Abstract
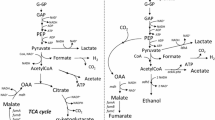
Two different approaches to activate the glyoxylate bypass in model Escherichia coli K-12 strains for succinate biosynthesis during dual-phase fermentation in minimal glucose media were examined. Inactivation of IclR and FadR, the transcriptional regulators of the aceBAK operon, were insufficient for the involvement of the glyoxylate bypass in anaerobic succinate biosynthesis by strains grown aerobically under glucose-abundant conditions. In contrast, the strains that constitutively expressed the aceEF-lpdA operon coding for the pyruvate dehydrogenase complex could partially synthesise succinate anaerobically via the glyoxylate bypass, even in the presence of intact regulators. The results suggest that the intensive acetyl-CoA formation in the strains constitutively expressing pyruvate dehydrogenase matches the physiological conditions that favour the activation of the glyoxylate bypass.
Similar content being viewed by others
References
Cheng KK, Zhao XB, Zeng J, Zhang JA (2012) Biotechnological production of succinic acid: current state and perspectives. Biofpr J 6:302–318
Choudhary MK, Yoon JM, Gonzalez R, Shanks JV (2011) Re-examination of metabolic fluxes in Escherichia coli during anaerobic fermentation of glucose using 13C labeling experiments and 2-dimensional nuclear magnetic resonance (NMR) spectroscopy. Biotech Bioprocess Eng 16:419–437
Cronan JE Jr, Laporte D (1996) Tricarboxylic acid cycle and glyoxylate bypass. In: Neidhardt F (ed) Escherichia coli and Salmonella: cellular and molecular biology, 2nd edn. ASM Press, Washington, pp 206–216
Datsenko KA, Wanner BL (2000) One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc Natl Acad Sci USA 97:6640–6645
Farmer WR, Liao JC (1997) Reduction of aerobic acetate production by Escherichia coli. Appl Environ Microbiol 63:3205–3210
Fischer E, Sauer U (2003) A novel metabolic cycle catalyzes glucose oxidation and anaplerosis in hungry Escherichia coli. J Biol Chem 278:46446–46451
Gui L, Sunnarborg A, Pan B, LaPorte DC (1996a) Autoregulation of iclR, the gene encoding the repressor of the glyoxylate bypass operon. J Bacteriol 178:321–324
Gui L, Sunnarborg A, LaPorte DC (1996b) Regulated expression of a repressor protein: FadR activates iclR. J Bacteriol 178:4704–4709
Gulevich AYu, Skorokhodova AYu, Ermishev VYu, Krylov AA, Minaeva NI, Polonskaya ZM, Zimenkov DV, Biryukova IV, Mashko SV (2009) A new method for the construction of translationally coupled operons in a bacterial chromosome. Mol Biol (Mosk) 43:505–514
Jiang M, Liu S, Ma J, Chen K, Yu L, Yue F, Wei P (2010) Effect of growth phase feeding strategies on succinate production by metabolically engineered E. coli. Appl Environ Microbiol 76:1298–1300
Katashkina JI, Skorokhodova AY, Zimenkov DV, Gulevich AY, Minaeva NI, Doroshenko VG, Biryukova IV, Mashko SV (2005) Tuning the expression level of a gene located on a bacterial chromosome. Mol Biol (Mosk) 39:719–726
Martinez I, Bennett GN, San KY (2010) Metabolic impact of the level of aeration during cell growth on anaerobic succinate production by an engineered Escherichia coli strain. Metab Eng 12:499–509
Phue JN, Shiloach J (2004) Transcription levels of key metabolic genes are the cause for different glucose utilization pathways in E. coli B (BL21) and E. coli K (JM109). J Biotechnol 109:21–30
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Press, Cold Spring Harbor
Skorokhodova AY, Gulevich AY, Morzhakova AA, Shakulov RS, Debabov VG (2011) Anaerobic synthesis of succinic acid by recombinant Escherichia coli strains with activated NAD+-reducing pyruvate dehydrogenase complex. Appl Biochem Microbiol 47:373–380
Thakker C, Martinez I, San KY, Bennett GN (2012) Succinate production in Escherichia coli. Biotechnol J 7:213–224
Vemuri GN, Eiteman MA, Altman E (2002a) Effects of growth mode and pyruvate carboxylase on succinic acid production by metabolically engineered strains of Escherichia coli. Appl Environ Microbiol 68:1715–1727
Vemuri GN, Eiteman MA, Altman E (2002b) Succinate production in dual-phase Escherichia coli fermentations depends on the time of transition from aerobic to anaerobic conditions. J Ind Microbiol Biotechnol 28:325–332
Waegeman H, Beauprez J, Moens H, Maertens J, De Mey M, Foulquié-Moreno MR, Heijnen JJ, Charlier D, Soetaert W (2011) Effect of iclR and arcA knockouts on biomass formation and metabolic fluxes in Escherichia coli K12 and its implications on understanding the metabolism of Escherichia coli BL21 (DE3). BMC Microbiol 11:70
Acknowledgments
This work was supported by a grant (16.512.11.2037) and, in part, by a grant (16.552.11.7029) from the Ministry of Education and Science of the Russian Federation.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Skorokhodova, A.Y., Gulevich, A.Y., Morzhakova, A.A. et al. Comparison of different approaches to activate the glyoxylate bypass in Escherichia coli K-12 for succinate biosynthesis during dual-phase fermentation in minimal glucose media. Biotechnol Lett 35, 577–583 (2013). https://doi.org/10.1007/s10529-012-1108-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-012-1108-z