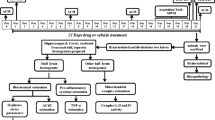
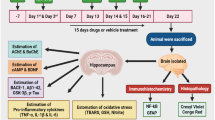
Abstract
We had previously reported that neuroinflammation and memory impairment associated with intracerebroventricular streptozotocin (ICV STZ) injection in rats was due to glial activation and modulation of the N-methyl-D-aspartate (NMDA) receptor function. However, the exact role of the NMDA receptor and the molecules associated with downstream calcium ion signaling in STZ-induced astroglial activation is not known. Thus, in the present study, Memantine (an NMDA receptor antagonist) and Ibuprofen (an anti-inflammatory drug) were used as the pharmacological tool to investigate the molecular mechanisms involved in STZ-induced astroglial inflammation. We have studied the effect of STZ (100 μM) treatment for 24 h on NMDA receptor subunits (NR1, NR2A, and NR2B) expression and its associated calcium ion regulated molecules calcium/calmodulin-dependent protein kinase II subunit α (CaMKIIα), cyclic AMP-response element-binding (CREB) protein, Calpain, and Caspase 3. We have found a significant increase in the expression of NR1, NR2B, Calpain, and Caspase 3 expression, whereas a decrease in the level of NR2A, CaMKIIα, and CREB protein expression after 24 h of STZ treatment. These results indicate that STZ altered the NMDA receptor subunit expression and its downstream calcium (Ca2+) ion signaling molecules. We have also found that both Memantine (5 µM) and Ibuprofen (200 μM) significantly prevented the STZ-induced change in CaMKIIα, CREB, Calpain, and Caspase 3 expressions in C6 astrocytoma cells. Interestingly, only Memantine (and not Ibuprofen) was able to prevent the changes in NMDA receptor subunit expression in STZ-treated astrocytoma cells. STZ treatment also increased the level of glial fibrillary acidic protein (GFAP), tumor necrosis factor-alpha (TNF-α), inducible nitric oxide synthase (iNOS), and decreased the level of interleukin-10 (IL-10), indicating inflammatory condition, which was restored by both Memantine and Ibuprofen. These results suggest that both Memantine and Ibuprofen exert anti-inflammatory effect against STZ-induced astroglial activation and neuroinflammation via modulation of NMDA receptor-associated downstream calcium signaling cascade. However, only Memantine (not Ibuprofen) was able to revert STZ-induced changes in NMDA receptor subunit expression.
Graphic abstract









Similar content being viewed by others
References
Alley GM et al (2010) Memantine lowers amyloid-β peptide levels in neuronal cultures and in APP/PS1 transgenic mice. J Neurosci Res 88:143–154. https://doi.org/10.1002/jnr.22172
Altarejos JY, Montminy M (2011) CREB and the CRTC co-activators: sensors for hormonal and metabolic signals. Nat Rev Mol Cell Biol 12:141–151. https://doi.org/10.1038/nrm3072
Araque A, Li N, Doyle RT, Haydon PG (2000) SNARE protein-dependent glutamate release from astrocytes. J Neuroscience 20:666–673
Ashpole NM, Chawla AR, Martin MP, Brustovetsky T, Brustovetsky N, Hudmon A (2013a) Loss of calcium/calmodulin-dependent protein kinase II activity in cortical astrocytes decreases glutamate uptake and induces neurotoxic release of ATP. J Biol Chem 288:14599–14611. https://doi.org/10.1074/jbc.M113.466235
Blanke ML, VanDongen AMJ (2009) Frontiers in Neuroscience, Activation Mechanisms of the NMDA Receptor. In: Van Dongen AM (ed) Biology of the NMDA Receptor. CRC Press/Taylor & Francis Taylor & Francis Group, LLC., Boca Raton (FL)
Blasko I, Apochal A, Boeck G, Hartmann T, Grubeck-Loebenstein B, Ransmayr G (2001) Ibuprofen decreases cytokine-induced amyloid beta production in neuronal cells. Neurobiol Dis 8:1094–1101. https://doi.org/10.1006/nbdi.2001.0451
Bonni A, Brunet A, West AE, Datta SR, Takasu MA, Greenberg ME (1999) Cell survival promoted by the Ras-MAPK signaling pathway by transcription-dependent and -independent mechanisms. Science (New York, NY) 286:1358–1362. https://doi.org/10.1126/science.286.5443.1358
Bukowska A, Lendeckel U, Bode-Boger SM, Goette A (2012) Physiologic and pathophysiologic role of calpain: implications for the occurrence of atrial fibrillation. Cardiovasc Ther 30:e115–127. https://doi.org/10.1111/j.1755-5922.2010.00245.x
Chang YC, Kim HW, Rapoport SI, Rao JS (2008) Chronic NMDA administration increases neuroinflammatory markers in rat frontal cortex: cross-talk between excitotoxicity and neuroinflammation. Neurochem Res 33:2318–2323. https://doi.org/10.1007/s11064-008-9731-8
Conti F, DeBiasi S, Minelli A, Melone M (1996) Expression of NR1 and NR2A/B subunits of the NMDA receptor in cortical astrocytes. Glia 17:254–258. https://doi.org/10.1002/(sici)1098-1136(199607)17:3<254:aid-glia7>3.0.co;2-0
Deak T, Bellamy C, D’Agostino LG (2003) Exposure to forced swim stress does not alter central production of IL-1. Brain Res 972:53–63. https://doi.org/10.1016/S0006-8993(03)02485-5
Dzamba D, Honsa P, Anderova M (2013) NMDA Receptors in Glial Cells: Pending Questions. Curr Neuropharmacol 11:250–262. https://doi.org/10.2174/1570159X11311030002
Graham ML, Janecek JL, Kittredge JA, Hering BJ, Schuurman HJ (2011) The streptozotocin-induced diabetic nude mouse model: differences between animals from different sources. Comp Med 61:356–360
Green SJ et al (1994) Nitric oxide: cytokine-regulation of nitric oxide in host resistance to intracellular pathogens. Immunol Letts 43:87–94. https://doi.org/10.1016/0165-2478(94)00158-8
Hewett SJ, Csernansky CA, Choi DW (1994) Selective potentiation of NMDA-induced neuronal injury following induction of astrocytic iNOS. Neuron 13:487–494. https://doi.org/10.1016/0896-6273(94)90362-x
Ifuku M, Katafuchi T, Mawatari S, Noda M, Miake K, Sugiyama M, Fujino T (2012) Anti-inflammatory/anti-amyloidogenic effects of plasmalogens in lipopolysaccharide-induced neuroinflammation in adult mice. J Neuroinflamm 9:197. https://doi.org/10.1186/1742-2094-9-197
Jaturapatporn D, Isaac MG, McCleery J, Tabet N (2012) Aspirin, steroidal and non-steroidal anti-inflammatory drugs for the treatment of Alzheimer's disease. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD006378.pub2
König N et al (2003) Calpain 3 is expressed in astrocytes of rat and Microcebus brain. J Chem Neuroanat 25:129–136. https://doi.org/10.1016/S0891-0618(02)00102-3
Kovács KA, Steullet P, Steinmann M, Do KQ, Magistretti PJ, Halfon O, Cardinaux J-R (2007) TORC1 is a calcium- and cAMP-sensitive coincidence detector involved in hippocampal long-term synaptic plasticity. Proc Nat Acad Sci 104:4700. https://doi.org/10.1073/pnas.0607524104
Lau CG, Takeuchi K, Rodenas-Ruano A, Takayasu Y, Murphy J, Bennett MV, Zukin RS (2009) Regulation of NMDA receptor Ca2+ signalling and synaptic plasticity. Biochem Soc Trans 37:1369–1374. https://doi.org/10.1042/bst0371369
Layne E (1957) Spectrophotometric and turbidimetric methods for measuring proteins. In: Methods in Enzymology, vol 3. Academic Press, pp 447–454. https://doi.org/10.1016/S0076-6879(57)03413-8
Lee MC, Ting KK, Adams S, Brew BJ, Chung R, Guillemin GJ (2010) Characterisation of the expression of NMDA receptors in human astrocytes. PLoS ONE 5:e14123. https://doi.org/10.1371/journal.pone.0014123
Leloup L, Mazeres G, Daury L, Cottin P, Brustis JJ (2006) Involvement of calpains in growth factor-mediated migration. Int J Biochem Cell Biol 38:2049–2063. https://doi.org/10.1016/j.biocel.2006.04.012
Lichtenstein MP, Carriba P, Baltrons MA, Wojciak-Stothard B, Peterson JR, García A, Galea E (2010) Secretase-independent and RhoGTPase/PAK/ERK-dependent regulation of cytoskeleton dynamics in astrocytes by NSAIDs and derivatives. J Alzheimer's Dis 22:1135–1155. https://doi.org/10.3233/jad-2010-101332
Mishra SK, Singh S, Shukla S, Shukla R (2018) Intracerebroventricular streptozotocin impairs adult neurogenesis and cognitive functions via regulating neuroinflammation and insulin signaling in adult rats. Neurochem Int 113:56–68. https://doi.org/10.1016/j.neuint.2017.11.012
Palygin O, Lalo U, Pankratov Y (2011) Distinct pharmacological and functional properties of NMDA receptors in mouse cortical astrocytes. Br J Pharmacol 163:1755–1766. https://doi.org/10.1111/j.1476-5381.2011.01374.x
Rai S, Kamat PK, Nath C, Shukla R (2013) A study on neuroinflammation and NMDA receptor function in STZ (ICV) induced memory impaired rats. J Neuroimmunol 254:1–9. https://doi.org/10.1016/j.jneuroim.2012.08.008
Rai S, Kamat PK, Nath C, Shukla R (2014) Glial activation and post-synaptic neurotoxicity: The key events in Streptozotocin (ICV) induced memory impairment in rats. Pharmacol Biochem Behav 117:104–117. https://doi.org/10.1016/j.pbb.2013.11.035
Rajasekar N, Dwivedi S, Nath C, Hanif K, Shukla R (2014) Protection of streptozotocin induced insulin receptor dysfunction, neuroinflammation and amyloidogenesis in astrocytes by insulin. Neuropharmacology 86:337–352. https://doi.org/10.1016/j.neuropharm.2014.08.013
Ricci S, Fuso A, Ippoliti F, Businaro R (2012) Stress-induced cytokines and neuronal dysfunction in Alzheimer's disease. J Alzheimer's Dis 28:11–24. https://doi.org/10.3233/jad-2011-110821
Schousboe A, Waagepetersen HS (2005) Role of astrocytes in glutamate homeostasis: implications for excitotoxicity. Neurotox Res 8:221–225. https://doi.org/10.1007/bf03033975
Shaywitz AJ, Greenberg ME (1999) CREB: a stimulus-induced transcription factor activated by a diverse array of extracellular signals. Ann Rev Biochem 68:821–861. https://doi.org/10.1146/annurev.biochem.68.1.821
Skowrońska K, Obara-Michlewska M, Zielińska M, Albrecht J (2019) NMDA receptors in astrocytes search for roles in neurotransmission and astrocytic homeostasis. Int J Mol Sci 20:309. https://doi.org/10.3390/ijms20020309
Stratman NC, Carter DB, Sethy VH (1997) Ibuprofen: effect on inducible nitric oxide synthase. Brain Res Mol Brain Res 50:107–112. https://doi.org/10.1016/s0169-328x(97)00168-x
Swarnkar S, Singh S, Goswami P, Mathur R, Patro IK, Nath C (2012) Astrocyte activation: a key step in rotenone induced cytotoxicity and DNA damage. Neurochem Res 37:2178–2189. https://doi.org/10.1007/s11064-012-0841-y
Swarnkar S, Goswami P, Kamat PK, Patro IK, Singh S, Nath C (2013) Rotenone-induced neurotoxicity in rat brain areas: a study on neuronal and neuronal supportive cells. Neuroscience 230:172–183. https://doi.org/10.1016/j.neuroscience.2012.10.034
Takeuchi Y, Yamamoto H, Fukunaga K, Miyakawa T, Miyamoto E (2000) Identification of the isoforms of Ca2+/calmodulin-dependent protein kinase II in rat astrocytes and their subcellular localization. J Neurochem 74:2557–2567. https://doi.org/10.1046/j.1471-4159.2000.0742557.x
Thalhammer A, Rudhard Y, Tigaret CM, Volynski KE, Rusakov DA, Schoepfer R (2006) CaMKII translocation requires local NMDA receptor-mediated Ca2+ signaling. EMBO J 25:5873–5883. https://doi.org/10.1038/sj.emboj.7601420
Uttara B, Singh AV, Zamboni P, Mahajan RT (2009) Oxidative stress and neurodegenerative diseases: a review of upstream and downstream antioxidant therapeutic options. Curr Neuropharmacol 7:65–74
Vosler PS, Brennan CS, Chen J (2008) Calpain-mediated signaling mechanisms in neuronal injury and neurodegeneration. Mol Neurobiol 38:78–100. https://doi.org/10.1007/s12035-008-8036-
Walton MR, Dragunow I (2000) Is CREB a key to neuronal survival? Trends Neurosci 23:48–53. https://doi.org/10.1016/s0166-2236(99)01500-3
Wang DD, Bordey A (2008) The astrocyte odyssey. Prog Neurobiol 86:342–367. https://doi.org/10.1016/j.pneurobio.2008.09.015
Wu LJ et al (2005) Upregulation of forebrain NMDA NR2B receptors contributes to behavioral sensitization after inflammation. J Neurosci 25:11107–11116. https://doi.org/10.1523/jneurosci.1678-05.2005
Zhang S, Wu M, Peng C, Zhao G, Gu R (2017) GFAP expression in injured astrocytes in rats. Experim Ther Med 14:1905–1908. https://doi.org/10.3892/etm.2017.4760
Acknowledgements
The authors are grateful to the Director, CSIR-Central Drug Research Institute (CDRI), Lucknow, for his constant support and guidance. Authors also acknowledge Council of Scientific and Industrial Research (CSIR), New Delhi, India, for providing financial support to perform this research work in CDRI.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mishra, S.K., Hidau, M. & Rai, S. Memantine and Ibuprofen pretreatment exerts anti-inflammatory effect against streptozotocin-induced astroglial inflammation via modulation of NMDA receptor-associated downstream calcium ion signaling . Inflammopharmacol 29, 183–192 (2021). https://doi.org/10.1007/s10787-020-00760-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10787-020-00760-0