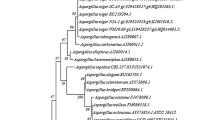
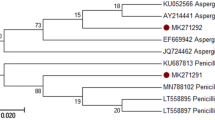
Abstract
The increasing awareness of importance of xylanases in various industrial processes has led to intensive research in effective production of xylanases. In this study, eight fungal strains were screened for the production of xylanases in submerged (SmF) and solid state fermentations (SSF) in corncob-based media. Positive results of reddish orange halo-zones of hydrolysis were obtained for qualitative screening. In SmF, xylanase activity from Aspergillus fumigatus SD5A yielded highest activity (50.55 U/ml) in 168 h, while Aspergillus flavus SD4A, A. fumigatus L1, Fusarium solani SD3C, Aspergillus niger L3, Trichoderma longibrachiatum L2, Botryodiplodia sp. L5 and A. flavus L4, showed xylanase activities ranging from 10.38 to 44.81 U/ml. In SSF, xylanase activities ranged from 12.30 to 48.63 U/g in 120 h, with the highest activity obtained from A. fumigatus L1. Optimum temperatures for xylanases obtained in SmF ranged from 55 to 70 °C, while that of SSF ranged from 50 to 70 °C. Also, the optimum pH for xylanases obtained in both SmF and SSF ranged from 5.0 to 7.0. All the fungi did not produce aflatoxin on neutral red desiccated coconut agar. The fungal xylanases improved dough rising of bread and clarification of orange juice by 1.87–2.2-folds and 58.12–74.22% respectively. The results reported here showed that the range of characteristics exhibited by the xylanases could make them widely applicable in various industries, and also an important way for the valorization of agricultural waste.









Similar content being viewed by others
References
Kumar, L., Kumar, D., Nagar, S., Gupta, R., Garg, N., Kuhad, R.C., Gupta, V.K.: Modulation of xylanase production from alkaliphilic Bacillus pumilus VLK-1 through process optimization and temperature shift operation. 3 Biotech. 4, 345–356 (2014)
Won-Jae, C., Da Yeon, P., Yong-Keun, C., Soon-Kwang, H.: A novel alkaliphilic xylanase from the newly isolated mesophilic Bacillus sp. MX47: production, purification, and characterization. Appl. Biochem. Biotechnol. 168(4), 899–909 (2012)
Silva, L.A.O., Terrasan, C.R.F., Carmona, E.C.: Purification and characterization of xylanases from Trichoderma inhamatum. Electron. J. Biotechnol. 18, 307–313 (2015)
Beg, Q.K., Kapoor, M., Mahajan, L., Hoondal, G.S.: Microbial xylanases and their industrial applications: a review. Appl. Microbiol. Biotechnol. 56, 326–338 (2001)
Shallom, D., Shoham, Y.: Microbial hemicellulases. Curr. Opin. Microbiol. 6, 219–228 (2003)
Svarachorn, A.: Production of fungal–xylanase using agricultural waste by solid state fermentation. J. Sci. Res. Chulalongkon Univ. 24(1), 13–20 (1999)
Kanimozhi, K., Nagalakshmi, P.K.: Xylanase production from Aspergillus niger by solid state fermentation using agricultural waste as substrate. Inter. J. Curr. Microbiol. Appl. Sci. 3(3), 437–446 (2014)
Goulart, A.J., Carmona, E.C., Monti, R.: Partial purification and properties of cellulase-free alkaline xylanase produced by Rhizopus stolonifer in solid-state fermentation. Braz. Arch. Biol. Technol. 48, 327–333 (2005)
Yegin, S., Buyukkileci, A.O., Sargin, S., Goksungur, Y.: Exploitation of agricultural wastes and by-products for production of Aureobasidium pullulans Y-2311-1 xylanase: screening, bioprocess optimization and scale up. Waste Biomass Valoriz. (2016). doi:10.1007/s12649-016-9646-6
Bedford, M.R., Classen, H.L., 1992. The influence of dietary xylanase on intestinal viscosity and molecular weight distribution of carbohydrates in rye-fed broiler chick. In: Visser, J., Beldman, G., van Someren, M.A.K., Voragen, A.G.J. (eds) Xylans and xylanases, pp 361–370. Elsevier, Amsterdam.
Wong, K.K.Y., Saddler, J.N.: Trichoderma xylanases, their properties and purification. Crit. Rev. Biotechnol. 12, 413–435 (1992)
Beg, Q.K., Bhushan, B., Kapoor, M., Hoondal, G.S.: Enhanced production of a thermostable xylanase from Streptomyces sp. QG-11-3 and its application in biobleaching of eucalyptus kraft pulp. Enzyme Microb. Technol. 27, 459–466 (2000)
Harris, A.D., Ramalingam, C.: Xylanase and its application in food Industry: a review. J. Exper. Sci. 1(7), 1–11 (2010)
Kapoor, M., Beg, Q.K., Bhushan, B., Singh, K., Dadhich, K.S., Hoondal, G.S.: Application of an alkaline and thermostable polygalacturonase from Bacillus sp. MG-cp-2 in degumming of ramie (Boehmeria nivea) and sunn hemp (Crotalaria juncea) bast fibers. Process Biochem. 36, 803–807 (2001)
Lateef, A., Oloke, J.K., Gueguim Kana, E.B., Oyeniyi, S.O., Onifade, O.R., Oyeleye, A.O., Oladosu, O.C., Oyelami, A.O.: Improving the quality of agro-wastes by solid-state fermentation: enhanced antioxidant activities and nutritional qualities. World J. Microbiol. Biotechnol. 24, 2369–2374 (2008)
Lateef, A., Ojo, M.O.: Public health issues in the processing of Cassava (Manihot esculenta) for production ‘Lafun’ and the application of hazard analysis control measures. Qual. Assur. Saf. Crops Foods. 8, 165–177 (2016)
Mohammed, I.J.: Screening of fungi isolated from environmental samples for xylanase and cellulase production. Inter. Scholarly Res. Notices: Microbiol. (2013). doi:10.1155/2013/283423
Domsch, K.H., Gams, W., erson, T.H.: Paecilomyces. In: Compendium of soil fungi, pp. 530–532. Academic Press, London (1980)
Lateef, A., Gueguim Kana, E.B.: Utilization of cassava wastes in the production of fructosyltransferase by Rhizopus stolonifer LAU 07. Rom. Biotechnol. Lett. 17(3), 7309–7316 (2012)
Sridevi, B., Charya, M.A.S.: Isolation, identification and screening of potential cellulase-free xylanase producing fungi. Afr. J. Biotechnol. 10(22), 4624–4630 (2011)
Bailey, M.J., Biely, P., Poutanen, K.: Interlaboratory testing for methods of assay of xylanase activity. J. Biotechnol. 23, 257–270 (1992)
Bajaj, B.K., Abbass, M.: Studies on an alkali-thermostable xylanase from Aspergillus fumigatus MA28. 3 Biotech. 1, 161–171 (2011)
Ahmed, S., Imdad, S.S., Jamil, A.: Comparative study for the kinetics of extracellular xylanases from Trichoderma harzianum and. Chaetomium thermophilum. Electron. J. Biotechnol. 15(3), 1–8 (2012)
Ahmed, S.A., Saleh, S.A.A., Mostafaa, F.A., El Atya A.A.: Characterization and valuable applications of xylanase from endophytic fungus Aspergillus terreus KP900973: isolated from Corchorus olitorius. Biocatal. Agric. Biotechnol. (2016).doi:10.1016/j.bcab.2016.05.015
Atanda, O.O., Ogunrinu, M.C., Olorunfemi, F.M.: A neutral red dessicated coconut agar for rapid detection of aflatoxigenic fungi and visual determination of aflatoxins. World Mycotoxin J. 4, 147–155 (2011)
Taher, I.B., Bennour, H., Fickers, P., Hassouna, M.: Valorization of potato peels residues on cellulase production using a mixed culture of Aspergillus niger ATCC 16404 and Trichoderma reesei DSMZ 970. Waste and Biomass Valoriz. (2016). doi:10.1007/s12649-016-9558-5
Ganaie, M.A., Lateef, A., Gupta, U.S.: Enzymatic trends of fructooligosaccharides production by microorganisms. Appl. Biochem. Biotechnol. 172(4), 2143–2159 (2014)
Shankar, J.: An overview of toxins in Aspergillus associated with pathogenesis. Inter. J. Life Sci. Biotechnol. Pharma. Res. 2(2), 16–31 (2013)
Ramírez-Camejo, L.A., Zuluaga-Montero, A., Lázaro-Escudero, M.A., Hernández-Kendall, V.N., Bayman, P.: Phylogeography of the cosmopolitan fungus Aspergillus flavus: is everything everywhere? Fungal Biol. 116(3), 452–463 (2012)
De Guadalupe Moctezuma-Zárate, M., Vargas-Morales, J.M., Cárdenas-González, J.F., Martínez-Juárez, V.M., Acosta-Rodríguez, I.: Induction of extracellular lytic enzymes by Fusarium solani. Adv. Microbiol. 3, 24–30 (2013)
Gupta, V.J., Gaur, R., Yadava, S.K., Darmwal, N.S.: Optimization of xylanase production from free and immobilized cells of Fusarium solani F7. BioResources. 4(3), 932–945 (2009)
Arabi, M.I.E, Bakri, Y., Jawhar, M.: Extracellular xylanase production by Fusarium species in solid state fermentation. Polish J. Microbiol. 60(3), 209–212 (2011)
Nagar, S., Mittal, A., Gupta, V.K.: Enzymatic clarification of fruit juices (apple, pineapple, and tomato) using purified Bacillus pumilus SV-85S xylanase. Biotechnol. Bioprocess Eng. 17, 1165–1175 (2012)
Sudan, R., Bajaj, B.K.: Production and biochemical characterization of xylanase from an alkalitolerant novel species Aspergillus niveus RS2. World J. Microbiol. Biotechnol. 23, 491–500 (2007)
Chapla, D., Divecha, J., Madamwar, D., Shah, A.: Utilization of agro-industrial waste for xylanase production by Aspergillus foetidus MTCC 4898 under solid state fermentation and its application in saccharification. Biochem. Eng. J. 49(3), 361–369 (2010)
Antoine, A.A., Jacqueline, D., Thonart, P.: Xylanase production by Penicillium canescens on soy oil cake in solid-state fermentation. Appl. Biochem. Biotechnol. 160, 50–62 (2010)
Nair, S.G., Sindhu, R., Shankar, S.: Purification and biochemical characterization of two xylanases from Aspergillus sydowii SBS45. Appl. Biochem. Biotechnol. 149, 229–243 (2008)
De Carvalho Peixoto-Nogueira, S., Michelin, M., Betini, J.H.A., Jorge, J.A., Terenzi, H.F., de Moraes, M.D.L.T.: Production of xylanase by Aspergilli using alternative carbon sources: application of the crude extract on cellulose pulp biobleaching. J. Ind. Microbiol. Biotechnol. 36, 149–155 (2009)
Bajaj, B.K., Singh, N.P.: Production of xylanase from an alkalitolerant sp. 7b under solid-state fermentation, its purification, and characterization. Appl. Biochem. Biotechnol. 162, 1804–1818 (2010)
Tallapragada, P., Venkatesh, K.: Isolation, identification and optimization of xylanase enzyme produced by Aspergillus niger under submerged fermentation. J. Microbiol. Biotechnol. Res. 1(4), 137–147 (2011)
Murthy, P.S., Naidu, M.M.: Production and application of xylanase from Penicillium sp. utilizing coffee by-products. Food Bioprocess Technol. 5(2), 657–664 (2012)
Ruckmani, A., Rajendran, A.: Production of cellulase-poor xylanase by an alkalitolerant strain of Aspergillus flavus. Indian J. Microbiol. 41, 115–118 (2001)
Polizeli, M.L.T.M., Rizzati, A.C.S., Monti, R., Terenzi, H.F., Jorge, J.A., Amorin, D.S.: Xylanases from fungi: Properties and industrial applications. Appl. Biochem. Biotechnol. 67(5), 577–591 (2005)
Srinivasan, M.C., Rele, M.V.: Microbial xylanases for paper industry. Curr. Sci. 77, 137–142 (1999)
Dutta, T., Sengupta, R., Sahoo, R., Sinha-Ray, S., Bhattacharjee, A., Ghosh, S.: A novel cellulase-free alkaliphilic xylanase from alkalitolerant Penicillium citrinum: production, purification and characterization. Lett. Appl. Microbiol. 44, 206–211 (2007)
Huang, L., Hseu, T.H., Wey, T.T.: Purification and characterization of an endoxylanase from Trichoderma koningii G-39. Biochemical J. 278(2), 329–333 (1991)
Raj, K.C., Chandra, T.S.: Purification and characterization of xylanase from alkali-tolerant Aspergillus fischeri Fxn1. FEMS Microbiol. Lett. 145(3), 457–461 (1996)
Lucena-neto, S.A., Ferreira-filho, E.X.: Purification and characterization of a new xylanase from Humicola grisea var. Thermoidea. Braz. J. Microbiol. 35(1–2), 86–90 (2004)
Bakri, Y., Masson, M., Thonart, P.: Isolation and identification of two new fungal strains for xylanase production. Appl. Biochem. Biotechnol. 162, 1626–1634 (2010)
Maalej, I., Belhaj, I., Masmoudi, N.F., Belghith, H.: Highly thermostable xylanase of the thermophilic fungus Talaromyces thermophilus: purification and characterization. Appl. Biochem. Biotechnol. 158, 200–212 (2009)
Kulkarni, N., Shendye, A., Rao, M.: Molecular and biotechnological aspects of xylanases. FEMS Microbiol. Rev. 23(4), 411–456 (1999)
Franco, P.F., Ferreira, H.M., Filho, E.X.F.: Production and characterization of hemicellulase activities from Trichoderma harzianum strain T4. Biotechnol. Appl. Biochem. 40(3), 255–259 (2004)
Chipeta, Z.A., Preez, J.C., Szakacs, G., Christopher, L.: Xylanase production by fungal strains on spent sulphite liquor. Appl. Microbiol. Biotechnol. 69(1), 71–78 (2005)
Subramanian, S., Prema, P.: Cellulase-free xylanases from Bacillus and other microorganisms. FEMS Microbiol. Lett. 183, 1–7 (2000)
Fialho, M.B., Carmona, E.C.: Purification and characterization of xylanases from Aspergillus giganteus. Folia Microbiol. 49(1), 13–18 (2004)
Ratanachomsri, U., Sriprang, R., Sornlek, W., Buaban, B., Champreda, V., Tanapongpipat, S., Eurwilaichitr, L.: Thermostable xylanase from Marasmius sp.: purification and characterization. J. Biochem. Mol. Biol. 39(1), 105–110 (2006)
Butt, M.S., Nadeem, M.T., Ahmad, Z., Sultan, M.T.: Xylanases and their applications in baking industry. Food Technol. Biotechnol. 46, 22–31 (2008)
Kaltsa, O., Georgopoulos, T., Yanniotis, S., Mandala, L.: Effect of enzyme blends and dough strengthening emulsifier on extending the shelf life of sandwich bread applying response surface methodology. Inter. J. Innov. Res. Sci. Eng. Technol. 3(4), 149–160 (2013)
Bajaj, B.K., Manhas, K.: Production and characterization of xylanase from Bacillus licheniformis P11(C) with potential for fruit juice and bakery industry. Biocatal. Agric. Biotechnol. 1, 330–337 (2012)
Dhiman, S.S., Garg, G., Sharma, J., Mahajan, R.: Characterization of statistically produced xylanase for enrichment of fruit juice clarification process. New Biotechnol. 28(6), 746–755 (2011)
Rai, P., Majumdar, G.C., Gupta, S.D., De, S.: Optimizing pectinase usage in pretreatment of mosambi juice for clarification by response surface methodology. J. Food Eng. 64, 397–403 (2003)
Kumar, L., Nagar, S., Mittal, A., Garg, N., Gupta, V.K.: Immobilization of xylanase purified from Bacillus pumilus VLK-1 and its application in enrichment of orange and grape juices. J. Food Sci. Technol. 51(9), 1737–1749 (2014)
Uçan, F., Akyildiz, A., Agçam, E.: Effects of different enzymes and concentrations in the production of clarified lemon juice. J. Food Processing. (2014).doi:10.1155/2014/215854
Pal, A., Khanum, F.: Efficacy of xylanase purified from Aspergillus niger DFR-5 alone and in combination with pectinase and cellulase to improve yield and clarity of pineapple juice. J. Food Sci. Technol. 48(5), 560–568 (2011)
Díaz, A.B., Alvarado, O., de Ory, I., Caro, I., Blandino, A.: Valorization of grape pomace and orange peels: improved production of hydrolytic enzymes for the clarification of orange juice. Food Bioprod. Process. 91, 580–586 (2013)
Olfa, E., Mondher, M., Issam, S., Ferid, L., Nejib, N.M.: Induction, properties and application of xylanase activity from Sclerotinia sclerotiorum S2 fungus. J. Food Chem. 31, 96–107 (2007)
Shah, S.: Optimization of an enzyme assisted process for juice extraction and clarification from litchis (Litchi chinensis Sonn.). Inter. J. Food Eng. 3(3), 1–17 (2007)
Ahmad, I., Jha, Y.K., Anurag, R.K.: Optimization of enzymic extraction process for higher yield and clarity of guava juice. J. Food Sci. Technol. 46, 307–311 (2009)
Lateef, A., Gueguim-Kana, E.B.: Quality assessment and hazard analysis in the small-scale production of poultry feeds in Ogbomoso, Southwest, Nigeria. Qual. Assur. Saf. Crops Foods. 6(1), 105–113 (2014)
Moss, M.O.: Risk assessment for aflatoxins in food stuffs. Inter. Biodeterior. Biodegrad. 50(3–4), 137–142 (2002)
Saleemullah, A.L., Khalil, I.A., Shah, H.: Aflatoxin contents of stored and artificially inoculated cereals and nuts. Food Chem. 98, 699–703 (2006)
Abbas, H.K., Shier, W.T., Horn, B.W., Weaver, M.A.: Cultural methods for aflatoxin detection. J. Toxicol. 23(2&3), 295–315 (2004)
Atanda, O.O., Akpan, I., Enikuomehin, O.A.: Palm kernel agar: an alternative culture medium for rapid detection of aflatoxins in agricultural commodities. Afr. J. Biotechnol. 5, 1029–1033 (2006)
Ezekiel, C.N., Adetunji, M.C., Atanda, O.O., Frisvad, J.C., Houbraken, J., Samson, R.A.: Phenotypic differentiation of species from Aspergillus section Flavi on neutral red desiccated coconut agar. World Mycotoxin J. 7(3), 335–344 (2014)
Ezekiel, C.N., Kayode, F.O., Fapohunda, S.O., Olorunfemi, M.F., Kponi, B.T.: Aflatoxigenic moulds and aflatoxins in street-vended snacks in Lagos, Nigeria. Internet J. Food Saf. 14, 83–88 (2012)
Acknowledgements
AL is grateful to the authority of LAUTECH, Ogbomoso, Nigeria for the provision of some of the facilities used in the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Elegbede, J.A., Lateef, A. Valorization of Corn-Cob by Fungal Isolates for Production of Xylanase in Submerged and Solid State Fermentation Media and Potential Biotechnological Applications. Waste Biomass Valor 9, 1273–1287 (2018). https://doi.org/10.1007/s12649-017-9932-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-017-9932-y