Abstract
Aerial algae are considered to be highly tolerant of and adaptable to severe conditions including radiation, desiccation, high temperatures, and nutrient deficiency, compared with those from aquatic habitats. There are considerable variations in the fatty acid (FA) composition of aerial microalgae from dry environments. A new species with a high lipid level was found on concrete surfaces and was identified as Coccomyxa sp. KGU-D001 (Trebouxiophyceae). This study characterized its FA content and profile in a bath culture. The alga showed a constant specific growth rate (0.26 day−1) ranging in light intensity from 20 to 80 μmol photons m−2 s−1. The algal cells started to form oil bodies in the early stationary phase of growth, and oil bodies occupied most of the cells during the late stationary phase when the cells accumulated 27 % total fatty acids (TFA). The process of lipid body formation accumulating large amounts of triacylglycerols (TAG) appeared to be very unusual in response to stress conditions persisting for a relatively long culture time (50 days). This study could indicate that aerial microalgae will be a candidate for biodiesel production when a new cultivation method is developed using extreme stresses such as nutritional deficiency and/or desiccation.





Similar content being viewed by others
References
Sharma, Y. C., Singh, B., & Upadhyay, S. N. (2008). Advancements in development and characterization of biodiesel: a review. Fuel, 57, 2355–2373.
Mata, T. M., Martins, A. A., & Caetano, N. S. (2010). Microalgae for biodiesel production and other application: a review. Renewable and Sustainable Energy Reviews, 14, 217–232.
Ahmad, A. L., Mat Yasin, N. H., Derek, C. J. C., & Lim, J. K. (2011). Microalgae as a sustainable energy source for biodiesel production: a review. Renewable and Sustainable Energy Reviews, 15, 584–593.
Rao, A. R., Dayananda, C., Sarada, R., Shamala, T. R., & Ravishankar, G. A. (2007). Effect of salinity on growth of green alga Botryococcus braunii and its constituents. Bioresource Technology, 98, 560–564.
Ruangsomboon, S. (2012). Effect of light, nutrient, cultivation time and salinity on lipid production of newly isolated strain of the green microalga, Botryococcus braunii KMITL 2. Bioresource Technology, 109, 261–265.
Yeesang, C., & Cheirsilp, B. (2011). Effect of nitrogen, salt, and iron content in the growth medium and light intensity on lipid production by microalgae isolated from freshwater sources in Thailand. Bioresource Technology, 102, 3034–3040.
McGinnis, K. M., Dempster, T. A., & Sommerfeld, M. R. (1997). Characterization of the growth and lipid content of the diatom Chaetoceros muelleri. Journal of Applied Phycology, 9, 19–24.
Satoh, A., Ichii, K., Matsumoto, M., Kubota, C., Nemoto, M., Tanaka, M., Yoshino, T., Matsunaga, T., & Tanaka, T. (2013). A process design and productivity evaluation for oil production by indoor mass cultivation of a marine diatom, Fistulifera sp. JPCC DA0580. Bioresource Technology, 137, 132–138.
Chaffin, J. D., Mishra, S., Kuhaneck, R. M., Heckathorn, S. A., & Bridgeman, T. B. (2012). Environmental controls on growth and lipid content for the freshwater diatom, Fragilaria capucina: a candidate for biofuel production. Journal of Applied Phycology, 24, 1045–1051.
Feng, D., Chen, Z., Xue, S., & Zhang, W. (2011). Increased lipid production of the marine oleaginous microalgae Isochrysis zhangjiangensis (Chrysophyta) by nitrogen supplement. Bioresource Technology, 102, 6710–6716.
Takagi, M., Watanabe, K., Yamaberi, K., & Yoshida, T. (2000). Limited feeding of potassium nitrate for intracellular lipid and triglyceride accumulation of Nannochloris sp. UTEX LB1999. Applied Microbiology and Biotechnology, 54, 112–117.
Su, C. H., Chien, L. J., Gomes, J., Lin, Y. S., Yu, Y. K., Liou, J. S., & Syu, R. J. (2011). Factors affecting lipid accumulation by Nannochloropsis oculata in a two-stage cultivation process. Journal of Applied Phycology, 23, 903–908.
Mansour, M. P., Frampton, D. M. F., Nichols, P. D., Volkman, J. K., & Blackburn, S. I. (2005). Lipid and fatty acid yield of nine stationary-phase microalgae: applications and unusual C24-C28 polyunsaturated fatty acids. Journal of Applied Phycology, 17, 287–300.
Gouveia, L., & Oliveira, A. C. (2009). Microalgae as a raw material for biofuels production. Journal of Industrial Microbiology & Biotechnology, 36, 269–274.
Matsunaga, T., Matsumoto, M., Maeda, Y., Sugiyama, H., Sato, R., & Tanaka, T. (2009). Characterization of marine microalga, Scenedesmus sp. strain JPCC GA0024 toward biofuel production. Biotechnology Letters, 31, 1367–1372.
Liu, J., Yuan, C., Hu, G., & Li, F. (2012). Effect of light intensity on the growth and lipid accumulation of microalga Scenedesmus sp. 11–1 under nitrogen limitation. Applied Biochemistry and Biotechnology, 166, 2127–2137.
Abe, K., Takizawa, H., Kimura, S., & Hirano, M. (2004). Characteristics of chlorophyll formation of the aerial microalga Coelastrella striolata var. multistriata and its application of environmental biomonitoring. Journal of Bioscience and Bioengineering, 98, 34–39.
Abe, K., Hattori, H., & Hirano, M. (2007). Accumulation and antioxidant activity of secondary carotenoids in the aerial microalga Coelastrella striolata var. multistriata. Food Chemistry, 100, 656–661.
Gustavs, L., Eggert, A., Michalik, D., & Karsten, U. (2010). Physiological and biochemical responses of green microalgae from different habitats to osmotic and matric stress. Protoplasma, 243, 3–14.
Přibyl, P., Cepák, V., & Zachleder, V. (2012). Production of lipids in 10 strains of Chlorella and Parachlorella, and enhanced lipid productivity in Chlorella vulgaris. Applied Microbiology and Biotechnology, 94, 549–561.
Shiratake, T., Sato, A., Minoda, A., Tsuzuki, M., & Sato, N. (2013). Air-drying of cells, the novel conditions for stimulated synthesis of triacylglycerol in a green alga, Chlorella kessleri. PloS One, 8, 1–8.
Medlin, L., Elwood, H. J., Stickel, S., & Sogin, M. L. (1988). The characterization of enzymatically amplified eukaryotic 16S-like rRNA-coding regions. Gene, 71, 491–499.
Hamby, R. K., Sims, L., Issel, L., & Zimmer, E. (1988). Direct ribosomal RNA sequencing: optimization of extraction and sequencing methods for work with higher plants. Plant Molecular Biology Reporter, 6, 175–192.
Altschul, S. F., Gish, W., Miller, W., Myers, E. W., & Lipman, D. J. (1990). Basic local alignment search tool. Journal of Molecular Biology, 215, 403–410.
Greenspan, P., Mayer, E. P., & Fowler, S. D. (1985). Nile red: a selective fluorescent stain for intracellular lipid droplets. Journal of Cell Biology, 100, 965–973.
Makarova, V. V., Kosourov, S., Krendeleva, T. E., Semin, B. K., Kukarskikh, G. P., Rubin, A. B., Sayre, R. T., Ghirardi, M. L., & Seibert, M. (2007). Photoproduction of hydrogen by sulfur-deprived C. reinhardtii mutants with impaired Photosystem II photochemical activity. Photosynthesis Research, 94, 79–89.
Mackinney, G. (1941). Absorption of light by chlorophyll solution. Journal of Biological Chemistry, 140, 315–322.
Bligh, E. G., & Dyer, W. J. (1959). A rapid method of total lipid extraction and purification. Canadian Journal of Biochemistry and Physiology, 37, 911–917.
Davidi, L., Katz, A., & Pick, U. (2012). Characterization of major lipid droplet proteins from Dunaliella. Planta, 236, 19–33.
Parida, A. K., Dagaonkar, V. S., Phalak, M. S., Umalkar, G. V., & Aurangabadkar, L. P. (2007). Alterations in photosynthetic pigments, protein and osmotic components in cotton genotypes subjected to short-term drought stress followed by recovery. Plant Biotechnology Reports, 1, 37–48.
Converti, A., Casazza, A. A., Ortiz, E. Y., Perego, P., & Borghi, M. D. (2009). Effect of temperature and nitrogen concentration on the growth and lipid content of Nannochloropsis oculata and Chlorella vulgaris for biodiesel production. Chemical Engineering and Processing: Process Intensification, 48, 1146–1151.
Chiu, S. Y., Kao, C. Y., Tsai, M. T., Ong, S. C., Chen, C. H., & Lin, C. S. (2009). Lipid accumulation and CO2 utilization of Nannochloropsis oculata in response to CO2 aeration. Bioresource Technology, 100, 833–838.
Ben-Amotz, A., & Tornabene, T. G. (1985). Chemical profile of selected species of microalgae with emphasis on lipids. Journal of Phycology, 21, 72–81.
Zhekisheva, M., Boussiba, S., Khozin-Goldberg, U., Zarka, A., & Cohen, Z. (2002). Accumulation of oleic acid in Heamatococcus pluvialis (Chlorophyceae) under nitrogen starvation or high light is correlated with that of astaxanthin esters. Journal of Phycology, 38, 325–331.
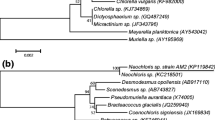
Msanne, J., Xu, D., Konda, A. R., Casas-Mollano, J. A., Awada, T., Cahoon, E. B., & Cerutti, H. (2012). Metabolic and gene expression changes triggered by nitrogen deprivation in the photoautotrophically grown microalgae Chlamydomonas reinhardtii and Coccomyxa sp. C-169. Phytochemistry, 75, 50–59.
Abe, K., Mihara, H., & Hirano, M. (1998). Characteristics of growth and carotenoid accumulation of the aerial microalga Trentepohlia aurea in liquid culture. Journal of Marine Biotechnology, 6, 53–58.
Siaut, M., Cuiné, S., Cagnon, C., Fessler, B., Nguyen, M., Carrier, P., Beyly, A., Beisson, F., Triantaphylidès, C., Li-Beisson, Y., & Peltier, G. (2011). Oil accumulation in the model green alga Chlamydomonas reinhardtii: characterization, variability between common laboratory strains and relationship with starch reserves. BMC Biotechnology, 11, 7.
European Standard EN 14214. (2004). Automotive fuels-fatty acid methyl esters (FAME) for diesel engines-requirements and test methods.
Guzmán, H. M., de la JaraValido, A., Duarte, L. C., & Presmanes, K. F. (2011). Analysis of interspecific variation in relative fatty acid composition: use of flow cytometry to estimate unsaturation index and relative polyunsaturated fatty acid content in microalgae. Journal of Applied Phycology, 23, 7–15.
Ratledge, C. (2004). Fatty acid biosynthesis in microorganisms being used for single cell oil production. Biochimie, 86, 807–815.
Guarnieri, M. T., Nag, A., Smolinski, S. L., Darzins, A., Seibert, M., & Pienkos, P. T. (2011). Examination of triacylglycerol biosynthetic pathways via de novo transcriptomic and proteomic analyses in an unsequenced microalga. PloS One, 6, 1–11.
Hu, C. W., Chuang, L. T., Yu, P. C., & Chen, C. N. N. (2013). Pigment production by a new thermotolerant microalga Coelastrella sp. F50. Food Chemistry, 138, 2071–2078.
Acknowledgments
The authors would like to thank Prof. Hideaki Miyashita and Dr Satoshi Ohkubo (Department of Interdisciplinary Environment, Graduate School of Human and Environmental Studies, Kyoto University) for their scientific support in identification of the isolated strain. This study was supported in part by a grant from the Strategic Research Foundation Grant-aided Project for Private Universities from the Ministry of Education, Culture, Sport, Science, and Technology, Japan (MEXT), 2014–2018 (S1411005).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abe, K., Ishiwatari, T., Wakamatsu, M. et al. Fatty Acid Content and Profile of the Aerial Microalga Coccomyxa sp. Isolated from Dry Environments. Appl Biochem Biotechnol 174, 1724–1735 (2014). https://doi.org/10.1007/s12010-014-1181-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-1181-y