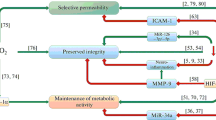
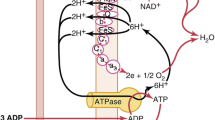
Abstract
Oxygen (O2) supplementation is commonly used to treat hypoxia in patients with respiratory failure. However, indiscriminate use can lead to hyperoxia, a condition detrimental to living tissues, particularly the brain. The brain is sensitive to reactive oxygen species (ROS) and inflammation caused by high concentrations of O2, which can result in brain damage and mitochondrial dysfunction, common features of neurodegenerative disorders. Hyperoxia leads to increased production of ROS, causing oxidative stress, an imbalance between oxidants and antioxidants, which can damage tissues. The brain is particularly vulnerable to oxidative stress due to its lipid composition, high O2 consumption rate, and low levels of antioxidant enzymes. Moreover, hyperoxia can cause vasoconstriction and decreased O2 supply to the brain, posing a challenge to redox balance and neurodegenerative processes. Studies have shown that the severity of hyperoxia-induced brain damage varies with inspired O2 concentration and duration of exposure. Therefore, careful evaluation of the balance between benefits and risks of O2 supplementation, especially in clinical settings, is crucial.

Similar content being viewed by others
Data Availability
Not applicable.
References
Andresen JH, Saugstad OD (2020) Oxygen metabolism and oxygenation of the newborn. Semin Fetal Neonatal Med 25:101078. https://doi.org/10.1016/j.siny.2020.101078
Bailey DM (2019a) Oxygen, evolution and redox signalling in the human brain; quantum in the quotidian. J Physiol 597:15–28. https://doi.org/10.1113/JP276814
Bailey DM (2019b) Oxygen is rocket fuel for the human brain; a radical perspective! J Physiol 597:659–660. https://doi.org/10.1113/JP277475
Barichello T, Lemos JC, Generoso JS et al (2011) Oxidative stress, Cytokine/Chemokine and disruption of blood–brain barrier in neonate rats after Meningitis by Streptococcus agalactiae. Neurochem Res 36:1922–1930. https://doi.org/10.1007/s11064-011-0514-2
Beasley R, Chien J, Douglas J et al (2015) Thoracic society of Australia and New Zealand oxygen guidelines for acute oxygen use in adults: swimming between the flags. Respirology 20:1182–1191. https://doi.org/10.1111/resp.12620
Bonfante S, Della Giustina A, Danielski LG et al (2020) Stanniocalcin-1 ameliorates cerebral ischemia by decrease oxidative stress and blood brain barrier permeability. Microvasc Res 128:103956. https://doi.org/10.1016/j.mvr.2019.103956
Brugniaux JV, Coombs GB, Barak OF et al (2018) Highs and lows of hyperoxia: physiological, performance, and clinical aspects. Am J Physiol Integr Comp Physiol 315:R1–R27. https://doi.org/10.1152/ajpregu.00165.2017
Burtscher J, Mallet RT, Pialoux V et al (2022) Adaptive responses to Hypoxia and/or Hyperoxia in humans. Antioxid Redox Signal 37:887–912. https://doi.org/10.1089/ars.2021.0280
Cancelier AC, Petronilho F, Reinke A et al (2009) Inflammatory and oxidative parameters in cord blood as diagnostic of early-onset neonatal sepsis: a case-control study. Pediatr Crit Care Med 10:467–471. https://doi.org/10.1097/PCC.0b013e318198b0e3
Carvalho D, Petronilho F, Vuolo F et al (2008) The nociceptin/orphanin FQ-NOP receptor antagonist effects on an animal model of sepsis. Intensive Care Med 34:2284–2290. https://doi.org/10.1007/s00134-008-1313-3
Cenini G, Lloret A, Cascella R (2019) Oxidative stress in neurodegenerative diseases: from a mitochondrial point of View. Oxid Med Cell Longev 2019:1–18. https://doi.org/10.1155/2019/2105607
Cheung HM, Yew DTW (2020) Neuroprotective mechanisms of Ginkgo biloba against oxidative stress. Oxidative stress and Dietary antioxidants in Neurological diseases. Elsevier, pp 271–290
Chu DK, Kim LHY, Young PJ et al (2018) Mortality and morbidity in acutely ill adults treated with liberal versus conservative oxygen therapy (IOTA): a systematic review and meta-analysis. Lancet 391:1693–1705. https://doi.org/10.1016/S0140-6736(18)30479-3
Dal-Pizzol F, Ritter C, Cassol-Jr OJ et al (2010) Oxidative mechanisms of Brain Dysfunction during Sepsis. Neurochem Res 35:1–12. https://doi.org/10.1007/s11064-009-0043-4
Damiani E, Donati A, Girardis M (2018) Oxygen in the critically ill. Curr Opin Anaesthesiol 31:129–135. https://doi.org/10.1097/ACO.0000000000000559
Davies KJA (1995) Oxidative stress: the paradox of aerobic life. Biochem Soc Symp 61:1–31. https://doi.org/10.1042/bss0610001
DeFreitas MJ, Katsoufis CP, Benny M et al (2022) Educational Review: the impact of perinatal oxidative stress on the developing kidney. Front Pediatr 10. https://doi.org/10.3389/fped.2022.853722
Di Meo S, Venditti P (2020) Evolution of the knowledge of free radicals and other oxidants. Oxid Med Cell Longev 2020:1–32. https://doi.org/10.1155/2020/9829176
Echevarria C, Steer J, Wason J, Bourke S (2021) Oxygen therapy and inpatient mortality in COPD exacerbation. Emerg Med J 38:170–177. https://doi.org/10.1136/emermed-2019-209257
Endesfelder S, Weichelt U, Strauß E et al (2017) Neuroprotection by Caffeine in Hyperoxia-Induced neonatal brain Injury. Int J Mol Sci 18:187. https://doi.org/10.3390/ijms18010187
Fedorova M, Bollineni RC, Hoffmann R (2014) Protein carbonylation as a major hallmark of oxidative damage: update of analytical strategies. Mass Spectrom Rev 33:79–97. https://doi.org/10.1002/mas.21381
Gibbons TD, Dempsey JA, Thomas KN et al (2022) Contribution of the carotid body to thermally mediated hyperventilation in humans. J Physiol 600:3603–3624. https://doi.org/10.1113/JP282918
Giridharan VV, Masud F, Petronilho F et al (2019) Infection-Induced systemic inflammation is a potential driver of Alzheimer’s Disease Progression. Front Aging Neurosci 11. https://doi.org/10.3389/fnagi.2019.00122
Giustina A, Della, Bonfante S, Zarbato GF et al (2018) Dimethyl Fumarate modulates oxidative stress and inflammation in organs after Sepsis in rats. Inflammation 41:315–327. https://doi.org/10.1007/s10753-017-0689-z
Grieco DL, Maggiore SM, Roca O et al (2021) Non-invasive ventilatory support and high-flow nasal oxygen as first-line treatment of acute hypoxemic respiratory failure and ARDS. Intensive Care Med 47:851–866. https://doi.org/10.1007/s00134-021-06459-2
Hadanny A, Efrati S (2020) The Hyperoxic-Hypoxic Paradox. Biomolecules 10:958. https://doi.org/10.3390/biom10060958
Halliwell B, Gutteridge JMC (2007) Free radicals in biology and medicine, 4th edn. Oxford University Press
Halliwell B, Gutteridge JMC (2015) Free radicals in Biology and Medicine, 5th edn. Oxford University Press, Oxford, UK
Hals I, Ohki T, Singh R et al (2017) Hyperoxia reduces insulin release and induces mitochondrial dysfunction with possible implications for hyperoxic treatment of neonates. Physiol Rep 5:e13447. https://doi.org/10.14814/phy2.13447
Haynes RL, Folkerth RD, Keefe RJ et al (2003) Nitrosative and oxidative Injury to Premyelinating oligodendrocytes in Periventricular Leukomalacia. J Neuropathol Exp Neurol 62:441–450. https://doi.org/10.1093/jnen/62.5.441
Hegeduš I, Milić J, Ćosić A et al (2017) Cerebrovascular reactivity in acute hyperoxia in patients with acute ischaemic stroke. Brain Inj 31:560–566. https://doi.org/10.1080/02699052.2017.1280853
Herrera-Campos AB, Zamudio-Martinez E, Delgado-Bellido D et al (2022) Implications of Hyperoxia over the Tumor Microenvironment: an overview highlighting the importance of the Immune System. Cancers (Basel) 14:2740. https://doi.org/10.3390/cancers14112740
Hess HW, Hostler D, Clemency BM et al (2021) Carotid body chemosensitivity is not attenuated during cold water diving. Am J Physiol Integr Comp Physiol 321:R197–R207. https://doi.org/10.1152/ajpregu.00202.2020
Hu T, Wang J, Wang S et al (2019) Effects of the duration of postresuscitation hyperoxic ventilation on neurological outcome and survival in an asphyxial cardiac arrest rat model. Sci Rep 9:16500. https://doi.org/10.1038/s41598-019-52477-y
Iturriaga R, Alcayaga J, Chapleau MW, Somers VK (2021) Carotid body chemoreceptors: physiology, pathology, and implications for health and disease. Physiol Rev 101:1177–1235. https://doi.org/10.1152/physrev.00039.2019
Kang L, Dong W, Ruan Y et al (2019) The molecular mechanism of Sirt1 Signaling Pathway in Brain Injury of Newborn rats exposed to Hyperoxia. Biol Pharm Bull 42:1854–1860. https://doi.org/10.1248/bpb.b19-00382
Kapadia V, Wyckoff MH (2018) Oxygen therapy in the delivery room. Clin Perinatol 45:293–306. https://doi.org/10.1016/j.clp.2018.01.014
Körpınar Ş, Uzun H (2019) The effects of hyperbaric oxygen at different pressures on oxidative stress and antioxidant status in rats. Med (B Aires) 55:205. https://doi.org/10.3390/medicina55050205
Kwak DJ, Kwak SD, Gauda EB (2006) The Effect of Hyperoxia on reactive oxygen species (ROS) in rat petrosal ganglion neurons during Development using Organotypic slices. Pediatr Res 60:371–376. https://doi.org/10.1203/01.pdr.0000239817.39407.61
Lacasse Y, Casaburi R, Sliwinski P et al (2022) Home oxygen for moderate hypoxaemia in chronic obstructive pulmonary disease: a systematic review and meta-analysis. Lancet Respir Med 10:1029–1037. https://doi.org/10.1016/S2213-2600(22)00179-5
Machado RS, Tenfen L, Joaquim L et al (2022) Hyperoxia by short-term promotes oxidative damage and mitochondrial dysfunction in rat brain. Respir Physiol Neurobiol 306:103963. https://doi.org/10.1016/j.resp.2022.103963
Mackle D, Beasley R, Bellomo R et al (2020) Conservative oxygen therapy during mechanical ventilation in the ICU. N Engl J Med 382:989–998. https://doi.org/10.1056/NEJMoa1903297
Mancardi D, Ottolenghi S, Attanasio U et al (2022) Janus, or the Inevitable Battle between too much and too little oxygen. Antioxid Redox Signal 37:972–989. https://doi.org/10.1089/ars.2021.0232
Martin DS, Grocott MPW (2013) Oxygen therapy in critical illness. Crit Care Med 41:423–432. https://doi.org/10.1097/CCM.0b013e31826a44f6
Mattos JD, Campos MO, Rocha MP et al (2019) Human brain blood flow and metabolism during isocapnic hyperoxia: the role of reactive oxygen species. J Physiol 597:741–755. https://doi.org/10.1113/JP277122
McEvoy JW (2018) Excess oxygen in acute illness: adding fuel to the fire. Lancet 391:1640–1642. https://doi.org/10.1016/S0140-6736(18)30546-4
Modun D, Krnic M, Vukovic J et al (2012) Plasma nitrite concentration decreases after hyperoxia-induced oxidative stress in healthy humans. Clin Physiol Funct Imaging 32:404–408. https://doi.org/10.1111/j.1475-097X.2012.01133.x
Mutch WAC, El-Gabalawy R, Ryner L et al (2020) Brain BOLD MRI O2 and CO2 stress testing: implications for perioperative neurocognitive disorder following surgery. Crit Care 24. https://doi.org/10.1186/s13054-020-2800-3
Nayernia Z, Jaquet V, Krause K-H (2014) New insights on NOX enzymes in the Central Nervous System. Antioxid Redox Signal 20:2815–2837. https://doi.org/10.1089/ars.2013.5703
Netto MB, de Oliveira Junior AN, Goldim M et al (2018) Oxidative stress and mitochondrial dysfunction contributes to postoperative cognitive dysfunction in elderly rats. Brain Behav Immun 73:661–669. https://doi.org/10.1016/j.bbi.2018.07.016
Ni Y-N, Wang Y-M, Liang B-M, Liang Z-A (2019) The effect of hyperoxia on mortality in critically ill patients: a systematic review and meta analysis. BMC Pulm Med 19:53. https://doi.org/10.1186/s12890-019-0810-1
O’Driscoll B, Howard L, Earis J et al (2017) BTS guidelines for oxygen use in healthcare. Thorax 72:43–45
Obermeier B, Daneman R, Ransohoff RM (2013) Development, maintenance and disruption of the blood-brain barrier. Nat Med 19:1584–1596. https://doi.org/10.1038/nm.3407
Onodera K, Omoi N-O, Fukui K et al (2003) Oxidative damage of rat cerebral cortex and Hippocampus, and changes in Antioxidative Defense systems caused by Hyperoxia. Free Radic Res 37:367–372. https://doi.org/10.1080/1071576031000090019
Ottolenghi S, Sabbatini G, Brizzolari A et al (2020) Hyperoxia and oxidative stress in anesthesia and critical care medicine. Minerva Anestesiol 86:64–75. https://doi.org/10.23736/S0375-9393.19.13906-5
Page D, Ablordeppey E, Wessman BT et al (2018) Emergency department hyperoxia is associated with increased mortality in mechanically ventilated patients: a cohort study. Crit Care 22:9. https://doi.org/10.1186/s13054-017-1926-4
Panfoli I, Candiano G, Malova M et al (2018) Oxidative stress as a primary risk factor for brain damage in Preterm newborns. Front Pediatr 6. https://doi.org/10.3389/fped.2018.00369
Perez M, Robbins ME, Revhaug C, Saugstad OD (2019) Oxygen radical disease in the newborn, revisited: oxidative stress and disease in the newborn period. Free Radic Biol Med 142:61–72. https://doi.org/10.1016/j.freeradbiomed.2019.03.035
Perrone S, Laschi E, Buonocore G (2020) Oxidative stress biomarkers in the perinatal period: diagnostic and prognostic value. Semin Fetal Neonatal Med 25:101087. https://doi.org/10.1016/j.siny.2020.101087
Pneumonia H (2005) Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med 171:388–416. https://doi.org/10.1164/rccm.200405-644ST
Polat İ, Cilaker Mıcılı S, Çalışır M et al (2020) Neuroprotective effects of Lacosamide and Memantine on Hyperoxic Brain Injury in rats. Neurochem Res 45:1920–1929. https://doi.org/10.1007/s11064-020-03056-5
Prabhakar NR, Peng Y-J, Nanduri J (2020) Hypoxia-inducible factors and obstructive sleep apnea. J Clin Invest 130:5042–5051. https://doi.org/10.1172/JCI137560
Prabhakar NR, Peng Y-J, Nanduri J (2022) Adaptive cardiorespiratory changes to chronic continuous and intermittent hypoxia. In: Handbook Of Clinical Neurology. pp 103–123
Radi R (2018) Oxygen radicals, nitric oxide, and peroxynitrite: Redox pathways in molecular medicine. Proc Natl Acad Sci 115:5839–5848. https://doi.org/10.1073/pnas.1804932115
Rasouli M, Fattahi R, Nuoroozi G et al (2023) The role of oxygen tension in cell fate and regenerative medicine: implications of hypoxia/hyperoxia and free radicals. https://doi.org/10.1007/s10561-023-10099-9. Cell Tissue Bank
Reich B, Hoeber D, Bendix I, Felderhoff-Mueser U (2016) Hyperoxia and the immature brain. Dev Neurosci 38:311–330. https://doi.org/10.1159/000454917
Reidy BTG, Whyte P, Neligan PJ (2020) Is oxygen toxic? In: Deutschman C, Neligan P (eds) Evidence-based practice of critical care, 3rd edn. Elsevier, Ontario, pp 36–42e1
Réus GZ, Carlessi AS, Titus SE et al (2015) A single dose of S -ketamine induces long‐term antidepressant effects and decreases oxidative stress in adulthood rats following maternal deprivation. Dev Neurobiol 75:1268–1281. https://doi.org/10.1002/dneu.22283
Ricard J-D, Roca O, Lemiale V et al (2020) Use of nasal high flow oxygen during acute respiratory failure. Intensive Care Med 46:2238–2247. https://doi.org/10.1007/s00134-020-06228-7
Riley CM, Sciurba FC (2019) Diagnosis and Outpatient Management of Chronic Obstructive Pulmonary Disease. JAMA 321:786. https://doi.org/10.1001/jama.2019.0131
Rincon F, Kang J, Maltenfort M et al (2014) Association between Hyperoxia and Mortality after Stroke. Crit Care Med 42:387–396. https://doi.org/10.1097/CCM.0b013e3182a27732
Semenza GL, Prabhakar NR (2018) The role of hypoxia-inducible factors in carotid body (patho) physiology. J Physiol 596:2977–2983. https://doi.org/10.1113/JP275696
Siemieniuk RAC, Chu DK, Kim LH-Y et al (2018) Oxygen therapy for acutely ill medical patients: a clinical practice guideline. https://doi.org/10.1136/bmj.k4169. BMJ k4169
Sies H, Berndt C, Jones DP (2017) Oxidative stress. Annu Rev Biochem 86:715–748. https://doi.org/10.1146/annurev-biochem-061516-045037
Singh A, Kukreti R, Saso L, Kukreti S (2019) Oxidative stress: a key modulator in neurodegenerative diseases. Molecules 24:1583. https://doi.org/10.3390/molecules24081583
Smit B, Smulders YM, van der Wouden JC et al (2018) Hemodynamic effects of acute hyperoxia: systematic review and meta-analysis. Crit Care 22:45. https://doi.org/10.1186/s13054-018-1968-2
Soriano SG, McCann ME (2020) Is Anesthesia Bad for the brain? Current knowledge on the impact of anesthetics on the developing brain. Anesthesiol Clin 38:477–492. https://doi.org/10.1016/j.anclin.2020.05.007
Stocco E, Barbon S, Tortorella C et al (2020) Growth factors in the carotid Body—An update. Int J Mol Sci 21:7267. https://doi.org/10.3390/ijms21197267
Suh J-Y, Cho G, Song Y et al (2018) Hyperoxia-Induced ∆R 1. Stroke 49:3012–3019. https://doi.org/10.1161/STROKEAHA.118.021469
Suzuki S, Eastwood GM, Peck L et al (2013) Current oxygen management in mechanically ventilated patients: a prospective observational cohort study. J Crit Care 28:647–654. https://doi.org/10.1016/j.jcrc.2013.03.010
Terraneo L, Paroni R, Bianciardi P et al (2017) Brain adaptation to hypoxia and hyperoxia in mice. Redox Biol 11:12–20. https://doi.org/10.1016/j.redox.2016.10.018
Tewari D, Sah AN, Bawari S et al (2020) Role of nitric oxide in neurodegeneration: function, regulation, and inhibition. Curr Neuropharmacol 19:114–126. https://doi.org/10.2174/1570159X18666200429001549
Torres-Cuevas I, Parra-Llorca A, Sánchez-Illana A et al (2017) Oxygen and oxidative stress in the perinatal period. Redox Biol 12:674–681. https://doi.org/10.1016/j.redox.2017.03.011
Uzun A-B, Iliescu MG, Stanciu L-E et al (2023) Effectiveness of intermittent hypoxia–hyperoxia therapy in different pathologies with possible metabolic implications. Metabolites 13:181. https://doi.org/10.3390/metabo13020181
Victor VM, Rocha M, De la Fuente M (2004) Immune cells: free radicals and antioxidants in sepsis. Int Immunopharmacol 4:327–347. https://doi.org/10.1016/j.intimp.2004.01.020
Wilson DF, Matschinsky FM (2019) Hyperbaric oxygen toxicity in brain: a case of hyperoxia induced hypoglycemic brain syndrome. Med Hypotheses 132:109375. https://doi.org/10.1016/j.mehy.2019.109375
Xiong M, Zhao Y, Mo H et al (2021) Intermittent hypoxia increases ROS/HIF-1α ‘related oxidative stress and inflammation and worsens bleomycin-induced pulmonary fibrosis in adult male C57BL/6J mice. Int Immunopharmacol 100:108165. https://doi.org/10.1016/j.intimp.2021.108165
Yamamoto N, Oyaizu T, Enomoto M et al (2020) VEGF and bFGF induction by nitric oxide is associated with hyperbaric oxygen-induced angiogenesis and muscle regeneration. Sci Rep 10:2744. https://doi.org/10.1038/s41598-020-59615-x
Zaja-Milatovic S, Gupta RC (2020) Excitotoxicity, oxidative stress, and neuronal injury. In: Handbook of Toxicology of Chemical Warfare Agents, 3rd ed. Elsevier, pp 795–810
Zhilyaev SY, Moskvin AN, Platonova TF et al (2003) Hyperoxic vasoconstriction in the brain is mediated by inactivation of nitric oxide by Superoxide Anions. Neurosci Behav Physiol 33:783–787. https://doi.org/10.1023/a:1025145331149
Acknowledgements
Not applicable.
Funding
All work was independently funded by the authors.
Author information
Authors and Affiliations
Contributions
RSM, KM, LJ, RWQ, GTZ, and FP wrote the review article. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Simon Machado, R., Mathias, K., Joaquim, L. et al. Hyperoxia and brain: the link between necessity and injury from a molecular perspective. Neurotox Res 42, 25 (2024). https://doi.org/10.1007/s12640-024-00702-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12640-024-00702-6