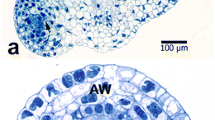
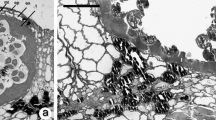
Abstract
This article describes the complete microsporogenesis and pollen formation in cassava (Manihot esculenta Crantz) at the various developmental stages (pollen mother cell, meiosis, tetrads, early free spore, mid uninucleate, late uninucleate, binucleate and mature pollen grain). Light microscopy, transmission electron microscopy and confocal laser scanning microscopy were used for the study. Floral bud size and other inflorescence characteristics were correlated with specific stages of the microspore development. This association allows a rapid selection of floral buds with similar microspore developmental stages, useful when a large number of homogeneous cells are needed for analysis and for in vitro induction of androgenesis. This article also compares methods for digestion the exine wall in microspores.
Similar content being viewed by others
Abbreviations
- CLSM:
-
confocal laser scanning microscope
- FISH:
-
fluorescent in situ hybridization
- DAPI:
-
4′-6-diamidino-2-phenylindole
- PMC:
-
pollen mother cell
- EFS:
-
early free spore
- T:
-
tapetum
- M:
-
middle layer
- EN:
-
endothecium
- E:
-
anther epidermis
References
Alves, C.A.A.: Cassava botany and physiology. — In: Hillocks, R.J., Tresh J.M., Bellotti A.C. (ed.): Cassava: Biology, Production and Utilization. Pp. 67–89. CABI Publishing, London 2002.
Barinova, J., Zhexembekova, M., Barsova, E., Lukyanov, S., Heberle-Bors, E., Touraev, A.: Anthirinum majus mircospore maturation and transient transformation in vitro. — J. exp. Bot. 53: 1119–1129, 2002.
Berry, P.E., Cordeiro, I., Wiedenhoeft, A.C., Vitorino-Cruz, M.A., Ribes de Lima, L.: Brasiliocroton, a new crotonoid genus of Euhphorbiacea from Eastern Brazil. — Syst. Bot. 30: 357–365, 2005.
Blackmore, S., Wortley, A.H., Skvarla, J.J., Rowley, J.R.: Pollen wall development in flowering plants. — New Phytol. 174:483–498, 2007.
Boavida, L.C., Becker J.D., Feijó J.A.: The making of gametes in higher plants. — Int. J. dev. Biol. 49: 595–614, 2005.
Ceballos, H., Fregene, M., Pérez, J.C., Morante, N, Calle, F.: Cassava genetic improvement. — In: Kang, M.S., Priyadarshan P.M. (ed.) Breeding Major Food Staples. Pp. 365–391, Blackwell Publishing, Ames 2007.
Ceballos, H., Iglesias, C.A., Pérez J.C., Dixon A.G.O.: Cassava breeding: opportunities. — Plant mol. Biol. 56: 503–515, 2004.
Chen, C.B., Xu, Y.Y., Ma, H., Chong, K.: Cell biological characterization of male meiosis and pollen development in rice. — J. Integr. Plant Biol. 47: 734–744, 2005.
Custódio, L., Carneiro, M.F., Romano, A.: Microsporogenesis and anther culture in carob tree (Ceratonia siliqua L.). — Sci. Hort.104: 65–77, 2005.
Da Silva, R.M., Bandel, G., Faraldo, M.I.F., Martins, P.S.: Biología reprodutiva de etnovariedades de mandioca. — Sci. Agr. 58: 101–107, 2001. [In Span.]
De Carvalho, R.D., Guerra, M.: Cytogenetics of Manihot esculenta Crantz (cassava) and eight related species. — Hereditas 136: 159–168, 2002.
De Souza, M.M., Pereira, T.N.S.: Development of pollen grain in yellow passion-fruit (Passiflora edulis f. flavicarpa; Passifloraceae). — Genet. mol. Biol. 23: 469–473, 2000.
Esau, K.: Anatomy of Seed Plants. — Wiley, New York 1977.
Fan, Z., Armstrong, K.C., Keller, W.A.: Development of microspores in vivo and in vitro in Brassica napus L. — Protoplasma 147: 191–199, 1988.
Goldberg, R.B., Beals, T.P., Sanders, P.M.: Anther development: basic principles and practical applications. — Plant Cell 5:1217–1229, 1993.
Halsey, M.E., Olsen, K.M., Taylor, N.J., Chavarriaga-Aguirre, P.: Reproductive biology of cassava (Manihot escultenta Crantz) and isolation of experimental field trials. — Crop Sci. 48: 49–58, 2008.
Heslop-Harrison, J.: Cytoplasm connections between angiosperm meiocytes. — Ann. Bot. 30: 221–230, 1966.
Hrabina, M., Jain, K., Gouyon, B.: Cross-reactivity between pollen allergens from common Pooideae grasses and cultivated cereals. — Clin. exp. Allergy Rev. 8: 18–20, 2008.
Huang, B., Bird, S., Kemble, R., Simmonds, D., Keller, W., Miki, B.: Effects of culture density, conditioned medium and feeder cultures on microspore embryogenesis in Brassica napus L. cv. Topas. — Plant Cell Rep. 8: 594–597, 1990.
Jennings, D.L.: Variation in pollen and ovule fertility in varieties of cassava, and the effect of interspecific crossing on fertility. — Euphytica 12: 69–76, 1963.
Kasperbauer, M.J., Wilson, H.M:. Haploid plant production and use. — USDA Technol. Bull. 1586: 33–39, 1979.
Kernan, Z., Ferrie, A.M.R.: Microspore embryogenesis and the development of a double haploidy protocol for cow cockle (Saponaria vaccaria). — Plant Cell Rep. 25: 274–280, 2006.
Khan, F.A., Ahmad, S., Siddiqui, S.A.: Microsporogenesis and development of male gametophyte in some Solanum species. — Beitr. Biol. Pflanz. 66: 1–7, 1991.
Kindiger, B., Beckett, J.B.: A hematoxilin staining procedure for maize pollen grain chromosomes. — Stain Technol. 60: 265–269, 1985.
Koltunow, A.M., Truettner, J., Cox, K.H., Wallroth, M., Goldberg, R.B.: Different temporal and spatial gene expression. — Plant Cell 2: 1201–1224, 1990.
Koti, S., Reddy, K.R., Kakani, V.G., Zhao, D., Reddy, V.R.: Soybean (Glycine max) pollen germination characteristics, flower and pollen morphology in response to enhanced ultraviolet-B radiation. — Ann. Bot. 94: 855–864, 2004.
Lauxen, M.S., Kaltchuk-Santos, E., Hu, C., Callegari-Jacquesi, S.M., Bodanese-Zanettini, M.H.: Association between floral bud size and developmental stage in soybean microspores. — Braz. Arch. Biol. Technol. 46: 515–520, 2003.
Lee, Y., Kim, E.S., Choi, Y. Hwang, I., Staiger, J., Chung, Y.Y., Lee, Y.: The Arabidopsis phosphatildylinositol-3-kinase is important for pollen development. — Plant Physiol. 147:1886–1897, 2008.
Li, D.X., Lin, M.Z., Wang, Y.Y., Tian, H.Q.: Synergid: a key link in fertilization of angiosperms. — Biol. Plant. 53: 401–407, 2009.
Liu, W., Zheng, M.Y., Polle, E.A., Konzak, C.F.: Highly efficient doubled-haploid production in wheat (Triticum aestivum L.) via induced microspore embryogenesis. — Crop Sci. 42: 686–692, 2002.
Mariani, C., Beuckeleer, M.D., Truettner, J., Leemans, J., Goldberg, R.B.: Induction of male sterility in plants by a chimaric ribonuclease gene. — Nature 347: 737–741, 1990.
Mascarenhas J.P.: The biochemistry of angiosperm pollen development. — Bot. Rev. 41: 259–314, 1975.
McCormick, S.: Control of male gametophyte development. — Plant Cell 16(Suppl.): S142–S153, 2004.
Nowack, M.K., Grini, P.E., Jakoby, M.J., Lafos, M., Koncz, C., Schnittger, A.: A positive signal from the fertilization of the egg cell sets off endosperm proliferation in angiosperm embryogenesis. — Nat. Genet. 38: 63–67, 2006.
Ospina, B., Ceballos, H.: La yuca en el tercer milenio.[Cassava in the Third Milenium.] — CIAT Publication, Cali 2002. [In Span.]
Owen, H.A., Makaroff, C.A.: Ultrastructure of microsporogenesis and microgametogenesis in Arabidopsis thaliana (L.) Heynh. ecotype Wassilewskija (Brassicaceae). — Protoplasma 185: 7–21, 1995.
Ressayre, A., Raquin, C., Mignot, A., Godelle, B, Gouyon, P.H.: Correlated variation in microtubule distribution, callose deposition during male post-meiotic cytokinesis, and pollen aperture number across Nicotiana species (Solanaceae). — Amer. J.Bot. 89: 393–400, 2002.
Reynolds, E.S.: The use of lead citrate at high pH as an electron-opaque stain for electron microscopy. — J. cell. Biol. 17: 208–213, 1963.
Rowley, J.R., Claugher, D., Skvarla, J.J.: Structure of the exine in Artemisia vulgaris (Asteraceae): a review. — Taiwania 44:1–21, 1999.
Scott, R.J., Spielman, M., Dickinson, H.G.: Stamen structure and function. — Plant Cell 16(Suppl.): S46–S60, 2004.
Scott, R., Hodge, R., Paul, W., Draper, J.: The molecular biology of anther differentiation. — Plant Sci. 80: 167–191, 1991.
Summers, W.L., Jaramillo, J., Bailey, T.: Microspore developmental stage and anther length influence the induction of tomato anther callus. — HortScience 27: 838–840, 1992.
Taskin, K.M., Turgut, K. Scott, R.J.: Apomeiotic pollen mother cell development in the apomictic Boechera species. — Biol. Plant. 53: 468–474, 2009.
Tomasi, P., Dierig, D.A., Backhaus, R.A., Pigg, K.B.: Floral bud and mean petal length as morphological predictors of microspore cytological stage in Lesquerella. — HortScience 34: 1269–1270, 1999.
Widholm, J.M.: The use of fluorescein diacetate and phenolsaphranine for determining viability of the cultured plant cells. — Stain Technol. 47: 189–194, 1972.
Acknowledgements
The excellent technical support on microscopy provided by José Arroyave is hereby recognized. Valuable comments by Dr. Stephen Blackmore on an earlier version of this manuscript have been incorporated. This work was supported through the grant project No. 2003 FS 121 and No. 2006 FS 062 by The Rockefeller Foundation, NY, USA, and Research Fellow Partnership Programme (RFPP), Switzerland.
Author information
Authors and Affiliations
Corresponding author
Additional information
Previously at International Center for Tropical Agriculture
Rights and permissions
About this article
Cite this article
Wang, C., Lentini, Z., Tabares, E. et al. Microsporogenesis and pollen formation in cassava. Biol Plant 55, 469–478 (2011). https://doi.org/10.1007/s10535-011-0112-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10535-011-0112-9