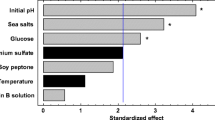
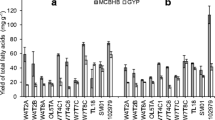
Abstract
Marine microheterotrophs thraustochytrids are emerging as a potential source for commercial production of polyunsaturated fatty acids (PUFA) that have nutritional and pharmacological values. With prospective demand for PUFAs increasing, biotechnological companies are looking for potential increases in those valuable products. However, high levels of NaCl in the culture media required for optimal thraustochytrid growth and PUFA production poses a significant problem to the biotechnological industry due to corrosion of fermenters calling for a need to reduce the amount of NaCl in the culture media, without imposing penalties on growth and yield of cultured organisms. Earlier, as reported by Shabala et al. (Environ Microbiol 11:1835–1843, 2009), we have shown that thraustochytrids use sodium predominantly for osmotic adjustment purposes and, as such, can be grown in low-salt environment without growth penalties, providing the media osmolality is adjusted. In this study, we verify if that conclusion, made for one specific strain and osmolyte only, is applicable to the larger number of strains and organic osmotica, as well as address the issue of yield quality (e.g., PUFA production in low-saline media). Using mannitol and sucrose for osmotic adjustment of the growth media enabled us to reduce NaCl concentration down to 1 mM; this is 15–100-fold lower than any method proposed so far. At the same time, the yield of essential PUFAs was increased by 15 to 20 %. Taken together, these results suggest that the proposed method can be used in industrial fermenters for commercial PUFA production.


Similar content being viewed by others
References
Arafiles KHV, Alcantara JCO, Batoon JAL, Galura FS, Cordero PRF, Leaño EM, Dedeles GR (2011) Cultural optimization of thraustochytrids for biomass and fatty acid production. Mycosphere 2:521–531
Bahnweg G (1979) Studies on the physiology of Thraustochytriales. I. Growth requirements and nitrogen nutrition of Thraustochytrium spp., Schizochytrium sp., Japonochytrium sp., Ulkenia spp., and Labyrinthuloides spp. Veroff Inst Meeresforsch. Bremerhaven 17:245–268
Bajpai PK, Bajpai P, Ward OP (1991) Optimization of production of docosahexaenoic acid (DHA) by Thraustochytrium aureum ATCC 34304. J Amer Oil Chem Soc 68:509–514
Barclay WR (1999) Reducing corrosion in a fermentor by providing sodium with a non-chloride sodium salt. US Patent 6,410,281, 14 Dec 1999
Barclay WR, Meager KM, Abril JR (1994) Heterotrophic production of long chain omega-3 fatty acids utilizing algae and algaelike microorganisms. J Appl Phycol 6:123–129
Barclay WR, Weaver C, Metz J (2005) Development of a docosahexaenoic acid production technology using Schizochytrium: a historical perspective. In: Cohen Z, Ratledge C (eds) Single cell oil. AOCS, Champaign
Batbatan CG, Hepowit NL, Oclarit JM (2011) Developmental historicity and saccharide heterotrophy of Schizochytrium sp OT01: implication of docosahexaenoic acid production for biotechnological applications. Asia Life Sci 20:289–305
Burja AM, Radianingtyas H, Windust A, Barrow CJ (2006) Isolation and characterization of polyunsaturated fatty acid producing Thraustochytrium species: screening of strains and optimization of omega-3 production. Appl Microbiol Biotechnol 72:1161–1169
Carter CG, Bransden MP, Lewis TE, Nichols PD (2003) Potential of thraustochytrids to partially replace fish oil in Atlantic salmon feeds. Marine Biotechnol 5:480–492
Garrill A, Clipson NJW, Jennings DH (1992) Preliminary observations on the monovalent cation relations of Thraustochytrium aureum, a fungus requiring sodium for growth. Mycol Res 96:295–304
Goldstein S (1963) Development and nutrition of new species of Thraustochytrium. Am J Bot 50:271–279
Honda D, Yokochi T, Nakahara T, Ragukumar S, Nakagiri A, Schaumann K, Higashihara T (1999) Molecular phylogeny of labyrnthulids and thraustochytrids based on sequencing of 18S ribosomal RNA gene. J Eukar Microbiol 46:637–647
Horrocks LA, Yeo YK (1999) Health benefits of docosahexaenoic acid (DHA). Pharmacol Res 40:211–225
Iida I, Nakahara T, Yokochi T, Kamisaka Y, Yagi H, Yamaoka M, Suzuki O (1996) Improvement of docosahexaenoic acid production in a culture of Thraustochytrium aereum by medium optimisation. J Ferment Bioeng 81:76–78
Jakobsen AN, Aasen IM, Strom AR (2007) Endogenously synthesized (−)-proto-quercitol and glycine betaine are principal compatible solutes of Schizochytrium sp strain S8 (ATCC 20889) and three new isolates of phylogenetically related thraustochytrids. Appl Environ Microbiol 73:5848–5856
James MJ, Ursin VM, Cleland LG (2003) Metabolism of stearidonic acid in human subjects: comparison with the metabolism of other n-3 fatty acids. Am J Clin Nutrition 77:1140–1145
Kamagata Y, Tamaki H (2005) Cultivation of uncultured fastidious microbes. Microb Environ 20:85–91
Leaño EM, Gapasin RSJ, Polohan B, Vrijmoed LLP (2003) Growth and fatty acid production of thraustochytrids from Panay mangroves, Philippines. Fungal Diversity 12:111–122
Lewis TE, Nichols PD, McMeekin TA (1999) The biotechnological potential of thraustochytrids. Mar Biotechnol 1:580–587
Lewis T, Nichols PD, McMeekin TA (2000) Evaluation of extraction methods for recovery of fatty acids from lipidproducing microheterotrophs. J Microbiol Methods 43:107–116
Li ZY, Ward OP (1994) Production of docosahexaenoic acid (DHA) by Thraustocytrium roseum. J Ind Microbiol 13:238–341
Lopez-Garcia P, Rodriguez-Valera, Pedros-Allo C, Moreira D (2001) Unexpected diversity of small eukaryotes in deep-sea Antarctic plankton. Nature 409:603–607
Nagano N, Taoka Y, Honda D, Hayashi M (2009) Optimization of culture conditions for growth and docosahexaenoic acid production by a marine thraustochytrid, Aurantiochytrium limacinum mh0186. J Oleo Sci 58:623–628
Nichols DS, Hart P, Nichols PD, McMeekin TA (1996) Enrichment of the rotifer Brachionus plicatilis fed an Antarctic bacterium containing polyunsaturated fatty acids. Aquaculture 147:115–125
Quilodrán B, Hinzpeter I, Quiroz A, Shene C (2009) Evaluation of liquid residues from beer and potato processing for the production of docosahexaenoic acid (C22:6n-3, DHA) by native thraustochytrid strains. World J Microbiol Biotechnol 21:2121–2128
Raghukumar S (2008) Thraustochytrid marine protists: production of PUFAs and other emerging technologies. Mar Biotechnol 10:631–640
Ratledge C (2012) Omega-3 biotechnology: errors and omissions. Biotechnol Adv 30:1746–1747
Shabala L, Shabala S, Ross T, McMeekin T (2001) Membrane transport activity and ultradian ion flux oscillations associated with cell cycle of Thraustochytrium sp. Aust J Plant Physiol 28:87–99
Shabala L, McMeekin T, Shabala S (2009) Osmotic adjustment and requirement for sodium in marine protist thraustochytrid. Environ Microbiol 11:1835–1843
Shene C, Leyton A, Esparza Y, Flores L, Quilodrán B, Hinzpeter I, Rubilar M (2010) Microbial oils and fatty acids: effect of carbon source on docosahexaenoic acid (C22:6 N-3, DHA) production by thraustochytrid strains. J Soil Sci Plant Nutr 10(3):207–216
Simopoulos AP (1999) Omega-3 fatty acids in health and disease and in growth and development. Am J Clin Nutrition 54:438–463
Singh A, Ward OP (1996) Production of high yields of docosahexaenoic acid by Thraustochytrium roseum ATCC 28210. J Indust Microbiol 16:370–373
Unagul P, Assantachai C, Phadungruengluij S, Suphantharika M, Verduyn C (2005) Properties of the docosahexaenoic acid-producer Schizochytrium mangrovei Sk-02: effects of glucose, temperature and salinity and their interaction. Bot Marina 48:387–394
Unagul P, Assantachai C, Phadungruengluij S, Pongsuteeragul T, Suphantharika M, Verduyn C (2006) Biomass and docosahexaenoic acid formation by Schizochytrium mangrovei Sk-02 at low salt concentrations. Bot Marina 49:182–190
Unagul P, Assantachai C, Phadungruengluij S, Suphantharika M, Tanticharoen M, Verduyn V (2007) Coconut water as a medium additive for the production of docosahexaenoic acid (C22:6n3) by Schizochytrium mangrovei Sk-02. Bioresource Technol 98:281–287
Ward OP, Singh A (2005) Omega-3/6 fatty acids: alternative sources of production. Process Biochem 40:3627–3652
White DC, Davis WM, Nickels JS, King JD, Bobbie RJ (1979) Determination of the sedimentary microbial biomass by extractable lipid phosphate. Oecologia 40:51–62
Yokochi T, Honda D, Higashihara T, Nakahara T (1998) Optimization of docosahexaenoic acid production by Schizochytrium limacinum SR21. Appl Microbiol Biotechnol 49:72–76
Yamasaki T, Aki T, Shinozaki M, Taguchi M, Kawamoto S, Ono K (2006) Utilization of shochu distillery wastewater for production of polyunsaturated fatty acids and xanthophylls using thraustochytrid. J Biosci Bioeng 102:323–327
Zhou L, Lu YH, Zhou MH, Zhao XW (2007) Enhanced production of docosahexaenoic acid using Schizochytrium sp by optimization of medium components. J Chem Eng Japan 40:1093–1100
Acknowledgments
This work was supported by ARC Discovery and University of Tasmania research grants to Prof. Sergey Shabala. We thank Dr. T. Lewis for his contribution to fatty acid analyses and information on strains origin.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shabala, L., McMeekin, T. & Shabala, S. Thraustochytrids Can Be Grown in Low-Salt Media Without Affecting PUFA Production. Mar Biotechnol 15, 437–444 (2013). https://doi.org/10.1007/s10126-013-9499-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-013-9499-y