Abstract
Background
Parkinson’s disease (PD) is a common neurodegenerative movement disorder, but the pathogenesis is still unclear. Long non-coding RNAs (lncRNAs) have been reported to play a prominent role in PD.
Objective
This study is designed to explore the role and mechanism of long intergenic non-coding RNA 00943 (LINC00943) in the N-methyl-4-phenylpyridine (MPP+)-inducted PD model.
Methods
LINC00943, microRNA-7-5p (miR-7-5p), and the chemokine (C-X-C motif) ligand 12 (CXCL12, also referred to as SDF-1) level were examined by real-time quantitative polymerase chain reaction (RT-qPCR). Cell viability and apoptosis were analyzed by 3-(4, 5-dimethyl-2-thiazolyl)-2, 5-diphenyl-2-H-tetrazolium bromide (MTT), and flow cytometry assays, severally. Protein levels of interleukin-1β (IL-1β), tumor necrosis factor-α (TNF-α), and CXCL12 were assessed by western blot assay. The ROS generation and SOD activity were detected by the corresponding kits. The binding relationship between miR-7-5p and LINC00943 or CXCL12 was predicted by Starbase and then verified by a dual-luciferase reporter and RNA Immunoprecipitation (RIP) assays.
Results
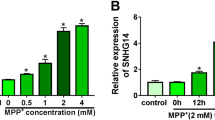
LINC00943 and CXCL12 were increased, and miR-7-5p was decreased in MPP+-inducted SK-N-SH cells. LINC00943 silencing promoted cell viability, and repressed apoptosis and the inflammatory response in MPP+-treated SK-N-SH cells. The mechanical analysis discovered that LINC00943 acted as a sponge of miR-7-5p to regulate CXCL12 expression.
Conclusions
LINC00943 knockdown could attenuate MPP+-triggered neuron injury by regulating the miR-7-5p/CXCL12 axis, hinting at a promising therapeutic target for PD treatment.





Similar content being viewed by others
References
Anderson FL, Coffey MM, Berwin BL, Havrda MC (2018) Inflammasomes: an emerging mechanism translating environmental toxicant exposure into neuroinflammation in Parkinson’s disease. Toxicol Sci 166(1):3–15
Cao B, Wang T, Qu Q, Kang T, Yang Q (2018) Long Noncoding RNA SNHG1 promotes neuroinflammation in Parkinson’s disease via regulating miR-7/NLRP3 pathway. Neuroscience 388:118–127
Charvin D, Medori R, Hauser RA, Rascol O (2018) Therapeutic strategies for Parkinson disease: beyond dopaminergic drugs. Nat Rev Drug Discov 17(11):804–822
Cui B, Guo X, You Y, Fu R (2019) Farrerol attenuates MPP(+) -induced inflammatory response by TLR4 signaling in a microglia cell line. Phytother Res 33(4):1134–1141
Feng Z, Zhang L, Wang S, Hong Q (2020) Circular RNA circDLGAP4 exerts neuroprotective effects via modulating miR-134-5p/CREB pathway in Parkinson’s disease. Biochem Biophys Res Commun 522(2):388–394
Ghosh A, Tyson T, George S, Hildebrandt EN, Steiner JA, Madaj Z, Schulz E, Machiela E, McDonald WG, Escobar Galvis ML et al (2016) Mitochondrial pyruvate carrier regulates autophagy, inflammation, and neurodegeneration in experimental models of Parkinson’s disease. Sci Transl Med 8(368):368ra174
Homayoun H (2018) Parkinson disease. Ann Intern Med 169(5):Itc33–Itc48
Ji BS, Gao Y (2008) Protective effect of trihexyphenidyl on hydrogen peroxide-induced oxidative damage in PC12 cells. Neurosci Lett 437(1):50–54
Jiang X, Jin T (2019) Current progress of mitochondrial quality control pathways underlying the pathogenesis of Parkinson’s disease. Oxid Med Cell Longev 2019:1–11
Kraus TFJ, Haider M, Spanner J, Steinmaurer M, Dietinger V, Kretzschmar HA (2017) Altered long noncoding RNA expression precedes the course of Parkinson’s disease-a preliminary report. Mol Neurobiol 54(4):2869–2877
Lev N, Melamed E, Offen D (2003) Apoptosis and Parkinson’s disease. Prog Neuropsychopharmacol Biol Psychiatry 27(2):245–250
Li D, Yang H, Ma J, Luo S, Chen S, Gu Q (2018) MicroRNA-30e regulates neuroinflammation in MPTP model of Parkinson’s disease by targeting Nlrp3. Hum Cell 31(2):106–115
Li Y, Niu M, Zhao A, Kang W, Chen Z, Luo N, Zhou L, Zhu X, Lu L, Liu J (2019) CXCL12 is involved in α-synuclein-triggered neuroinflammation of Parkinson’s disease. J Neuroinflamm 16(1):263
Liu R, Li F, Zhao W (2020) Long noncoding RNA NEAT1 knockdown inhibits MPP(+)-induced apoptosis, inflammation and cytotoxicity in SK-N-SH cells by regulating miR-212-5p/RAB3IP axis. Neurosci Lett 731:135060
Lu M, Sun WL, Shen J, Wei M, Chen B, Qi YJ, Xu CS (2018) LncRNA-UCA1 promotes PD development by upregulating SNCA. Eur Rev Med Pharmacol Sci 22(22):7908–7915
Lv Q, Wang Z, Zhong Z, Huang W (2020) Role of long noncoding RNAs in Parkinson’s disease: putative biomarkers and therapeutic targets. Parkinsons Dis 2020:1–12
Ma L, Bajic VB, Zhang Z (2013) On the classification of long non-coding RNAs. RNA Biol 10(6):925–933
Ma CL, Su L, Xie JJ, Long JX, Wu P, Gu L (2014) The prevalence and incidence of Parkinson’s disease in China: a systematic review and meta-analysis. J Neural Transm (Vienna) 121(2):123–134
Ma J, Wang Z, Chen S, Sun W, Gu Q, Li D, Zheng J, Yang H, Li X (2020) EphA1 activation induces neuropathological changes in a mouse model of Parkinson’s disease through the CXCL12/CXCR4 signaling pathway. Mol Neurobiol 58(3):913–915
Majidinia M, Mihanfar A, Rahbarghazi R, Nourazarian A, Bagca B, Avci ÇB (2016) The roles of non-coding RNAs in Parkinson’s disease. Mol Biol Rep 43(11):1193–1204
Puspita L, Chung SY, Shim JW (2017) Oxidative stress and cellular pathologies in Parkinson’s disease. Mol Brain 10(1):53
Ransohoff RM (2016) How neuroinflammation contributes to neurodegeneration. Science 353(6301):777–783
Riva P, Ratti A, Venturin M (2016) The long non-coding RNAs in neurodegenerative diseases: novel mechanisms of pathogenesis. Curr Alzheimer Res 13(11):1219–1231
Shen DF, Qi HP, Ma C, Chang MX, Zhang WN, Song RR (2020) Astaxanthin suppresses endoplasmic reticulum stress and protects against neuron damage in Parkinson’s disease by regulating miR-7/SNCA axis. Neurosci Res 165:51–60
Tan C, Liu X, Chen J (2018) Microarray analysis of the molecular mechanism involved in Parkinson’s disease. p 1590465
Timotijević G, Mostarica Stojković M, Miljković D (2012) CXCL12: role in neuroinflammation. Int J Biochem Cell Biol 44(6):838–841
Wang Z, Sun L, Jia K, Wang H, Wang X (2019) miR-9-5p modulates the progression of Parkinson’s disease by targeting SIRT1. Neurosci Lett 701:226–233
Wang R, Li Q, He Y, Yang Y, Ma Q, Li C (2020) miR-29c-3p inhibits microglial NLRP3 inflammasome activation by targeting NFAT5 in Parkinson’s disease. Genes Cells 25(6):364–374
Wilusz JE, Sunwoo H, Spector DL (2009) Long noncoding RNAs: functional surprises from the RNA world. Genes Dev 23(13):1494–1504
Xu X, Zhuang C, Wu Z, Qiu H, Feng H, Wu J (2018) LincRNA-p21 Inhibits cell viability and promotes cell apoptosis in Parkinson’s disease through activating α-synuclein expression. Biomed Res Int 2018:1–10
Yoon JH, Abdelmohsen K, Gorospe M (2014) Functional interactions among microRNAs and long noncoding RNAs. Semin Cell Dev Biol 34:9–14
Zhao J, Li H, Chang N (2020) LncRNA HOTAIR promotes MPP+-induced neuronal injury in Parkinson’s disease by regulating the miR-874-5p/ATG10 axis. Excli J 19:1141–1153
Zhou S, Zhang D, Guo J, Chen Z, Chen Y, Zhang J (2020) Deficiency of NEAT1 prevented MPP(+)-induced inflammatory response, oxidative stress and apoptosis in dopaminergic SK-N-SH neuroblastoma cells via miR-1277-5p/ARHGAP26 axis. Brain Res 1750:147156
Zhu J, Deng J, Zhang L, Zhao J, Zhou F, Liu N, Cai R, Wu J, Shu B, Qi S (2020) Reconstruction of lncRNA-miRNA-mRNA network based on competitive endogenous RNA reveals functional lncRNAs in skin cutaneous melanoma. BMC Cancer 20(1):927
Funding
There is no funding to report.
Author information
Authors and Affiliations
Contributions
HL and BW participated in the design of the work, methodology, data interpretation, and drafted the manuscript. QL participated in the collection of data and analysis for the work, carried out the statistical analysis. BC participated in the methodology, data interpretation. Hui Yang participated in data interpretation and methodology.
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest between authors. Han Lian, Baohua Wang, Quan Lu, Bin Chen and Hui Yang declare that they have no conflict of interest.
Ethics approval and consent to participate
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
13258_2021_1084_MOESM1_ESM.tif
Figure S1 Downregulation of miR-7-5p abrogated the effects of LINC00943 knockdown on cell viability, apoptosis, and the inflammatory response in MPP+-inducted SK-N-SH cells. SK-N-SH cells were treated with MPP++si-NC, MPP++si-LINC00943, MPP++si-LINC00943 + inhibitor NC, and MPP++si-LINC00943 + miR-7-5p inhibitor. (A) Cell viability was detected by MTT assay in treated SK-N-SH cells. (B) Apoptosis rate was analyzed by flow cytometry assay in treated SK-N-SH cells. (C and D) Protein levels of IL-1β and TNF-α were monitored by western blot assays in treated SK-N-SH cells. (E and F) The ROS generation and SOD activity were evaluated by the corresponding kits in treated SK-N-SH cells. (ANOVA with Tukey’s tests). N = 3, *P < 0.05, **P < 0.01, ***P < 0.001
13258_2021_1084_MOESM2_ESM.tif
Figure S2 MiR-7-5p upregulation promoted cell viability, and repressed apoptosis and inflammatory response by regulating CXCL12 in MPP+-inducted SK-N-SH cells. SK-N-SH cells were treated with MPP++miRNA NC, MPP++miR-7-5p mimic, MPP++miR-7-5p mimic + pc-NC, and MPP++miR-7-5p mimic + pc-CXCL12. (A) Cell viability of treated SK-N-SH cells was detected. (B) The apoptosis rate of treated SK-N-SH cells was examined. (C and D) Protein levels of IL-1β and TNF-α in treated SK-N-SH cells were determined. (E and F) ROS generation and SOD activity of treated SK-N-SH cells were analyzed. (ANOVA with Tukey’s tests). N = 3, *P < 0.05, **P < 0.01
Rights and permissions
About this article
Cite this article
Lian, H., Wang, B., Lu, Q. et al. LINC00943 knockdown exerts neuroprotective effects in Parkinson’s disease through regulates CXCL12 expression by sponging miR-7-5p. Genes Genom 43, 797–805 (2021). https://doi.org/10.1007/s13258-021-01084-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-021-01084-1