Abstract
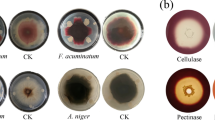
The biodiversity of wheat-associated bacteria from the northern hills zone of India was deciphered. A total of 247 bacteria was isolated from five different sites. Analysis of these bacteria by amplified ribosomal DNA restriction analysis (ARDRA) using three restriction enzymes, AluI, MspI and HaeIII, led to the grouping of these isolates into 19–33 clusters for the different sites at 75 % similarity index. 16S rRNA gene based phylogenetic analysis revealed that 65 %, 26 %, 8 % and 1 % bacteria belonged to four phyla, namely Proteobacteria, Firmicutes, Actinobacteria and Bacteroidetes, respectively. Overall, 28 % of the total morphotypes belonged to Pseudomonas followed by Bacillus (20 %), Stenotrophomonas (9 %), Methylobacterium (8 %), Arthrobacter (7 %), Pantoea (4 %), Achromobacter, Acinetobacter, Exiguobacterium and Staphylococcus (3 %), Enterobacter, Providencia, Klebsiella and Leclercia (2 %), Brevundimonas, Flavobacterium, Kocuria, Kluyvera and Planococcus (1 %). Representative strains from each cluster were screened in vitro for plant growth promoting traits, which included solubilisation of phosphorus, potassium and zinc; production of ammonia, hydrogen cyanide, indole-3-acetic acid and siderophore; nitrogen fixation, 1-aminocyclopropane-1-carboxylate deaminase activity and biocontrol against Fusarium graminearum, Rhizoctonia solani and Macrophomina phaseolina. Cold-adapted isolates may have application as inoculants for plant growth promotion and biocontrol agents for crops growing under cold climatic conditions.





Similar content being viewed by others
References
Aleksandrov VG, Blagodyr RN, Ilev IP (1967) Liberation of phosphoric acid from apatite by silicate bacteria. Mikrobiol Z 29:111–114
Anzai Y, Kim H, Park JY, Wakabayashi H, Oyaizu H (2000) Phylogenetic affiliation of the pseudomonads based on 16S rRNA sequence. Int J Syst Evol Microbiol 50:1563–1589
Araújo WL, Marcon J, Maccheroni W, van Elsas JD, van Vuurde JW, Azevedo JL (2002) Diversity of endophytic bacterial populations and their interaction with Xylella fastidiosa in citrus plants. Appl Environ Microbiol 68:4906–4914
Bais HP, Weir TL, Perry LG, Gilroy S, Vivanco JM (2006) The role of root exudates in rhizosphere interactions with plants and other organisms. Annu Rev Plant Biol 57:233–266
Bakker AW, Schippers B (1987) Microbial cyanide production in the rhizosphere in relation to potato yield reduction and Pseudomonas SPP-mediated plant growth-stimulation. Soil Biol Biochem 19:451–457
Bargabus R, Zidack N, Sherwood J, Jacobsen B (2002) Characterisation of systemic resistance in sugar beet elicited by a non-pathogenic, phyllosphere-colonizing Bacillus mycoides, biological control agent. Physiol Mol Plant Pathol 61:289–298
Beneduzi A, Peres D, da Costa PB, Bodanese Zanettini MH, Passaglia LMP (2008) Genetic and phenotypic diversity of plant-growth-promoting bacilli isolated from wheat fields in southern Brazil. Res Microbiol 159:244–250
Bisht SC, Mishra PK, Joshi GK (2013) Genetic and functional diversity among root-associated psychrotrophic Pseudomonad’s isolated from the Himalayan plants. Arch Microbiol 195:605–615
Boddey R, De Oliveira O, Urquiaga S, Reis V, De Olivares F, Baldani V, Döbereiner J (1995) Biological nitrogen fixation associated with sugar cane and rice: contributions and prospects for improvement. Plant Soil 174:195–209
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bray RH, Kurtz L (1945) Determination of total, organic, and available forms of phosphorus in soils. Soil Sci 59:39–46
Bric JM, Bostock RM, Silverstone SE (1991) Rapid in situ assay for indoleacetic acid production by bacteria immobilized on a nitrocellulose membrane. Appl Environ Microbiol 57:535–538
Brown ME, Burlingham SK (1968) Production of plant growth substances by Azotobacter chroococcum. J Gen Microbiol 53:135–144
Cappucino JC, Sherman N (1992) Nitrogen cycle. In: Microbiology: a laboratory manual, 4th edn. Benjamin/Cumming, New York, pp 311–312
Compant S, Duffy B, Nowak J, Clément C, Barka EA (2005) Use of plant growth-promoting bacteria for biocontrol of plant diseases: principles, mechanisms of action, and future prospects. Appl Environ Microbiol 71:4951–4959
Conn VM, Franco CM (2004) Effect of microbial inoculants on the indigenous actinobacterial endophyte population in the roots of wheat as determined by terminal restriction fragment length polymorphism. Appl Environ Microbiol 70:6407–6413
Coroler L, Elomari M, Hoste B, Gillis M, Izard D, Leclerc H (1996) Pseudomonas rhodesiae sp. nov., a new species isolated from natural mineral waters. Syst Appl Microbiol 19:600–607
Dey R, Pal K, Bhatt D, Chauhan S (2004) Growth promotion and yield enhancement of peanut (Arachis hypogaea L.) by application of plant growth-promoting rhizobacteria. Microbiol Res 159:371–394
Edwards U, Rogall T, Blöcker H, Emde M, Böttger EC (1989) Isolation and direct complete nucleotide determination of entire genes. Characterization of a gene coding for 16S ribosomal RNA. Nucleic Acids Res 17:7843–7853
Ghosh S, Penterman JN, Little RD, Chavez R, Glick BR (2003) Three newly isolated plant growth-promoting bacilli facilitate the seedling growth of canola. Brassica campestris. Plant Physiol Biochem 41:277–281
Goldstein AH (1994) Involvement of the quinoprotein glucose dehydrogenase in the solubilization of exogenous phosphates by gram-negative bacteria. In: Torriani-Gorni A, Yagil E, Silver S (eds) Phosphate in microorganisms: cellular and molecular biology. ASM Press, Washington, pp 197–203
Grover M, Ali SZ, Sandhya V, Rasul A, Venkateswarlu B (2011) Role of microorganisms in adaptation of agriculture crops to abiotic stresses. World J Microbiol Biotechnol 27:1231–1240
Gulati A, Rahi P, Vyas P (2008) Characterization of phosphate-solubilizing fluorescent pseudomonads from the rhizosphere of seabuckthorn growing in the cold deserts of Himalayas. Curr Microbiol 56:73–79
Gupta HS, Kant L (2012) Wheat improvement in Northern hills of India. Agric Res 1:100–116
Han SO, New P (1998) Variation in nitrogen fixing ability among natural isolates of Azospirillum. Microb Ecol 36:193–201
Hantsis-Zacharov E, Halpern M (2007) Culturable psychrotrophic bacterial communities in raw milk and their proteolytic and lipolytic traits. Appl Environ Microbiol 73:7162–7168
Holland MA, Polacco JC (1994) PPFMs and other covert contaminants: is there more to plant physiology than just plant? Annu Rev Plant Biol 45:197–209
Holland MA, Davis R, Moffitt S, O’Laughlin K, Peach D, Sussan S, Wimbrow L, Tayman B (2000) Using “leaf prints” to investigate a common bacterium. Am Biol Teach 62:128–131
Hu X, Chen J, Guo J (2006) Two phosphate and potassium-solubilizing bacteria isolated from Tianmu mountain, Zhejiang, China. World J Microbiol Biotechnol 22:983–990
Jaccard P (1912) The distribution of the flora in the alpine zone. New Phytol 11:37–50
Jacobson CB, Pasternak J, Glick BR (1994) Partial purification and characterization of 1-aminocyclopropane-1-carboxylate deaminase from the plant growth promoting rhizobacterium Pseudomonas putida GR12-2. Can J Microbiol 40:1019–1025
Jha P, Kumar A (2009) Characterization of novel plant growth promoting endophytic bacterium Achromobacter xylosoxidans from wheat plant. Microb Ecol 58:179–188
Kloepper J, Schroth M, Miller T (1980) Effects of rhizosphere colonization by plant growth-promoting rhizobacteria on potato plant development and yield. Phytopathology 70:1078–1082
Kumar M, Yadav AN, Tiwari R, Prasanna R, Saxena AK (2013) Deciphering the diversity of culturable thermotolerant bacteria from Manikaran hot springs. Ann Microbiol 64:741–751
Kumar M, Yadav AN, Tiwari R, Prasanna R, Saxena AK (2014) Evaluating the diversity of culturable thermotolerant bacteria from four hot springs of India. J Biodivers Bioprospect Dev 1:127. doi:10.4172/ijbbd.1000127
Liu Y, Zuo S, Zou Y, Wang J, Song W (2013) Investigation on diversity and population succession dynamics of endophytic bacteria from seeds of maize (Zea mays L., Nongda108) at different growth stages. Ann Microbiol 63:71–79
Madhaiyan M, Poonguzhali S (2014) Methylobacterium pseudosasicola sp. nov. and Methylobacterium phyllostachyos sp. nov., two novel species isolated from bamboo leaf surfaces. Int J Syst Evol Microbiol 64:2376–2384. doi:10.1099/ijs.0.057232-0
Magnani G, Didonet C, Cruz L, Picheth C, Pedrosa F, Souza E (2010) Diversity of endophytic bacteria in Brazilian sugarcane. Genet Mol Res 9:250–258
McInroy JA, Kloepper JW (1995) Population dynamics of endophytic bacteria in field-grown sweet corn and cotton. Can J Microbiol 41:895–901
Mehta S, Nautiyal CS (2001) An efficient method for qualitative screening of phosphate-solubilizing bacteria. Curr Microbiol 43:51–56
Misaghi I, Donndelinger C (1990) Endophytic bacteria in symptom-free cotton plants. Phytopathology 80:808–811
Mishra PK, Bisht SC, Ruwari P, Selvakumar G, Joshi GK, Bisht JK, Bhatt JC, Gupta HS (2011) Alleviation of cold stress in inoculated wheat (Triticum aestivum L.) seedlings with psychrotolerant Pseudomonads from NW Himalayas. Arch Microbiol 193:497–513
Monib M, Zahra MK, Abdel Al, Heggo A (1984) Role of silicate bacteria in releasing K and Si from biotite and orthoclase. Soil biology and conservation of the biosphere. In: Szegi, J. (ed) Akademiai Kiado, Budapest, pp 733–743
Murphy J, Riley J (1962) A modified single solution method for the determination of phosphate in natural waters. Anal Chim Acta 27:31–36
Nejad P, Johnson PA (2000) Endophytic bacteria induce growth promotion and wilt disease suppression in oilseed rape and tomato. Biol Control 18:208–215
Pandey S, Singh S, Yadav AN, Nain L, Saxena AK (2013) Phylogenetic diversity and characterization of novel and efficient cellulase producing bacterial isolates from various extreme environments. Biosci Biotechnol Biochem 77:1474–1480
Patten CL, Glick BR (2002) Role of Pseudomonas putida indoleacetic acid in development of the host plant root system. Appl Environ Microbiol 68:3795–3801
Rico A, Ortiz-Barredo A, Ritter E, Murillo J (2004) Genetic characterization of Erwinia amylovora strains by amplified fragment length polymorphism. J Appl Microbiol 96:302–310
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Saravanan VS, Osborne J, Madhaiyan M, Mathew L, Chung J, Ahn K, Sa T (2007) Zinc metal solubilization by Gluconacetobacter diazotrophicus and induction of pleomorphic cells. J Microbiol Biotechnol 17:1477–1482
Schwyn B, Neilands J (1987) Universal chemical assay for the detection and determination of siderophores. Anal Biochem 160:47–56
Selvakumar G, Joshi P, Suyal P, Mishra PK, Joshi GK, Bisht JK, Bhatt JC, Gupta HS (2011) Pseudomonas lurida M2RH3 (MTCC 9245), a psychrotolerant bacterium from the Uttarakhand Himalayas, solubilizes phosphate and promotes wheat seedling growth. World J Microbiol Biotechnol 27:1129–1135
Sheng XF (2005) Growth promotion and increased potassium uptake of cotton andrape by a potassium releasing strain ofBacillus edaphicus. Soil Biol Biochem 37:1918–1922
Sijam K, Dikin A (2005) Biochemical and physiological characterization of Burkholderia cepacia as biological control agent. Int J Agric Biol 7:385–388
Sturz A, Christie B, Matheson B, Arsenault W, Buchanan N (1999) Endophytic bacterial communities in the periderm of potato tubers and their potential to improve resistance to soil-borne plant pathogens. Plant Pathol 48:360–369
Sun L, Lu Z, Bie X, Lu F, Yang S (2006) Isolation and characterization of a co-producer of fengycins and surfactins, endophytic Bacillus amyloliquefaciens ES-2, from Scutellaria baicalensis Georgi. World J Microbiol Biotechnol 22:1259–1266
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Thomas P, Soly TA (2009) Endophytic bacteria associated with growing shoot tips of banana (Musa sp.) cv. Grand naine and the affinity of endophytes to the host. Microb Ecol 58:952–964
Tilak KVBR, Ranganayaki N, Pal KK, De R, Saxena AK, Nautiyal CS, Mittal S, Tripathi AK, Johri BN (2005) Diversity of plant growth and soil health supporting bacteria. Curr Sci 89:136–150
Verma P, Yadav AN, Kazy SK, Saxena AK, Suman A (2014) Evaluating the diversity and phylogeny of plant growth promoting bacteria associated with wheat (Triticum aestivum) growing in central zone of India. Int J Curr Microbiol Appl Sci 3:432–447
Vyas P, Rahi P, Gulati A (2009) Stress tolerance and genetic variability of phosphate-solubilizing fluorescent Pseudomonas from the cold deserts of the trans-Himalayas. Microb Ecol 58:425–434
Walkley A, Black IA (1934) An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Sci 37:29–38
Yadav AN, Sachan SG, Verma P, Saxena AK (2014a) Prospecting cold deserts of north western Himalayas for microbial diversity and plant growth promoting attributes. J Biosci Bioeng. doi:10.1016/j.jbiosc.2014.11.006
Yadav AN, Sachan SG, Verma P, Tyagi SP, Kaushik R, Saxena AK (2014b) Culturable diversity and functional annotation of psychrotrophic bacteria from cold desert of Leh Ladakh (India). World J Microbiol Biotechnol. doi:10.1007/s11274-014-1768-z
Yadav AN, Verma P, Kumar M, Pal KK, Dey R, Gupta A, Padaria JC, Gujar GT, Kumar S, Suman A, Prasanna R, Saxena AK (2014c) Diversity and phylogenetic profiling of niche-specific Bacilli from extreme environments of India. Ann Microbiol. doi:10.1007/s13213-014-0897-9
Yang J, Kloepper JW, Ryu C-M (2009) Rhizosphere bacteria help plants tolerate abiotic stress. Trends Plant Sci 14:1–4
Zinniel DK, Lambrecht P, Harris NB, Feng Z, Kuczmarski D, Higley P, Ishimaru CA, Arunakumari A, Barletta RG, Vidaver AK (2002) Isolation and characterization of endophytic colonizing bacteria from agronomic crops and prairie plants. Appl Environ Microbiol 68:2198–2208
Acknowledgements
The authors are grateful to the Division of Microbiology, Indian Agricultural Research Institute (IARI), New Delhi and Department of Biotechnology (DBT), Ministry of Science and Technology for providing the facilities and financial support needed to undertake this investigation.
Declaration
The experiments undertaken comply with the current laws of India, the country where the investigation was undertaken. There are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Priyanka Verma and Ajar Nath Yadav contributed equally to the present work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary table 1
(DOC 38 kb)
Supplementary table 2
(DOC 124 kb)
Rights and permissions
About this article
Cite this article
Verma, P., Yadav, A.N., Khannam, K.S. et al. Assessment of genetic diversity and plant growth promoting attributes of psychrotolerant bacteria allied with wheat (Triticum aestivum) from the northern hills zone of India. Ann Microbiol 65, 1885–1899 (2015). https://doi.org/10.1007/s13213-014-1027-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13213-014-1027-4