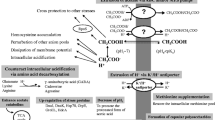
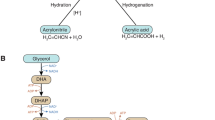
Abstract
Acetic acid bacteria are versatile organisms converting a number of carbon sources into biomolecules of industrial interest. Such properties, together with the need to limit chemical syntheses in favor of more sustainable biological processes, make acetic acid bacteria appropriate organisms for food, chemical, medical, pharmaceutical and engineering applications. At current, well-established bioprocesses by acetic acid bacteria are those derived from the oxidative pathways that lead to organic acids, ketones and sugar derivates. Whereas emerging applications include biopolymers, such as bacterial cellulose and fructans, which are getting an increasing interest for the biotechnological industry. However, considering the industrial demand of high performing bioprocesses, the production yield of metabolites obtained by acetic acid bacteria, is still not satisfying. In this paper we review the major acetic acid bacteria industrial applications, considering the current status of bioprocesses. We will also describe new biotechnological advances in order to optimize the industrial production, offering also an overview on future directions.
Similar content being viewed by others
References
Abdel-Fattah AM, Gamal-Eldeen AM, Helmy WA, Esawy MA (2012) Antitumor and antioxidant activities of levan and its derivative from the isolate Bacillus subtilis NRC1aza. Carbohydr Polym 89:314–322
Adachi O, Yakushi T (2016) Membrane-bound dehydrogenases of acetic acid bacteria. In: Matsushita H, Toyama N, Tonouchi N, Okamoto-Kainuma A (eds) Acetic acid bacteria ecology and physiology. Springer, Japan, pp 273–297. https://doi.org/10.1007/978-4-431-55933-7_13
Adachi O, Tanasupawat S, Yoshihara N, Toyama H, Matsushita K (2003) 3-Dehydroquinate production by oxidative fermentation and further conversion of 3-dehydroquinate to the intermediates in the shikimate pathway. Biosci Biotechnol Biochem 67:2124–2131. https://doi.org/10.1271/bbb.67.2124
Ameyama M, Osada K, Shinagawa E, Matsushita K, Adachi O (1981) Purification and characterization of aldehyde dehydrogenase of Acetobacter aceti. Agric Biol Chem 45:1889–1890. https://doi.org/10.1271/bbb1961.45.1889
Ano Y, Shinagawa E, Adachi O, Toyama H, Yakushi T, Matsushita K (2011) Selective, high conversion of d-glucose to 5-keto-d-gluoconate by Gluconobacter suboxydans. Biosci Biotechnol Biochem 75:586–589. https://doi.org/10.1271/bbb.100701
Bartowsky EJ, Henschke PA (2008) Acetic acid bacteria spoilage of bottled red wine: a review. Int J Food Microbiol 125:60–70. https://doi.org/10.1016/j.ijfoodmicro.2007.10.016
Bello FD, Hertel C, Hammes W, Walter J (2001) In vitro study of prebiotic properties of levan-type exopolysaccharides from Lactobacilli and non-digestible carbohydrates using denaturing gradient gel electrophoresis. Syst Appl Microbiol 24:232–237. https://doi.org/10.1078/0723-2020-00033
Benigar E, Dogsa I, Stopar D, Jamnik A, Cigiæ IK, Tomšiè M (2014) Structure and dynamics of a polysaccharide matrix: aqueous solutions of bacterial levan. Langmuir 30:4172–4182. https://doi.org/10.1021/la500830j
Brandt JU, Jakob F, Jürgen B, Geissler AJ, Vogel RF (2016) Dissection of exopolysaccharide biosynthesis in Kozakia baliensis. Microb Cell Fact 15:170. https://doi.org/10.1186/s12934-016-0572-x
Cañete-Rodríguez AM, Santos-Dueñas IM, Jiménez JE, Ehrenreich HA, Liebl W, García-García I (2016) Gluconic acid: properties, production methods and applications-an excellent opportunity for agro-industrial by-products and waste bio-valorization. Process Biochem 51:1891–1903. https://doi.org/10.1016/j.procbio.2016.08.028
Chambert R, Treboul G, Dedonder R (1974) Kinetic studies of levansucrase of Bacillus subtilis. Eur J Biochem 41:285. https://doi.org/10.1111/j.1432-1033.1974.tb03269.x
Chozhavendhan S, Kumar RP, Elavazhagan S, Barathiraja B, Jayakumar M, Varjani SJ (2018) Utilization of crude glycerol from biodiesel industry for the production of value-added bioproducts. In: Singhania R, Agarwal R, Kumar R, Sukumaran R (eds) Waste to wealth. Energy, environment, and sustainability. Springer, Singapore, pp 65–82. https://doi.org/10.1007/978-981-10-7431-8_4
De Roos J, De Vuyst L (2018) Acetic acid bacteria in fermented foods and beverages. Curr Opin Microbiol 49:115–119. https://doi.org/10.1016/j.copbio.2017.08.007
De Vero L, Gullo M, Giudici P (2010) Acetic acid bacteria, biotechnological applications. Encycl Ind Biotechnol. https://doi.org/10.1002/9780470054581.eib003
De Vuyst L, Vanderveken F, Van de Ven S, Degeest B (1998) Production by and isolation of exopolysaccharides from Streptococcus thermophiles grown in milk medium and evidence for their growth-associated biosynthesis. J Appl Microbiol 84:1059–1068. https://doi.org/10.1046/j.1365-2672.1998.00445.x
Dikshit PK, Moholkar VS (2018) Batch and repeated-batch fermentation for 1,3-dihydroxyacetone production from waste glycerol using free, immobilized and resting Gluconobacter oxydans cells. Waste Biomass Valori. https://doi.org/10.1007/s12649-018-0307-9
Donot F, Fontana A, Baccou JC, Schorr-Galindo S (2012) Microbial exopolysaccharides: main examples of synthesis, excretion, genetics and extraction. Carbohydr Polym 87:951–962. https://doi.org/10.1016/j.carbpol.2011.08.083
Esawy MA, Ahmed EF, Helmy WA, Mansour NM, El-Senousy WM, El-Safty MM (2011) Production of levansucrase from novel honey Bacillus subtilis isolates capable of producing antiviral levans. Carbohydr Polym 86:823–830. https://doi.org/10.1016/j.carbpol.2011.05.035
Gao L, Hu Y, Liu J, Du G, Zhou J, Chen J (2014) Stepwise metabolic engineering of Gluconobacter oxydans WSH-003 for the direct production of 2-keto-l-gulonic acid from d-sorbitol. Metab Eng 24:30–37. https://doi.org/10.1016/j.ymben.2014.04.003
García-García I, Cañete-Rodríguez AM, Santos-Dueñas IM, Jiménez Hornero JE, Ehrenreich A, Liebl W, García-Martínez T, Mauricio JC (2017) Biotechnologically relevant features of gluconic acid production by acetic acid bacteria. Acetic Acid Bact 6(6458):7–12. https://doi.org/10.4081/aab.2017.6458
Gätgens C, Degner U, Bringer-Meyer S, Herrmann U (2007) Biotransformation of glycerol to dihydroxyacetone by recombinant Gluconobacter oxydans DSM 2343. Appl Microbiol Biotechnol 76:553–559. https://doi.org/10.1007/s00253-007-1003-z
Giudici P, Gullo M, Solieri L (2009) Traditional balsamic vinegar. In: Solieri L, Giudici P (eds) Vinegars of the world. Springer, Milano, pp 157–177. https://doi.org/10.1007/978-88-470-0866-3_10
Giudici P, De Vero L, Gullo M, Solieri L, Lemmetti F (2016) Fermentation strategy to produce high gluconate vinegar. Acetic Acid Bact 5(6067):1–6. https://doi.org/10.4081/aab.2016.6067
Gullo M, Giudici P (2008) Acetic acid bacteria in traditional balsamic vinegar: phenotypic traits relevant for starter cultures selection. Int J Food Microbiol 125:46–53. https://doi.org/10.1016/j.ijfoodmicro.2007.11.076
Gullo M, De Vero L, Giudici P (2009) Succession of selected strains of Acetobacter pasteurianus and other acetic acid bacteria in traditional balsamic vinegar. Appl Environ Microbiol 75:2585–2589. https://doi.org/10.1128/AEM.02249-08
Gullo M, Verzelloni E, Canonico M (2014) Aerobic submerged fermentation by acetic acid bacteria for vinegar production: process and biotechnological aspects. Process Biochem 49:1571–1579. https://doi.org/10.1016/j.procbio.2014.07.003
Gullo M, Zanichelli G, Verzelloni E, Lemmetti F, Giudici P (2016) Feasible acetic acid fermentations of alcoholic and sugary substrates in combined operation mode. Process Biochem 51:1129–1139. https://doi.org/10.1016/j.procbio.2016.05.018
Gullo M, Sola A, Zanichelli G, Montorsi M, Messori M, Giudici P (2017) Increased production of bacterial cellulose as starting point for scaled-up applications. Appl Microbiol Biotechnol 101:8115–8127. https://doi.org/10.1007/s00253-017-8539-3
Gullo M, La China S, Falcone PM, Giudici P (2018) Biotechnological production of cellulose by acetic acid bacteria: current state and perspectives. Appl Microbial Biotechnol. https://doi.org/10.1007/s00253-018-9164-5
Han YW, Clarke MA (1990) Production and characterization of microbial levan. J Agric Food Chem 38:393–396. https://doi.org/10.1021/jf00092a011
Hekmat D, Bauer R, Fricke J (2003) Optimization of the microbial synthesis of dihydroxyacetone from glycerol with Gluconobacter oxydans. Bioprocess Biosyst Eng 26:109–116. https://doi.org/10.1007/s00449-003-0338-9
Herrmann U, Merfort M, Jeude M, Bringer-Meyer S, Sahm H (2004) Biotransformation of glucose to 5-keto-d-gluconic acid by recombinant Gluconobacter oxydans DSM 2343. Appl Microbiol Biotechnol 64:86–90. https://doi.org/10.1007/s00253-003-1455-8
Hidalgo C, Vegas C, Mateo E, Tesfaye W, Cerezo AB, Callejón RM, Poblet M, Guillamón JM, Mas A, Torija MJ (2010) Effect of barrel design and the inoculation of Acetobacter pasteurianus in wine vinegar production. Int J Food Microbiol 141:56–62. https://doi.org/10.1016/j.ijfoodmicro.2010.04.018
Hoshino T, Sugisawa T, Tazoe M, Shinjoh M, Fujiwara A (1990) Metabolic pathway for 2-keto-l-gulonic acid formation in Gluconobacter melanogenus IFO 3293. Agric Biol Chem 54:1211–1218. https://doi.org/10.1080/00021369.1990.10870126
Jakob F, Meissner D, Vogel RF (2012a) Comparison of novel GH 68 levansucrases of levan-overproducing Gluconobacter species. Acetic Acid Bact 1:e2. https://doi.org/10.4081/aab.2012.e2
Jakob F, Steger S, Vogel RF (2012b) Influence of novel fructans produced by selected acetic acid bacteria on the volume and texture of wheat breads. Eur Food Res Technol 234(3):493–499. https://doi.org/10.1007/s00217-011-1658-7
Jakob F, Phaff A, Novoa-Carballal R, Rübsam H, Becker T, Vogel RF (2013) Structural analysis of fructans produced by acetic acid bacteria reveals a relation to hydrocolloidal function. Carbohydr Polym 92:1234–1242. https://doi.org/10.1016/j.carbpol.2012.10.054
Jiménez-Hornero JE, Santos-Dueñas IM, García-García I (2009) Optimization of biotechnological processes. The acetic acid fermentation. Part I: the proposed model. Biochem Eng J 45:1–6. https://doi.org/10.1016/j.bej.2009.01.009
Jonas R, Farah LF (1998) Production and application of microbial cellulose. Polym Degrad Stabil 59:101–106. https://doi.org/10.1016/S0141-3910(97)00197-3
Krystynowicz A, Czaja W, Wiktorowska-Jezierska A, Gonçalves-Miśkiewicz M, Turkiewicz M, Bielecki S (2002) Factors affecting the yield and properties of bacterial cellulose. J Ind Microbiol Biotechnol 29:189–195. https://doi.org/10.1038/sj.jim.7000303
Ku H, Wang H, Pattarachaiyakoop N, Trada M (2011) A review of tensile properties of natural fiber reinforced polymer composites. Compos B 42:856–873. https://doi.org/10.1016/j.compositesb.2011.01.010
Kuo CH, Lin PJ, Lee CK (2010) Enzymatic saccharification of dissolution pretreated waste cellulosic fabrics for bacterial cellulose production by Gluconacetobacter xylinus. J Chem Technol Biotechnol 85:1346–1352. https://doi.org/10.1002/jctb.2439
Kuo CH, Chen JH, Liou BK, Lee CK (2015a) Utilization of acetate buffer to improve bacterial cellulose production by Gluconacetobacter xylinus. Food Hydrocoll 53:98–103. https://doi.org/10.1016/j.foodhyd.2014.12.034
Kuo CH, Teng HY, Lee CK (2015b) Knock-out of glucose dehydrogenase gene in Gluconacetobacter xylinus for bacterial cellulose production enhancement. Biotechnol Bioprocess E 20:18–25
Li MH, Wu J, Liu X, Lin JP, Wei DZ, Chen H (2010) Enhanced production of dihydroxyacetone from glycerol by overexpression of glycerol dehydrogenase in an alcohol dehydrogenase-deficient mutant of Gluconobacter oxydans. Bioresour Technol 101:8294–8299. https://doi.org/10.1016/j.biortech.2010.05.065
Li K, Mao X, Liu L, Lin J, Sun M, Wei D, Yang S (2016) Overexpression of membrane-bound gluconate-2-dehydrogenase to enhance the production of 2-keto-d-gluconic acid by Gluconobacter oxydans. Microb Cell Fact 15:121. https://doi.org/10.1186/s12934-016-0521-8
Lu L, Wei L, Zhu K, Wei D, Hua Q (2012) Combining metabolic engineering and adaptive evolution to enhance the production of dihydroxyacetone from glycerol by Gluconobacter oxydans in a low-cost way. Bioresour Technol 17:317–324. https://doi.org/10.1016/j.biortech.2012.03.013
Lux S, Siebenhofer M (2013) Synthesis of lactic acid from dihydroxyacetone: use of alkaline-earth metal hydroxides. Catal Sci Technol 3:1380–1385. https://doi.org/10.1039/C3CY20859A
Macauley S, McNeil B, Harvey LM (2001) The genus Gluconobacter and its applications in biotechnology. Crit Rev Biotechnol 21:1–25. https://doi.org/10.1080/20013891081665
Mamlouk D, Gullo M (2013) Acetic acid bacteria: physiology and carbon sources oxidation. Indian J Microbiol 53:377–384. https://doi.org/10.1007/s12088-013-0414-z
Mas A, Torija MJ, García-Parrilla MC, Troncoso AM (2014) Acetic acid bacteria and the production and quality of wine vinegar. Sci World J. https://doi.org/10.1155/2014/394671
Matsushita K, Toyama H, Adachi O (1994) Respiratory chains and bioenergetics of acetic acid bacteria. Adv Microb Physiol 36:247–301. https://doi.org/10.1016/S0065-2911(08)60181-2
Matsutani M, Nishikura M, Saichana N, Hatano T, Masud-Tippayasak U, Theergool G, Yakushi T, Matsushita K (2013) Adaptive mutation of Acetobacter pasteurianus SKU1108 enhances acetic acid fermentation ability at high temperature. J Biotechnol 165:109–119. https://doi.org/10.1016/j.jbiotec.2013.03.006
Mendez C, Salas JA (2001) The role of ABC transporters in antibiotic-producing organisms: drug secretion and resistance mechanisms. Res Microbiol 152:341–350. https://doi.org/10.1016/S0923-2508(01)01205-0
Méndez-Lorenzo L, Porras-Domínguez JR, Raga-Carbajal E, Olvera C, Rodríguez-Alegría ME, Carrillo-Nava E, Costas M, López Munguía A (2015) Intrinsic levanase activity of Bacillus subtilis 168 Levansucrase (SacB). ed. PLoS ONE 10:e0143394. https://doi.org/10.1371/journal.pone.0143394
Merfort M, Herrmann U, Bringer-Meyer S, Sahm H (2006) High-yield 5-keto-d-gluconic acid formation is mediated by soluble and membrane-bound gluconate-5-dehydrogenases of Gluconobacter oxydans. Appl Microbiol Biotechnol 73:443–451. https://doi.org/10.1007/s00253-006-0467-6
Muongman P, Opasanon S, Suwanchot S, Thangthed O (2011) Efficiency of microbial cellulose dressing in partial-thickness burn wounds. J Am Coll Cert Wound Special 3:16–19. https://doi.org/10.1016/j.jcws.2011.04.001
Murali N, Fernandez S, Ahring BK (2017) Fermentation of wet-exploded corn stover for the production of volatile fatty acids. Bioresour Technol 227:197–204. https://doi.org/10.1016/j.biortech.2016.12.012
Ndoye B, Lebecque S, Dubois-Dauphin R, Tounkara L, Guiro AT, Kere C, Diawara B, Thonart P (2006) Thermoresistant properties of acetic acids bacteria isolated from tropical products of Sub-Saharan Africa and destined to industrial vinegar. Enzyme Microb Technol 39:916–923. https://doi.org/10.1016/j.enzmictec.2006.01.020
Oikawa T, Morino T, Ameyama M (1995a) Production of cellulose from n-arabitol by Acetobacter xylinum KU-l. Biosci Biotechnol Biochem 59:1564–1565. https://doi.org/10.1271/bbb.59.1564
Oikawa T, Ohtori T, Ameyama M (1995b) Production of cellulose from n-mannitol by Acetobacter xylinum KU-l. Biosci Biotechnol Biochem 59:331–332. https://doi.org/10.1271/bbb.59.331
Okamoto-Kainuma A, Ishikawa M, Nakamura H, Fukazawa S, Tanaka N, Yamagami K, Koizumi Y (2011) Characterization of rpoH in Acetobacter pasteurianus NBR3283. J Biosci Bioeng 111:429–432. https://doi.org/10.1016/j.jbiosc.2010.12.016
Pagliaro M, Ciriminna R, Kimura H, Rossi M, Pina CD (2007) From glycerol to value-added products. Angew Chem 46:4434–4440. https://doi.org/10.1002/anie.200604694
Pappenberger G, Hohmann HP (2014) Industrial production of l-ascorbic acid (vitamin C) and d-isoascorbic acid. Adv Biochem Eng Biotechnol 143:143–188. https://doi.org/10.1007/10_2013_243
Qi Z, Wang W, Yang H, Xia X, Yu X (2013) Mutation of Acetobacter pasteurianus by UV irradiation under acidic stress for high-acidity vinegar fermentation. Int J Food Sci Technol 49:468–476. https://doi.org/10.1111/ijfs.12324
QYResearch Reports (2017). https://www.qyresearchreports.com/. Accessed 5 April 2018
Rairakhwada D, Pal AK, Bhathena ZP, Sahu NP, Jha A, Mukherjee SC (2007) Dietary microbial levan enhances cellular non-specific immunity and survival of common carp (Cyprinus carpio) juveniles. Fish Shellfish Immunol 22:477–486. https://doi.org/10.1016/j.fsi.2006.06.005
Roberfroid MB, Van Loo JA, Gibson GR (1998) The bifidogenic nature of chicory inulin and its hydrolysis products. J Nutr 128:11–19. https://doi.org/10.1093/jn/128.1.11
Roehr M, Kubicek CP, Kominek J (1996) Gluconic acid. In: Rehm HJ, Reed G (eds) Biotechnology, product of primary metabolism. Verlag Chemie, Weinheim, pp 347–362. https://doi.org/10.1002/9783527620999.ch10f
Rogers P, Chen JS, Zidwick MJ (2006) Organic acid and solvent production Part I: Acetic, lactic, gluconic, succinic and polyhydroxyalkanoic acids. In: Dworkin M (ed) Prokaryotes. Springer, New York, pp 511–755. https://doi.org/10.1007/978-3-642-31331-8_23
Ross P, Mayer R, Benziman M (1991) Cellulose biosynthesis and function in bacteria. Microbiol Rev 55:35–58
Rühmkorf C, Jungkunz S, Wagner M, Vogel RF (2012) Optimization of homoexopolysaccharide formation by lactobacilli in gluten-free sourdoughs. Food Microbiol 32:286–294. https://doi.org/10.1016/j.fm.2012.07.002
Ruka D, Simon GP, Dean KM (2012) Altering the growth conditions of Gluconacetobacter xylinus to maximize the yield of bacterial cellulose. Carbohydr Polym 89:613–622. https://doi.org/10.1016/j.carbpol.2012.03.059
Saichana N, Matsushita K, Adachi O, Frebort I, Frebortova J (2015) Acetic acid bacteria: a group of bacteria with versatile biotechnological applications. Biotechnol Adv 33:1260–1271. https://doi.org/10.1016/j.biotechadv.2014.12.001
Sainz F, Navarro D, Mateo E, Torija MJ, Mas A (2016) Comparison of d-gluconic acid production in selected strains of acetic acid bacteria. Int J Food Microbiol 222:40–47. https://doi.org/10.1016/j.ijfoodmicro.2016.01.015
Saito Y, Ishii Y, Hayashi H, Imao Y et al (1997) Cloning of genes coding for l-sorbose and l-sorbosone dehydrogenases from Gluconobacter oxydans and microbial production of 2-keto-l-gulonate, a precursor of l-ascorbic acid, in a recombinant G. oxydans strain. Appl Environ Microbiol 63:454–460
Sengun Y, Karabiyikli S (2011) Importance of acetic acid bacteria in food industry. Food Control 22:647–656. https://doi.org/10.1016/j.foodcont.2010.11.008
Shi L, Li K, Zhang H, Liu X, Lin J, Wei D (2014) Identification of a novel promoter gHp0169 for gene expression in Gluconobacter oxydans. J Biotechnol 175:69–74. https://doi.org/10.1016/j.jbiotec.2014.01.035
Shinagawa E, Matsushita K, Adachi O, Ameyama M (1983) Selective production of 5-keto-d-gluconate by Gluconobacter strains. J Ferment Technol 61:359–363
Shinjoh M, Toyama H (2016) Industrial application of acetic acid bacteria (Vitamin C and others). In: Matsushita K, Toyama H, Tonouchi N, Okamoto-Kainuma A (eds) Acetic acid bacteria ecology and physiology. Springer, Japan, pp 321–338. https://doi.org/10.1007/978-4-431-55933-7_15
Sima F, Mutlu EC, Eroglu MS, Sima LE, Serban N, Ristoscu C, Petrescu SM, Oner ET, Mihailescu IN (2011) Levan nanostructured thin films by MAPLE assembling. Biomacromol 12:2251–2256. https://doi.org/10.1021/bm200340b
Singh OV, Kumar R (2007) Biotechnological production of gluconic acid: future implications. Appl Microbiol Biotechnol 75:713–722. https://doi.org/10.1007/s00253-007-0851-x
Sokollek SJ, Hammes WP (1997) Description of a starter culture preparation for vinegar fermentation. Syst Appl Microbiol 20:481–491. https://doi.org/10.1016/S0723-2020(97)80017-3
Son HJ, Heo MS, Kim YG, Lee SJ (2001) Optimization of fermentation conditions for the production of bacterial cellulose by a newly isolated Acetobacter sp. A9 in shaking cultures. Biotechnol Appl Biochem 33:1–5. https://doi.org/10.1042/BA20000065
Sonoyama T, Tani H, Matsuda K, Kageyama B, Tanimoto M, Kobayashi K, Yagi S, Kyotani H, Mitsushima K (1982) Production of 2-keto-l-gulonic acid from d-glucose by two-stage fermentation. Appl Environ Microbiol 43:1064–1069
Srikanth R, Siddartha G, Sundhar Reddy CH, Harish BS, Janaki Ramaiah M, Uppuluri KB (2015) Antioxidant and anti-inflammatory levan produced from Acetobacter xylinum NCIM2526 and its statistical optimization. Carbohydr Polym 123:8–16. https://doi.org/10.1016/j.carbpol.2014.12.079
Stottmeister U, Aurich A, Wilde H, Andersch J, Schemidt S, Sicker D (2005) White biotechnology for green chemistry: fermentative 2-oxocarboxylic acids as novel building blocks for subsequent chemical syntheses. J Ind Microbiol Biotechnol 32:651. https://doi.org/10.1007/s10295-005-0254-x
Sugisawa T, Hoshino T, Nomura S, Fujiwara A (1991) Isolation and characterization of membrane-bound l-sorbose dehydrogenase from Gluconobacter melanogenus UV10. Agric Biol Chem 55:363–370. https://doi.org/10.1080/00021369.1991.10870609
Thompson DN, Hamilton MA (2001) Production of bacterial cellulose from alternate feedstocks. Appl Biochem Biotechnol 91–93:503–513. https://doi.org/10.1385/ABAB:91-93:1-9:503
Toyama H, Soemphol W, Moonmangmee D, Adachi O, Matsushita K (2005) Molecular properties of membrane-bound FAD-containing D-sorbitol dehydrogenase from thermotolerant Gluconobacter frateurii isolated from Thailand. Biosci Biotechnol Biochem 69:1120–1129. https://doi.org/10.1271/bbb.69.1120
Ua-Arak T, Jakob F, Vogel RF (2016) Characterization of growth and exopolysaccharide production of selected acetic acid bacteria in buckwheat sourdoughs. Int J Food Microbiol 239:103–112. https://doi.org/10.1016/j.ijfoodmicro.2016.04.009
Ua-Arak T, Jakob F, Vogel RF (2017) Modulation of size distributions and functional properties in gluten-free baking of Gluconobacter albidus TMW 2.1191 levan in fermentations with controlled pH. Front Microbiol 8:807. https://doi.org/10.3389/fmicb.2017.00807
Vidra A, Németh Á (2018) Bio-produced acetic acid: a review. Period Polytech Chem 62(3):245–256. https://doi.org/10.3311/PPch.11004
Wei SH, Song QX, Wei DZ (2007) Repeated use of immobilized Gluconobacter oxydans cells for conversion of glycerol to dihydroxyacetone. Prep Biochem Biotechnol 37:67–76. https://doi.org/10.1080/10826060601040954
Wu JJ, Gullo M, Chen FS, Giudici P (2010) Diversity of Acetobacter pasteurianus strains isolated from solid-state fermentation of cereal vinegars. Curr Microbiol 60:280–286. https://doi.org/10.1007/s00284-009-9538-0
Xu S, Wang W, Du G, Zhou J, Chen J (2014) Enhanced production of l-sorbose from d-sorbitol by improving the mRNA abundance of sorbitol dehydrogenase in Gluconobacter oxydans WSH-003. Microb Cell Fact 13:146. https://doi.org/10.1186/s12934-014-0146-8
Yoo S, Yoon E, Cha E, Lee H (2004) Antitumor activity of levan polysaccharides from selected microorganisms. Int J Biol Macromol 34:37–41. https://doi.org/10.1016/j.ijbiomac.2004.01.002
Zheng Y, Wang J, Bai X, Chang Y, Mou J, Song J, Min W (2018) Improving the acetic acid tolerance and fermentation of Acetobacter pasteurianus by nucleotide excision repair protein UvrA. Appl Microbiol Biotechnol. https://doi.org/10.1007/s00253-018-9066-6
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
La China, S., Zanichelli, G., De Vero, L. et al. Oxidative fermentations and exopolysaccharides production by acetic acid bacteria: a mini review. Biotechnol Lett 40, 1289–1302 (2018). https://doi.org/10.1007/s10529-018-2591-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-018-2591-7