Abstract
Purpose
The Limoncocha lagoon, inside a Ramsar site in the Ecuadorian Amazon, increasingly hosts ecotourism and energy development activities. This work estimates the local sediments’ baseline of As, Ba, Cd, Co, Cr, Cu, Mo, Ni, Pb, V and Zn using four methods. This makes it possible to apply single and integrated pollution indices to estimate the contamination level and the ecological risk of the sediments.
Methods
Seven sites were analysed for metal(oid)s by ICP-MS. The local baseline was estimated using the relative cumulative frequency method, the iterative 2σ- and 4σ-outlier-techniques and the normalisation method to a “conservative” element. Combinations of single Cf and Ef and integrated indices (NPI, mNPI, mCd, MEQ, RI, mPELq, mERMq and TRI) were applied.
Results
The relative cumulative frequency method had the best performance for the geochemical baselines. Cf and Ef indices classify sediments from a “low contamination” remote site to the “moderate contamination” and “minor enrichment” of the remaining sites due to As, Cd and Zn concentrations. The overall analysis of the integrated indices results in Ni, Zn and Cu being identified as priority pollutants because they have occasionally been associated with adverse biological effects in the centre of the lagoon and in the most anthropised areas. These are classified as moderately polluted with a medium–low priority risk level.
Conclusions
This work provides sediment baseline and contamination indicators for pollutants, which could be incorporated into the sediment quality assessment and monitoring programme of the Limoncocha lagoon. Ni, Cd and Cr due to the high baseline values in comparison with SQGs, and Cu and Zn due to their potential risk, should be of special attention.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Wetlands are recognised as some of the most valuable green spaces due to their rich biodiversity and the high number of ecosystem services they provide as the essential building blocks for nature conservation (Dudley 2008; Hernandez-Gonzalez et al. 2019). Wetlands contribute to climate regulation and to climate change mitigation as they are responsible for carbon sequestration, improving water quality, flood control, aquifer loading and unloading, food supply, biodiversity maintenance and pollution control (Millennium Ecosystem Assessment 2005). Such complex environmental systems are supported on the equilibrium among environmental compartments such as water bodies, sediments, soils, air and biota, which are regulated by several controlling factors.
The sediment compartment is a relatively heterogeneous matrix of materials in terms of its physical, chemical and biological characteristics (Håkanson 1992). Bed sediments are formed by the deposition of organic and inorganic particles that accumulate on the bottom of water bodies such as lakes and lagoons. This environmental compartment plays an important role in aquatic ecosystems, influencing biogeochemical cycles and reflecting the current quality of the entire ecosystem (de Andrade et al. 2018). These natural components of the ecosystem are sinks, carriers and sometimes an important secondary source of pollutants in the environment, since some potential pollutants may be mobilised, through physicochemical and biological processes, to the water column (Nemati et al. 2011). Metals are a major cause for concern, due to their characteristics such as toxicity, persistence, bioavailability and bioaccumulation (Ali et al. 2019). Their occurrence in the environment results from natural and anthropogenic activities and can be associated with sediments.
Therefore, sediments, which require appropriate protection and management due to their high ecological value, may reflect the current quality of the entire ecosystem. Pollution indices are widely considered as explicit enforcement tools to quantify the degree of total metal pollution and assess the ecological risk of metals in sediments as well as in the environmental protection regulatory process (Alvarez-Guerra et al. 2007a, 2007b). In this context, the Ramsar Convention (or the Convention on Wetlands) was one of the first international conservation agreements in promoting globally wise use and management of wetlands. Kingsford et al. (2021) highlighted recently that wetlands of international importance require more attention focused on a vision and objectives, with regular reporting of key indicators to guide management and track ecological characteristics and ecosystem change effectively.
Many values of pollution indices are calculated based on local baseline and/or reference geochemical backgrounds and results can vary widely depending on the background used. Geochemical background values are a relative measure to distinguish between concentrations of natural elements or compounds and anthropogenically influenced concentrations in collective real samples (Matschullat et al. 2000; Dung et al. 2013). The geochemical background values correspond to the concentration of potentially toxic elements before industrialisation and/or the levels that reflect natural processes not influenced by human activities (Reimann et al. 2005; Teng et al. 2009; Wang et al. 2019a). However, due to natural variability, the generalised anthropic contribution caused by population growth, industrialisation or urbanisation, and the effects of global change, it is difficult to quantify a true background value beyond doubts (Karim et al. 2015).
An element’s local baseline provides the means to distinguish between the natural origin and the anthropogenic origin of the element in an environmental compartment (Teng et al. 2009). Local baselines represent surface material concentrations that are time and area specific (Wang et al. 2019a). The difference between the local baseline and the geochemical background value for a specific metal in the sediment is this: a background is the natural concentration of the metal, and a baseline is the concentration determined at a specific time and therefore includes natural and human influences (Wei and Wen 2012).
Knowledge of the baseline is essential to define contamination, identify the source of contamination and establish reliable criteria of environmental quality in sediments and other environmental compartments (Sojka et al. 2021). Lastly, it plays a crucial role in managing environmental pollution. However, developing the baseline is not straightforward, as there is no single standard or recommended methodology. Statistical methods are a widely used tool for the determination of the baseline in different environmental compartments. Reimann et al. (2018) established the geochemical background and thresholds for 53 elements at the European scale, analysing 2108 horizon soil samples in 33 countries under the GEMA project. The paper proposed and discusses several methods such as Median + 2MAD, the cumulative probability distribution (CPD) upper break in the data distribution, the 98th percentile and the Tukey inner fence (TIF) among others, as soil guideline values are specifically related to the risk-based values defined in some European countries for soil. The paper concludes that the CPD, 98th percentile and TIF methods are useful to estimate geochemical threshold values and identify locations with an unusually high concentration of elements. The paper by Reimann et al. (2018) discusses the use of the single Mean + 2SD method; this approach has been used by several authors (e.g. Matschullat et al. 2000; Reimann and Garret 2005), but it has also been considered outdated because it does not consider the multimodal nature of data sets, among other shortcomings (e.g. Reimann and Filzmoser 2000). However, the iterative 2σ-technique has been used by several authors (Matschullat et al. 2000; Zhuang et al. 2018; Wang et al. 2019a) to set up an approximated normal distribution around the mode value of the original data set. Sojka et al. (2021) consider 13 different statistical methods for the determination of the baseline of potential pollutants, including rare earth elements, from the sediments of 24 lakes of a natural park in Poland; Wang et al. (2019a) determine the baseline of lake sediment metals using the relative cumulative frequency method and the iterative 2σ-technique. Matschullat et al. (2000) propose two iterative statistical methods (the iterative 2σ- and 4σ-outlier-techniques) and the relative cumulative frequency method to determine the baseline values of different metals in different environmental compartments. The relative cumulative frequency method has been used to obtain the geochemical background and threshold of metals in sediments. The upper end of the relative cumulative frequency distribution (RCF) curve that “first deviates” from the regression line was chosen as the upper inflection point (Karim et al. 2015; Reimann et al. 2018).
The method of normalising element concentrations to a conservative element has also been widely used (Teng et al. 2009; Wei and Wen 2012; Zhou et al. 2019; Sojka et al. 2021). The procedure of choosing the appropriate Al concentration for normalisation in soils is described in detail in Birch (2020) and applied in the World Harbour Project (WHP) from a wide range of environments in multiple locations across the globe (Birch et al. 2020).
Once both the baseline and the reference element values have been defined, a high number of indices can be applied in order to assess the quality of the sediments under study. However, the assessment results have proven to be highly dependent on the reference values used (Maanan et al. 2015; Kowalska et al. 2018). Therefore, the use of a strategy based on the application of a combination of single and integrated indices founded on different and on-site specific criteria has been widely used in order to obtain a more accurate assessment of the studied area (Dung et al. 2013; Sakan et al. 2015; Saddik et al. 2019). Some of the most used indexes are the single indices based on the concentration of the reference element or/and the background or baseline concentration, such as the pollution factor (Cf) (Håkanson 1980), the geoaccumulation index (Igeo) (Müller 1969) and enrichment factor (EF) (Chen et al. 2007). Integrated indices can be divided into a different number of groups: based on background or baseline values, such as the mean enrichment quotient MEQ (Birch and Olmos 2008), the degree of contamination (Cd) (Håkanson 1980) and its modified version (mCd) (Abrahim and Parker 2008), the pollution load index (PLI) (Tomlinson et al. 1980) and the Nemerow pollution index (NPI) (Nemerow 1991); based on the reference element and the background or baseline concentration just like the modified Nemerow pollution index (mNPI) (Brady et al. 2015); indices including toxic response as the potential ecological risk index (RI) (Håkanson 1980); and indices based on the sediment quality guidelines (SQGs) such as the toxic risk index (TRI) (Zhang et al. 2016) and the mean SQGs quotients such as the mERMq and the mPELq (Long et al. 1995; Long and MacDonald 1998).
The Reserva Biologica Limoncocha (Ramsar site no. 956) in Ecuador is characterised by its abundant biodiversity comprising several ecosystems. The area surrounding the Limoncocha lagoon, which is the largest water body in the area, is a predominantly wet tropical forest (Ramsar 2020). The lagoon is the main tourism attraction in the reserve area, and facilities are being developed for ecotourism. Anthropic development including energy development – as manifested by increased oil exploration – as well as ecotourism is a driver of change in Amazonian ecosystems in general and the Limoncocha lagoon in particular (Anderson et al. 2019). The monitoring of contaminants and the environmental risk assessment are essential for environmental protection of the Limoncocha lagoon.
A recent study conducted by Carrillo et al. (2021) has shown the potential toxic elements in the soils and sediments of the Limoncocha Biological Reserve, but does not discuss freshwater sediment quality guidelines (SQGs) based indices in any depth. In the present work, two additional methods for the determination of the sediment baseline are discussed. In addition, the variability of the sediment pollutant contents (minimum and maximum obtained concentrations) is taken into account to analyse the environmental meaning of a higher number of applied indices more deeply. For that, we collected surface sediments from the Limoncocha lagoon, the largest body of water inside the Ramsar site Reserva Biologica Limoncocha, which is under increasing human pressure. The objectives of this work are (1) to estimate baseline values based on the distribution of background concentrations to identify locations with an unusually high element concentration of As, Ba, Cd, Co, Cr, Cu, Mo, Ni, Pb, V and Zn, using three statistical and conservative reference element methods, and (2) to determine the contamination level and to evaluate the ecological risk of the Limoncocha lagoon sediments using single and integrated pollution indices.
2 Materials and methods
2.1 Study area
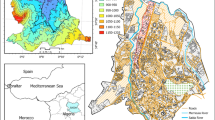
The Limoncocha lagoon (2.3 km2) is the main body of water in the Limoncocha Biological Reserve, a Ramsar site of 46.13 km2 located on the north-eastern side of the Ecuadorian Amazon, in the south-western region of the province of Sucumbíos (Fig. 1). The Pishira and Playayacu rivers drain into the Limoncocha lagoon that is connected to the difficult to access, a small body of water called Yanacocha, which is rarely visited due to the Kichwa community’s superstitions (Armas and Lasso 2011). Surrounding the lagoon, several ecosystems coexist highlighting the native wet tropical forest in the South and the agricultural mosaic on the North Bank, respectively.
The Limoncocha lagoon, which is very rich in fish stocks, is a source of natural resources to the indigenous Kichwa community to which a subsistence farming, mostly with banana and yucca cultivations, must be added. The Limoncocha lagoon is an increasingly popular tourist attraction in the reserve, and facilities are being developed for ecotourism as well as for research, education and environmental interpretation, attracting rising numbers of visitors. The region around the lagoon has experienced a very rapid and constant growth of population, agriculture (particularly palm), aquaculture and ecotourism since 2010, which has brought great pressure to bear in this protected environment.
The Limoncocha village, which is located in the eastern corner of the lagoon and had 6817 inhabitants in 2019, depends mainly on biodiversity to survive (GAD 2021); the elimination of excreta is carried out through latrines and in the open air with a very limited urban waste collection system in this village. A central production facility (CPF) of the Petroamazonas oil company is located northwest of the lagoon between the mouths of the Playayacu and Pishira rivers (Fig. 1).
2.2 Sample collection and analysis
Selection of the sampling sites was influenced by the requirements of accessibility, biogeophysical homogeneity, and areas with different degrees of anthropic influence. Seven sampling sites were chosen to provide a sampling density of 2.17 samples/km2 that is higher than most of the protected sites previously studied (see Table 1), leading to a broad spatial coverage of the Limoncocha lagoon. The sediment sampling sites (SE1–SE7) and their detailed description are shown in Fig. 1; SE1, SE3, SE4 and SE5 are stations positioned along the banks of the Limoncocha lagoon and confluent with the mouth of the Playayacu and Pishira rivers. SE2 is located in the centre of the lagoon, and SE6 and SE7 are located in the CPF at the headwaters of the Playayacu and Pishira rivers, respectively. Two surface (5 cm depth) sediment samples from each sampling station selected (Fig. 1) were collected; each of them was taken in the form of mixed composite samples within a square of 1 m2 by using a plastic spatula. The sample from the central area of the lagoon (SE2) was collected using a plastic core sampler with a surface layer of 5 cm for the analysis. Sediments were collected once in the rainy season (December 2018) and again in the dry season (July 2019), totalling 28 surface sediment samples. The samples were stored in closed plastic containers previously washed with 1:1 HNO3 and rinsed with distilled water. The samples were kept on ice until arrival at the laboratory. Once in the laboratory, the samples were dried at room temperature; then, they were ground and passed through a 230-mesh sieve. Grain size fraction (< 63 µm) was homogenised and stored in polyethylene plastic bags.
The analysis of the major elements Al, Fe, Mn, Ca, K, Mg, Na, P, Si and Ti was carried out at Activation Laboratories Ltd., in Canada, using the major elements fusion ICP package (Code WRA/ICPOES-4B). The lithium metaborate/tetraborate fusion technique was employed. The resulting molten bead was rapidly digested in a weak nitric acid solution, and the analysis was carried out using ICP and ICPMS. Certified reference materials NIST 694, DNC-1, W-2a, SY4 and BIR-1a were used to obtain recoveries from 86.2 to 104.3%.
The rest of metal (Ba, Cd, Co, Cr, Cu, Mo, Ni, Pb, V and Zn) and metalloid (As) concentrations were determined using ICP-MS (Thermo iCAP-Q) in our laboratories. The metals were extracted with a microwave acid digestion system (CEM model Mars 5) according to the SW-846 EPA Method 3051A (USEPA 1998), which involved the digestion of 0.1 g of a sediment sample with a mix of 9 mL of nitric acid (65%, Suprapur quality) and 3 mL of hydrochloric acid (30%, Suprapur quality) in Teflon vessels. After the digestions, the samples were diluted to 50 mL using Milli-Q water and then analysed. The accuracy of the analysis was checked with certified reference materials. The best certified material recoveries of each analyte assessed are shown in Table 2.
2.3 Establishing local baseline
In the present study, the local baseline is determined considering four methods: the relative cumulative frequency method, the iterative 2σ-technique, the iterative 4σ-outlier-technique and the normalisation method to a “conservative” element.
2.3.1 Relative cumulative frequency method
The relative cumulative frequency method is based on the different slopes of the relative cumulative frequency–content of elements fitting curves of the sampling sites (Wei and Wen 2012; Wang et al. 2019c). Zero, one or two inflections appeared in the fitting curves of all of the sampling sites to separate the unpolluted and polluted samples. If the cumulative frequency curve approximated a straight line and no inflection point existed, the baseline value is calculated from the average value of all the data. If only a higher inflection point is found, then the baseline is obtained from the data below the inflection value. If two inflections occurred, the lower inflection point represents the upper limit of the natural origin concentrations, and the higher inflection point represents the lower limit of abnormal concentrations (Zhou et al. 2019). The Kolmogorov–Smirnov test was applied to check the normal distributions of the concentrations (or the logarithm of the concentrations) (Karim et al. 2015; Tian et al. 2017). To determine the baseline value, a linear regression method described by Wei and Wen (2012) was employed. The inflection with low content represents the upper limit of the natural background concentrations, and the baseline is obtained from the data below the inflection value. In this study, low inflections were determined using linear regression of the cumulative frequency method, the data sets in the cumulative frequency were tested for linearity of distribution, the maximum value was removed to obtain the linearity of the data (this procedure was repeated until the two criteria above were met), and the maximum value within the regression line will represent low inflection. The condition of the linearity of distribution in this study was p < 0.05 and R2 > 0.95, proposed by several authors (Karim et al. 2015; Tian et al. 2017; Niu et al. 2019; Zhou et al. 2019; Jiang et al. 2020). The average element concentrations of these data were selected as the local baselines providing values that allow managers to monitor sediment quality under a conservative approach rather than considering the inflection point value as the threshold to take actions such as dredging and/or remediation.
2.3.2 Iterative 2σ-technique
The mean and standard deviation is calculated for the original data set. All values beyond the mean ± 2σ interval are omitted. This procedure is repeated until all the remaining values lie within this range. The mean ± 2σ calculated from the resulting subcollective is considered to reflect the local baseline. This technique set up an approximated normal distribution around the mode value of the original data set (Matschullat et al. 2000; Zhuang et al. 2018; Wang et al. 2019a). The average element concentrations resulting from the mean ± 2σ were selected as the local baselines.
2.3.3 Iterative 4σ-outlier-technique
The iterative method 4σ-outlier-technique defines the geochemical baseline of the elements within the normal distribution range from a mathematical point of view. First, the mean value and standard deviation (σ) of the initial data column are calculated, and values beyond the range of [mean ± 4σ] are dropped. Subgroups of values are generated from the remaining data set to eliminate possible outliers from it. The mean ± 2σ of this subgroup will be the normal range for the baseline (Matschullat et al. 2000). Subgroups are created to take into account the value of the frequencies, selecting the lowest values within the subgroup and eliminating the remaining subgroups.
2.3.4 Normalisation method
The measured element concentrations are normalised to a “conservative” element. This is done by linear regression so that the baseline can be estimated for each point that confirms the regression conditions. Samples that exceed the confidence interval (95%) will be classified as anthropogenically influenced. They are removed from the data set, and a new linear equation is constructed with the updated data set until all data falls within the 95% confidence band. The normalising element must be an important component of one or more of the main trace metal carriers and reflect its granular variability in the sediments; therefore, the elements of Al, Ti, Fe, Y, Eu, Ce, Sc and others are commonly used as elements of standardisation (Teng et al. 2009; Wei and Wen 2012; Zhou et al. 2019). In this study, an analysis and a discussion of results are performed to select type and concentration of the conservative element.
2.4 Sediment contamination and potential risk assessment methodology
Table 3 shows a detailed description of the indices considered to estimate the contamination level and the potential ecological risk of the potentially toxic elements in the sediments of the Limoncocha lagoon studied. From the numerous available indices for the classification of sediments site (j) concerning potentially toxic elements pollution (i), a combination of single and integrated risk assessment indices was used in the present study, to determine the state of contamination in the Limoncocha lagoon and tributaries. The contamination factor (\({\mathrm{Cf}}_{i,j}\)) and the enrichment factor (\({\mathrm{Ef}}_{i, j}\)) are used as individual indices to calculate the degree of contamination of each potentially toxic element at a given site. However, these sediment quality indicators either define a qualitative threshold or focus on the ecological risk assessment of a single element. Potentially toxic element pollution in the environment generally occurs in the form of complex mixtures. Therefore, the mean enrichment quotient (MEQ), the modified degree of contamination (mCd), the Nemerow pollution index (\({\mathrm{NPI}}_{j}\)), the modified Nemerow pollution index (\({\mathrm{mNPI}}_{j}\)), and the potential ecological risk index (\({\mathrm{RI}}_{j}\)) are used as integrated indices to determine the degree of contamination of the set of pollutants at a given site. In addition, the evaluation of the sediment quality with respect to potentially toxic elements is determined using indices based on the freshwater sediment quality guidelines (SQGs). In this approach, the probable median effect level quotient \({\mathrm{mPELq}}_{\mathrm{j}}\), the effective range median mean quotient \({\mathrm{mERMq}}_{\mathrm{j}}\) and the toxic risk index, \({\mathrm{TRI}}_{j},\) based on TEL and PEL guidelines have been used. The mean SQGs quotients have been widely used to determine the indicative toxicity of lake and lagoon sediments as a simple and easily understandable numerical index based on SQGs for metals in freshwater ecosystems (Alvarez-Guerra et al. 2010; Hu et al. 2018; Soliman et al. 2019; Zhuang et al. 2019; Christophoridis et al. 2020; Magesh et al. 2021).
Hierarchical cluster analysis (HCA) was used to identify groups of sediment samples according to the values of the eight integrated indices, which were represented visually as dendograms of the sampling stations using Ward’s method with squared Euclidean distance as a measure of similarity. Statistical analyses were performed with the aid of the statistical software Minitab®.
3 Results and discussion
3.1 Major and potentially toxic elements in the sediments from the lagoon and tributaries
The concentration of the major and the potentially toxic elements in the sediments set as mean ± σ and median ± MAD values, together with the concentration range, 1st and 3rd quartile, CV and CV* statistical parameters are shown in Table 4. CV* is a robust, non-parametric estimate that it is not affected by the presence of outliers (Reimann and De Caritat 2005). The order of variability of the distribution of the trace elements (CV*) is Co (7.42%) < V (7.71%) < Ni (10.2%) < Mo (11.5%) < Ba (13.9%) < Cu (17.8%) < Cr (20.7%) < Zn (23.1%) < Pb (25.1%) < As (31.1%) < Cd (35.7%). Note that only As and Cd show higher CV* values than the highest values of the major elements (K (27.8%), Ti (27.4%) and Mg (26.4%)). This suggests that the variability of the potentially toxic elements can be considered low except for As and Cd.
Other authors (Guo et al. 2012; Karim et al. 2015; Niu et al. 2019) suggest using the coefficient of variation (CV) to study the variability in the distribution of the elements. They consider relatively low CV values of the elements dominated by natural sources, while the CV values of elements affected by anthropogenic sources are quite high (Guo et al. 2012; Niu et al. 2019). These authors classify the degree of variability as CV < 20% with low variability, 20% ≤ CV ≤ 50% with moderate variability, CV > 50% with high variability and CV > 100% with exceptionally high variability (Qing et al. 2015; Niu et al. 2019). In the present study, according to this classification, Mo (17.3%) shows a low degree of variability; Co (21.6%), Cu (21.7%), Cr (24.3%); Ba (30.6), Mo (33.1%) and Pb (37.0%) indicate a moderate degree of variability; and As (56.0%), Cd (58.2%) and Zn (62.5%) a high variability. Most of the major elements studied show a moderate degree of variability with P (21.2%), Fe (22.8%), Ti (26.0%), Mg (27.3%), Na (27.8%), K (34.2%) and Mn (48.8%). The low CV and CV* values of Al (CV = 7.05%, CV* = 6.75%), below 10% and below the CV and CV* values of the potentially toxic elements, indicate a low anthropogenic contribution, which is why Al can be considered the conservative element.
In the present work, only the fraction with a particle size smaller than 63 µm was analysed. This fraction is the most common of those used in the analysis of metals in sediments, and its use constitutes a standardisation method, which is especially useful if you want to make comparisons. The mean value of the Al concentration obtained during the two sampling campaigns for the analysed fraction (100% fine fraction) in each sampling station is what has been used as a normalisation element. This strategy is similar to some of the normalisation measures reported in the work of Birch et al. (2020). The Al content has not been made at a different percentage of fine fraction, and therefore, it should be used as a normaliser element cautiously. However, the relative similarity in regional distribution of geological formation has been previously shown, with the same type of soil with shales and sandstones as the geological formations present on the surface around the Limoncocha lagoon (Jarrin et al. 2017; GAD 2021). Analysis of the Al content at different percentages of fine fraction is needed in future studies to obtain more exact normalised values for each sampling site or even provide a common Al value for the study area.
The concentrations of most of the potentially toxic elements in the sediments of the Limoncocha lagoon and its tributaries are of the same order as other wetland and lagoon protected areas worldwide (Table 1). The concentration ranges of the elements studied differ by almost one order of magnitude in each of the protected areas shown in Table 1. The concentration values of Cu and Zn obtained from the Limoncocha lagoon are mostly higher than those observed in other protected areas, probably due to the antrophogenic activities in some areas of the lagoon. Special attention is paid to Ba and V that show concentrations that are one order of magnitude higher than the rarely reported values in protected areas (Castro et al. 2013; Sojka et al. 2021). Among other metals, V has been referenced as an indicator of oil spill pollution (Ogunlaja et al. 2019; Pratte et al. 2019; Sadeghi et al. 2019). High Ba concentrations have been correlated with the location of oil wells and are probably related to the use of barite (BaSO4) in drilling muds (Sharma et al. 1999; Yang et al. 2015). The relatively high concentrations obtained for these metals in the sediments may be related to the oil well drilling and extraction activities in the Limoncocha Biological Reserve (Fig. 1). Despite the levels of the concentrations analysed, a local baseline needs to be established to assess potential contamination by toxic elements. This would provide the means to distinguish between their natural or anthropogenic origin.
3.2 Establishing local sediment baselines for environmental management in the Limoncocha lagoon
The authors tested the TIF, Median + 2MAD, and P98 methods suggested by Reimann et al. (2018), in the present work. The three methods overestimate the baseline values for several of the potential contaminants studied (Table S1 and Fig. S1 in Supplementary Information). Using the P98 method, the baseline values are close to the maximum experimental value obtained; the CV between P98 and Maximum values range from 1.6 to 0.13%. This prevents us from using these methods in the present work.
The frequency and cumulative frequency as functions of the concentrations of the potentially toxic elements are shown in Fig. 2. The baseline values obtained from the four methods proposed (highlighted in coloured circles) are within the linear range of the relative cumulative frequency curve (white circles) for each element studied.
Frequency (in bars) and cumulative frequency (circles) versus the concentration of potentially toxic elements. The white circles () indicate the linear interval of the relative cumulative frequency curve and the coloured circles the baseline values obtained using the different statistical methods: (blue circle) relative cumulative frequency method, (red circle) iterative 2σ-technique, (yellow circle) iterative 4σ-outlier-technique, (green circle) normalisation method
The graphical determination of the baseline values by different methods is illustrated through the case of V in Fig. 3, while all the elements studied are shown in Figs. S2–S4 in the Supplementary Information. Figure S2 illustrates the concentration (Cd, Co, Cr, Cu, Ni and V elements with a positive Kolmogorov–Smirnov test) or the concentration logarithm (As, Ba, Mo, Pb and Zn) versus the relative cumulative frequency curve. In the present work, the concentration set of As, Ba, Mo, Pb and Zn shows a data distribution slightly skewed to the right, which is much slighter than that shown in Reimann et al. (2005), as shown in Fig. S3. The red circle indicates the local baseline value as the average concentration of the linear section under the inflection point of the curves that is displayed in Fig. 3a for V. Figure 3b and Fig. S4 represent the frequencies by classes and the cumulative frequency versus the concentration classes using the iterative 4σ-outlier-technique. The baseline value is determined from the grey bars, which correspond to the lower subgroups, as the mean ± 2σ of this subgroup. The orange arrow indicates the baseline concentration class calculated with this method. Figure 3c and Figure S5 show the concentrations of the potentially toxic elements against Al, once the outliers have been eliminated. The green circles represent the baseline values obtained by this method, calculated as the mean value of the subgroup of concentrations represented in the graphs (concentrations of potentially toxic elements after the outliers are removed).
Table S2 in the Supplementary information shows the concentration values and ranges for each local baseline obtained from the four statistical methods considered in this study. In the case of the relative cumulative frequency (RCF) method, the iterative 2σ-technique and the normalisation method, the baseline values correspond to the mean value of the range. However, for the iterative 4σ-outlier-technique, the baseline value corresponds to the mean ± 2σ. Table S2 also includes information about the standard deviation (σ), the coefficient of variation (CV) and the coefficient CV*. The lowest joint values of CV and CV* are obtained using the relative cumulative frequency method, except for Mo with the iterative 2σ-technique and Zn with the iterative 4σ-outliers technique. However, the selection of the relative cumulative frequency method in contrast to the 2σ- and 4σ-outlier-iterative techniques entails Mo and Zn baselines less than 7% higher. Therefore, the values obtained by means of the relative cumulative frequency method are selected as a baseline for the determination of the contamination indices. The gaps in the cumulative frequency baseline, in comparison with the other methods and measured as a percentage, are also shown in Fig. 4. It may be observed that except for As, the gaps are lower than ± 20% (red dashed lines). Gaps higher than ± 10% (green dashed lines) are shown for Zn using the iterative 2σ-technique; Ba, Mo and Pb use the iterative 4σ-outlier-technique and Mo with the normalisation method.
Another possible approach to assess the quality of the sediments from pollution indices is to use the average composition of the upper continental crust (UCC) as a direct geochemical background. Three global reference values as the average distribution of the elements in the earth’s crust for the sedimentary rocks (Turekian and Wedepohl 1961) and the average compositions of the upper continental crust proposed by Taylor and McLennan (1995) and Rudnick and Gao (2003) were used to compare against the RCF local baseline values obtained (Table S3). On the one hand, the RCF values of Ba, Cr and Pb are lower than the UCC values; on the contrary, Cd, Cu and Zn RCF values are higher than all the UCC values considered; finally, the As, Co, Mo, Ni and V values are lower or higher depending on the UCC reference. On the other hand, the geochemical background is not a constant value over time due to the natural processes taking place at the interface of water and sediment. Additionally, the geochemical background value is local or regional rather than global, and the value adopted affects the quality of the assessment of the local environment. A strategy based on the application of a combination of single and integrated indices founded on a local baseline instead of a global reference is used in the present work. The local baseline is the concentration determined at a specific time and therefore can include natural and human influences; further, a local baseline establishes reliable criteria for the continuous assessment and monitoring of a sediment’s quality. This strategy has been widely adopted in order to obtain a more accurate assessment of the area studied (Dung et al. 2013; Maanan et al. 2015; Sakan et al. 2015; Kowalska et al. 2018; Saddik et al. 2019).
3.3 Environmental risk of potentially toxic elements in sediments of the Limoncocha lagoon
From the numerous indices available to assess the pollution level and the potential toxicity of mixtures of contaminants in sediments, as well as for their classification as to potentially toxic elements pollution, the widely used and well-documented Cf, Ef, NPI, mNPI, RI, mCd, MEQ, mPELq, mERMq and TRI indices were employed in the present study (Table 3).
Two single indices were applied: the contamination factor (Cf), based on the element baseline value, and the enrichment factor (Ef), which additionally takes into account the concentration as well as the baseline of a reference element. In addition, eight integrated indices were used: the modified degree of contamination mCd and the Nemerow pollution index (NPI) which integrated Cf in their formulations; the mean enrichment quotient (MEQ) and the modified Nemerow pollution index (mNPI) based both on the Ef instead of the Cf and the potential ecological risk (RI) which takes into account the toxic response factor for each element studied; and finally the mPELq, mERMq and TRI indices obtained without the use of baseline values but based on the widely applied freshwater SQGs. Figure 5 shows the values of the indices studied and the classification at each sampling site using a colour code. Values of the single and integrated indices obtained are shown in the Supplementary Information (Table S4-S5).
Sediment pollution indices and their classification at each sediment site. Cf:
 Cf < 1 low contamination,
Cf < 1 low contamination,
 1 < Cf < 3 moderate contamination; Ef:
1 < Cf < 3 moderate contamination; Ef:
 Ef < 1 no enrichment,
Ef < 1 no enrichment,
 1 < Ef < 3 minor enrichment; NPI:
1 < Ef < 3 minor enrichment; NPI:
 0.7 < NPI < 1 warning limit,
0.7 < NPI < 1 warning limit,
 1 < NPI < 2 slight pollution,
1 < NPI < 2 slight pollution,
 2 < NPI < 3 moderate pollution; mNPI:
2 < NPI < 3 moderate pollution; mNPI:
 mNPI < 1 unpolluted,
mNPI < 1 unpolluted,
 1 < mNPI < 2 slightly polluted,
1 < mNPI < 2 slightly polluted,
 2 < NPI < 3 moderate pollution; mCd:
2 < NPI < 3 moderate pollution; mCd:
 mCd < 1.5 nil to very low degree of contamination,
mCd < 1.5 nil to very low degree of contamination,
 1.5 < mCd < 2 low degree of contamination; MEQ:
1.5 < mCd < 2 low degree of contamination; MEQ:
 MEQ < 1.5 category 1 (no enrichment),
MEQ < 1.5 category 1 (no enrichment),
 1.5 < MEQ < 3 category 2 (minor enrichment); RI:
1.5 < MEQ < 3 category 2 (minor enrichment); RI:
 RI < 1 low potential risk; mPELq::
RI < 1 low potential risk; mPELq::
 0.1 < mPELq < 1.5; mERMq:
0.1 < mPELq < 1.5; mERMq:
 0.1 < mERMq < 0.5 medium–low priority risk level; TRI:
0.1 < mERMq < 0.5 medium–low priority risk level; TRI:
 TRI < 5 no toxic risk,
TRI < 5 no toxic risk,
 5 < TRI < 10 low toxic risk
5 < TRI < 10 low toxic risk
Values of \({\mathrm{Cf}}_{i, j}\)< 3 were found for all the contaminants, indicating “low contamination” or “moderate contamination” for all the contaminants and sampling sites (Fig. 5). “Moderate contamination” (1 < \({\mathrm{Cf}}_{i, j}\) < 3) was found for As and Pb at SE1, SE2, SE3 and S4; meanwhile, the remaining sites show moderate contamination due to the different combination of metals without a common pattern (Table S4). At site SE5, all trace elements show “low contamination,” while sites SE1 and SE2 show the lowest number of elements with “low contamination”. The Cf of V and Co are the main elements responsible for characterising the highest number of sites (six and five sites respectively) as “moderate contamination” although three values of Cf for V are close to a value of 1. If the minimum concentration analysed in the sediments is taken into account, just As, Mo and Pb at the SE2 site show Cf > 1.5 (Table S4).
The \({\mathrm{Ef}}_{i,j}\) values for the sediment samples in this study are lower than 2.4. The classification varied from the “non-enriched” S5 sample to the remaining sediments classified as samples with “minor enrichment” (Fig. 5). The elements responsible for the “minor enrichment” were a different combination of metals without a common pattern, as the Cf behaviour indicates (Table S4).
According to Zhang and Liu (2002) and Luo et al. (2019), an \({\mathrm{Ef}}_{\mathrm{i},\mathrm{j}}\) >1.5 suggests that the source of the metals is more anthropogenic than from crustal origin. The SE1 site (due to As and Cd), the SE2 site (due to As, Cd, Zn, Pb, Mo) and the SE6 site (due to Zn and Ba) could be classified as sites contaminated by metals with a possible anthropogenic origin. If the minimum concentration analysed is taken into account, just As at the SE1 and SE2 sites, and Mo and Pb at the SE2 site show Ef > 1.5 (Table S4).
Cf and Ef classify the sediment sites with the same contamination level, showing the same elements as being responsible for that categorisation. The number of elements responsible for the contamination at each study area leads to a decreasing order SE2 = SE1 > SE6 = SE7 = SE4 > SE3 > > SE5, where the centre of the lagoon and the most anthropised areas have a greater number of potential toxic elements in the sediments than the most remote areas. As and Zn have the highest values in both indices indicating that these elements are more likely to be anthropogenic. Despite this analysis, the low Cf and Ef values obtained suggest that the possible sources of the element studied may be of various natures (anthropogenic and crustal), making additional temporal series necessary to fix the potential sources.
The mNPI index allows more sediment class graduation than NPI and uses an enrichment factor which takes into consideration diverse sediment behaviour due to the use of a normalisation element (Brady et al. 2015; Duodu et al. 2016). However, both the NPI and mNPI integrated indices put the stations into three groups, with SE2 as “moderately polluted”, S5 as “unpolluted” or on the “warning limit” and the remaining sites (SE1, SE3, SE4, SE6 and SE7) as “slightly polluted”. In contrast, RI, mPELq, mERMq and TRI classify homogeneously all the sediment stations as “Low potential risk (RI < 150)”, “Medium–low priority risk level (0.1 < mERMq < 0.5 and 0.1 < mPELq < 1.5)” and “Low toxic risk (5 < TRI < 10)”; these classifications remain uniform although the maximum concentrations analysed were used to obtain the indices (Fig. 5 and Table S4). The mCd and MEQ indices are very alike due to the similarity of the Cf and Ef values they are based on respectively. Both indices classify all stations as having nil to a very low degree of contamination, using mCd and category 1 (no enrichment). Examining the results from MEQ, the exception is SE2 that a very small margin exceeds this first category and is classified as a low degree of contamination and category 2 (minor enrichment).
Most sediment sites have an index of RI < 150 and \({\mathrm{Er}}_{i,j}\)< 40, indicating a low ecological risk. Only the average concentrations of Cd at SE1 and SE2 and the maximum concentrations at SE4 and SE7 result in Er > 40 but maintain RI < 150 (Table S4), which is a warning with respect to the potential effects of this metal. The RI index, that takes into account the toxicity of the elements when assessing the risk, mainly matches with mERMq, which allows us to assess the potential toxicity of the mixtures of contaminants in sediments to receptor benthic organisms. The highest ecological risk factor \({\mathrm{Er}}_{i,j}\) corresponds to the elements Cd and As at all the stations following a decreasing order similar to the toxic response factor values that Tri shows in Table 3.
The probable median effect level quotient mPELq and the effective range median mean quotient mERMq, as well as the toxic risk index (TRI), were used to gauge the potential biological effects of the composite potentially toxic elements: As, Cd, Cr, Cu, Ni, Pb and Zn on freshwater sediments based on individual threshold effect and probable effect concentrations (Long and MacDonald 1998; MacDonald et al. 2000). A comparative analysis of the indices is applied, which is based on SQGs as shown in Fig. 6 and Table S4.
The mPELq values that represent the concentrations above which adverse effects are expected to occur frequently are all below the individual PEL value. All index values, even those obtained using maximum concentrations, fall within the range of 0.1 < mPELq < 1.5, thus classifying the stations as “Medium–low priority risk level” and following the decreasing sequence SE2 > SE6 = SE1 > SE7 > SE4 = SE3 > SE5. However, values between TEL and PEL are obtained due to Cu at all stations, to Ni at all stations except SE5, due to As and Cd at SE1 and SE2, Zn at SE2 and SE6 and due to Cr at SE4. These results rank these stations as sites and pollutants “that can occasionally be associated with adverse biological effects”. As is shown in Fig. 6, \({\mathrm{PEL}}_{i, j}\) values of Cd and Pb are the lowest at all stations, followed by As at four stations. On the contrary, Ni shows the highest \({\mathrm{PEL}}_{i, j}\) at all stations except at SE2 where the Zn quotient is the highest.
In relation to mERMq, all the index values, even those obtained using maximum concentrations, fall within the range 0.1 < mERMq < 0.5 classifying the stations as “Medium–low priority risk level” and follow a decreasing sequence of SE2 > SE6 > SE1 > SE7 > SE4 > SE3 > SE5 similar to mPELq. The highest values of mERMq are found at SE2 and SE6 where Zn is the main element responsible for such high values. Values between ERL and ERM are obtained due to Cu at all stations and Zn at the SE2 and SE6 stations, ranking these stations as sites and pollutants “that can occasionally be associated with adverse biological effects”. The ecological risk decreases in the following sequence Ni > Cu ≈ Zn > > Cr > Pb ≈ As ≈ Cd, as is common at most of the sediment sites (Fig. 6).
The toxic risk index (TRI), which assesses the integrated toxic risk based on both the TEL and PEL effects of pollutants (Zhang et al. 2016; Gao et al. 2018), shows values in the range of 5 ≤ TRI < 10 classifying all stations as a “low toxic risk” except SE5 that shows no toxic risk (Fig. 5). The TRI values by station followed a decreasing sequence of SE2 > SE1 > SE6 > SE7 > SE4 = SE3 > SE5 similar to the previously applied quotients. The TRIij values of Pb and Cd are the lowest at all stations, but contrary to these quotients, Cu shows the highest values at all stations except at SE6 where the Zn index is slightly higher. The significant contribution of Cu to the TRI was ascribed mainly to its relatively low TEL (Gao et al. 2018).
Neither TEL-PEL nor ERL-ERM takes into account baseline concentrations as a reference. In the Limoncocha lagoon sediments, the Ni baseline value exceeded TEL, and the Cd and Cr baselines were close to the TEL value. Therefore, even without the contribution of human activities, the concentrations of Ni, Cd and Cr would be considered high.
The indicators used in the present work to gauge the potential biological effects of the composite potentially toxic elements (RI index, mPELq, mERMq and TRI) are used as an initial approach to the environmental risk assessment in the absence of direct biological effects and bioavailability data. The mobility, bioavailability and the toxicity of elements in an ecosystem mainly depend on abiotic and biotic factors (Salomons and Förstner 1984; Saher and Siddiqui 2019). Bioavailability depends mostly on a sediment’s physicochemical characteristics, element speciation and concentration, the real environmental conditions in contact with sediments, the interactions and characteristics of the chemicals occurring in complex mixtures as well as on the way living organisms take up any pollutants (e.g. feeding strategy, metabolism rate). In the present work, the total chemical analysis of the sediment fine fraction smaller than 63 µm was considered to be an adequate estimate of exposure. Adjustments to account for bioavailability or chemical speciation can improve exposure estimates (Wenning et al. 2005; Alvarez-Guerra et al. 2010; Lécrivain et al. 2018). To determine the potential mobility, bioavailability and toxicity of the elements examined, more extensive studies including the analysis of element speciation, as well as the temporal variability in the sediments’ characteristics, are proposed. The determination of the bioavailable fraction of elements by sequential extraction or simultaneous extractable metal acid volatile sulphides procedures would be useful for this purpose. The indices applied indicate that the sediment poses low potential risk with a medium–low priority risk level; however, some elements occasionally are associated with adverse biological effects at some of the sites studied. In view of this, further investigation of heavy metal speciation and bioavailability is required to ascertain the extent of pollution in the study area.
The result of the cluster analysis conducted on the sampling sites according to the integrated indices calculated (Fig. 7) shows the formation of two major clusters, both of them with two different subgroups. The first cluster, with an 86.2% of similitude, comprises the most contaminated sediments at SE1 (located at a pier with high anthropic activity), SE6 (located at the head of the Playayacu river) and SE2 (located in the centre of the lake, which acts as a sink for contaminants) with the highest index values. SE1 and SE6 are clustered with more than 90% similitude, whereas SE2 is in a differentiated subcluster showing a different pollution classification due to the NPI, mNPI, mCd and MEQ indices. As, Cd, Cu, Ni and Zn are the elements mainly responsible for their “minor enrichment”, “moderate contamination” and “medium–low priority risk level” at these sediment sites. The second cluster, with a 79.6% of similitude, includes the low and moderately contaminated sampling stations, which are clustered in two subgroups according to their sediment index values. One subcluster with more than 99% of similitude is formed with sediments from SE3, SE4 and SE7 with an intermediate grade of contamination. These sediments were collected from the head and mouth of the river with a similar anthropogenic influence. The second subcluster includes SE5 that has the sediment with the lowest contamination, which is located in a remote area, accessible only by water and which connects to the small water body called Yanacocha (Fig. 1).
4 Conclusions
The levels of As, Ba, Cd, Co, Cr, Cu, Mo, Ni, Pb, V and Zn, in sediments of the Limoncocha lagoon, a body of water inside a Ramsar wetland of International importance, have been determined in this work. Local baselines of the potentially toxic elements studied have been determined by four methods, taking into consideration statistical and reference element approaches: the relative cumulative frequency method, the iterative 2σ-technique, the 4σ-outlier-technique and the normalisation method to Al as the “conservative” element. The comparative analysis of the baselines led to the conclusion that the relative cumulative frequency method, used for the determination of the contamination indices, worked better.
Single indices contamination factor (Cf) and enrichment factor (Ef) classify the sediment with the same contamination level, from “low contamination” and “non-enriched” at the remote SE5 site to “moderate contamination” and “minor enrichment” at the remaining sites. The elements responsible for such categorisation are mainly As, Cd and Zn, which demonstrate a high variability and the highest values at the centre of the lagoon. The most anthropised areas and the centre of the lagoon have a greater number of potential toxic elements in the sediments than the more remote areas. The potential ecological risk (RI) integrated indices obtained the mean quotients of mPELq and mERMq and TRI and homogeneously classify all the sediment stations as “Low potential risk” or “Medium–low priority risk level”. On the contrary, the NPI and mNPI indices differentiate three groups of stations, with the site in the centre of the lagoon as “moderately polluted”, the northern remote site as “unpolluted” or at the “warning limit” and the remaining sites as “slightly polluted”. The overall analysis of the integrated indices points to Ni, Zn and Cu as being the priority pollutants because they can occasionally be associated with adverse biological effects at the SE2, SE1 and SE6 sites, which are classified as being moderately polluted. The values obtained show a mixture of potential sources for the contamination from crustal to anthropogenic, probably due to agricultural and oil activities, as well as urban wastewater discharges. Most sediment sites show a low-moderate pollution level and medium–low priority risk, which, added to the rising human pressure in the wetland, recommends using indices for the continuous assessment and monitoring of the sediment quality of the protected Limoncocha lagoon.
Analysis of the Al content at a different percentage of fine fraction to improve normalisation, as well as the investigation of potentially toxic elements speciation and bioavailability in the sediment of the Limoncocha lagoon, is proposed for further investigation.
References
Abrahim GMS, Parker RJ (2008) Assessment of heavy metal enrichment factorsand the degree of contamination in marine sediments from Tamaki Estuary, Auckland, New Zealand. Environ Monit Assess 136:227–238.https://doi.org/10.1007/s10661-007-9678-2
Ali H, Khan E, Ilahi I (2019) Environmental chemistry and ecotoxicology of hazardous heavy metals: environmental persistence, toxicity, and bioaccumulation. J Chem 2019:6730305. https://doi.org/10.1155/2019/6730305
Alvarez-Guerra M, Viguri JR, Casado-Martínez C, del Valls A (2007a) Sediment quality assessment and dredged material management in Spain: Part I, application Of Sediment Quality Guidelines In The Bay of Santander. Integr Environ Assess Manag 3:529–538. https://doi.org/10.1897/IEAM_2007-016.1
Alvarez-Guerra M, Viguri JR, Casado-Martínez C, del Valls A (2007b) Sediment quality assessment and dredged material management in Spain: Part II, analysis of action levels for dredged material management and application to the Bay of Cádiz. Integr Environ Assess Manag 3:539–551
Alvarez-Guerra M, González-Piñuela C, Andrés A et al (2008) Assessment of self-organizing map artificial neural networks for the classification of sediment quality. Environ Int 34:782–790. https://doi.org/10.1016/j.envint.2008.01.006
Alvarez-Guerra M, Ballabio D, Amigo JM, Bro R, Viguri JR (2010) Development of models for predicting toxicity from sediment chemistry by partial least squares-discriminant analysis and counter-propagation artificial neural networks. Environ Pollut 158:607–614. https://doi.org/10.1016/j.envpol.2009.08.007
Anderson EP, Osborne T, Maldonado-Ocampo JA et al (2019) Energy development reveals blind spots for ecosystem conservation in the Amazon Basin. Front Ecol Environ 17:521–529. https://doi.org/10.1002/fee.2114
Armas MF, Lasso S (2011) Plan de Manejo de la Reserva Biológica Limoncocha, Ministerio del Ambiente, Ecuador
Birch GF, Olmos MA (2008) Sediment-bound heavy metals as indicators of human influence and biological risk in coastal water bodies. ICES J Mar Sci 65:1407–1413. https://doi.org/10.1093/icesjms/fsn139
Birch GF (2020) An assessment of aluminum and iron in normalisation and enrichment procedures for environmental assessment of marine sediment. Sci Total Environ 727(2020):138123. https://doi.org/10.1016/j.scitotenv.2020.138123
Birch GF, Gunns TJ, Olmos M (2015) Sediment-bound metals as indicators of anthropogenic change in estuarine environments. Mar Pollut Bull 101:243–257. https://doi.org/10.1016/j.marpolbul.2015.09.056
Birch GF, Lee JH, Tanner E et al (2020) Sediment metal enrichment and ecological risk assessment of ten ports and estuaries in the World Harbours Project. Mar Pollut Bull 155:111129. https://doi.org/10.1016/j.marpolbul.2020.111129
Brady JP, Ayoko GA, Martens WN, Goonetilleke A (2015) Development of a hybrid pollution index for heavy metals in marine and estuarine sediments. Environ Monit Assess 187:306. https://doi.org/10.1007/s10661-015-4563-x
Carrillo KC, Drouet JC, Rodríguez-Romero A et al (2021) Spatial distribution and level of contamination of potentially toxic elements in sediments and soils of a biological reserve wetland, northern Amazon region of Ecuador. J Environ Manage 289.https://doi.org/10.1016/j.jenvman.2021.112495
Castro JE, Fernandez AM, Gonzalez-Caccia V, Gardinali PR (2013) Concentration of trace metals in sediments and soils from protected lands in south Florida: background levels and risk evaluation. Environ Monit Assess 185:6311–6332. https://doi.org/10.1007/s10661-012-3027-9
Chen CW, Kao CM, Chen CF, Di DC (2007) Distribution and accumulation of heavy metals in the sediments of Kaohsiung Harbor. Taiwan Chemosphere 66:1431–1440. https://doi.org/10.1016/j.chemosphere.2006.09.030
Christophoridis C, Evgenakis E, Bourliva A et al (2020) Concentration, fractionation, and ecological risk assessment of heavy metals and phosphorus in surface sediments from lakes in N. Greece. Environ Geochem Health 42:2747–2769.https://doi.org/10.1007/s10653-019-00509-x
de Andrade LC, Tiecher T, de Oliveira JS et al (2018) Sediment pollution in margins of the Lake Guaíba, Southern Brazil. Environ Monit Assess 190:1–13. https://doi.org/10.1007/s10661-017-6365-9
Dudley N (2008) Guidelines for applying protected area management categories. IUCN WCPA’s best practice protected area guidelines series. Gland, Switzerland. https://portals.iucn.org/library/sites/library/files/documents/pag-021.pdf. Accessed 9 March 2021
Dung TTT, Cappuyns V, Swennen R, Phung NK (2013) From geochemical background determination to pollution assessment of heavy metals in sediments and soils. Rev Environ Sci Biotechnol 12:335–353. https://doi.org/10.1007/s11157-013-9315-1
Duodu GO, Goonetilleke A, Ayoko GA (2016) Comparison of pollution indices for the assessment of heavy metal in Brisbane River sediment. Environ Pollut 219:1077–1091. https://doi.org/10.1016/j.envpol.2016.09.008
El-Metwally MEA, Darwish DH, Dar MA (2021) Spatial distribution and contamination assessment of heavy metals in surface sediments of Lake Burullus. Egypt Arab J Geosci 14:19. https://doi.org/10.1007/s12517-020-06149-1
Elmaci A, Teksoy A, Topaç FO et al (2007) Assessment of heavy metals in Lake Uluabat, Turkey. African J Biotechnol 6:2236–2244. https://doi.org/10.5897/ajb2007.000-2351
Engin MS, Uyanik A, Cay S (2017) Investigation of trace metals distribution in water, sediments and wetland plants of Kızılırmak Delta, Turkey. Int J Sediment Res 32:90–97. https://doi.org/10.1016/j.ijsrc.2016.03.004
GAD (2021) Gobierno Autonomo Descrentralizado Limoncocha (Autonomous Government Decrentralized Limoncocha). Actualización del plan de desarrollo y ordenamiento territorial de la parroquia Limoncocha (Updating of the development plan and territorial ordering of the Limoncocha parish). https://gadlimoncocha.gob.ec. Accessed 4 June 2021
Gao L, Wang Z, Li S, Chen J (2018) Bioavailability and toxicity of trace metals (Cd, Cr, Cu, Ni, and Zn) in sediment cores from the Shima River, South China. Chemosphere 192:31–42. https://doi.org/10.1016/j.chemosphere.2017.10.110
Guo G, Wu F, Xie F, Zhang R (2012) Spatial distribution and pollution assessment of heavy metals in urban soils from southwest China. J Environ Sci 24:410–418. https://doi.org/10.1016/S1001-0742(11)60762-6
Håkanson L (1980) An ecological risk index for aquatic pollution control. A Sedimentological Approach Water Res 14:975–1001
Håkanson L (1992) Sediment variability. In: Burton GA (ed) Sediment toxicity assessment. Lewis Publishers, Boca Raton, pp 19–36
Hernandez Gonzalez LM, Rivera VA, Phillips CB et al (2019) Characterization of soil profiles and elemental concentrations reveals deposition of heavy metals and phosphorus in a Chicago-area nature preserve, Gensburg Markham Prairie. J Soils Sediments 19:3817–3831. https://doi.org/10.1007/s11368-019-02315-5
Hu J, Zhou S, Wu P, Qu K (2017) Assessment of the distribution, bioavailability and ecological risks of heavy metals in the lake water and surface sediments of the Caohai plateau wetland, China. PLoS ONE 12:1–15. https://doi.org/10.1371/journal.pone.0189295
Hu C, Yang X, Dong J, Zhang X (2018) Heavy metal concentrations and chemical fractions in sediment from Swan Lagoon, China: their relation to the physiochemical properties of sediment. Chemosphere 209:848–856. https://doi.org/10.1016/j.chemosphere.2018.06.113
Huang B, Guo Z, Xiao X et al (2019) Changes in chemical fractions and ecological risk prediction of heavy metals in estuarine sediments of Chunfeng Lake estuary, China. Mar Pollut Bull 138:575–583. https://doi.org/10.1016/j.marpolbul.2018.12.015
Imran U, Ullah A, Shaikh K (2020) Pollution loads and ecological risk assessment of metals and a metalloid in the surface sediment of keenjhar lake, pakistan. Polish J Environ Stud 29:3629–3641. https://doi.org/10.15244/pjoes/117659
Jarrín AE, Salazar JG, Martínez-Fresneda M (2017) Pollution risk evaluation of the aquifers of the Limoncocha Biological Reserve, Ecuadorian Amazon. Rev Ambient Agua 12:652–665 (In Spanish) https://doi.org/10.4136/ambi-agua.2030
Jiang HH, Cai LM, Wen HH (2020) Luo J (2020) Characterizing pollution and source identification of heavy metals in soils using geochemical baseline and PMF approach. Sci Rep-UK 10:6460. https://doi.org/10.1038/s41598-020-63604-5
Kalita S, Sarma HP, Devi A (2019) Sediment characterisation and spatial distribution of heavy metals in the sediment of a tropical freshwater wetland of Indo-Burmese province. Environ Pollut 250:969–980. https://doi.org/10.1016/j.envpol.2019.04.112
Karim Z, Qureshi BA, Mumtaz M (2015) Geochemical baseline determination and pollution assessment of heavy metals in urban soils of Karachi, Pakistan. Ecol Indic 48:358–364. https://doi.org/10.1016/j.ecolind.2014.08.032
Kingsford RT, Bino G, Finlayson CM et al (2021) Ramsar wetlands of international importance–improving conservation outcomes. Front Environ Sci 9:1–6. https://doi.org/10.3389/fenvs.2021.643367
Kostka A, Leśniak A (2021) Natural and anthropogenic origin of metals in lacustrine sediments; assessment and consequences—a case study of wigry lake (Poland). Minerals 11:1–22. https://doi.org/10.3390/min11020158
Kowalska JB, Mazurek R, Gąsiorek M, Zaleski T (2018) Pollution indices as useful tools for the comprehensive evaluation of the degree of soil contamination–a review. Environ Geochem Health 40:2395–2420. https://doi.org/10.1007/s10653-018-0106-z
Lécrivain N, Aurenche V, Cottinb N et al (2018) Multi-contamination (heavy metals, polychlorinated biphenyls and polycyclic aromatic hydrocarbons) of littoral sediments and the associated ecological risk assessment in a large lake in France (Lake Bourget). Sci Total Environ 619–620:854–865. https://doi.org/10.1016/j.scitotenv.2017.11.151
Long ER, MacDonald DD (1998) Recommended uses of empirically derived, sediment quality guidelines for marine and estuarine ecosystems. Hum Ecol Risk Assess 4:1019–1039. https://doi.org/10.1080/10807039891284956
Long ER, Macdonald DD, Smith SL, Calder FD (1995) Incidence of adverse biological effects within ranges of chemical concentrations in marine and estuarine sediments. Environ Manage 19:81–97. https://doi.org/10.1007/BF02472006
Luo L, Mei K, Qu L et al (2019) Assessment of the geographical detector method for investigating heavy metal source apportionment in an urban watershed of Eastern China. Sci Total Environ 653:714–722. https://doi.org/10.1016/j.scitotenv.2018.10.424
Maanan M, Saddik M, Maanan M et al (2015) Environmental and ecological risk assessment of heavy metals in sediments of Nador lagoon, Morocco. Ecol Indic 48:616–626. https://doi.org/10.1016/j.ecolind.2014.09.034
MacDonald DD, Ingersoll CG, Berger TA (2000) Development and evaluation of consensus-based sediment quality guidelines for freshwater ecosystems. Arch Environ Contam Toxicol 39:20–31. https://doi.org/10.1007/s002440010075
Magesh NS, Tiwari A, Botsa SM, da Lima LT (2021) Hazardous heavy metals in the pristine lacustrine systems of Antarctica: insights from PMF model and ERA techniques. J Hazard Mater 412:125263. https://doi.org/10.1016/j.jhazmat.2021.125263
Matschullat J, Ottenstein R, Reimann C (2000) Geochemical background-can we calculate it? Environ Geol 39:990–1000. https://doi.org/10.1007/s002549900084
Mayanglambam B, Neelam SS (2020) Geochemistry and pollution status of surface sediments of Loktak Lake, Manipur, India. SN Appl Sci 2:1–16. https://doi.org/10.1007/s42452-020-03903-8
Millennium Ecosystem Assessment (2005) Ecosystems and human well-being: wetlands and water synthesis. World Resources Institute, Washington, DC. https://www.millenniumassessment.org/documents/document.358.aspx.pdf. Accessed 9 March 2021
Morillo J, Usero J, Rojas R (2008) Fractionation of metals and As in sediments from a biosphere reserve (Odiel salt marshes) affected by acidic mine drainage. Environ Monit Assess 139:329–337. https://doi.org/10.1007/s10661-007-9839-3
Müller G (1969) Index of geo-accumulation in sediments of the Rhine River. GeoJournal 2:108–118
Nemati K, Bakar NKA, Abas MR, Sobhanzadeh E (2011) Speciation of heavy metals by modified BCR sequential extraction procedure in different depths of sediments from Sungai Buloh, Selangor, Malaysia. J Hazard Mater 192:402–410. https://doi.org/10.1016/j.jhazmat.2011.05.039
Nemerow NL (1991) Stream, lake, estuary, and ocean pollution. United States, New York
Niu S, Gao L, Wang X (2019) Characterization of contamination levels of heavy metals in agricultural soils using geochemical baseline concentrations. J Soils Sediments 19:1697–1707. https://doi.org/10.1007/s11368-018-2190-1
Ogunlaja A, Ogunlaja OO, Okewole DM, Morenikeji OA (2019) Risk assessment and source identification of heavy metal contamination by multivariate and hazard index analyses of a pipeline vandalised area in Lagos State, Nigeria. Sci Total Environ 651:2943–2952. https://doi.org/10.1016/j.scitotenv.2018.09.386
Pitacco V, Mistri M, Ferrari CR et al (2020) Multiannual trend of micro-pollutants in sediments and benthic community response in a mediterranean lagoon (Sacca di Goro, Italy). Water (switzerland) 12:1074. https://doi.org/10.3390/W12041074
Pratte S, Bao K, Shen J et al (2019) Centennial records of cadmium and lead in NE China lake sediments. Sci Total Environ 657:548–557. https://doi.org/10.1016/j.scitotenv.2018.11.407
Qing X, Yutong Z, Shenggao Lu (2015) Assessment of heavy metal pollution and human health risk in urban soils of steel industrial city (Anshan), Liaoning, Northeast China. Ecotoxicol Environ Saf 120:377–385. https://doi.org/10.1016/j.ecoenv.2015.06.019
Ramsar (2020) The list of wetlands of international importance. Published 2 February 2020 pp. 55. Available at: https://www.ramsar.org/sites/default/files/documents/library/sitelist.pdf
Reimann C, Fabian K, Birke M et al (2018) GEMAS: establishing geochemical background and threshold for 53 chemical elements in European agricultural soil. Appl Geochem 88:302–318. https://doi.org/10.1016/j.apgeochem.2017.01.021
Reimann C, De Caritat P (2005) Distinguishing between natural and anthropogenic sources for elements in the environment: regional geochemical surveys versus enrichment factors. Sci Total Environ 337:91–107. https://doi.org/10.1016/j.scitotenv.2004.06.011
Reimann C, Garrett RG (2005) Geochemical background e concept and reality. Sci Total Environ 350:12–27. https://doi.org/10.1016/j.scitotenv.2005.01.047
Reimann C, Filzmoser P, Garrett RG (2005) Background and threshold: critical comparison of methods of determination. Sci Total Environ 346:1–16. https://doi.org/10.1016/j.scitotenv.2004.11.023
Reimann C, Filzmoser P (2000) Normal and lognormal data distribution in geochemistry: death of a myth. Consequences for the statistical treatment of geochemical and environmental data. Environ Geol 39:1001–1014. https://doi.org/10.1007/s002549900081
Rudnick RL, Gao S (2003) Composition of the continental crust. Treatise Geochem 3:1–64. https://doi.org/10.1016/B0-08-043751-6/03016-4
Saddik M, Fadili A, Makan A (2019) Assessment of heavy metal contamination in surface sediments along the Mediterranean coast of Morocco. Environ Monit Assess 191.https://doi.org/10.1007/s10661-019-7332-4
Sadeghi P, Loghmani M, Afsa E (2019) Trace element concentrations, ecological and health risk assessment in sediment and marine fish Otolithes ruber in Oman Sea. Iran Mar Pollut Bull 140:248–254. https://doi.org/10.1016/j.marpolbul.2019.01.048
Saeedi M, Jamshidi-Zanjani A (2015) Development of a new aggregative index to assess potential effect of metals pollution in aquatic sediments. Ecol Indic 58:235–243. https://doi.org/10.1016/j.ecolind.2015.05.047
Saher NU, Siddiqui AS (2019) Occurrence of heavy metals in sediment and their bioaccumulation in sentinel crab (Macrophthalmus depressus) from highly impacted coastal zone. Chemosphere 221:89–98. https://doi.org/10.1016/j.chemosphere.2019.01.008
Sakan S, Dević G, Relić D et al (2015) Evaluation of sediment contamination with heavy metals: the importance of determining appropriate background content and suitable element for normalization. Environ Geochem Health 37:97–113. https://doi.org/10.1007/s10653-014-9633-4
Salomons W, Förstner U (1984) Metals in the hydrocycle. In Chapter 2: “interactions with ligands, particulate matter and organisms”, pp.55. Springer-Verlag. Berlin
Sharma VK, Rhudy KB, Koening R, Vazquez FG (1999) Metals in sediments of the Upper Laguna Madre. Mar Pollut Bull 38:1221–1226. https://doi.org/10.1016/S0025-326X(99)00166-6
Sojka M, Choiński A, Ptak M, Siepak M (2021) Causes of variations of trace and rare earth elements concentration in lakes bottom sediments in the Bory Tucholskie National Park, Poland. Sci Rep 11:1–18. https://doi.org/10.1038/s41598-020-80137-z
Soliman NF, Younis AM, Elkady EM (2019) An insight into fractionation, toxicity, mobility and source apportionment of metals in sediments from El Temsah Lake, Suez Canal. Chemosphere 222:165–174. https://doi.org/10.1016/j.chemosphere.2019.01.009
Taylor SR, McLennan SM (1995) The geochemical evolution of the continental crust. Rev Geophys 33:241–265. https://doi.org/10.1029/95RG00262
Teng Y, Ni S, Wang J, Niu L (2009) Geochemical baseline of trace elements in the sediment in Dexing area, South China. Environ Geol 57:1649–1660. https://doi.org/10.1007/s00254-008-1446-2
Tian K, Huang B, Xing Z, Hu W (2017) Geochemical baseline establishment and ecological risk evaluation of heavy metals in greenhouse soils from Dongtai, China. Ecol Indic 72:510–520. https://doi.org/10.1016/j.ecolind.2016.08.037
Tomlinson DL, Wilson JG, Harris CR, Jeffrey DW (1980) Problems in the assessment of heavy-metal levels in estuaries and the formation of a pollution index. Helgolander Meeresun 33:566–575. https://doi.org/10.1007/BF02414780
Turekian KK, Wedepohl KH (1961) Distribution of the elements in ome major units of the Earth’s Crust. Geol Soc Am Bull 72:175–192
USEPA–United States Environmental Protection Agency (1998) Method 3051A—microwave assisted acid digestion of sediments, sludges, soils and oils. http://www.epa.gov/SW-846/pdfs/3051a.pdf
Wang S, Wang W, Chen J et al (2019a) Geochemical baseline establishment and pollution source determination of heavy metals in lake sediments: a case study in Lihu Lake, China. Sci Total Environ 657:978–986. https://doi.org/10.1016/j.scitotenv.2018.12.098
Wang W, Wang S, Chen J et al (2019b) Combined use of diffusive gradients in thin film, high-resolution dialysis technique and traditional methods to assess pollution and bioavailability of sediment metals of lake wetlands in Taihu Lake Basin. Sci Total Environ 671:28–40. https://doi.org/10.1016/j.scitotenv.2019.03.053
Wang Z, Zhou J, Zhang C et al (2019c) A comprehensive risk assessment of metals in riverine surface sediments across the rural-urban interface of a rapidly developing watershed. Environ Pollut 245:1022–1030. https://doi.org/10.1016/j.envpol.2018.11.078
Wei C, Wen H (2012) Geochemical baselines of heavy metals in the sediments of two large freshwater lakes in China: implications for contamination character and history. Environ Geochem Health 34:737–748. https://doi.org/10.1007/s10653-012-9492-9
Wenning RJ, Batley GE, Ingersoll CG, Moore DW (Eds.) (2005) Use of sediment quality guidelines and related tools for the assessment of contaminated sediments. SETAC Press (Society of Environmental Toxicology and Chemistry), Pensacola, FL
Yang J, Wang W, Zhao M et al (2015) Spatial distribution and historical trends of heavy metals in the sediments of petroleum producing regions of the Beibu Gulf, China. Mar Pollut Bull 91:87–95. https://doi.org/10.1016/j.marpolbul.2014.12.023
Zhang G, Bai J, Zhao Q et al (2016) Heavy metals in wetland soils along a wetland-forming chronosequence in the Yellow River Delta of China: levels, sources and toxic risks. Ecol Indic 69:331–339. https://doi.org/10.1016/j.ecolind.2016.04.042
Zhang J, Liu CL (2002) Riverine composition and estuarine geochemistry of particulate metals in China - weathering features, anthropogenic impact and chemical fluxes. Estuar Coast Shelf Sci 54:1051–1070. https://doi.org/10.1006/ecss.2001.0879
Zhou Y, Gao L, Xu D, Gao B (2019) Geochemical baseline establishment, environmental impact and health risk assessment of vanadium in lake sediments, China. Sci Total Environ 660:1338–1345. https://doi.org/10.1016/j.scitotenv.2019.01.093
Zhuang W, Wang Q, Tang L et al (2018) A new ecological risk assessment index for metal elements in sediments based on receptor model, speciation, and toxicity coefficient by taking the Nansihu Lake as an example. Ecol Indic 89:725–737. https://doi.org/10.1016/j.ecolind.2018.02.033
Zhuang W, Ying SC, Frie AL et al (2019) Distribution, pollution status, and source apportionment of trace metals in lake sediments under the influence of the South-to-North Water Transfer Project, China. Sci Total Environ 671:108–118. https://doi.org/10.1016/j.scitotenv.2019.03.306
Acknowledgements
Dr Araceli Rodríguez-Romero is supported by the Spanish grant Juan de la Cierva Incorporación referenced as IJC2018–037545-I.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature.
Author information
Authors and Affiliations
Contributions
KCC carried out the research plan and collected and analysed the data; ARR and ATS: methodology and investigation, review and editing; GRG: formal analysis, writing–review and editing; JRVF: conceptualisation, writing–review and editing. All authors have read and agreed to the present version of this manuscript.
Corresponding author
Additional information
Responsible editor: Tomas Matys Grygar
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Carrillo, K.C., Rodríguez-Romero, A., Tovar-Sánchez, A. et al. Geochemical baseline establishment, contamination level and ecological risk assessment of metals and As in the Limoncocha lagoon sediments, Ecuadorian Amazon region. J Soils Sediments 22, 293–315 (2022). https://doi.org/10.1007/s11368-021-03084-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11368-021-03084-w