparative analysis of linear and non-linear method of estimating the sorption isotherm parameters for malachite green onto activated carbon. J Hazard Mater 136:197–202. doi:10.1016/j.jhazmat.2005.09.018
Kumar KV (2007) Optimum sorption isotherm by linear and non-linear methods for malachite green onto lemon peel. Dyes Pigments 74:595–597. doi:10.1016/j.dyepig.2006.03.026
Kumar KV, Porkodi K (2007) Batch adsorber design for different solution volume/adsorbent mass ratios using the experimental equilibrium data with fixed solution volume/adsorbent mass ratio of malachite green onto orange peel. Dyes Pigments 74:590–594. doi:10.1016/j.dyepig.2006.03.024
Kumar KV, Sivanesan S (2006) Pseudo second order kinetics and pseudo isotherms for malachite green onto activated carbon: comparison of linear and non-linear regression methods. J Hazard Mater 136:721–726. doi:10.1016/j.jhazmat.2006.01.003
Kumar KV, Sivanesan S, Ramamurthi V (2005) Adsorption of malachite green onto Pithophora sp., a fresh water algae: equilibrium and kinetic modelling. Process Biochem 40:2865–2872. doi:10.1016/j.procbio.2005.01.007
Kumar KY, Muralidhara HB, Nayaka YA et al (2013) Low-cost synthesis of metal oxide nanoparticles and their application in adsorption of commercial dye and heavy metal ion in aqueous solution. Powder Technol 246:125–136. doi:10.1016/j.powtec.2013.05.017
Kurniawan A, Sutiono H, Indraswati N, Ismadji S (2012) Removal of basic dyes in binary system by adsorption using rarasaponin–bentonite: revisited of extended Langmuir model. Chem Eng J 189–190:264–274. doi:10.1016/j.cej.2012.02.070
Kushwaha AK, Gupta N, Chattopadhyaya MC (2014) Removal of cationic methylene blue and malachite green dyes from aqueous solution by waste materials of Daucus carota. J Saudi Chem Soc 18:200–207. doi:10.1016/j.jscs.2011.06.011
Lata H, Gupta RK, Garg VK (2008) Removal of basic dye from aqueous solution using chemically modified parthenium hysterophorus linn. Biomass Chem Eng Commun 195:1185–1199. doi:10.1080/00986440701691103
Lee Y-C, Kim EJ, Yang J-W, Shin H-J (2011) Removal of malachite green by adsorption and precipitation using aminopropyl functionalized magnesium phyllosilicate. J Hazard Mater 192:62–70. doi:10.1016/j.jhazmat.2011.04.094
Lee Y-C, Kim J-Y, Shin H-J (2013) Removal of malachite green (MG) from aqueous solutions by adsorption, precipitation, and alkaline fading using talc. Sep Sci Technol 48:1093–1101. doi:10.1080/01496395.2012.723100
Levin L, Papinutti L, Forchiassin F (2004) Evaluation of Argentinean white rot fungi for their ability to produce lignin-modifying enzymes and decolorize industrial dyes. Bioresour Technol 94:169–176. doi:10.1016/j.biortech.2003.12.002
Li X, Li Y, Zhang S, Ye Z (2012) Preparation and characterization of new foam adsorbents of poly(vinyl alcohol)/chitosan composites and their removal for dye and heavy metal from aqueous solution. Chem Eng J 183:88–97. doi:10.1016/j.cej.2011.12.025
Lin K-YA, Chang H-A (2015) Ultra-high adsorption capacity of zeolitic imidazole framework-67 (ZIF-67) for removal of malachite green from water. Chemosphere. doi:10.1016/j.chemosphere.2015.01.041
Liu Y, Ohko Y, Zhang R et al (2010) Degradation of malachite green on Pd/WO3 photocatalysts under simulated solar light. J Hazard Mater 184:386–391. doi:10.1016/j.jhazmat.2010.08.047
Low LW, Teng TT, Rafatullah M et al (2013) Adsorption studies of methylene blue and malachite green from aqueous solutions by pretreated lignocellulosic materials. Sep Sci Technol 48:1688–1698. doi:10.1080/01496395.2012.756912
Lv X, Xu Y, Lv K, Zhang G (2005) Photo-assisted degradation of anionic and cationic dyes over iron(III)-loaded resin in the presence of hydrogen peroxide. J Photochem Photobiol Chem 173:121–127. doi:10.1016/j.jphotochem.2005.01.011
Mahto TK, Chowdhuri AR, Sahu SK (2014) Polyaniline-functionalized magnetic nanoparticles for the removal of toxic dye from wastewater. J Appl Polym Sci 131:1–9. doi:10.1002/app.40840
Makeswari M, Santhi T (2013) Removal of malachite green dye from aqueous solutions onto microwave assisted zinc chloride chemical activated epicarp of Ricinus communis. J Water Resour Prot 5:222–238. doi:10.4236/jwarp.2013.52023
Malana MA, Ijaz S, Ashiq MN (2010) Removal of various dyes from aqueous media onto polymeric gels by adsorption process: their kinetics and thermodynamics. Desalination 263:249–257. doi:10.1016/j.desal.2010.06.066
Malik R, Ramteke DS, Wate SR (2007) Adsorption of malachite green on groundnut shell waste based powdered activated carbon. Waste Manag 27:1129–1138. doi:10.1016/j.wasman.2006.06.009
Mall ID, Srivastava VC, Agarwal NK, Mishra IM (2005) Adsorptive removal of malachite green dye from aqueous solution by bagasse fly ash and activated carbon-kinetic study and equilibrium isotherm analyses. Colloids Surf Physicochem Eng Asp 264:17–28. doi:10.1016/j.colsurfa.2005.03.027
Man LW, Kumar P, Teng TT, Wasewar KL (2012) Design of experiments for malachite green dye removal from wastewater using thermolysis—coagulation–flocculation. Desalin Water Treat 40:260–271. doi:10.1080/19443994.2012.671257
Mane VS, Babu PVV (2011) Studies on the adsorption of brilliant green dye from aqueous solution onto low-cost NaOH treated saw dust. Desalination 273:321–329. doi:10.1016/j.desal.2011.01.049
Mansa RF, Sipaut CS, Rahman IA et al (2016) Preparation of glycine–modified silica nanoparticles for the adsorption of malachite green dye. J Porous Mater 23:35–46. doi:10.1007/s10934-015-0053-3
Méndez A, Fernández F, Gascó G (2007) Removal of malachite green using carbon-based adsorbents. Desalination 206:147–153. doi:10.1016/j.desal.2006.03.564
Meng L, Zhang X, Tang Y et al (2015) Hierarchically porous silicon-carbon-nitrogen hybrid materials towards highly efficient and selective adsorption of organic dyes. Sci Rep 5:7910. doi:10.1038/srep07910
Mirzajani R, Ahmadi S (2015) Melamine supported magnetic iron oxide nanoparticles (Fe3O4@Mel) for spectrophotometric determination of malachite green in water samples and fish tissues. J Ind Eng Chem 23:171–178. doi:10.1016/j.jiec.2014.08.011
Mishra RR, Chandran P, Khan SS (2014) Equilibrium and kinetic studies on adsorptive removal of malachite green by the citrate-stabilized magnetite nanoparticles. RSC Adv 4:51787–51793. doi:10.1039/C4RA07651F
Mittal A (2006) Adsorption kinetics of removal of a toxic dye, malachite green, from wastewater by using hen feathers. J Hazard Mater 133:196–202. doi:10.1016/j.jhazmat.2005.10.017
Mittal A, Krishnan L, Gupta VK (2005) Removal and recovery of malachite green from wastewater using an agricultural waste material, de-oiled soya. Sep Purif Technol 43:125–133. doi:10.1016/j.seppur.2004.10.010
Mittal A, Mittal J, Malviya A, Gupta VK (2009) Adsorptive removal of hazardous anionic dye “Congo Red” from wastewater using waste materials and recovery by desorption. J Colloid Interface Sci 340:16–26. doi:10.1016/j.jcis.2009.08.019
Modirshahla N, Behnajady MA (2006) Photooxidative degradation of malachite green (MG) by UV/H2O2: influence of operational parameters and kinetic modeling. Dyes Pigments 70:54–59. doi:10.1016/j.dyepig.2005.04.012
Mohammadi A, Daemi H, Barikani M (2014) Fast removal of malachite green dye using novel superparamagnetic sodium alginate-coated Fe3O4 nanoparticles. Int J Biol Macromol 69:447–455. doi:10.1016/j.ijbiomac.2014.05.042
Mondal S (2008) Methods of dye removal from dye house effluent—an overview. Environ Eng Sci 25:383–396. doi:10.1089/ees.2007.0049
Moumeni O, Hamdaoui O (2012) Intensification of sonochemical degradation of malachite green by bromide ions. Ultrason Sonochem 19:404–409. doi:10.1016/j.ultsonch.2011.08.008
Nandi BK, Goswami A, Purkait MK (2009) Adsorption characteristics of brilliant green dye on kaolin. J Hazard Mater 161:387–395. doi:10.1016/j.jhazmat.2008.03.110
Nethaji S, Sivasamy A, Kumar RV, Mandal AB (2013) Preparation of char from lotus seed biomass and the exploration of its dye removal capacity through batch and column adsorption studies. Environ Sci Pollut Res Int 20:3670–3678. doi:10.1007/s11356-012-1267-4
Nethaji S, Sivasamy A, Mandal AB (2012) Adsorption isotherms, kinetics and mechanism for the adsorption of cationic and anionic dyes onto carbonaceous particles prepared from Juglans regia shell biomass. Int J Environ Sci Technol 10:231–242. doi:10.1007/s13762-012-0112-0
Nethaji S, Sivasamy A, Thennarasu G, Saravanan S (2010) Adsorption of malachite green dye onto activated carbon derived from Borassus aethiopum flower biomass. J Hazard Mater 181:271–280. doi:10.1016/j.jhazmat.2010.05.008
Ng HW, Lee LY, Chan WL et al (2015) Luffa acutangula peel as an effective natural biosorbent for malachite green removal in aqueous media: equilibrium, kinetic and thermodynamic investigations. Desalin Water Treat 57:7302–7311. doi:10.1080/19443994.2015.1016460
Nidheesh PV, Gandhimathi R, Ramesh ST (2013) Degradation of dyes from aqueous solution by Fenton processes: a review. Environ Sci Pollut Res 20:2099–2132. doi:10.1007/s11356-012-1385-z
Odom TW, Huang J-L, Kim P, Lieber CM (1998) Atomic structure and electronic properties of single-walled carbon nanotubes. Nature 391:62–64. doi:10.1038/34145
Oladoja NA, Aliu YD (2009) Snail shell as coagulant aid in the alum precipitation of malachite green from aqua system. J Hazard Mater 164:1496–1502. doi:10.1016/j.jhazmat.2008.09.114
Onal Y (2006) Kinetics of adsorption of dyes from aqueous solution using activated carbon prepared from waste apricot. J Hazard Mater 137:1719–1728. doi:10.1016/j.jhazmat.2006.05.036
Önal Y, Akmil-Başar C, Eren D et al (2006) Adsorption kinetics of malachite green onto activated carbon prepared from Tunçbilek lignite. J Hazard Mater 128:150–157. doi:10.1016/j.jhazmat.2005.07.055
Önal Y, Akmil-Başar C, Sarıcı-Özdemir Ç (2007) Investigation kinetics mechanisms of adsorption malachite green onto activated carbon. J Hazard Mater 146:194–203. doi:10.1016/j.jhazmat.2006.12.006
Özdemir A, Keskin CS (2009) Removal of a binary dye mixture of congo red and malachite green from aqueous solutions using a bentonite adsorbent. Clays Clay Miner 57:695–705. doi:10.1346/CCMN.2009.0570603
Ozdes D, Gundogdu A, Duran C, Senturk HB (2010) Evaluation of adsorption characteristics of malachite green onto Almond Shell (Prunus dulcis). Sep Sci Technol 45:2076–2085. doi:10.1080/01496395.2010.504479
Pan X, Zhang D (2009) Removal of malachite green from water by Firmiana simplex wood fiber. Electron J Biotechnol 12:9–10
Pandit P, Basu S (2004) Removal of ionic dyes from water by solvent extraction using reverse micelles. Environ Sci Technol 38:2435–2442
Papinutti L, Mouso N, Forchiassin F (2006) Removal and degradation of the fungicide dye malachite green from aqueous solution using the system wheat bran–Fomes sclerodermeus. Enzyme Microb Technol 39:848–853. doi:10.1016/j.enzmictec.2006.01.013
Parida SK, Dash S, Patel S, Mishra BK (2006) Adsorption of organic molecules on silica surface. Adv Colloid Interface Sci 121:77–110. doi:10.1016/j.cis.2006.05.028
Patil MR, Shrivastava VS (2014) Adsorption of malachite green by polyaniline–nickel ferrite magnetic nanocomposite: an isotherm and kinetic study. Appl Nanosci 5:809–816. doi:10.1007/s13204-014-0383-5
Paul M, Pal N, Bhaumik A (2012) Selective adsorption and release of cationic organic dye molecules on mesoporous borosilicates. Mater Sci Eng C Mater Biol Appl 32:1461–1468. doi:10.1016/j.msec.2012.04.026
Pearce CI, Lloyd JR, Guthrie JT (2003) The removal of colour from textile wastewater using whole bacterial cells: a review. Dyes Pigments 58:179–196. doi:10.1016/S0143-7208(03)00064-0
Pehlivan E, Arslan G (2007) Removal of metal ions using lignite in aqueous solution—low cost biosorbents. Fuel Process Technol 88:99–106. doi:10.1016/j.fuproc.2006.09.004
Pirsaheb M, Shahmoradi B, Khosravi T et al (2015) Solar degradation of malachite green using nickel-doped TiO2 nanocatalysts. Desalin Water Treat 57:9881–9888. doi:10.1080/19443994.2015.1033764
Podstawczyk D, Witek-Krowiak A, Chojnacka K, Sadowski Z (2014) Biosorption of malachite green by eggshells: mechanism identification and process optimization. Bioresour Technol 160:161–165. doi:10.1016/j.biortech.2014.01.015
Porkodi K, Vasanth Kumar K (2007) Equilibrium, kinetics and mechanism modeling and simulation of basic and acid dyes sorption onto jute fiber carbon: eosin yellow, malachite green and crystal violet single component systems. J Hazard Mater 143:311–327. doi:10.1016/j.jhazmat.2006.09.029
Pourjavadi A, Doulabi M, Doroudian M (2014) Adsorption characteristics of malachite green dye onto novel kappa-carrageenan-g-polyacrylic acid/TiO2–NH2 hydrogel nanocomposite. J Iran Chem Soc 11:1057–1065. doi:10.1007/s13738-013-0374-6
Pradeep Sekhar C, Kalidhasan S, Rajesh V, Rajesh N (2009) Bio-polymer adsorbent for the removal of malachite green from aqueous solution. Chemosphere 77:842–847. doi:10.1016/j.chemosphere.2009.07.068
Pradhan AC, Parida KM, Nanda B (2011) Enhanced photocatalytic and adsorptive degradation of organic dyes by mesoporous Cu/Al2O3-MCM-41: intra-particle mesoporosity, electron transfer and OH radical generation under visible light. Dalton Trans Camb Engl 40:7348–7356. doi:10.1039/c1dt10363f
Prado AGS, Costa LL (2009) Photocatalytic decouloration of malachite green dye by application of TiO2 nanotubes. J Hazard Mater 169:297–301. doi:10.1016/j.jhazmat.2009.03.076
Rahman IA, Saad B, Shaidan S, Sya Rizal ES (2005) Adsorption characteristics of malachite green on activated carbon derived from rice husks produced by chemical–thermal process. Bioresour Technol 96:1578–1583. doi:10.1016/j.biortech.2004.12.015
Rajabi HR, Khani O, Shamsipur M, Vatanpour V (2013) High-performance pure and Fe3+-ion doped ZnS quantum dots as green nanophotocatalysts for the removal of malachite green under UV-light irradiation. J Hazard Mater 250–251:370–378. doi:10.1016/j.jhazmat.2013.02.007
Rajabi M, Mirza B, Mahanpoor K et al (2016) Adsorption of malachite green from aqueous solution by carboxylate group functionalized multi-walled carbon nanotubes: determination of equilibrium and kinetics parameters. J Ind Eng Chem 34:130–138. doi:10.1016/j.jiec.2015.11.001
Rajesh Kannan R, Rajasimman M, Rajamohan N, Sivaprakash B (2010) Equilibrium and kinetic studies on sorption of malachite green using hydrilla verticillata biomass. Int J Environ Res 4:817–824
Rajeshkannan R, Rajamohan N, Rajasimman M (2009) Removal of malachite green from aqueous solution by sorption on hydrilla verticillata biomass using response surface methodology. Front Chem Eng China 3:146–154. doi:10.1007/s11705-009-0007-x
Rajeshkannan R, Rajasimman M, Rajamohan N (2011) Decolourization of malachite green using tamarind seed: optimization, isotherm and kinetic studies. Chem Ind Chem Eng Quart 17:67–79. doi:10.2298/CICEQ100716056R
Rajeshkannan R, Rajasimman M, Rajamohan N (2010) Removal of malachite green from aqueous solution using hydrilla verticillata-optimization, equilibrium and kinetic studies. Int J Civ Environ Eng 2:222–229
Rajeshwarisivaraj Sivakumar S, Senthilkumar P, Subburam V (2001) Carbon from Cassava peel, an agricultural waste, as an adsorbent in the removal of dyes and metal ions from aqueous solution. Bioresour Technol 80:233–235. doi:10.1016/S0960-8524(00)00179-6
Rajeshwarisivaraj, Subburam V (2002) Activated parthenium carbon as an adsorbent for the removal of dyes and heavy metal ions from aqueous solution. Bioresour Technol 85:205–206. doi:10.1016/S0960-8524(02)00089-5
Rajgopal S, Karthikeyan T, Prakash Kumar BG, Miranda LR (2006) Utilization of fluidized bed reactor for the production of adsorbents in removal of malachite green. Chem Eng J 116:211–217. doi:10.1016/j.cej.2005.09.026
Ramaraju B, Manoj Kumar Reddy P, Subrahmanyam C (2014) Low cost adsorbents from agricultural waste for removal of dyes. Environ Prog Sustain Energy 33:38–46. doi:10.1002/ep.11742
Rauf MA, Bukallah SB, Hamour FA, Nasir AS (2008) Adsorption of dyes from aqueous solutions onto sand and their kinetic behavior. Chem Eng J 137:238–243. doi:10.1016/j.cej.2007.04.025
Raval NP, Shah PU, Ladha DG et al (2016a) Comparative study of chitin and chitosan beads for the adsorption of hazardous anionic azo dye Congo Red from wastewater. Desalin Water Treat 57:9247–9262. doi:10.1080/19443994.2015.1027959
Raval NP, Shah PU, Shah NK (2016b) Nanoparticles loaded biopolymer as effective adsorbent for adsorptive removal of malachite green from aqueous solution. Water Conserv Sci Eng 1:69–81. doi:10.1007/s41101-016-0004-0
Raval NP, Shah PU, Shah NK (2016c) Adsorptive amputation of hazardous azo dye Congo red from wastewater: a critical review. Environ Sci Pollut Res 23:14810–14853. doi:10.1007/s11356-016-6970-0
Raval NP, Shah PU, Shah NK (2016d) Adsorptive removal of nickel(II) ions from aqueous environment: a review. J Environ Manag 179:1–20. doi:10.1016/j.jenvman.2016.04.045
Ren S, Guo J, Zeng G, Sun G (2006) Decolorization of triphenylmethane, azo, and anthraquinone dyes by a newly isolated Aeromonas hydrophila strain. Appl Microbiol Biotechnol 72:1316–1321. doi:10.1007/s00253-006-0418-2
Robaina NF, Soriano S, Cassella RJ (2009) Polyurethane foam loaded with SDS for the adsorption of cationic dyes from aqueous medium: multivariate optimization of the loading process. J Hazard Mater 167:653–659. doi:10.1016/j.jhazmat.2009.01.033
Robinson T, McMullan G, Marchant R, Nigam P (2001) Remediation of dyes in textile effluent: a critical review on current treatment technologies with a proposed alternative. Bioresour Technol 77:247–255. doi:10.1016/S0960-8524(00)00080-8
Rogaieh Z, Gharbani P (2013) Study on malachite green dye removal by adsorption onto activated nano-alumina. Appl Chem Environ 3:71–77
Rong X, Qiu F, Qin J et al (2014) Removal of malachite green from the contaminated water using a water-soluble melamine/maleic anhydride sorbent. J Ind Eng Chem 20:3808–3814. doi:10.1016/j.jiec.2013.12.083
Roosta M, Ghaedi M, Shokri N et al (2014) Optimization of the combined ultrasonic assisted/adsorption method for the removal of malachite green by gold nanoparticles loaded on activated carbon: experimental design. Spectrochim Acta A Mol Biomol Spectrosc 118:55–65. doi:10.1016/j.saa.2013.08.082
Safarik I, Lunackova P, Mosiniewicz-Szablewska E et al (2007) Adsorption of water-soluble organic dyes on ferrofluid-modified sawdust. Holzforschung 61:247–253. doi:10.1515/HF.2007.060
Šafařík I, Šafaříková M, Vrchotová N (1995) Study of sorption of triphenylmethane dyes on a magnetic carrier bearing an immobilized copper phthalocyanine dye. Collect Czechoslov Chem Commun 60:34–42. doi:10.1135/cccc19950034
Šafařík I, Šafaříková M (2002) Detection of low concentrations of malachite green and crystal violet in water. Water Res 36:196–200. doi:10.1016/S0043-1354(01)00243-3
Saha P, Chowdhury S, Gupta S et al (2010a) Assessment on the removal of malachite green using tamarind fruit shell as biosorbent. CLEAN—Soil Air Water 38:437–445. doi:10.1002/clen.200900234
Saha P, Chowdhury S, Gupta S, Kumar I (2010b) Insight into adsorption equilibrium, kinetics and thermodynamics of malachite green onto clayey soil of Indian origin. Chem Eng J 165:874–882. doi:10.1016/j.cej.2010.10.048
Saha S, Pal A (2014) Microporous assembly of MnO2 nanosheets for malachite green degradation. Sep Purif Technol 134:26–36. doi:10.1016/j.seppur.2014.07.021
Saha S, Wang JM, Pal A (2012) Nano silver impregnation on commercial TiO2 and a comparative photocatalytic account to degrade malachite green. Sep Purif Technol 89:147–159. doi:10.1016/j.seppur.2012.01.012
Saleh TA, Gupta VK (2011) Functionalization of tungsten oxide into MWCNT and its application for sunlight-induced degradation of rhodamine B. J Colloid Interface Sci 362:337–344. doi:10.1016/j.jcis.2011.06.081
Salima A, Benaouda B, Noureddine B, Duclaux L (2013) Application of Ulva lactuca and Systoceira stricta algae-based activated carbons to hazardous cationic dyes removal from industrial effluents. Water Res 47:3375–3388. doi:10.1016/j.watres.2013.03.038
Salleh MAM, Mahmoud DK, Karim WAWA, Idris A (2011) Cationic and anionic dye adsorption by agricultural solid wastes: a comprehensive review. Desalination 280:1–13. doi:10.1016/j.desal.2011.07.019
Samiey B, Toosi AR (2010) Adsorption of malachite green on silica gel: effects of NaCl, pH and 2-propanol. J Hazard Mater 184:739–745. doi:10.1016/j.jhazmat.2010.08.101
Sánchez PCM, Pariente JP (2011) Zeolites and ordered porous solids: fundamentals and applications. Editorial Universitat Politècnica de València, Valencia
Sand LB, Mumpton FA (1978) Natural zeolites: occurrence, properties, and use. Pergamon Press Inc, Elmsford
Santhi T, Manonmani S (2011) Malachite green removal from aqueous solution by the peel of cucumis sativa fruit. CLEAN—Soil Air Water 39:162–170. doi:10.1002/clen.201000077
Santhi T, Manonmani S, Smitha T (2010a) Kinetics and isotherm studies on cationic dyes adsorption onto annona squmosa seed activated carbon. Int J Eng Sci Technol 2:287–295
Santhi T, Manonmani S, Smitha T (2010b) Removal of malachite green from aqueous solution by activated carbon prepared from the epicarp of Ricinus communis by adsorption. J Hazard Mater 179:178–186. doi:10.1016/j.jhazmat.2010.02.076
Saravanan R, Gupta VK, Prakash T et al (2013) Synthesis, characterization and photocatalytic activity of novel Hg doped ZnO nanorods prepared by thermal decomposition method. J Mol Liq 178:88–93. doi:10.1016/j.molliq.2012.11.012
Saravanan R, Prakash T, Gupta VK, Stephen A (2014) Tailoring the electrical and dielectric properties of ZnO nanorods by substitution. J Mol Liq 193:160–165. doi:10.1016/j.molliq.2013.12.029
Saravanan R, Sacari E, Gracia F et al (2016) Conducting PANI stimulated ZnO system for visible light photocatalytic degradation of coloured dyes. J Mol Liq 221:1029–1033. doi:10.1016/j.molliq.2016.06.074
Sarkar AK, Pal A, Ghorai S et al (2014) Efficient removal of malachite green dye using biodegradable graft copolymer derived from amylopectin and poly(acrylic acid). Carbohydr Polym 111:108–115. doi:10.1016/j.carbpol.2014.04.042
Sartape AS, Mandhare AM, Jadhav VV et al (2015) Removal of malachite green dye from aqueous solution with adsorption technique using Limonia acidissima (wood apple) shell as low cost adsorbent. Arab J Chem. doi:10.1016/j.arabjc.2013.12.019
Savva I, Marinica O, Papatryfonos CA et al (2015) Evaluation of electrospun polymer–Fe3O4 nanocomposite mats in malachite green adsorption. RSC Adv 5:16484–16496. doi:10.1039/C4RA16938G
Sayılkan F, Asiltürk M, Tatar P et al (2007a) Photocatalytic performance of Sn-doped TiO2 nanostructured mono and double layer thin films for malachite green dye degradation under UV and vis-lights. J Hazard Mater 144:140–146. doi:10.1016/j.jhazmat.2006.10.011
Sayılkan F, Asiltürk M, Tatar P et al (2007b) Preparation of re-usable photocatalytic filter for degradation of malachite green dye under UV and vis-irradiation. J Hazard Mater 148:735–744. doi:10.1016/j.jhazmat.2007.03.036
Sayılkan F, Asiltürk M, Tatar P et al (2008) Photocatalytic performance of Sn-doped TiO2 nanostructured thin films for photocatalytic degradation of malachite green dye under UV and VIS-lights. Mater Res Bull 43:127–134. doi:10.1016/j.materresbull.2007.02.012
Schmidt D, Shah D, Giannelis EP (2002) New advances in polymer/layered silicate nanocomposites. Curr Opin Solid State Mater Sci 6:205–212. doi:10.1016/S1359-0286(02)00049-9
Sharma P, Kaur H, Sharma M, Sahore V (2011) A review on applicability of naturally available adsorbents for the removal of hazardous dyes from aqueous waste. Environ Monit Assess 183:151–195. doi:10.1007/s10661-011-1914-0
Sharma YC (2011) Adsorption characteristics of a low-cost activated carbon for the reclamation of colored effluents containing malachite green. J Chem Eng Data 56:478–484. doi:10.1021/je1008922
Sharma YC, Singh B, Uma (2009) Fast removal of malachite green by adsorption on rice husk activated carbon. Open Environ Pollut Toxicol J 1:74–78. doi:10.2174/1876397900901010074
Shedbalkar U, Jadhav JP (2011) Detoxification of malachite green and textile industrial effluent by Penicillium ochrochloron. Biotechnol Bioprocess Eng 16:196–204. doi:10.1007/s12257-010-0069-0
Shi Q, Zhang J, Zhang C et al (2010) Preparation of activated carbon from cattail and its application for dyes removal. J Environ Sci China 22:91–97
Shirmardi M, Mahvi AH, Hashemzadeh B et al (2013) The adsorption of malachite green (MG) as a cationic dye onto functionalized multi walled carbon nanotubes. Korean J Chem Eng 30:1603–1608. doi:10.1007/s11814-013-0080-1
Singh A, Manju Rani S, Bishnoi NR (2012) Malachite green dye decolorization on immobilized dead yeast cells employing sequential design of experiments. Ecol Eng 47:291–296. doi:10.1016/j.ecoleng.2012.07.001
Singh DK, Rastogi K (2004) Adsorptive removal of basic dyes from aqueous phase onto activated carbon of used tea leaves: a kinetic and thermodynamic study. J Environ Sci Eng 46:293–302
Sivashankar R, Sathya AB, Vasantharaj K, Sivasubramanian V (2014) Magnetic composite an environmental super adsorbent for dye sequestration—a review. Environ Nanotechnol Monit Manag 1–2:36–49. doi:10.1016/j.enmm.2014.06.001
Solís M, Solís A, Pérez HI et al (2012) Microbial decolouration of azo dyes: a review. Process Biochem 47:1723–1748. doi:10.1016/j.procbio.2012.08.014
Sonawane GH, Shrivastava VS (2009) Kinetics of decolourization of malachite green from aqueous medium by maize cob (Zea maize): an agricultural solid waste. Desalination 247:430–441. doi:10.1016/j.desal.2009.01.006
Sonawane SH, Chaudhari PL, Ghodke SA et al (2009) Ultrasound assisted synthesis of polyacrylic acid-nanoclay nanocomposite and its application in sonosorption studies of malachite green dye. Ultrason Sonochem 16:351–355. doi:10.1016/j.ultsonch.2008.10.008
Srinivasan A, Viraraghavan T (2010) Decolorization of dye wastewaters by biosorbents: a review. J Environ Manag 91:1915–1929. doi:10.1016/j.jenvman.2010.05.003
Srivastava R, Rupainwar DC (2011) A comparative evaluation for adsorption of dye on Neem bark and Mango bark powder. Indian J Technol 18:67–75
Stephen Inbaraj B, Chiu CP, Ho GH et al (2006) Removal of cationic dyes from aqueous solution using an anionic poly-γ-glutamic acid-based adsorbent. J Hazard Mater 137:226–234. doi:10.1016/j.jhazmat.2006.01.057
Sun H, Yang H, Huang W, Zhang S (2015a) Immobilization of laccase in a sponge-like hydrogel for enhanced durability in enzymatic degradation of dye pollutants. J Colloid Interface Sci 450:353–360. doi:10.1016/j.jcis.2015.03.037
Sun L, Hu S, Sun H et al (2015b) Malachite green adsorption onto Fe3O4@SiO2-NH2: isotherms, kinetic and process optimization. RSC Adv 5:11837–11844. doi:10.1039/C4RA13402H
Sun X-F, Wang S-G, Liu X-W et al (2008) Biosorption of malachite green from aqueous solutions onto aerobic granules: kinetic and equilibrium studies. Bioresour Technol 99:3475–3483. doi:10.1016/j.biortech.2007.07.055
Suresh T, Annadurai G (2013) Synthesis, characterization and photocatalytic degradation of malachite green dye using titanium dioxide nanoparticles. Int J Res Environ Sci Technol 3:71–77
Suwandi AC, Indraswati N, Ismadji S (2012) Adsorption of N-methylated diaminotriphenilmethane dye (malachite green) on natural rarasaponin modified kaolin. Desalin Water Treat 41:342–355. doi:10.1080/19443994.2012.664738
Syed SP (2011) Study of the removal of malachite green from aqueous solution by using solid agricultural waste. Res J Chem Sci 1:1
Tahir H, Hammed U, Sultan M, Jahanzeb Q (2010) Batch adsorption technique for the removal of malachite green and fast green dyes by using montmorillonite clay as adsorbent. Afr J Biotechnol 9:8206–8214
Tahir H, Sultan M, Jahanzeb Q (2009) Remediation of azo dyes by using household used black tea as an adsorbent. Afr J Biotechnol. doi:10.4314/ajb.v8i15.61858
Tahir SS, Rauf N (2006) Removal of a cationic dye from aqueous solutions by adsorption onto bentonite clay. Chemosphere 63:1842–1848. doi:10.1016/j.chemosphere.2005.10.033
Tang H, Zhou W, Zhang L (2012) Adsorption isotherms and kinetics studies of malachite green on chitin hydrogels. J Hazard Mater 209–210:218–225. doi:10.1016/j.jhazmat.2012.01.010
Tang Y, Hu T, Zeng Y et al (2014) Effective adsorption of cationic dyes by lignin sulfonate polymer based on simple emulsion polymerization: isotherm and kinetic studies. RSC Adv 5:3757–3766. doi:10.1039/C4RA12229A
Tanthapanichakoon W, Ariyadejwanich P, Japthong P et al (2005) Adsorption–desorption characteristics of phenol and reactive dyes from aqueous solution on mesoporous activated carbon prepared from waste tires. Water Res 39:1347–1353. doi:10.1016/j.watres.2004.12.044
Tavakol M, Azar PA, Tehrani MS, Ghaedi M (2015) Silver nanoparticles loaded on activated carbon as a novels adsorbent for the competitive removal of malachite green and methylene blue. Int J Life Sci 9:75–92. doi:10.3126/ijls.v9i2.12053
Tavlieva MP, Genieva SD, Georgieva VG, Vlaev LT (2013) Kinetic study of brilliant green adsorption from aqueous solution onto white rice husk ash. J Colloid Interface Sci 409:112–122. doi:10.1016/j.jcis.2013.07.052
Tayade RJ, Surolia PK, Kulkarni RG, Jasra RV (2007) Photocatalytic degradation of dyes and organic contaminants in water using nanocrystalline anatase and rutile TiO2. Sci Technol Adv Mater 8:455–462. doi:10.1016/j.stam.2007.05.006
Tehrani-Bagha AR, Nikkar H, Mahmoodi NM et al (2011) The sorption of cationic dyes onto kaolin: kinetic, isotherm and thermodynamic studies. Desalination 266:274–280. doi:10.1016/j.desal.2010.08.036
Tian Y, Liu P, Wang X, Lin H (2011) Adsorption of malachite green from aqueous solutions onto ordered mesoporous carbons. Chem Eng J 171:1263–1269. doi:10.1016/j.cej.2011.05.040
Tolia J, Chakraborty M, Murthy Z (2012) Photocatalytic degradation of malachite green dye using doped and undoped ZnS nanoparticles. Pol J Chem Technol 14:16–21. doi:10.2478/v10026-012-0065-6
Tsai W-T, Chen H-R (2010) Removal of malachite green from aqueous solution using low-cost chlorella-based biomass. J Hazard Mater 175:844–849. doi:10.1016/j.jhazmat.2009.10.087
Uma, Banerjee S, Sharma YC (2013) Equilibrium and kinetic studies for removal of malachite green from aqueous solution by a low cost activated carbon. J Ind Eng Chem 19:1099–1105. doi:10.1016/j.jiec.2012.11.030
Vasanth Kumar K, Ramamurthi V, Sivanesan S (2006) Biosorption of malachite green, a cationic dye onto Pithophora sp., a fresh water algae. Dyes Pigments 69:102–107. doi:10.1016/j.dyepig.2005.02.005
Vasanth Kumar K, Sivanesan S (2007) Isotherms for Malachite Green onto rubber wood (Hevea brasiliensis) sawdust: comparison of linear and non-linear methods. Dyes Pigments 72:124–129. doi:10.1016/j.dyepig.2005.07.021
Vasu AE (2008) Studies on the removal of rhodamine B and malachite green from aqueous solutions by activated carbon. J Chem 5:844–852. doi:10.1155/2008/271615
Vijayaraghavan K, Premkumar Y, Jegan J (2015) Malachite green and crystal violet biosorption onto coco-peat: characterization and removal studies. Desalin Water Treat 57:6423–6431. doi:10.1080/19443994.2015.1011709
Volesky B (1990) Biosorption of heavy metals. CRC Press, Boca Raton
Wan Ngah WS, Ariff NFM, Hashim A, Hanafiah MAKM (2010) Malachite green adsorption onto chitosan coated bentonite beads: isotherms, kinetics and mechanism. CLEAN—Soil Air Water 38:394–400. doi:10.1002/clen.200900251
Wan Ngah WS, Teong LC, Hanafiah MAKM (2011) Adsorption of dyes and heavy metal ions by chitosan composites: a review. Carbohydr Polym 83:1446–1456. doi:10.1016/j.carbpol.2010.11.004
Wang D, Liu L, Jiang X et al (2015) Adsorption and removal of malachite green from aqueous solution using magnetic β-cyclodextrin-graphene oxide nanocomposites as adsorbents. Colloids Surf Physicochem Eng Asp 466:166–173. doi:10.1016/j.colsurfa.2014.11.021
Wang H, Yuan X, Zeng G et al (2014a) Removal of malachite green dye from wastewater by different organic acid-modified natural adsorbent: kinetics, equilibriums, mechanisms, practical application, and disposal of dye-loaded adsorbent. Environ Sci Pollut Res Int 21:11552–11564. doi:10.1007/s11356-014-3025-2
Wang H-Y, Gao H-W (2009) Preparation of calcium oxalate-bromopyrogallol red inclusion sorbent and application to treatment of cationic dye and heavy metal wastewaters. Environ Sci Pollut Res Int 16:339–347. doi:10.1007/s11356-008-0070-8
Wang J, Zhao G, Li Y et al (2014b) One-step fabrication of functionalized magnetic adsorbents with large surface area and their adsorption for dye and heavy metal ions. Dalton Trans Camb Engl 43:11637–11645. doi:10.1039/c4dt00694a
Wang L, Zhang J, Zhao R et al (2010) Adsorption of basic dyes on activated carbon prepared from Polygonum orientale Linn: equilibrium, kinetic and thermodynamic studies. Desalination 254:68–74. doi:10.1016/j.desal.2009.12.012
Wang Q, Chen C, Zhao D et al (2008a) Change of adsorption modes of dyes on fluorinated TiO2 and its effect on photocatalytic degradation of dyes under visible irradiation. Langmuir ACS J Surf Colloids 24:7338–7345. doi:10.1021/la800313s
Wang S, Ariyanto E (2007) Competitive adsorption of malachite green and Pb ions on natural zeolite. J Colloid Interface Sci 314:25–31. doi:10.1016/j.jcis.2007.05.032
Wang XS (2009) Invasive freshwater macrophyte alligator weed: novel adsorbent for removal of malachite green from aqueous solution. Water Air Soil Pollut 206:215–223. doi:10.1007/s11270-009-0097-6
Wang XS, Zhou Y, Jiang Y, Sun C (2008b) The removal of basic dyes from aqueous solutions using agricultural by-products. J Hazard Mater 157:374–385. doi:10.1016/j.jhazmat.2008.01.004
Waring DR, Hallas G (eds) (1990) The chemistry and application of dyes. Springer US, Boston
Wei A, Liu B, Zhao H et al (2014) Synthesis and formation mechanism of flowerlike architectures assembled from ultrathin NiO nanoflakes and their adsorption to malachite green and acid red in water. Chem Eng J 239:141–148. doi:10.1016/j.cej.2013.10.079
Weng X, Huang L, Chen Z et al (2013) Synthesis of iron-based nanoparticles by green tea extract and their degradation of malachite. Ind Crops Prod 51:342–347. doi:10.1016/j.indcrop.2013.09.024
Witek-Krowiak A (2013) Biosorption of malachite green from aqueous solutions by pine sawdust: equilibrium, kinetics and the effect of process parameters. Desalin Water Treat 51:3284–3294. doi:10.1080/19443994.2012.749053
Witek-Krowiak A, Szafran RG, Modelski S, Dawiec A (2012) Removal of cationic dyes from aqueous solutions using microspherical particles of fly ash. Water Environ Res Res Publ Water Environ Fed 84:162–169
Wu J, Jung B-G, Kim K-S et al (2009) Isolation and characterization of Pseudomonas otitidis WL-13 and its capacity to decolorize triphenylmethane dyes. J Environ Sci China 21:960–964
Wu X, Wang Y, Liu J et al (2010) Study of malachite green adsorption onto natural zeolite in a fixed-bed column. Desalin Water Treat 20:228–233. doi:10.5004/dwt.2010.1212
Xing Y, Deng D (2009) Enhanced adsorption of malachite green by EDTAD-modified sugarcane bagasse. Sep Sci Technol 44:2117–2131. doi:10.1080/01496390902775588
Xing Y, Wang G (2009) Poly(methacrylic acid)-modified sugarcane bagasse for enhanced adsorption of cationic dye. Environ Technol 30:611–619. doi:10.1080/09593330902838098
Xu R, Jia M, Zhang Y, Li F (2012) Sorption of malachite green on vinyl-modified mesoporous poly(acrylic acid)/SiO2 composite nanofiber membranes. Microporous Mesoporous Mater 149:111–118. doi:10.1016/j.micromeso.2011.08.024
Yagub MT, Sen TK, Afroze S, Ang HM (2014) Dye and its removal from aqueous solution by adsorption: a review. Adv Colloid Interface Sci 209:172–184. doi:10.1016/j.cis.2014.04.002
Yang C-H, Shih M-C, Chiu H-C, Huang K-S (2014) Magnetic Pycnoporus sanguineus-loaded alginate composite beads for removing dye from aqueous solutions. Mol Basel Switz 19:8276–8288. doi:10.3390/molecules19068276
Yang J, Chen C, Ji H et al (2005) Mechanism of TiO2-assisted photocatalytic degradation of dyes under visible irradiation: photoelectrocatalytic study by TiO2-film electrodes. J Phys Chem B 109:21900–21907. doi:10.1021/jp0540914
Yu J, Chi R, He Z, Qi Y (2011) Adsorption performances of cationic dyes from aqueous solution on pyromellitic dianhydride modified sugarcane bagasse. Sep Sci Technol 46:452–459. doi:10.1080/01496395.2010.510125
Zeng Y, Zhao L, Wu W et al (2013) Enhanced adsorption of malachite green onto carbon nanotube/polyaniline composites. J Appl Polym Sci 127:2475–2482. doi:10.1002/app.37947
Zhang G, Yi L, Deng H, Sun P (2014a) Dyes adsorption using a synthetic carboxymethyl cellulose-acrylic acid adsorbent. J Environ Sci China 26:1203–1211. doi:10.1016/S1001-0742(13)60513-6
Zhang H, Tang Y, Liu X et al (2011) Improved adsorptive capacity of pine wood decayed by fungi Poria cocos for removal of malachite green from aqueous solutions. Desalination 274:97–104. doi:10.1016/j.desal.2011.01.077
Zhang J, Li Y, Zhang C, Jing Y (2008) Adsorption of malachite green from aqueous solution onto carbon prepared from Arundo donax root. J Hazard Mater 150:774–782. doi:10.1016/j.jhazmat.2007.05.036
Zhang L, Zhang H, Guo W, Tian Y (2014b) Removal of malachite green and crystal violet cationic dyes from aqueous solution using activated sintering process red mud. Appl Clay Sci 93–94:85–93. doi:10.1016/j.clay.2014.03.004
Zhang X, Yu H, Yang H et al (2015) Graphene oxide caged in cellulose microbeads for removal of malachite green dye from aqueous solution. J Colloid Interface Sci 437:277–282. doi:10.1016/j.jcis.2014.09.048
Zhang X, Zhang S, Pan B et al (2012) Simple fabrication of polymer-based Trametes versicolor laccase for decolorization of malachite green. Bioresour Technol 115:16–20. doi:10.1016/j.biortech.2011.11.063
Zhou X-J, Guo W-Q, Yang S-S et al (2013) Ultrasonic-assisted ozone oxidation process of triphenylmethane dye degradation: evidence for the promotion effects of ultrasonic on malachite green decolorization and degradation mechanism. Bioresour Technol 128:827–830. doi:10.1016/j.biortech.2012.10.086
Zhou Y, Min Y, Qiao H et al (2015) Improved removal of malachite green from aqueous solution using chemically modified cellulose by anhydride. Int J Biol Macromol 74:271–277. doi:10.1016/j.ijbiomac.2014.12.020
Zou W, Gao S, Zou X, Bai H (2013) Adsorption of neutral red and malachite green onto grapefruit peel in single and binary systems. Water Environ Res Res Publ Water Environ Fed 85:466–477
Acknowledgements
The authors gratefully acknowledge the financial assistance provided by the INSPIRE Programme under the Assured Opportunity for Research Careers (AORC) scheme, funded by the Department of Science and Technology (DST) (Sanction Order No.: DST/INSPIRE Fellowship/2013/66).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Raval, N.P., Shah, P.U. & Shah, N.K. Malachite green “a cationic dye” and its removal from aqueous solution by adsorption. Appl Water Sci 7, 3407–3445 (2017). https://doi.org/10.1007/s13201-016-0512-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13201-016-0512-2