Abstract
Purpose
Post-traumatic disc degeneration (DD) is currently investigated with models not fully matching the clinical condition, in particular post-traumatic loading of the disc is not considered. Therefore, the aim was to establish an in vitro burst fracture model that more closely mimics the in vivo situation by including post-traumatic physiological loading and to investigate DD under these conditions.
Methods
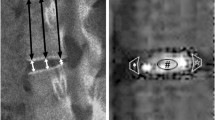
72 rabbit spinal segments (disc/endplates + 1/3 of adjacent vertebrae) were harvested from T8/9 to L5/6 and assigned to control (n = 36) or trauma groups (n = 36). Burst fractures were induced at day 0 in the trauma group using a dropped-weight device. From day 1 to 28, all specimens were cultured at 37 °C and were dynamically loaded daily (~1 MPa nominal pressure, 1 Hz, 2,500 cycles). At day 1, 7, 14, and 28, 9 specimens from each group were taken for analysis: histology (n = 2), total disc glycosaminoglycan (GAG) content (n = 3) normalized to DNA, and qPCR of DD marker genes (n = 4) in the nucleus pulposus and the annulus fibrosus.
Results
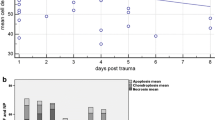
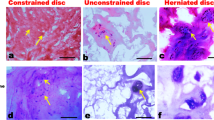
Burst fracture with post-traumatic physiological loading resulted in a 65 % loss of GAG/DNA by day 28. Histological sections confirmed the remodeling of the matrix. Catabolic (MMP-1/-3), pro-apoptotic (TNF-α, fas ligand), and pro-inflammatory (IL-1/-6, iNOS) gene transcription was substantially up-regulated in the nucleus after the trauma and did not normalize to control within 28 days. Similar results were found for the annulus on lower levels.
Conclusion
An in vitro burst fracture model with physiological post-traumatic loading was established. Under these conditions, burst spinal segments undergo strong and persistent degenerative changes.





Similar content being viewed by others
References
Luoma K, Riihimäki H, Luukkonen R et al (2000) Low back pain in relation to lumbar disc degeneration. Spine 25:487–492
Peng B, Chen J, Kuang Z et al (2009) Diagnosis and surgical treatment of back pain originating from endplate. Eur Spine J 18:1035–1040
Adams MA, Stefanakis M, Dolan P (2010) Healing of a painful intervertebral disc should not be confused with reversing disc degeneration: implications for physical therapies for discogenic back pain. Clin Biomech 25:961–971
Denis F (1983) The three column spine and its significance in the classification of acute thoracolumbar spinal injuries. Spine 8:817–831
Kerttula LI, Serlo WS, Tervonen OA et al (2000) Post-traumatic findings of the spine after earlier vertebral fracture in young patients. Spine 25:1104–1108
Heyde C-E, Tschoeke SK, Hellmuth M et al (2006) Trauma induces apoptosis in human thoracolumbar intervertebral discs. BMC Clin Pathol 6:5
Singh K, Masuda K, An HS (2005) Animal models for human disc degeneration. Spine J 5:267S–279S
Haschtmann D, Stoyanov JV, Gédet P, Ferguson SJ (2008) Vertebral endplate trauma induces disc cell apoptosis and promotes organ degeneration in vitro. Eur Spine J 17:289–299
Dudli S, Haschtmann D, Ferguson SJ (2012) Fracture of the vertebral endplates, but not equienergetic impact load, promotes disc degeneration in vitro. J Orthop Res 30:809–816
Dudli S, Ferguson SJ, Haschtmann D (2014) Severity and pattern of posttraumatic intervertebral disc degeneration depends on the type of injury. Spine J. doi:10.1016/j.spinee.2013.07.488
Chan SCW, Ferguson SJ, Gantenbein-Ritter B (2011) The effects of dynamic loading on the intervertebral disc. Eur Spine J 20:1796–1812
Reitmaier S, Schmidt H, Ihler R et al (2013) Preliminary investigations on intradiscal pressures during daily activities: an in vivo study using the merino sheep. PLoS One 8:e69610
Wilke HJ, Neef P, Caimi M, Hoogland T, Claes LE (1999) New in vivo measurements of pressures in the intervertebral disc in daily life. Spine 24:755–762
Sato K, Kikuchi S, Yonezawa T (1999) In vivo intradiscal pressure measurement in healthy individuals and in patients with ongoing back problems. Spine 24:2468–2474
MacLean JJ, Lee CR, Alini M, Iatridis JC (2005) The effects of short-term load duration on anabolic and catabolic gene expression in the rat tail intervertebral disc. J Orthop Res 23:1120–1127
Lotz JC, Colliou OK, Chin JR et al (1998) Compression-induced degeneration of the intervertebral disc: an in vivo mouse model and finite-element study. Spine 23:2493–2506
Enobakhare BO, Bader DL, Lee DA (1996) Quantification of sulfated glycosaminoglycans in chondrocyte/alginate cultures, by use of 1,9-dimethylmethylene blue. Anal Biochem 243:189–191
Haschtmann D, Stoyanov JV, Ettinger L et al (2006) Establishment of a novel intervertebral disc/endplate culture model: analysis of an ex vivo in vitro whole-organ rabbit culture system. Spine 31:2918–2925
Guevara I, Iwanejko J, Dembińska-Kieć A et al (1998) Determination of nitrite/nitrate in human biological material by the simple Griess reaction. Clin Chim Acta 274:177–188
Van Lint P, Libert C (2007) Chemokine and cytokine processing by matrix metalloproteinases and its effect on leukocyte migration and inflammation. J Leukoc Biol 82:1375–1381
Hoyland JA, Le Maitre CL, Freemont AJ (2008) Investigation of the role of IL-1 and TNF in matrix degradation in the intervertebral disc. Rheumatology 47:809–814
Haro H, Crawford HC, Fingleton B et al (2000) Matrix metalloproteinase-3-dependent generation of a macrophage chemoattractant in a model of herniated disc resorption. J Clin Invest 105:133–141
Wuertz K, Vo N, Kletsas D, Boos N (2012) Inflammatory and catabolic signalling in intervertebral discs: the roles of NF-κB and MAP kinases. Eur Cell Mater 23:103–119 (discussion 119–120)
Cui L-Y, Liu S-L, Ding Y et al (2007) IL-1beta sensitizes rat intervertebral disc cells to fas ligand mediated apoptosis in vitro. Acta Pharmacol Sin 28:1671–1676
Wang J, Tang T, Yang H et al (2007) The expression of fas ligand on normal and stabbed-disc cells in a rabbit model of intervertebral disc degeneration: a possible pathogenesis. J Neurosurg Spine 6:425–430
O’Connell J, Houston A, Bennett MW et al (2001) Immune privilege or inflammation? Insights into the fas ligand enigma. Nat Med 7:271–274
Burke JG, Watson RWG, Conhyea D et al (2003) Human nucleus pulposis can respond to a pro-inflammatory stimulus. Spine (Phila Pa 1976) 28:2685–2693
Zhao C-Q, Jiang L-S, Dai L-Y (2006) Programmed cell death in intervertebral disc degeneration. Apoptosis 11:2079–2088
Anderson DG, Izzo MW, Vaccaro AR et al (2002) Comparative gene expression profiling of normal and degenerative discs. Spine 27:1291–1296
Yang F, Leung VYL, Luk KDK et al (2009) Injury-induced sequential transformation of notochordal nucleus pulposus to chondrogenic and fibrocartilaginous phenotype in the mouse. J Pathol 218:113–121
Walsh AJL, Bradford DS, Lotz J (2004) In vivo growth factor treatment of degenerated intervertebral discs. Spine (Phila Pa 1976) 29:156–163
Saklatvala J (1986) Tumour necrosis factor alpha stimulates resorption and inhibits synthesis of proteoglycan in cartilage. Nature 322:547–549
Lowenstein CJ, Padalko E (2004) iNOS (NOS2) at a glance. J Cell Sci 117:2865–2867
Adams MA, Dolan P (2012) Intervertebral disc degeneration: evidence for two distinct phenotypes. J Anat 221:497–506
Ulrich JA, Liebenberg EC, Thuillier DU, Lotz J (2007) ISSLS prize winner: repeated disc injury causes persistent inflammation. Spine 32:2812–2819
Shamji MF, Setton LA, Jarvis W et al (2010) Proinflammatory cytokine expression profile in degenerated and herniated human intervertebral disc tissues. Arthr Rheum 62:1974–1982
Acknowledgments
We thank Jochen Walser and Peter Schwilch for their help with the design of the loading device. This study was funded by the Swiss National Science Foundation #310000-122105.
Conflict of interest
None of the authors has any potential conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dudli, S., Haschtmann, D. & Ferguson, S.J. Persistent degenerative changes in the intervertebral disc after burst fracture in an in vitro model mimicking physiological post-traumatic conditions. Eur Spine J 24, 1901–1908 (2015). https://doi.org/10.1007/s00586-014-3301-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-014-3301-3