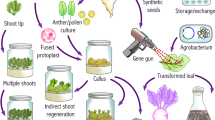
Abstract
Synthetic seeds are artificially encapsulated propagules that mimic true seeds in agriculture. Although a variety of plant materials, such as shoot tips, axillary buds, callus, micro cuttings, and protocorm-like bodies, are used in the production of synthetic seeds, somatic embryos are the most widely used explants in the production of these seeds. Synthetic seeds compete with traditional approaches to preserve the germplasm of threatened plant species. The resulting progenies are the true clones of the main plant, thus preserving the intactness of the genetic background. Due to poor germination and low seed amount, wild Beta species are exposed to the risk of extinction. Wild relatives of Beta have agronomically important properties such as resistance to diseases and abiotic stresses. Numerous attempts have been made to give these traits to sugar beet crop through conventional breeding methods. Despite the importance of synthetic seed for wild beets, it has not yet been investigated. The production of synthetic seeds ensures the conservation and availability of wild germplasm of the genus Beta for cytogenetic and breeding studies.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Abe J, Tsuda C (1987) Genetic analysis for isozyme variation in the section Vulgares, genus Beta. Jpn J Breed 37(3):253–261
Ahmad N, Anis M (2010) Direct plant regeneration from encapsulated nodal segments of Vitex negundo. Biol Plant 54:748–752
Ahmad N, Faisal M, Fatima N et al (2012) Encapsulation of microcuttings for propagation and short-term preservation in Ruta graveolens L.: a plant with high medicinal value. Acta Physiol Plant 34:2303–2310
Alatar A, Faisal M (2012) Encapsulation of Rauvolfia tetraphylla microshoots as artificial seeds and evaluation of genetic fidelity using RAPD and ISSR markers. J Med Plants Res 6:1367–1374
Alcaraz G, Geuter T, Laillet G et al (1998) Sugar beet pollen biology. In: Proceedings of 61th IIRB congress, Brussels, pp 393–399
Alibert B, Sellier H, Souvré A (2003) Flux de gènes entre la betteravesucrière GM et lesbetteravesrudéralesdansles conditions de la production de semencesau champ. In: Proceedings of 66th IIRB congress, Brussels
Altınok HH (2012) The disease prevalence and severity of Cercospora leaf spot in sugar beet cultivations in Kayseri. Derim 29(2):33–45
Ara H, Jaiswal U, Jaiswal VS (2000) Synthetic seed: prospects and limitations. Curr Sci 78:1438–1444
Attree SM, Pomeroy MK, Fowke LC (1994) Production of vigorous, desiccation tolerant white spruce (Picea-glauca (moench) voss) synthetic seeds in a bioreactor. Plant Cell Rep 13:601–606
Bapat VA, Rao PS (1990) In vivo growth of encapsulated axillary buds of mulberry (Morus-indica L.). Plant Cell Tissue Organ Cult 20:69–70
Bapat VA, Mhatre M, Rao PS (1987) Propagation of Morus indica L. (Mulberry) by encapsulated shoot buds. Plant Cell Rep 1987(6):393–395
Bewley JD, Black M (1985) Seeds: physiology of development and germination. Plenum, New York, p 367
Biancardi E, De Biaggi M (1979) Beta maritima L. in the Po Delta. In: Atti Convegno Tecnico Internazionale sulla Bieticoltura in Commemorazione di Ottavio Munerati. ISCI, Rovigo, pp 183–185
Biancardi E, McGrath JM, Panella LW et al (2010) Sugar beet. In: Bradshaw JE (ed) Root and tuber crop. Springer, New York, pp 173–219
Biancardi E, Panella LW et al (2012) Beta maritima: the origin of beets. Springer, New York
Boughey CL (1981) Evolutionary and taxonomic studies on wild and cultivated beets. PhD Thesis, University of Birmingham
Capuano G, Piccioni E, Standardi A (1998) Effect of different treatments on the conversion of M.26 apple rootstock synthetic seeds obtained from encapsulated apical and axillary micropropagated buds. J Hortic Sci Biotechnol 73:299–305
Cartes P, Castellanos H, Ríos D et al (2009) Encapsulated somatic embryos and zygotic embryos for obtaining artificial seeds of rauli-beech (Nothofagus alpina (Poepp. and Endl.) oerst.). Chil J Agric Res 69:112–118
Castellanos H, Sánchez-Olate M, Ríos YD (2004) Embriogénesis somática recurrente en raulí (Nothofagus alpine (Poepp. et Endl.) Oerst). Segundo Congreso Chileno de Ciencias Forestales, Valdivia, Chile. 10–12 de noviembre. Universidad Austral de Chile, Valdivia, p 36
Chand S, Singh AK (2004) Plant regeneration from encapsulated nodal segments of Dalbergia sissoo Roxb, a timber-yielding leguminous tree species. J Plant Physiol 161:237–243
Coons GH (1954) The wild species of Beta. Proc Am Soc Sugar Beet Technol 8:142–147
Coons GH (1975) Interspecific hybrids between Beta vulgaris L. and the wild species of Beta. J Am Soc Sugar Beet Technol 18:281–306
Cuguen J, Arnaud JF, Delescluse M et al (2005) Gene flow within the Beta species complex: genetic diversity of weed and wild sea beet populations within the French sugar beet production area. Adv Sugar Beet Res 6:103–115
Daud M, Taha MZ, Hasbullah AZ (2008) Artificial seed production from encapsulated micro shoots of Saintpaulia ionantha Wendl. (African Violet). J Appl Sci 8:4662–4667
De Bock TSM (1986) The genus Beta: domestication, taxonomy and interspecific hybridization for plant breeding. Acta Hortic 182:335–343
Doney DL (1993) Broadening the genetic base of sugarbeet. J Sugar Beet Res 30:209–220
Doney DL, Whitney ED (1990) Genetic enhancement in Beta for disease resistance using wild relatives: a strong case for the value of genetic conservation. Econ Bot 44:445–451
Drew RLK (1979) A cheap, simple apparatus for growing large batches of plant tissue in submerged liquid culture plant. Sci Lett 17:227–236
Elliott MC, Weston GD (1993) Biology and physiology of the sugar beet plant. In: Cooke DA, Scott RK (eds) The sugar beet crop: science into practice. Chapman and Hall, London, pp 37–66
Engelmann F, Takagi H (2000) Cryopreservation of tropical plant germplasm: current research progress and application. In: Proceedings of an international workshop. International Plant Genetic Resources Institute (IPGRI), Tsukuba, Japan
Ergül A, Khabbazi SD, Oğuz MÇ et al (2018) In vitro multiplication of wild relatives in genus Beta conserves the invaluable threatened germplasms. Plant Cell Tissue Organ Cult:1–7
Faisal M, Anis M (2007) Regeneration of plants from alginate-encapsulated shoots of Tylophora indica (Burm. F.) Merrill., an endangered medicinal plant. J Hortic Sci Biotechnol 82:351–354
Ford-Lloyd BV (2005) Sources of genetic variation, genus Beta. In: Biancardi E, Campbell LG, Skaracis GN, De Biaggi M (eds) Genetics and breeding of sugar beet. Science, Enfield, pp 25–33
Frese L (2010) Conservation and access to sugarbeet germplasm. Sugar Technol 12(3–4):207–219
Frese L, Desprez B, Ziegler D (2001) Potential of genetic resources and breeding strategies for base-broadening in Beta. Broadening the genetic base of crop production. IPGRI/FAO, Rome, pp 295–309
Ganapathi TR, Suprasanna P, Bapat VA et al (1992) Propagation of banana through encapsulated shoot tips. Plant Cell Rep 11:571–575
Gantait S, Kundu S, Ali N et al (2015) Synthetic seed production of medicinal plants: a review on influence of explants, encapsulation agent and matrix. Acta Physiol Plant 37(5):98
Gidner S, Lennefors BL, Nilsson NO et al (2005) QTL mapping of BNYVV resistance from the WB41 source in sugar beet. Genome 48:279–285
Grimmer MK, Kraft T, Francis SA et al (2008a) QTL mapping of BNYVV resistance from the WB258 source in sugar beet. Plant Breed 127(6):650–652
Grimmer MK, Bean KMR, Qi A et al (2008b) The action of three Beet yellows virus resistance QTLs depends on alleles at a novel genetic locus that controls symptom development. Plant Breed 127(4):391–397
Heijbroek W, McFarlane JS, Doney DL (1977) Breeding for tolerance to beet-cyst eelworm Heterodera schachtii Schm. in sugarbeet. Euphytica 26(3):557–564
Heijbroek W, Roelands AJ, De Jong JH et al (1988) Sugar beets homozygous for resistance to beet cyst nematode (Heterodera schachtii Schm.), developed from monosomic additions of Beta procumbens to B. vulgaris. Euphytica 38:121–131
Hung CD, Trueman SJ (2012a) Alginate encapsulation of shoot tips and nodal segments for short-term storage and distribution of the eucalypt Corymbia torelliana x C. citriodora. Acta Physiol Plant 34:117–128
Hung CD, Trueman SJ (2012b) Preservation of encapsulated shoot tips and nodes of the tropical hardwoods Corymbia torelliana × C. citriodora and Khaya senegalensis. Plant Cell Tissue Organ Cult 109:341–352
Ikhlaq M, Hafiz IA, Micheli M et al (2010) In vitro storage of synthetic seeds: effect of different storage conditions and intervals on their conversion ability. Afr J Biotechnol 9:5712–5721
Janick J, Kim YH, Kitto S et al (1993) Desiccated synthetic seed. In: Redenbaugh K (ed) Synseeds. CRC, Boca Raton, FL, pp 11–33
Jassem B (1992) Species relationship in the genus Beta as revealed by crossing experiments. In: International beta genetic resources network. In: Report on the 2nd international beta genetic resources workshop held at the Institute for Crop Science and Plant Breeding, Braunschweig, Germany, 24–28 June 1991. International Crop Network Series No, vol 7, pp 55–61
Jung C, Wricke G (1987) Selection of diploid nematode-resistant sugar beet from monosomic addition lines. Plant Breed 98(3):205–214
Jung SJ, Yoon ES, Jeong JH et al (2004) Enhanced post-germinative growth of encapsulated somatic embryos of Siberian ginseng by carbohydrate addition to the encapsulation matrix. Plant Cell Rep 23:365–370
Kadereit G, Hohmann S, Kadereit JW (2006) A synopsis of Chenopodiaceae subfam. Betoideae and notes on the taxonomy of Beta. Willdenowia 36:9–19
Kavyashree R, Gayatri MC, Revanasiddaiah HM (2006) Propagation of mulberry variety-S54 by synseeds of axillary bud. Plant Cell Tissue Organ Cult 84:245–249
Keskin B (1964) Polymyxa betae n. sp., einParasit in den Wurzeln von Beta vulgaris Tournefort, besonders während der Jugendentwicklung der Zuckerrübe. Arch Microbiol 49(4):348–374
Kitto SL, Janick J (1982) Polyox as an artificial seed coat for a sexual embryo. HortScience 17:448
Kitto SL, Janick J (1985) Hardening increases survival of synthetically-coated asexual embryos of carrot. J Am Soc Hortic Sci 110:283–286
Lambardi M, Benelli C, Ozudogru EA (2006) Synthetic seed technology in ornamental plants. In: Teixeira da Silva JA (ed) Floriculture, ornamental and plant biotechnology, vol 2. Global Science Books, London, pp 347–354
Lange W, De Bock TS, Van Geyt JPC et al (1988) Monosomic additions in beet (Beta vulgaris) carrying extra chromosomes of B. procumbens. Theor Appl Genet 76(5):656–664
Lange W, Muller J, De Bock TS (1993) Virulence in the beet cyst nematode (Heterodera schachtii) versus some alien genes for resistance in beet. Fundam Appl Nematol 16:447–454
Larkin PJ, Davies PA, Tanner GJ (1998) Nurse culture of low number of Medicago and Nicotiana protoplasts using calcium alginate beads. Plant Sci 58:203–210
Lata H, Chandra S, Khan IA et al (2009) Propagation through alginate encapsulation of axillary buds of Cannabis sativa L. – an important medicinal plant. Physiol Mol Biol Plants 15:79–86
Leroy XJ, Leon K, Charles G et al (2000) Cauliflower somatic embryogenesis and analysis of regenerant stability by ISSRs. Plant Cell Rep 19:1102–1107
Letschert JPW (1993) Beta section Beta: biogeographical patterns of variation and taxonomy. Thesis. Wageningen Agricultural University Papers 93–1, p 155
Lewellen RT (1992) Use of plant introductions to improve populations and hybrids of sugarbeet. In: Use of plant introductions in Cultivar Development, Part 2. Crop Science Society of America, Madison, WI, pp 117–135
Lewellen RT (2000) Registration of powdery mildew resistant sugarbeet germplasms CP01 and CP02. Crop Sci 40:1515
Lewellen RT (2004) Registration of CP07 and CP08 sugarbeet germplasms with resistance to powdery mildew, rhizomania, and other diseases. Crop Sci 44(6):2276
Lewellen RT (2006) Registration of CN12 and CN72 sugarbeet germplasm populations with resistance to cyst nematode. Crop Sci 46(3):1414
Lewellen RT, Skoyen IO (1991) Improvement and performance of populations of sugarbeet × Beta maritima. J Sugar Beet Res 28:79
Lewellen RT, Whitney ED (1993) Registration of germplasm lines developed from composite crosses of sugarbeet× Beta maritima. Crop Sci 33(4):882–883
Lulsdorf MM, Tautorus TE, Kikcio SI et al (1993) Germination of encapsulated embryos of interior spruce (Picea-glauca-engelmannii complex) and black spruce (Picea-mariana Mill). Plant Cell Rep 12:385–389
Luterbacher M, Asher M, Deambrogio E et al (2004) Sources of resistance to diseases of sugar beet in related Beta germplasm: I. Foliar diseases. Euphytica 139:105–121
Magray MM, Wani KP, Chatto MA et al (2017) Synthetic seed technology. Int J Cur Microbiol Appl Sci 6(11):662–674
Marlander B, Lange T, Wulkow A (2011) Dispersal principles of sugar beet from seed to sugar with particular relation to genetically modified varieties. J Kult (J Cul Plants) 63:349–373
McCarter JP (2008) Molecular approaches toward resistance to plant-parasitic nematodes. In: Plant cell monographs. Springer, Berlin
McFarlane JS (1984) Final report: breeding for resistance to sugarbeet yellow wilt. Cooperative Agreement No. 58-9AHZ-0-520 between USDA-ARS, Washington, DC and Beet Sugar Development Foundation, Denver, CO
McGrann GR, Grimmer MK, Mutasa-Göttgens ES et al (2009) Progress towards the understanding and control of sugar beet rhizomania disease. Mol Plant Pathol 10(1):129–141
McGrath JM, Saccomani M, Stevanato P et al (2007) Beet. In: Kole C (ed) Genome mapping and molecular breeding in plants, vol 5. Springer, Berlin, pp 191–207
Mesbah M, Scholten OE, De Bock TS et al (1997) Chromosome localisation of genes for resistance to Heterodera schachtii, Cercospora beticola and Polymyxa betae using sets of Beta procumbens and B. patellaris derived monosomic additions in B. vulgaris. Euphytica 97(1):117–127
Micheli M, Pellegrino S, Piccioni E et al (2002) Effects of double encapsulation and coating on synthetic seed conversion in M.26 apple rootstock. J Microencapsul 19:347–356
Mishra J, Singh M, Palni LMS et al (2011) Assessment of genetic fidelity of encapsulated microshoots of Picrorhiza kurroa. Plant Cell Tissue Organ Cult 104:181–186
Munerati O (1932) Sull’incrocio della barbabietola coltivata con la beta selvaggia della costa adriatica. L’Industria Saccarifera Italiana 25:303–304
Munerati O, Mezzadroli G, Zapparoli TV (1913) Osservazioni sulla Beta maritima L. nel triennio 1910–1912. Staz Sper Agric Ital 46:415–445
Murashige T (1977) Plant cell and organ culture as horticultural practice. Acta Hortic 78:17–30
Naik SK, Chand PK (2006) Nutrient-alginate encapsulation of in vitro nodal segments of pomegranate (Punica granatum L.) for germplasm distribution and exchange. Sci Hortic 108:247–252
Nyende AB, Schittenhelm S, Mix-Wagner G et al (2005) Yield and canopy development of field grown potato plants derived from synthetic seeds. Eur J Agron 22:175–184
Owen FV (1942) Inheritance of cross- and self-sterility and self-fertility in Beta vulgaris. J Agric Res 64:679–698
Ozudogru EA, Kaya E, Kirdok E et al (2011) In vitro propagation from young and mature explants of thyme (Thymus vulgaris and T. longicaulis) resulting in genetically stable shoots. In Vitro Cell Dev Biol Plant 47(2):309–320
Panella L, Frese L (2000) Cercospora resistance in Beta species and the development of resistant sugarbeet lines. In: Asher MJC, Holtschulte B, Molard MR, Rosso F, Steinrücken G, Beckers R (eds) Cercospora beticola Sacc. biology, agronomic influence and control measures in sugar beet, Belgium, pp 163–176
Panella L, Lewellen RT (2007) Broadening the genetic base of sugarbeet: introgression from wild relatives. Euphytica 154:383–400
Pelsy F, Merdinoglu D (1996) Identification and mapping of random amplified polymorphic DNA markers linked to a rhizomania resistance gene in sugar beet (Beta vulgaris L.) by bulked segregant analysis. Plant Breed 115(5):371–377
Pinker I, Abdel-Rahman SSA (2005) Artificial seed for propagation of Dendranthema × grandiflora (Ramat.). Prop Orn Plants 5:186–191
Pintos B, Bueno MA, Cuenca B et al (2008) Synthetic seed production from encapsulated somatic embryos of cork oak (Quercus suber L.) and automated growth monitoring. Plant Cell Tissue Organ Cult 95:217–225
Pond S, Cameron S (2003) Tissue culture artificial seed. In: Thomas B (ed) Encyclopedia of applied plant sciences. Elsevier, Oxford, pp 1379–1388
Pourjavadi A, Barzegar SH, Mahdavinia GR (2006) MBA-cross linked Na-Alg/CMC as a smart full-polysaccharide superabsorbent hydrogels. Carbohydr Polym 66:386–395
Ravi D, Anand P (2012) Production and applications of artificial seeds: a review. Int Res J Biol Sci 1(5):74–78
Reddy MC, Murthy KSR, Pullaiah T (2012) Synthetic seeds: a review in agriculture and forestry. Afr J Biotechnol 11:14254–14275
Reinert J (1958) Morphogenese und ihre Kontrolle an Gewebekulturenaus Carotten. Naturwissenschaften 45(14):344–345
Ren H, Zhang Q, Lu H et al (2012) Wild plant species with extremely small populations require conservation and reintroduction in China. Ambio 41(8):913–917
Richardson KL (2018) Registration of sugar beet germplasm lines CN921–515 and CN921–516 with sugar beet cyst nematode resistance from Beta vulgaris subsp. maritima. J Plant Regist 12(2):264–269
Rihan HZ, Al-Issawi M, Al-Swedi F et al (2012) The effect of using PPM (plant preservative mixture) on the development of cauliflower microshoots and the quality of artificial seed produced. Sci Hortic 141:47–52
Rihan H, Kareem F, El-Mahrouk M et al (2017) Artificial seeds (principle, aspects and applications). Agronomy 7(4):71
Rizkalla AA, Badr-Elden AM, Ottai MES et al (2012) Development of artificial seed technology and preservation in sugar beet. Sugar Tech 14(3):312–320
Rossi V, Racca P, Giosue S (1995) Geophytopathological analysis of Cercospora leaf spot on sugarbeet in the Mediterranean area. Phytopathol Med:69–82
Roy B, Mandal AB (2008) Development of synthesis seed involving androgenic and pro-embryos in elite indica rice. Indian J Biotechnol 7:515–519
Saiprasad G (2001) Artificial seeds and their applications. Resonance 6:39–47
Saiprasad GVS, Polisetty R (2003) Propagation of three orchid genera using encapsulated protocorm-like bodies. In Vitro Cell Dev Biol Plant 39:42–48
Sakhanokho HF, Pounders CT, Blythe EK (2013) Alginate encapsulation of Begonia microshoots for short-term storage and distribution. Sci World J 13:1–7
Salentijn EMJ, Sandal NN, Lange W et al (1992) Isolation of DNA markers linked to a beet cyst nematode resistance locus in Beta patellaris and Beta procumbens. Mol Gen Genet MGG 235(2–3):432–440
Sandal NN, Salentijn EMJ, Kleine M et al (1997) Backcrossing of nematode-resistant sugar beet: a second nematode resistance gene at the locus containing Hs1pro–i. Mol Breed 3:471–480
Sarkar D, Naik PS (1998) Synseeds in potato: an investigation using nutrient-encapsulated in vitro nodal cutting segments. Sci Hortic 73:179–184
Saunders JW, Tsai CJ (1999) Production of somatic embryos and shoots from sugarbeet callus: Effects of abscisic acid, other growth regulators, nitrogen source, sucrose concentration and genotype. In Vitro Cell Dev Biol Plant 35(1):18–24
Savitsky H (1960) Viable diploid, triploid, and tetraploid hybrids between Beta vulgaris and species of the section Patellares. J Am Soc Sugar Beet Technol 11:215–235
Savitsky H (1975) Hybridization between Beta vulgaris and B. procumbens and transmission of nematode (Heterodera schachtii) resistance to sugar beet. Can J Genet Cytol 17:197–209
Scholten OE, De Bock TS, Klein-Lankhorst RM et al (1999) Inheritance of resistance to beet necrotic yellow vein virus in Beta vulgaris conferred by a second gene for resistance. Theor Appl Genet 99(3–4):740–746
Senaratna T, McKersie BD, Bowley SR (1990) Artificial seeds of alfalfa (Medicago sativa L.). Induction of desiccation tolerance in somatic embryos. In Vitro Cell Dev Biol Plant 16:85–90
Sharma S, Shahzad A, Jan N et al (2009a) In vitro studies on shoot regeneration through various explants and alginate-encapsulated nodal segments of Spilanthes mauritiana DC., an endangered medicinal herb. Int J Plant Dev Biol 3:56–61
Sharma S, Shahzad A, Sahai A (2009b) Artificial seeds for propagation and preservation of Spilanthes acmella (L.) Murr., a threatened pesticidal plant species. Int J Plant Dev Biol 3:62–64
Sharma S, Shahzad A, da Silva JAT (2013) Synseed technology—a complete synthesis. Biotechnol Adv 31(2):186–207
Singh AK, Chand S (2010) Plant regeneration from alginate encapsulated somatic embryos of Dalbergia sissoo Roxb. Indian J Biotechnol 9:319–324
Singh B, Sharma S, Rani G et al (2007) In vitro response of encapsulated and non-encapsulated somatic embryos of Kinnow mandarin (Citrus nobilis Lour × C. deliciosa Tenora). Plant Biotechnol Rep 1:101–107
Skaracis GN (2005) Molecular biology and biotechnology, genetic engineering. In: Biancardi E, Campbell LG, Skaracis GN, De Biaggi M (eds) Genetics and breeding of sugar beet. Science, Enfield, NH, pp 255–268
Skaracis GN, Biancardi E (2000) Breeding for Cercospora resistance in sugar beet. In: Asher MJC, Holtschulte B, Molard MR, Rosso F, Steinrücken G, Beckers R. (eds) Cercospora beticola Sacc. biology, agronomic influence and control measures in sugar beet. International Institute for Beet Research (IIRB), Brussels. Adv Sugar Beet Res Ser 2:177–195
Srivastava V, Khan SA, Banerjee S (2009) An evaluation of genetic fidelity of encapsulated microshoots of the medicinal plant: Cineraria maritima following six months of storage. Plant Cell Tissue Organ Cult 99:193–198
Steward FC, Mapes MO, Smith J (1958) Growth and organized development of cultured cells I. Growth and division of freely suspended cells. Am J Bot 45(9):693–703
Sundararaj SG, Agrawal A, Tyagi RK (2010) Encapsulation for in vitro short-term storage and exchange of ginger (Zingiber officinale Rosc.) germplasm. Sci Hortic 125:761–766
Tabassum B, Nasil IA, Farooq AM et al (2010) Viability assessment of in vitro produced synthetic seeds of cucumber. Afr J Biotechnol 9:7026–7032
Tamada T, Baba T (1973) Beet necrotic yellow vein virus from rhizomania affected sugar beet in Japan. Ann Phytopathol Soc Jpn 39:325–332
Van Geyt JPC, Oleo M, Lange W et al (1988) Monosomic additions in beet (Beta vulgaris) carrying extra chromosomes of Beta procumbens. Theor Appl Genet 76(4):577–586
Van Geyt JPC, Lange W, Oleo M et al (1990) Natural variation within the genus Beta and its possible use for breeding sugar beet: a review. Euphytica 49(1):57–76
Varshney A, Anis M (2014) Synseed conception for short-term storage, germplasm exchange and potentialities of regeneration genetically stable plantlets of desert date tree (Balanites aegyptiaca Del.). Agrofor Syst 88:321–329
Wang WG, Wang SH, Wu XA et al (2007) High frequency plantlet regeneration from callus and artificial seed production of rock plant Pogonatherum paniceum (Lam.) Hack. (Poaceae). Sci Hortic 113:196–201
Weiland J, Koch G (2004) Sugarbeet leaf spot disease (Cercospora beticola Sacc.). Mol Plant Pathol 5(3):157–166
Westcott RJ (1981) Tissue culture storage of potato germplasm. Use of growth retardants 2. Potato Res 24:343–352
Winkelmann T, Meyer L, Serek M (2004) Germination of encapsulated somatic embryos of Cyclamen persicum. Hortic Sci 39:1093–1097
Yu MH (1983) Sugar beet germplasm resistant to sugar beet nematode. Crop Sci 23:1021–1022
Yu MH (1989) Callus induction and differentiation from leaf explants of different species of the genus Beta. Crop Sci 29:205–209
Yu MH (1997) Registration of Mi-1 root-knot nematode resistant beet germplasm line. Crop Sci 37(1):295–295
Yu MH (2002) Registration of sugarbeet germplasm M1-3 resistant to root-knot nematode. Crop Sci 42(5):1756
Yu Y (2004) Genetics of Aphanomyces disease resistance in sugarbeet (Beta vulgaris), AFLP mapping and QTL analyses. PhD Dissertation. Michigan State University
Yu MH (2005) Cyst nematode. In: Biancardi E, Campbell LG, Skaracis GN, De Biaggi M (eds) Genetics and breeding of sugar beet. Science, Enfield, NH, pp 103–109
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Khabbazi, S.D., Yüksel Özmen, C., Ergül, A. (2019). Synthetic Seeds of Wild Beet: Basic Concepts and Related Methodologies. In: Faisal, M., Alatar, A. (eds) Synthetic Seeds . Springer, Cham. https://doi.org/10.1007/978-3-030-24631-0_18
Download citation
DOI: https://doi.org/10.1007/978-3-030-24631-0_18
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-24630-3
Online ISBN: 978-3-030-24631-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)