Abstract
Purpose of Review
The purpose of this review is to provide a summary on the status of the art of simultaneous perfusion-innervation imaging by CZT-SPECT and to clarify its potential applications in clinical practice.
Recent Findings
Introduction of CZT cameras represented a critical breakthrough in nuclear cardiology. Although it has been demonstrated that cardiac innervation may be easily assessed by CZT-SPECT, as for now, there are only few studies investigating cardiac perfusion and innervation with simultaneous dual-isotope imaging by CZT cameras.
Summary
Perfusion and innervation assessment by CZT camera is a feasible tool, allowing to reduce radiation dose exposure and acquisition time, thanks to CZT better spatial, temporal, and energy resolution, with higher compliance by patients. Moreover, this technique allows to integrate regional evaluation of myocardial perfusion/innervation status, providing more accurate information for risk stratification of different cardiac disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cardiac adrenergic innervation imaging by 123I-metaiodobenzylguanidine (mIBG) single-photon emission computed tomography (SPECT) is recognized as a useful tool in risk stratification and disease assessment in patients with heart failure (HF), ischemic heart disease (IHD), and other conditions affecting cardiac sympathetic activity [1,2,3,4,5]. AdreView Myocardial Imaging Evaluation in Heart Failure (ADMIRE-HF) is the first multicentric study to demonstrate that 123I-mIBG imaging-derived parameters, such as late heart-to-mediastinum ratio (HMR) and washout rate (WR), may suggest the presence of sympathetic dysfunction and are linked to the HF worsening, worst response to implantable cardiac device (ICD) therapy, and increased occurrence of fatal cardiac events [5]. The 123I-mIBG HMR and WR are easily obtained by a static planar acquisition of chest, including in the field of view (FOV) both heart and mediastinum. Moreover, a further SPECT acquisition may provide a regional evaluation of 123I-mIBG distribution allowing to combine and compare these findings with parameters obtained by myocardial perfusion imaging (MPI), such as extent and severity of myocardial perfusion defect and dyssynchrony [6,7,8]. Although the combined assessment of myocardial perfusion and innervation could provide more information compared to the only HMR evaluation in risk assessment of HF patients, the dual protocol with 123I-mIBG and a perfusion tracer is still not widely feasible in clinical practice, because of the high radiation exposure required by standard Anger camera and long SPECT acquisition time. On the other hand, there is increasing evidence that solid-state cameras with cadmium-zinc-telluride (CZT) detectors may overcome some of the standard Anger camera limitations, due to the higher spatial and energy resolution, providing the possibility for a low-dose protocol with higher image quality both for perfusion and innervation imaging, lower imaging acquisition time, and scatter correction availability [9,10,11,12]. Considering all these aspects, it is reasonable to expect that simultaneous global and regional myocardial perfusion, innervation, and functional assessment by CZT-SPECT may be a valuable tool, along with a significant benefit for patients [12, 13]. The field of application of simultaneous dual-isotope (SDI) cardiac imaging by CZT-SPECT seems to be wide but, as for our knowledge, is still uncharted and only few studies documented the value of CZT-SPECT SDI-derived parameters and the feasibility of this acquisition protocol in clinical practice [14•, 15•, 16•, 17•]. Thus, this review article aims to provide the state of the art of combined dual myocardial perfusion/innervation imaging, underling the effective advantages and potential applications of SDI by CZT-SPECT in clinical practice.
Technical Features of Dual Acquisition: Limitations and Possible Solutions
According to the recommendations of the EANM Cardiovascular Committee and the European Council of Nuclear Cardiology, 123I-mIBG imaging acquisition protocol with Anger camera consists of two planar acquisitions of the chest performed 15 min (early) and 4 h (delay) after 123I-mIBG administration, allowing the assessment of, respectively, early and late HMR and WR. These parameters are derived from planar images, drawing manually two regions of interest (ROIs), one on the heart and one on the mediastinum, and comparing each pixel’s radioactivity counts [1]. The protocol described in literature as simultaneous perfusion/innervation imaging by Anger camera consists in two separate imaging acquisitions, with the administration of a perfusion tracer, 99mTc-Sestamibi or 99mTc-tetrofosmin, after a variable interval time (ranging from 15 min to 7 days, according to different authors) since 123I-mIBG injection (Fig. 1) [5,6,7,8]. Despite that this technique including two separate imaging studies seemed quite easy to perform, its clinical use is still negatively affected by lack of standardization in injection dose, protocol acquisition, imaging time, and imaging elaboration, causing low reproducibility among different centers [18, 19]. One of the main limiting factors is due to the presence of 123I high-energy photon emission, over the main photopeak of 159 keV, which may penetrate collimator septa and affect image quality, especially when using low-energy (LE) collimators instead of medium energy (ME) ones [20,21,22]. This phenomenon, known as scatter effect, may be particularly impacting when acquiring perfusion/innervation studies in the same day, since the energy window of Anger camera is about 20% symmetrical and the main photopeak of 99mTc (140 keV) and 123I (159 keV) may overlap [23]. Thus, collimator choice, energy window width, and detector sensitivity have a dramatic impact on both image quality and semi-quantitative parameters’ reliability. The introduction of new CZT technology opened also new frontiers in the dual-isotope imaging approach. Two commercially available CZT cameras (D-SPECT by Spectrum Dynamics and DNM by GE) differ right in detector and collimator features: D-SPECT camera is equipped with 9 mobile detectors and tungsten wide-angle square parallel collimators, while GE camera features 19 fixed detectors with tungsten multi-pinhole collimators, both focused on heart [24, 25]. Both cameras collect data by list-mode acquisition, which allows to perform SDI acquisition and to obtain two different images in one acquisition (Fig. 2), retrospectively selecting specific asymmetric energy windows: D-SPECT energy windows for 123I is 8.5% asymmetrical centered on 159-keV photopeak, while 99mTc energy window is 10% asymmetrical centered on 140-keV photopeak; GE DNM energy windows for both 123I and 99mTc are 15% asymmetrical [14•, 15•, 16•, 17•]. In Table 1 are summarized main technical characteristics of D-SPECT and GE DNM compared to standard Anger camera [10, 26,27,28,29,30,31,32]. By the CZT-SPECT cameras, the derived HMR is not calculated on planar images but on a planar equivalent image, planogram, obtained with different modalities according to each CZT-camera. D-SPECT planogram is the sum of two series of 2D images for each angular position [28]. GE DNM planogram may be obtained by reprojection of planar equivalent images or by using trans axial images. The use of trans axial images determines that more reliable values can be obtained, as it may avoid some truncation artifacts related to the multi-pinhole collimation and affect reprojection planogram [15•]. When performing SDI by CZT-SPECT, early and late HMR are obtained drawing manually the ROIs (if using planogram) or the volumes of interest (if using trans axial images) including the heart and the mediastinum on perfusion images. Then, the ROI is automatically copied on 123I-mIBG images corrected or non-corrected for scatter (Fig. 3), and HMR is calculated. Accordingly, the values obtained may be corrected according to attenuation, scatter, or a conversion factor to make them comparable with standard Anger camera [22, 26, 28]. As showed in Figs. 1 and 2, the overall time for a combined acquisition is completely different using the two different protocols. It should be outlined that CZT cameras directly provide a SPECT acquisition of 10 min compared to a further acquisition by Anger camera of 30–45 min [10, 12]. Previous studies compared the two different CZT cameras to an Anger camera, using both phantoms and clinical data acquired separately or simultaneously, demonstrating that 99mTc crosstalk into 123I energy window is irrelevant when SDI acquisition is performed by CZT camera [26,27,28,29,30,31,32]. Moreover, early and late HMR values obtained by this technique showed good agreement with those obtained by Anger camera especially when a correction factor is applied to uniform CZT camera values to those obtained by ME collimators [29]. Similarly, perfusion and LV functional assessment by SDI CZT-SPECT showed high overall agreement with single isotope imaging results, especially when using 99mTc tracers, the crosstalk, scatter effect, and tailing effect of 123I into 99mTc photopeak are negligible [30]. In fact, it has been demonstrated that perfusion/innervation defect size and mismatch quantification are neither influenced by the type of CZT camera, nor by acquisition time or mode [15•]. Moreover, only a slight difference was found in LV volume assessment according to the type of camera, but this was not linked to the acquisition mode. Anyway, LV wall motion and thickening assessment showed a high agreement between perfusion-only and SDI acquisitions [30, 31]. However, as showed in Table 2, most of the different clinical studies available have provided combined data using a protocol with two different acquisitions, both by Anger camera and, as well, by CZT cameras. Thus, the main advantage to use a CZT camera allowing to perform two studies using one acquisition has not been fully utilized.
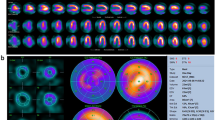
Example of low-dose simultaneous dual-isotope (SDI) protocol with one imaging acquisition by CZT camera [17•]
99mTc-sestamibi/123I-mIBG SDI planogram with and without scatter correction. A 99mTc-sestamibi and B 123I-mIBG non-scatter-corrected planograms; C 99mTc-sestamibi and D 123I-mIBG scatter-corrected planograms. White circular ROI positioned on LV, yellow squared ROI positioned on mediastinum, to obtain HMR
Clinical Applications: Emerging Evidence
From a clinical point of view, major goal of simultaneous imaging using dual-isotope approach allows to evaluate simultaneously global and regional perfusion/innervation defect severity, extent, and matching/mismatching, along with a functional assessment of LV parameters such LVEF, volumes, and mechanical dyssynchrony. Considering the promising results of previous phantom feasibility studies, researchers’ interest is growing on SDI CZT-SPECT (one imaging for two tracers) for possible clinical applications [26,27,28,29,30,31,32]. In Table 3 are summarized the most recent clinical studies on CZT-SPECT SDI protocol (Fig. 2). Bellevre et al. [14•] in the ADRECARD study first demonstrated the feasibility of CZT-SPECT SDI using 123I-mIBG and 99mTc-tetrofosmin by D-SPECT camera in a cohort of ischemic and non-ischemic HF patients. The original purpose of using a perfusion tracer was to better localize the heart in the FOV, considering that HF patients often show reduced 123I-mIBG uptake and dilated LV leading to not properly include the left ventricle in the camera FOV. The authors compared derived 123I-mIBG late HMR by D-SPECT SDI to HMR obtained by standard Anger camera phantom studies showing high agreement and outlining that SDI CZT-SPECT is feasible in patients with HF, providing reliable late HMR. Similarly, other studies compared late HMR obtained by GE DNM SDI and late HMR by standard Anger camera in a cohort of HF patients showing high agreement between the value obtained by the two different cameras, consistent with ADRECARD study results [16•, 30].
D’estanque et al. first demonstrated the advantages of CZT-SPECT SDI in patients with IHD and high risk for ventricular arrythmia (VA), using 201Tl and 123I-mIBG by GE DNM camera [15•]. IHD is characterized by both myocardial perfusion and adrenergic impairment, but usually LV 123I-mIBG uptake is reduced not only in necrotic regions (perfusion/innervation match), but also in ischemic and/or still viable regions (perfusion/innervation mismatch), which are more sensitive to catecholamine and may trigger VA. Using cardiac MRI as standard reference, the authors demonstrated that 201Tl and 123I-mIBG SDI is useful to evaluate the presence and the extent of these trigger zones, and to better stratify IHD patients according to their risk of developing VA. Moreover, this study highlighted the necessity of scatter correction for a reliable estimation of trigger zone extent, although clinical implications are not investigated yet. Recently, Assante et al. assessed the feasibility of 99mTc-Sestamibi and 123I-mIBG CZT-SPECT SDI with low-dose protocol in patients with HF [17•]. Besides global innervation evaluation by early and late HMR and WR, using 17-segment model, the authors derived also total perfusion and innervation defect size, expressed as percentage of LV, and calculated perfusion/innervation mismatch, as indicator of trigger zone extent. It has been demonstrated that low-dose CZT-SPECT SDI may provide good planar and SPECT images with significant reduction of dose exposure and acquisition time (Fig. 4) [12, 17•]. Moreover, parameters derived by SPECT analysis, such as the extent perfusion/innervation mismatch, demonstrated a strong correlation with 123I-mIBG WR, which reflects adrenergic neuronal retention system impairment and is an independent predictor of fatal cardiac events. These preliminary studies are promising and enforce the need for wide prospective multicentric standardized trials to standardize the technical aspects of this approach and its possible clinical usefulness.
SDI by CZT-SPECT in a patient with ischemic HF and preserved LVEF (50%). A Tomographic reconstructed images and B perfusion and innervation bulls’ eyes. Upper series represent late 123I-mIBG images; bottom series represent 99mTc-sestamibi rest MPI. Innervation defect on infero-lateral wall (red arrows) appears more severe than perfusion defect (white arrows) both at visual analysis and at semi-quantitative evaluation, suggesting a potential perfusion/innervation mismatch in infero-lateral region (yellow arrow)
Future Perspective: Let Us See Further!
Although favorable results of recent studies, a question remains to be addressed: in which patients’ categories SDI imaging could be useful? Indeed, CZT-SPECT SDI may play a role in HF patients, not only to better localize heart but also to better stratify patients according to their risk of cardiac events [2,3,4, 33]. The evidence that perfusion/innervation mismatch extent is not only correlated with trigger zones, responsible for the occurrence of VA, but also is an independent predictor of increased 123I-mIBG WR suggests that it may reflect the severity of neuronal retention system disease [17•]. Moreover, as demonstrated by previous studies, 123I-mIBG late HMR is independent prognostic factor for fatal cardiac events and ICD therapy response, improving the appropriacy of ICD implantation in HF patients [6, 8]. Thus, the combined evaluation of late 123I-mIBG HMR and perfusion/innervation mismatch by CZT-SPECT may provide a useful tool to further stratify HF patients according to risk of VA and to ICD therapy response [33,34,35,36,37,38]. Clearly, the technique should be further standardized, and large prospective multicentric studies are required to investigate the real added value of SDI-derived novel parameters in diagnosis and prognosis of patients with HF. Other patients’ categories could be involved in a SDI protocol, despite that lack of evidence till now does not support this hypothesis, considering that cardiac adrenergic innervation may result impaired in some conditions, like chronic kidney failure, obesity, and diabetes mellitus, where perfusion defect could be observed also in the absence of known CAD [2, 3, 39,40,41]. Some authors evaluated obesity impact on cardiac innervation assessment in a cohort of obese and non-obese HF patients concluding that obesity is associated with reduced early and late HMR [42]. Similarly, diabetes mellitus complications could include not only macrovascular and microvascular dysfunction, but also cardiac autonomic neuropathy, which may lead to silent myocardial ischemia and increased risk of fatal arrythmia [43, 44]. It has been demonstrated that regional assessment of cardiac 123I-mIBG distribution is useful in diabetic patients with known CAD where perfusion assessment has important implications [43]. Moreover, 123I-mIBG imaging resulted useful also in documenting diabetes therapy response and in risk stratification even before the onset of microvascular impairment [44]. In such clinical scenarios, the opportunity to perform serial simultaneous global and regional perfusion/innervation assessment in a single 10-min acquisition seems to be very cost-effective in the management of diabetic patients. Similarly, some evidence reported the use of 123I-mIBG imaging in diagnosing cardiomyophaties like cardiac amyloidosis or chronic Chagas disease, both conditions that may lead to HF and fatal arrhythmia [45,46,47,48]. Moreover, it has been demonstrated that a massive catecholamine release, which happens typically in Tako-Tsubo cardiomyopathy, may cause myocardial stunning and reduce myocardial 123I-mIBG uptake [49].
Since each of these conditions may require further myocardial perfusion assessment, it appears clear the need for large, multicentric, and standardized trials to assess the real diagnostic and prognostic added value of performing perfusion/innervation SDI imaging by CZT-SPECT in different clinical scenarios [50,51,52]. Lastly, considering that CZT cameras allow high-quality list-mode dynamic image acquisition for myocardial blood flow and perfusion reserve assessment [53, 54], some preliminary studies on animals evaluated the feasibility of dynamic innervation imaging combined with gated MPI performed immediately before 123I-mIBG administration [55, 56]. The authors concluded that dynamic 123I-mIBG kinetic analysis, considering tracer’s uptake peak, may integrate the standard evaluation parameters like late HMR and WR, adding more information of 123I-mIBG myocardial distribution [55].
Conclusions
Simultaneous assessment of myocardial perfusion and innervation by dual-isotope CZT-SPECT is a feasible technique, proved to have many possible advantages over single-isotope evaluation in HF management, and promising data are emerging about its usefulness in many cardiac and systemic disease involving myocardial perfusion and innervation impairment. CZT cameras allow a dramatic reduction in dose exposure and acquisition time, with great patient’s comfort and benefit. Thus, more studies are mandatory to standardize and validate this technique in clinical practice and to evaluate its real value in patients with cardiac disease. The farther the research goes, more applications may be implemented.
Data Availability
The authors declare that all data supporting the findings of this study are available within the paper and its supplementary information.
Abbreviations
- CZT:
-
Cadmium-zinc-telluride
- HF:
-
Heart failure
- HMR:
-
Heart to mediastinum ratio
- ICD:
-
Implantable cardiac device
- IHD:
-
Ischemic heart disease
- LV:
-
Left ventricle
- MPI:
-
Myocardial perfusion imaging
- SDI:
-
Simultaneous dual isotope
- SPECT:
-
Single-photon emission computed tomography
- mIBG:
-
Metaiodobenzylguanidine
- VA:
-
Ventricular arrythmia
- WR:
-
Washout rate
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Gimelli A, Liga R, Agostini D, Bengel FM, Ernst S, Hyafil F, et al. The role of myocardial innervation imaging in different clinical scenarios: an expert document of the European Association of Cardiovascular Imaging and Cardiovascular Committee of the European Association of Nuclear Medicine. Eur Heart J Cardiovasc Imaging. 2021;22(5):480–90. https://doi.org/10.1093/ehjci/jeab007.
Gargiulo P, Acampa W, Asile G, Abbate V, Nardi E, Marzano F, et al. 123I-MIBG imaging in heart failure: impact of comorbidities on cardiac sympathetic innervation. Eur J Nucl Med Mol Imaging. 2023;50(3):813–24. https://doi.org/10.1007/s00259-022-05941-3.
Pontico M, Brunotti G, Conte M, Corica F, Cosma L, De Angelis C, et al. The prognostic value of 123I-mIBG SPECT cardiac imaging in heart failure patients: a systematic review. J Nucl Cardiol. 2022;29(4):1799–809. https://doi.org/10.1007/s12350-020-02501-w.
Verschure DO, Nakajima K, Verberne HJ. Cardiac 123I-mIBG Imaging in Heart Failure. Pharmaceuticals (Basel). 2022;15(6):656. https://doi.org/10.3390/ph15060656.
Jacobson AF, Senior R, Cerqueira MD, Wong ND, Thomas GS, Lopez VA, et al. Myocardial iodine-123 meta-iodobenzylguanidine imaging and cardiac events in heart failure. Results of the prospective ADMIRE-HF (AdreView Myocardial Imaging for Risk Evaluation in Heart Failure) study. J Am Coll Cardiol. 2010; 55(20):2212–21 https://doi.org/10.1016/j.jacc.2010.01.014.
Hachamovitch R, Nutter B, Menon V, Cerqueira MD. Predicting risk versus predicting potential survival benefit using 123I-mIBG imaging in patients with systolic dysfunction eligible for implantable cardiac defibrillator implantation: analysis of data from the prospective ADMIRE-HF study. Circ Cardiovasc Imaging. 2015;12:e003110. https://doi.org/10.1161/CIRCIMAGING.114.003110.
Gimelli A, Liga R, Menichetti F, Soldati E, Bongiorni MG, Marzullo P. Interactions between myocardial sympathetic denervation and left ventricular mechanical dyssynchrony: a CZT analysis. J Nucl Cardiol. 2019;26(2):509–18. https://doi.org/10.1007/s12350-017-1036-3.
Mishkina AI, Saushkin VV, Atabekov TA, Sazonova SI, Shipulin VV, Massalha S, et al. The value of cardiac sympathetic activity and mechanical dyssynchrony as cardiac resynchronization therapy response predictors: comparison between patients with ischemic and non-ischemic heart failure. J Nucl Cardiol. 2023;30(1):371–82. https://doi.org/10.1007/s12350-022-03046-w.
Assante R, Acampa W. Dual isotope and multidetector camera: the best choices for a specific end-point. J Nucl Cardiol. 2017;24(4):1370–3. https://doi.org/10.1007/s12350-016-0520-5.
Cantoni V, Green R, Acampa W, Zampella E, Assante R, Nappi C, et al. Diagnostic performance of myocardial perfusion imaging with conventional and CZT single-photon emission computed tomography in detecting coronary artery disease: a meta-analysis. J Nucl Cardiol. 2021;28(2):698–715. https://doi.org/10.1007/s12350-019-01747-3.
Gimelli A, Liga R, Giorgetti A, Genovesi D, Marzullo P. Assessment of myocardial adrenergic innervation with a solid-state dedicated cardiac cadmium-zinc-telluride camera: first clinical experience. Eur Heart J Cardiovasc Imaging. 2014;15(5):575–85. https://doi.org/10.1093/ehjci/jet258.
Acampa W, Buechel RR, Gimelli A. Low dose in nuclear cardiology: state of the art in the era of new cadmium-zinc-telluride cameras. Eur Heart J Cardiovasc Imaging. 2016;17(6):591–5. https://doi.org/10.1093/ehjci/jew036.
Ben-Haim S, Kacperski K, Hain S, Van Gramberg D, Hutton BF, Erlandsson K, et al. Simultaneous dual-radionuclide myocardial perfusion imaging with a solid-state dedicated cardiac camera. Eur J Nucl Med Mol Imaging. 2010;37(9):1710–21. https://doi.org/10.1007/s00259-010-1441-1.
• Bellevre D, Manrique A, Legallois D, Bross S, Baavour R, Roth N, et al. First determination of the heart-to-mediastinum ratio using cardiac dual isotope (123I-MIBG/99mTc-tetrofosmin) CZT imaging in patients with heart failure: the ADRECARD study. Eur J Nucl Med Mol Imaging. 2015;42(12):1912–9. https://doi.org/10.1007/s00259-015-3141-3. This study is relevant as it is the first report of the feasibility of CZT-SPECT SDI protocols in clinical practice by D-SPECT camera.
• D’estanque E, Hedon C, Lattuca B, Bourdon A, Benkiran M, Verd A, et al. Optimization of a simultaneous dual-isotope 201Tl/123I-MIBG myocardial SPECT imaging protocol with a CZT camera for trigger zone assessment after myocardial infarction for routine clinical settings: are delayed acquisition and scatter correction necessary? J Nucl Cardiol. 2017; 24(4):1361-1369. https://doi.org/10.1007/s12350-016-0524-1. This study is relevant as evaluates the role of SPECT analysis in perfusion/innervation SDI imaging by CZT camera and investigates the real advantages of scatter correction.
• Blaire T, Bailliez A, Ben Bouallegue F, Bellevre D, Agostini D, Manrique A. Determination of the heart-to-mediastinum ratio of 123I-MIBG uptake using dual-isotope (123I-MIBG/99mTc-Tetrofosmin) multipinhole cadmium-zinc-telluride SPECT in patients with heart failure. J Nucl Med. 2018;59(2):251–8. https://doi.org/10.2967/jnumed.117.194373. This study is relevant as it first investigates the late HMR assessment by DNM camera, showing the need to use transaxial images and a linear correction equation to compare CZT-camera results to standard Anger camera.
• Assante R, D’Antonio A, Mannarino T, Nappi C, Gaudieri V, Zampella E, et al. Simultaneous assessment of myocardial perfusion and adrenergic innervation in patients with heart failure by low-dose dual-isotope CZT SPECT imaging. J Nucl Cardiol. 2022;29(6):3341–51. https://doi.org/10.1007/s12350-022-02951-4. This study is relevant as it demonstrates the feasibility of low-dose SDI protocol by CZT-SPECT in clinical practice, highlighting the possibility to obtain a full global and regional assessment of cardiac perfusion and innervation study in a single simultaneous acquisition time.
Verschure DO, Bongers V, Hagen P, Somsen GA, van Eck-Smit BF, Verberne H. Impact of a predefined mediastinal ROI on interobserver variability of planar 123I-MIBG heart-to-mediastinum ratio. J Nucl Cardiol. 2014;21(3):605–13. https://doi.org/10.1007/s12350-014-9854-z.
Pellegrino T, Petretta M, De Luca S, Paolillo S, Boemio A, Carotenuto R, et al. Observer reproducibility of results from a low-dose 123I-metaiodobenzylguanidine cardiac imaging protocol in patients with heart failure. Eur J Nucl Med Mol Imaging. 2013;40(10):1549–57. https://doi.org/10.1007/s00259-013-2461-4.
Brumberg J, Blazhenets G, Schröter N, Frings L, Jost WH, Lapa C, et al. Imaging cardiac sympathetic innervation with MIBG: linear conversion of the heart-to-mediastinum ratio between different collimators. EJNMMI Phys. 2019;6(1):12. https://doi.org/10.1186/s40658-019-0250-2.
Owenius R, Zanette M, Cella P. Variability in heart-to-mediastinum ratio from planar 123I-MIBG images of a thorax phantom for 6 common γ-camera models. J Nucl Med Technol. 2017;45(4):297–303. https://doi.org/10.2967/jnmt.117.196055.
Nakajima K, Okuda K, Yoshimura M, Matsuo S, Wakabayashi H, Imanishi Y, et al. Multicenter cross-calibration of I-123 metaiodobenzylguanidine heart-to-mediastinum ratios to overcome camera-collimator variations. J Nucl Cardiol. 2014;21(5):970–8. https://doi.org/10.1007/s12350-014-9916-2.
Verschure DO, de Wit TC, Bongers V, Hagen PJ, Sonneck-Koenne C, D’Aron J, et al. 123I-MIBG heart-to-mediastinum ratio is influenced by high-energy photon penetration of collimator septa from liver and lung activity. Nucl Med Commun. 2015;36(3):279–85. https://doi.org/10.1097/MNM.0000000000000238.
Erlandsson K, Kacperski K, van Gramberg D, Hutton BF. Performance evaluation of D-SPECT: a novel SPECT system for nuclear cardiology. Phys Med Biol. 2009;54(9):2635–49. https://doi.org/10.1088/0031-9155/54/9/003.
Gambhir SS, Berman DS, Ziffer J, Nagler M, Sandler M, Patton J, Hutton B, Sharir T, Haim SB, Haim SB. A novel high-sensitivity rapid-acquisition single-photon cardiac imaging camera. J Nucl Med. 2009;50(4):635–43. https://doi.org/10.2967/jnumed.108.060020.
Nakajima K, Okuda K, Yokoyama K, Yoneyama T, Tsuji S, Oda H, et al. Cross calibration of 123I-meta-iodobenzylguanidine heart-to-mediastinum ratio with D-SPECT planogram and Anger camera. Ann Nucl Med. 2017;31(8):605–15. https://doi.org/10.1007/s12149-017-1191-2.
Holstensson M, Erlandsson K, Poludniowski G, Ben-Haim S, Hutton BF. Model-based correction for scatter and tailing effects in simultaneous 99mTc and 123I imaging for a CdZnTe cardiac SPECT camera. Phys Med Biol. 2015;60(8):3045–63. https://doi.org/10.1088/0031-9155/60/8/3045.
Nakajima K, Verschure DO, Okuda K, Verberne HJ. Standardization of 123I-meta-iodobenzylguanidine myocardial sympathetic activity imaging: phantom calibration and clinical applications. Clin Transl Imaging. 2017;5(3):255–63. https://doi.org/10.1007/s40336-017-0230-2.
Verschure DO, Poel E, Nakajima K, Okuda K, van Eck-Smit BLF, Somsen GA, et al. A European myocardial 123I-mIBG cross-calibration phantom study. J Nucl Cardiol. 2018;25(4):1191–7. https://doi.org/10.1007/s12350-017-0782-6.
Blaire T, Bailliez A, Ben Bouallegue F, Bellevre D, Agostini D, Manrique A. First assessment of simultaneous dual isotope ((123)I/(99m)Tc) cardiac SPECT on two different CZT cameras: a phantom study. J Nucl Cardiol. 2018;25(5):1692–704. https://doi.org/10.1007/s12350-017-0841-z.
Niimi T, Nanasato M, Sugimoto M, Maeda H. Comparative cardiac phantom study using Tc-99m/I-123 and Tl-201/I-123 tracers with cadmium-zinc-telluride detector-based single-photon emission computed tomography. Nucl Med Mol Imaging. 2019;53(1):57–63. https://doi.org/10.1007/s13139-018-0559-0.
Kacperski K, Erlandsson K, Ben-Haim S, Hutton BF. Iterative deconvolution of simultaneous 99mTc and 201Tl projection data measured on a CdZnTe-based cardiac SPECT scanner. Phys Med Biol. 2011;56(5):1397–414. https://doi.org/10.1088/0031-9155/56/5/012.
Sood N, Al Badarin F, Parker M, Pullatt R, Jacobson AF, Bateman TM, et al. Resting perfusion MPI-SPECT combined with cardiac 123I-mIBG sympathetic innervation imaging improves prediction of arrhythmic events in non-ischemic cardiomyopathy patients: sub-study from the ADMIRE-HF trial. J Nucl Cardiol. 2013;20(5):813–20. https://doi.org/10.1007/s12350-013-9750-y.
Clements IP, Garcia EV, Chen J, Folks RD, Butler J, Jacobson AF. Quantitative iodine-123-metaiodobenzylguanidine (MIBG) SPECT imaging in heart failure with left ventricular systolic dysfunction: development and validation of automated procedures in conjunction with technetium-99m tetrofosmin myocardial perfusion SPECT. J Nucl Cardiol. 2016;23(3):425–35. https://doi.org/10.1007/s12350-015-0097-4.
Travin MI, Henzlova MJ, van Eck-Smit BLF, Jain D, Carrio I, Folks RD, et al. Assessment of (123)I-mIBG and (99 m)Tctetrofosmin single-photon emission computed tomographic images for the prediction of arrhythmic events in patients with ischemic heart failure: intermediate severity innervation defects are associated with higher arrhythmic risk. J Nucl Cardiol. 2017;24(2):377–91. https://doi.org/10.1007/s12350-015-0336-8.
Vauchot F, Ben Bouallègue F, Hedon C, Piot C, Roubille F, Mariano-Goulart D. Assessment of the area at risk after acute myocardial infarction using 123I-MIBG SPECT: comparison with the angiographic APPROACH-score. J Nucl Cardiol. 2018;25(2):572–80. https://doi.org/10.1007/s12350-016-0644-7.
Verschure DO, Poel E, Travin MI, Henzlova MJ, Jain D, Jacobson AF, et al. A simplified wall-based model for regional innervation/perfusion mismatch assessed by cardiac 123I-mIBG and rest 99mTc-tetrofosmin SPECT to predict arrhythmic events in ischaemic heart failure. Eur Heart J Cardiovasc Imaging. 2022;23(9):1201–9. https://doi.org/10.1093/ehjci/jeab132.
Zhou Y, Zhou W, Folks RD, Manatunga DN, Jacobson AF, Bax JJ, et al. I-123 mIBG and Tc-99m myocardial SPECT imaging to predict inducibility of ventricular arrhythmia on electrophysiology testing: a retrospective analysis. J Nucl Cardiol. 2014;21(5):913–20. https://doi.org/10.1007/s12350-014-9911-7.
Marsico F, Paolillo S, Gargiulo P, Parisi V, Nappi C, Assante R, et al. Renal function and cardiac adrenergic impairment in patients affected by heart failure. J Nucl Cardiol. 2021;28(5):2112–22. https://doi.org/10.1007/s12350-019-01975-7.
Cantoni V, Green R, Acampa W, Assante R, Zampella E, Nappi C, et al. Prognostic value of myocardial perfusion imaging in patients with chronic kidney disease: a systematic review and meta-analysis. J Nucl Cardiol. 2022;29(1):141–54. https://doi.org/10.1007/s12350-020-02449-x.
Amami K, Yamada S, Yoshihisa A, Kaneshiro T, Hijioka N, Nodera M, et al. Predictive impacts of chronic kidney disease and cardiac sympathetic nervous activity on lethal arrhythmic events in chronic heart failure. Ann Noninvasive Electrocardiol. 2022;27(1):e12900. https://doi.org/10.1111/anec.12900.
Komici K, Bencivenga L, Paolillo S, Gargiulo P, Formisano R, Assante R, et al. Impact of body mass index on cardiac adrenergic derangement in heart failure patients: a 123I-mIBG imaging study. Eur J Nucl Med Mol Imaging. 2020;47(7):1713–21. https://doi.org/10.1007/s00259-019-04658-0.
Mba CM, Nganou-Gnindjio C-N, Azabji-Kenfack M, Mfeukeu-Kuate L, Dehayem MY, Mbanya JC, et al. Short term optimization of glycaemic control using insulin improves sympatho-vagal tone activities in patients with type 2 diabetes. Diabetes Res Clin Pract. 2019;157:107875. https://doi.org/10.1016/j.diabres.2019.107875.
Sano M. A new class of drugs for heart failure: SGLT2 inhibitors reduce sympathetic overactivity. J Cardiol. 2018;71(5):471–6. https://doi.org/10.1016/j.jjcc.2017.12.004.
Piekarski E, Chequer R, Algalarrondo V, Eliahou L, Mahida B, Vigne J, et al. Cardiac denervation evidenced by MIBG occurs earlier than amyloid deposits detection by diphosphonate scintigraphy in TTR mutation carriers. Eur J Nucl Med Mol Imaging. 2018;45(7):1108–18. https://doi.org/10.1007/s00259-018-3963-x.
Dorbala S, Ando Y, Bokhari S, Dispenzieri A, Falk RH, Ferrari VA, et al. ASNC/AHA/ASE/EANM/HFSA/ISA/SCMR/SNMMI expert consensus recommendations for multimodality imaging in cardiac amyloidosis: part 2 of 2-Diagnostic criteria and appropriate utilization. J Nucl Cardiol. 2020;27(2):659–73. https://doi.org/10.1007/s12350-019-01761-5.
Slart R, Glaudemans A, Noordzij W, Bijzet J, Hazenberg BPC, Nienhuis HLA. Time for new imaging and therapeutic approaches in cardiac amyloidosis. Eur J Nucl Med Mol Imaging. 2019;46(7):1402–6. https://doi.org/10.1007/s00259-019-04325-4.
Gadioli LP, Miranda CH, Pintya AO, de Figueiredo AB, Schmidt A, Maciel BC, et al. The severity of ventricular arrhythmia correlates with the extent of myocardial sympathetic denervation, but not with myocardial fibrosis extent in chronic Chagas cardiomyopathy: Chagas disease, denervation and arrhythmia. J Nucl Cardiol. 2018;25(1):75–83. https://doi.org/10.1007/s12350-016-0556-6.
Christensen TE, Bang LE, Holmvang L, Skovgaard DC, Oturai DB, Søholm H, et al. I-MIBG scintigraphy in the subacute state of Takotsubo cardiomyopathy. JACC Cardiovasc Imaging. 2016;9(8):982–90. https://doi.org/10.1016/j.jcmg.2016.01.028.
Gimelli A, Pugliese NR, Bertasi M, Airò E, Bauleo C, Formichi B, et al. Cardio-pulmonary involvement in pulmonary arterial hypertension: a perfusion and innervation scintigraphic evaluation. J Nucl Cardiol. 2021;28(2):546–56. https://doi.org/10.1007/s12350-019-01689-w.
Stirrup J, Gregg S, Baavour R, Roth N, Breault C, Agostini D, et al. Hybrid solid-state SPECT/CT left atrial innervation imaging for identification of left atrial ganglionated plexi: technique and validation in patients with atrial fibrillation. J Nucl Cardiol. 2020;27(6):1939–50. https://doi.org/10.1007/s12350-018-01535-5.
Nappi C, Assante R, Zampella E, Gaudieri V, De Simini G, Giordano A, et al. Relationship between heart rate response and cardiac innervation in patients with suspected or known coronary artery disease. J Nucl Cardiol. 2021;28(6):2676–83. https://doi.org/10.1007/s12350-020-02091-7.
Acampa W, Zampella E, Assante R, Genova A, De Simini G, Mannarino T, D’Antonio A, Gaudieri V, Nappi C, Buongiorno P, Mainolfi CG, Petretta M, Cuocolo A. Quantification of myocardial perfusion reserve by CZT-SPECT: a head to head comparison with 82Rubidium PET imaging. J Nucl Cardiol. 2021;28(6):2827–39. https://doi.org/10.1007/s12350-020-02129-w. (Epub 2020 May 7 PMID: 32383083).
D’Antonio A, Assante R, Zampella E, Mannarino T, Buongiorno P, Cuocolo A, et al. Myocardial blood flow evaluation with dynamic cadmium-zinc-telluride single-photon emission computed tomography: bright and dark sides. Diagn Interv Imaging. 2023;S2211–5684(23):00025–6. https://doi.org/10.1016/j.diii.2023.02.001.
Tinti E, Positano V, Giorgetti A, Marzullo P. Feasibility of [(123)I]-meta-iodobenzylguanidine dynamic 3-D kinetic analysis in vivo using a CZT ultrafast camera: preliminary results. Eur J Nucl Med Mol Imaging. 2014;41(1):167–73. https://doi.org/10.1007/s00259-013-2549-x.
Giorgetti A, Burchielli S, Positano V, Kovalski G, Quaranta A, Genovesi D, et al. Dynamic 3D analysis of myocardial sympathetic innervation: an experimental study using 123I-MIBG and a CZT camera. J Nucl Med. 2015;56(3):464–9. https://doi.org/10.2967/jnumed.114.143669.
Funding
Open access funding provided by Università degli Studi di Napoli Federico II within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Human and Animal Rights
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
D’Antonio, A., Falzarano, M., Reina, F. et al. Cardiac Perfusion and Innervation Assessment by CZT-SPECT: Have a Look to Simultaneous Dual-isotope Protocol. Curr Cardiovasc Imaging Rep 16, 41–50 (2023). https://doi.org/10.1007/s12410-023-09578-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12410-023-09578-0