Abstract
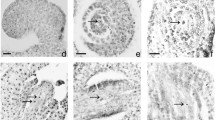
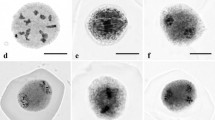
Camellia oleifera is believed to exhibit a complex intraspecific polyploidy phenomenon. Abnormal microsporogenesis can promote the formation of unreduced gametes in plants and lead to sexual polyploidy, so it is hypothesized that improper meiosis probably results in the formation of natural polyploidy in Camellia oleifera. In this study, based on the cytological observation of meiosis in pollen mother cells (PMCs), we found natural 2n pollen for the first time in Camellia oleifera, which may lead to the formation of natural polyploids by sexual polyploidization. Additionally, abnormal cytological behaviour during meiosis, including univalent chromosomes, extraequatorial chromosomes, early segregation, laggard chromosomes, chromosome stickiness, asynchronous meiosis and deviant cytokinesis (monad, dyads, triads), was observed, which could be the cause of 2n pollen formation. Moreover, we confirmed a relationship among the length–width ratio of flower buds, stylet length and microsporogenesis. This result suggested that we can immediately determine the microsporogenesis stages by phenotypic characteristics, which may be applicable to breeding advanced germplasm in Camellia oleifera.





Similar content being viewed by others
References
Bomblies K, Higgins JD, Yant L (2015) Meiosis evolves: adaptation to external and internal environments. New Phytol 208(2):306–323
Bretagnolle F, Thompson D (1995) Gametes with the somatic chromosome number: mechanisms of their formation and role in the evolution of autopolyploid plants. New Phytol 129(1):1–22
Brownfield L, Köhler C (2011) Unreduced gamete formation in plants: mechanisms and prospects. J Exp Bot 62(5):1659–1668
Camadro EL, Saffarano SK, Espinillo JC, Castro M, Simon PW (2008) Cytological mechanisms of 2n pollen formation in the wild potato Solanum okadae and pollen-pistil relations with the cultivated potato, Solanum tuberosum. Genet Resour Crop Evol 55(3):471–477
Chen C, Lyon MT, O’Malley D, Federici CT, Gmitter J, Gosser JW, Chaparro JX, Rosse ML, Gmitter FR Jr (2008) Origin and frequency of 2n gametes in Citrus sinensis × Poncirus trifoliata and their reciprocal crosses. Plant Sci 174(1):1–8
Cramer CS (1999) Laboratory techniques for determining ploidy in plants. HortTechnology 9(4):594–596
Custódio L, Carneiro MF, Romano A (2005) Microsporogenesis and anther culture in carob tree (Ceratonia siliqua L.). Sci Horticult 104(1):65–77.
De Storme N, Geelen D (2013) Sexual polyploidization in plants–cytological mechanisms and molecular regulation. New Phytol 198(3):670–684
De Storme N, Geelen D (2013a) Cytokinesis in plant male meiosis. Plant Signal Behav 8(3):e23394.
De Storme N, Copenhaver GP, Geelen D (2012) Production of diploid male gametes in Arabidopsis by cold-induced destabilization of postmeiotic radial microtubule arrays. Plant Physiol 160(4):1808–1826
Dewitte A, Eeckhaut T, Van Huylenbroeck J, Van Bockstaele E (2006) Flow cytometric detection of unreduced pollen in Begonia. Paper presented at the XXII International Eucarpia Symposium, Section Ornamentals, Breeding for Beauty 714.
Feng Y, Cai Y, Zhang X, Gao T, Mu X, Liang CL (2016) Microsporogenesis and male gametophyte development in Prunus mahaleb Linn. J Hortic Sci Biotechnol 91(5):514–519
Galbraith DW, Harkins KR, Maddox JM, Ayres NM, Sharma DP, Firoozabady E (1983) Rapid flow cytometric analysis of the cell cycle in intact plant tissues. Science 220(4601):1049–1051
García AV, Ortiz AM, Silvestri MC, Custodio A, Moretzsohn M, Lavia GI (2020) Occurrence of 2n microspore production in diploid interspecific hybrids between the wild parental species of peanut (Arachis hypogaea L., Leguminosae) and its relevance in the genetic origin of the cultigen. Crop Sci 60(5):2420–2436.
Han Z, Geng X, Du K, Xu C, Yao P, Bai F, Kang X (2018) Analysis of genetic composition and transmitted parental heterozygosity of natural 2n gametes in Populus tomentosa based on SSR markers. Planta 247(6):1407–1421
Harlan JR, deWet JM (1975) On Ö. Winge and a prayer: the origins of polyploidy. Bot Rev 41(4):361–390.
Hedhly A, Hormaza JI, Herrero M (2009) Global warming and sexual plant reproduction. Trends Plant Sci 14(1):30–36
Husband PKBC (2012) Using flow cytometry to estimate pollen DNA content: improved methodology and applications. Ann Bot 110(5):1067–1078
Jacob Y, Pierret V (1998) Pollen size and ploidy level in the genus Rosa. Paper presented at the XIX International Symposium on Improvement of Ornamental Plants 508, pp 289–292
Jauhar PP (2003) Formation of 2n gametes in durum wheat haploids: sexual polyploidization. Euphytica 133(1):81–94
Koltunow AM, Truettner J, Cox KH, Wallroth M, Goldberg RB (1990) Different temporal and spatial gene expression patterns occur during anther development. Plant Cell 2(12):1201–1224
Li Y, Wang Y, Wang P, Yang J, Kang X (2016) Induction of unreduced megaspores in Eucommia ulmoides by high temperature treatment during megasporogenesis. Euphytica 212(3):515–524
Loureiro J, Rodriguez E, Doležel J, Santos C (2007) Two new nuclear isolation buffers for plant DNA flow cytometry: a test with 37 species. Ann Bot 100(4):875–888
Lu M, Zhang P, Wang J, Kang X, Wu J, Wang X, Chen Y (2014) Induction of tetraploidy using high temperature exposure during the first zygote division in Populus adenopoda Maxim. Plant Growth Regul 72(3):279–287
Mable BK (2004) ‘Why polyploidy is rarer in animals than in plants’: myths and mechanisms. Biol J Lin Soc 82(4):453–466
Mao Y, Zhang P, Shi L, Dong C, Suo Y, Kang X (2013) The optimum conditions for inducing 2n pollen chromosome doubling by high temperature in Eucommia ulmoides. J Beijing Forest Univ 35(1):53–58
McCormick S (2004) Control of male gametophyte development. Plant Cell 16(suppl_1):S142–S153.
Okazaki K, Kurimoto K, Miyajima I, Enami A, Mizuochi H, Matsumoto Y, Ohya H (2005) Induction of 2 n pollen in tulips by arresting the meiotic process with nitrous oxide gas. Euphytica 143(1):101–114
Pan G, Zhou Y, Fowke LC, Wang H (2004) An efficient method for flow cytometric analysis of pollen and detection of 2n nuclei in Brassica napus pollen. Plant Cell Rep 23(4):196–202
Paterson AH, Bowers JE, Chapman BA (2004) Ancient polyploidization predating divergence of the cereals, and its consequences for comparative genomics. Proc Natl Acad Sci 101(26):9903–9908
Ramsey J (2007) Unreduced gametes and neopolyploids in natural populations of Achillea borealis (Asteraceae). Heredity 98(3):143–150
Ronceret A, Vielle JP (2015) Meiosis, unreduced gametes, and parthenogenesis: implications for engineering clonal seed formation in crops. Plant Reproduction 28(2):91–102
Sage TL, Bagha S, Lundsgaard-Nielsen V, Branch HA, Sultmanis S, Sage RF (2015) The effect of high temperature stress on male and female reproduction in plants. Field Crop Res 182:30–42
Scott R, Hodge R, Paul W, Draper J (1991) The molecular biology of anther differentiation. Plant Sci 80(1–2):167–191
Song X, Wang J, Sun P, Ma X, Yang Q, Hu J, Feng S (2020) Preferential gene retention increases the robustness of cold regulation in Brassicaceae and other plants after polyploidization. Horticult Res 7(1):1–15
Tang H, Bowers JE, Wang X, Ming R, Alam M, Paterson AH (2008) Synteny and collinearity in plant genomes. Science 320(5875):486–488
Taschetto OM, Pagliarini MS (2003) Occurrence of 2n and jumbo pollen in the Brazilian ginseng (Pfaffia glomerata and P. tuberosa). Euphytica 133(1):139–145.
Tian M, Zhang Y, Liu Y, Kang X, Zhang P (2018) High temperature exposure did not affect induced 2n pollen viability in Populus. Plant Cell Environ 41(6):1383–1393
Tondini F, Tavoletti S, Mariani A, Veronesi F (1993) A statistical approach to estimate the frequency of n, 2n and 4n pollen grains in diploid alfalfa. Euphytica 69(1):109–114
Veilleux R (1985) Diploid and polyploid gametes in crop plants: mechanisms of formation and utilization in plant breeding. AVI Publishing Co.
Wang C, Lentini Z, Tabares E, Quintero M, Ceballos H, Dedicova B, Zhang P (2011) Microsporogenesis and pollen formation in cassava. Biol Plant 55(3):469–478
Wang J, Li D-L, Kang X-Y (2012) Induction of unreduced megaspores with high temperature during megasporogenesis in Populus. Ann for Sci 69(1):59–67
Wang J, Kang X, Li D (2012) High temperature-induced triploid production during embryo sac development in Populus. Silvae Genetica 61(3):85–93
Wang J, Li D, Shang F, Kang X (2017) High temperature-induced production of unreduced pollen and its cytological effects in Populus. Sci Rep 7(1):1–12
Wu J, Shahid MQ, Guo H, Yin W, Chen Z, Wang L, Lu Y (2014) Comparative cytological and transcriptomic analysis of pollen development in autotetraploid and diploid rice. Plant Reprod 27(4):181–196
Xu FX, Chen DQ, Specht C (2013) Comparative microsporogenesis and anther development of selected species from Magnoliaceae. Nord J Bot 31(3):291–300
Yang J, Kang X (2015) Microsporogenesis and flower development in Eucalyptus urophylla × E. tereticornis. Breed Sci 65(2):138–144.
Yao PQ, Li GH, Long QY, He LG, Kang XY (2017) Microsporogenesis and induction of unreduced pollen with high temperatures in rubber tree clone RRIM 600. Forests 8(5):152
Ye TW, Li YM, Zhang J, Gong QY, Yuan DY, Xiao SX (2020) Ptimization of chromosome mounting technique and karyotype analysis of Camellia oleifera. J Nanjing Forest Univ 44(5):93–99
Younis A, Hwang Y-J, Lim K-B (2014) Exploitation of induced 2n-gametes for plant breeding. Plant Cell Rep 33(2):215–223
Zhang J, Liu XR, Zhang FX, Liu JX (2012) Microsporogenesis and development of the male gametophyte in Allium senescens L. (Liliaceae) in China. Plant System Evolut 298(9):1619–1624
Zhuang RL (2008) Camellia oleifera in China, 2nd edn. China Forestry Publishing House, Beijing
Acknowledgements
The authors would like to thank the staff of the anonymous reviewers and editors for their constructive comments.
Funding
The work has partially been financed by Hunan province Natural Science Foundation Youth Fund (2020JJ5969); Excellent Youth Scientific Research Project of Hunan province Education Department (18B170); Key project of Youth Science Foundation of Central South University of Forestry and Technology (201801030308); The National Key R&D Program of China (2018YFD1000603-1). The project was also supported by Key Laboratory for Germplasm Innovation and Utilization of Eco-economic Woody Plant Open Fund Project.
Author information
Authors and Affiliations
Contributions
ZH conceived the study; XZ and HT conducted the experiments; DY supervised the research; ZH wrote the manuscript; and all authors read, edited, and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The author(s) declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.

Rights and permissions
About this article
Cite this article
Zhang, X., Tong, H., Han, Z. et al. Cytological and morphology characteristics of natural microsporogenesis within Camellia oleifera. Physiol Mol Biol Plants 27, 959–968 (2021). https://doi.org/10.1007/s12298-021-01002-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-021-01002-5