Abstract
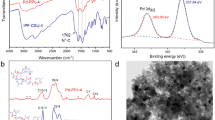
An efficient and stable palladium species catalyst immobilized on functionalized hyper–cross-linked polymers (HCPs-Pd) has been successfully developed and applied in the Suzuki–Miyaura coupling reaction of diverse types of aromatic halides with aryl boronic acid in this work. The results demonstrated that HCPs-Pd exhibited high catalytic activity, which benefited from the microporous structure of the catalyst guaranteed high dispersibility of active palladium, and high specific surface area, large pore volume, good chemical stability. Under optimal reaction conditions, 93.0% of biphenyl yield in the Suzuki–Miyaura reaction with bromobenzene (C6H5Br) and phenylboronic acid (C6H7BO2) as raw material was obtained. The good stability of the HCPs-II-Pd catalyst was verified by five cycles experiments. Perhaps this work provides new insights into the high-yield preparation of aromatic biphenyl compounds employing HCPs-Pd as an efficient and stable catalyst in the Suzuki–Miyaura reaction.
Graphical abstract












Similar content being viewed by others
Data availability
All data generated or analyzed in this study are included in this article.
References
Y. Gu, S.U. Son, T. Li, B. Tan, Adv. Funct. Mater. 31, 2008265 (2020)
Y. Liu, X. Fan, X. Jia, B. Zhang, H. Zhang, A. Zhang, Q. Zhang, J. Mater. Sci. 51, 8579 (2016)
L. Tan, B. Tan, Chem. Soc. Rev. 46, 3322 (2017)
A. Croce, G. Re, C. Bisio, G. Gatti, S. Coluccia, L. Marchese, Res. Chem. Intermed. 47, 419 (2021)
Z. Duan, Y. Wang, Q. Pan, Y. Xie, Z. Chen, Chin. J. Polym. Sci. 40, 310 (2022)
J. Li, X. Wang, G. Chen, D. Li, Y. Zhou, X. Yang, J. Wang, Appl. Catal. B Environ. 29, 87 (2012)
S. Gorji, R. Ghorbani-Vaghei, Appl. Organomet. Chem. 35, e6018 (2021)
C. Yue, Q. Xing, P. Sun, Z. Zhao, H. Lv, F. Li, Nat. Commun. 12, 1875 (2021)
T. Ratvijitvech, Polymer 14, 2749 (2022)
S. Sadjadi, M. Malmir, N. Pourmohammad, S. Ahmadi, M.M. Heravi, Res. Chem. Intermed. 45, 4349 (2019)
X. Huang, X. Hong, H. Lin, X. Cao, Q. Dang, S. Tang, D. Chen, Y. Zhang, Chem. Eng. J. 435, 134990 (2022)
H. Ya, L. Zhan, S. Gaa, H. Wana, Z. Ha, Y. Xu, K. Huang, J. Catal. 396, 342 (2021)
T. Mandal, M. Mondal, J. Choudhury, Organometallics 40, 2443 (2021)
K. Wang, W. Cui, Z. Bian, Y. Liu, S. Jiang, Y. Zhou, J. Wang, Appl. Catal. B Environ. 281, 119425 (2021)
Y. Zhang, L. Zhang, X. Zhang, D. Yang, C. Du, L. Wan, C. Au, J. Chen, M. Xie, New J. Chem. 44, 15202 (2020)
Y. Gu, S.U. Son, T. Li, B. Tan, Adv. Funct. Mater. 31, 2170082 (2021)
G. Xiong, S. Gao, Q. Zhang, B. Ren, L. You, F. Ding, Y. He, Y. Sun, Polymer 247, 124787 (2022)
J. Zhang, N. Liu, H. Gong, Q. Chen, H. Liu, Microp. Mesop. Mat. 336, 111836 (2022)
H.R. Penchah, P. Najafi, A. Ghaemi, H.G. Gilani, Environ. Progr. Sustain. 40, e1586 (2021)
R.A. Molla, P. Bhanja, K. Ghosh, S.S. Islam, S.M. Islam, A. Bhaumik, ChemCatChem 9, 1939 (2017)
W. Jiang, W. Sun, Y. Zhou, Y. Li, Res. Chem. Intermed. 45, 5535 (2019)
A. Blocher, F. Mayer, P. Schweng, T.M. Tikovits, N. Yousefi, R.T. Woodward, Mater. Adv. 3, 6335 (2022)
J. Gu, J. Zhang, D. Li, H. Yuan, Y. Chen, J. Chem. Technol. Biot. 94, 3073 (2019)
S.E. Lyubimov, A.A. Zvinchuk, A.A. Korlyukov, V.A. Davankov, O.P. Parenago, Petrol. Chem. 61, 76 (2021)
S.M. Nobre, A.L. Monteiro, J. Mol. Catal. A: Chem. 313, 65 (2009)
Z. Jia, K. Wang, B. Tan, Y. Gu, Adv. Synth. Catal. 359, 78 (2017)
A. Fihri, D. Luart, C. Len, A. Solhy, C. Chevrin, Y. Polshettiwar, Dalton Trans. 40, 3116 (2011)
H. Lin, H. Yao, X. Gao, L. Zhang, Q. Luo, Y. Ouyang, B. Xiang, S. Liu, D. Xiang, Chem. Lett. 50, 1879 (2021)
H. Lin, X. Gao, H. Yao, Q. Luo, B. Xiang, C. Liu, Y. Ouyang, N. Zhou, L. Zhang, D. Xian, Catal Sci. Technol. 11, 3676 (2021)
W. Xu, C. Liu, D. Xiang, Q. Luo, Y. Shu, H. Lin, Y. Hu, Z. Zhang, Y. Ouyang, RSC Adv. 9, 34395 (2019)
C. Liu, L. Zheng, D. Xiang, S. Liu, W. Xu, Q. Luo, Y. Shu, Y. Ouyang, H. Lin, RSC Adv. 10, 17123 (2020)
B. Li, Z. Guan, W. Wang, X. Yang, J. Hu, B. Tan, T. Li, Adv. Mater. 24, 3390 (2012)
Y. Zhi, K. Li, H. Xia, M. Xue, Y. Mu, X. Liu, J. Mater. Chem. A 5, 8697 (2017)
N.A. Nemygina, L.Z. Nikoshvili, M.G. Sulman, V.G. Matveeva, E.M. Sulman, Chem. Eng. Trans. 52, 691 (2015)
F. Mohajer, G.M. Ziarani, A. Badiei, Res. Chem. Intermed. 49, 1273 (2023)
Q. Fu, Y. Meng, Z. Fang, Q. Hu, L. Xu, W. Gao, X. Huang, Q. Xue, Y. Sun, F. Lu, A.C.S. Appl, Mater. Inter. 9, 2469 (2017)
P.S. Pharande, G.S. Rashinkar, D.M. Pore, Res. Chem. Intermed. 47, 4457 (2021)
V. Faria, D. Oliveira, M. Kurz, F. Gonçalves, C. Scheeren, G. Rosa, RSC Adv. 4, 13446 (2014)
G. Alvarenga, C. Ruas, J. Vicenti, F. Duarte, M. Gelesky, G. Rosa, J. Brazil. Chem. Soc. 27, 787 (2016)
R. Begum, Z. Farooqi, J. Xiao, E. Ahmed, A. Sharif, A. Irfan, J. Mol. Liq. 338, 116780 (2021)
Y. Dong, Z.Z. Wang, X. Li, T. Li, Y. Ren, W. Hu, L. Zhang, X. Zhang, C. Wei, J. Mol. Liq. 368, 120679 (2022)
M. Lin, S. Wang, J. Zhang, W. Luo, H. Liu, W. Wang, C. Su, J. Mol. Catal. A: Chem. 394, 33 (2014)
S. Xu, K. Song, T. Li, B. Tan, J. Mater. Chem. A 3, 1272 (2015)
K.S.W. Sing, D.H. Everett, R.A.W. Haul, L. Moscou, R.A. Pierotti, J. Rouquerol, T. Siemieniewska, Pure Appl. Chem. 57, 603 (1985)
N. Koukabi, M. Arghan, Res. Chem. Intermed. 48, 4553 (2022)
W. Lin, Y. Song, L. Wang, N. Li, Y. Fu, D. Chen, W. Zhu, F. Zhang, Catal. Lett. 153, 2368 (2023)
M. Ghabdian, M.A. Nasseri, A. Allahresani, A. Motavallizadehkakhky, Res. Chem. Intermed. 47, 1713 (2021)
D. Meng, J. Bi, Y. Dong, B. Hao, K. Qin, T. Li, D. Zhu, Chem. Commun. 56, 2889 (2020)
D. Rosa, B. Vargas, M. Silveira, C. Rosa, M. Martins, G. Rosa, Waste Biomass Valori. 10, 2285 (2018)
B. Wu, P. Lyu, K. Wang, X. Qiu, T. Liu, F. Zhang, H. Li, S. Xiao, Res. Chem. Intermed. 44, 6327 (2018)
S. Rana, S. Maddila, K. Yalagala, S.B. Jonnalagadda, Appl. Catal. A: Gen. 505, 539 (2015)
S. Sun, D. Pan, H. Huang, Z. Wang, Y. Xu, Y. Zhao, Res. Chem. Intermed. 48, 3129 (2022)
D. Xu, F. Wang, G. Yu, H. Zhao, J. Yang, M. Yuan, X. Zhang, Z. Dong, ChemCatChem 10, 4569 (2018)
H. Sato, T. Mameda, K. Nakai, T. Misaki, Y. Haruyama, S. Sonobe, T. Kubota, Y. Okamoto, T. Sugimura, Res. Chem. Intermed. 42, 31 (2016)
A. Modak, J. Su, W. Qiu, X. Liu, Catalysts 6, 161 (2016)
G. Chen, K. You, F. Zhao, Z. Chen, H. Luo, Res. Chem. Intermed. 48, 2593 (2022)
C. Xu, M. Hu, Q. Wang, Gu. Fan, Y. Wang, Y. Zhang, D. Gao, J. Bi, Dalton Trans. 47, 2561 (2018)
Z. Jia, K. Wang, T. Li, B. Tan, Y. Gu, Catal. Sci. Technol. 6, 4345 (2016)
F. Zhang, J. Jin, X. Zhong, S. Li, J. Niu, R. Li, J. Ma, Green Chem. 13, 1238 (2011)
V. Sadhasivam, M. Mathappan, M. Harikrishnan, C. Chithiraikumar, S. Murugesan, A. Siva, Res. Chem. Intermed. 44, 2853 (2018)
Y. Cao, Y. Wang, Y. Fu, F. Zhou, J. Huang, Sep. Purif. Technol. 322, 124272 (2023)
J. Zhang, K. Dong, W. Luo, H. Guan, Fuel 234, 664 (2018)
A. Balanta, C. Godard, C. Claver, Chem. Soc. Rev. 40, 4973 (2011)
V. Polshettiwar, A. Decottignies, C. Len, A. Fihri, Chemsuschem 3, 502 (2010)
S. Kotha, K. Lahiri, D. Kashinath, Tetrahedron 58, 9633 (2002)
Acknowledgements
The authors are grateful for the financial support of the Natural Science Foundation of Hunan Province (2021JJ30539, 2023JJ50450) and Innovative Training Project for College Students of China (S202310548066).
Funding
Funding was provided by Natural Science Foundation of Hunan Province (2021JJ30539, 2023JJ50450) and Innovative Training Project for College Students of China (S202310548066).
Author information
Authors and Affiliations
Contributions
LF contributed to conceptualization, investigation, data curation and writing—original draft. SL was involved in writing and editing and data curation. HZ contributed to conceptualization, investigation and data curation. GC was involved in conceptualization, methodology, formal analysis, investigation, writing—original draft, and writing—review and editing. DX contributed to conceptualization, supervision and project administration.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Feng, L., Lu, S., Zou, H. et al. Palladium catalyst immobilized on functionalized hyper–cross-linked polymers with 8-hydroxyquinoline as monomer for Suzuki–Miyaura coupling reactions. Res Chem Intermed 50, 2051–2066 (2024). https://doi.org/10.1007/s11164-024-05270-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-024-05270-0