Abstract
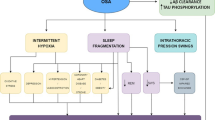
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder. The human brain is extremely sensitive to hypoxia, ischemia, and glucose depletion. Impaired delivery of oxygen in obstructive sleep apnea (OSA) alters neuronal homeostasis, induces pathology, and triggers neuronal degeneration/death. This article systematically delineates the steps in the complex cascade leading to AD, focusing on pathology caused by chronic intermittent hypoxia, hypertension, brain hypoperfusion, glucose dysmetabolism, and endothelial dysfunction. Hypoxia/hypoxemia underpins several pathological processes including sympathetic activation, chemoreflex activity, neuroinflammation, oxidative stress, and a host of perturbations leading to neurodegeneration. The arterial blood flow reduction in OSA is profound, being about 76 % in obstructive hypopneas and 80 % in obstructive apneas; this leads to cerebral ischemia promoting neuronal apoptosis in neocortex and brainstem. OSA pathology also includes gray matter loss in the frontal, parietal, temporal, and occipital cortices, the thalamus, hippocampus, and key brainstem nuclei including the nucleus tractus solitarius. (18)F-FDG PET studies on OSA and AD patients, and animal models of AD, have shown reduced cerebral glucose metabolism in the above mentioned brain regions. Owing to the pathological impact of hypoxia, hypertension, hypoperfusion and impaired glucose metabolism, the adverse cardiovascular, neurocirculatory and metabolic consequences upregulate amyloid beta generation and tau phosphorylation, and lead to memory/cognitive impairment—culminating in AD. The framework encompassing these factors provides a pragmatic neuropathological approach to explain onset of Alzheimer’s dementia. The basic tenets of the current paradigm should influence the design of therapeutic strategies to ameliorate AD.

Similar content being viewed by others
Abbreviations
- AGEs:
-
Advanced glucose endproducts
- AD:
-
Alzheimer’s disease
- Aβ:
-
Amyloid-β
- APP:
-
Amyloid precursor protein
- APAP:
-
Autotitrating positive airway pressure
- AHI:
-
Apnea-hypopnea index
- BACE:
-
β-site amyloid precursor protein cleavage enzyme
- BFB:
-
Basal forebrain
- BOLD:
-
Blood-oxygenation-level-dependent
- BP:
-
Blood pressure
- CAA:
-
Amyloid angiopathy
- CBFV:
-
Cerebral artery blood flow velocity
- CIC:
-
Circulating inflammatory cytokines
- CIH:
-
Chronic intermittent hypoxia
- CMRO2 :
-
Cerebral metabolic rate of oxygen
- COX-2:
-
Cycloxygenase-2
- CRP:
-
C-reactive protein
- CPAP:
-
Continuous positive airway pressure
- DM:
-
Type 2 diabetes mellitus
- DMV:
-
Dorsal motor nucleus V cranial nerve
- DMNV:
-
Dorsal motor nucleus of the vagus
- EDS:
-
Excessive daytime sleepiness
- ERC:
-
Entorhinal cortex
- GMV:
-
Gray matter volume
- HEP:
-
High energy phosphates
- HIF:
-
Hypoxia-inducible factor
- ICAM-1 and -2:
-
Intercellular adhesion molecule-1 and -2
- IGF-1R:
-
IGF-1 receptor
- IH:
-
Intermittent hypoxia
- IR:
-
Insulin receptor
- IL-1β:
-
Interleukin-1β
- LDL:
-
Low-density lipoprotein
- MMP:
-
Matrix metalloproteinase 2
- MTL:
-
Medial temporal lobe
- NMDARs:
-
N-methyl-D-aspartate receptors
- MCAV:
-
Middle cerebral artery velocity
- MCI:
-
Mild cognitive impairment
- NA:
-
Nucleus ambiguus
- NIRS:
-
Near-infrared spectroscopy
- NFTs:
-
Neurofibrillary tangles
- NO:
-
Nitric oxide
- NREM:
-
Non-rapid eye movement sleep
- NF-κB:
-
Nuclear factor kappa B
- NTS:
-
Nucleus of the solitary tract
- OSA:
-
Obstructive sleep apnea
- PCC:
-
Posterior cingulate cortex
- PECAM:
-
Platelet endothelial cell adhesion molecule-1
- PFC:
-
Prefrontal cortex
- RAGE:
-
AGE receptor
- RAS:
-
Renin-angiotensin system
- ROS:
-
Reactive oxygen species
- SAA:
-
Serum amyloid A
- SDB:
-
Sleep disordered breathing
- SHR:
-
Hypertensive rat
- SPs:
-
Amyloid senile plaques
- SPECT:
-
Single photon emission computed tomography
- TNF-α:
-
Tumor necrosis factor-α
- UA:
-
Upper airway
- VBM:
-
Voxel-based morphometry
- VEGF:
-
Vascular endothelial growth factor
References
Seubert P, Oltersdorf T, Lee MG, Barbour R, Blomquist C, Davis DL, Bryant K, Fritz LC, Galasko D, Thai LJ (1993) Secretion of β-amyloid precursor protein cleaved at the amino terminus of the β-amyloid peptide. Nature 361:260–263
Selkoe DJ (2011) Resolving controversies on the path to Alzheimer’s therapeutics. Nat Med 17:1060–1065
Daulatzai MA (2009) The pharyngeal landscape: its length and breadth. J Sleep Res 18:483–484
Daulatzai MA (2011) Role of Sensory Stimulation in Amelioration of Obstructive Sleep Apnea. Sleep Disorders 2011 (2011), Article ID 596879, 12 pp. doi:10.1155/2011/596879
Young T, Palta M, Dempsey J, Skatrud J, Weber S, Badr S (1993) The occurrence of sleep-disordered breathing among middle-aged adults. N Engl J Med 328:1230–1235
Daulatzai MA (2010) Early stages of pathogenesis in memory impairment during normal senescence and Alzheimer’s disease. J Alzheimers Dis 20:355–367
Ayappa I, Rapaport BS, Norman RG, Rapoport DM (2005) Immediate consequences of respiratory events in sleep disordered breathing. Sleep Med 6:123–130
Ronksley PE, Hemmelgarn BR, Heitman SJ, Hanly PJ, Faris PD, Quan H, Tsai WH (2009) Obstructive sleep apnoea is associated with diabetes in sleepy subjects. Thorax 64:834–839
Butt M, Dwivedi G, Khair O, Lip GY (2010) Obstructive sleep apnea and cardiovascular disease. Int J Cardiol 139:7–16
Bosc LV, Resta T, Walker B, Kanagy NL (2010) Mechanisms of intermittent hypoxia induced hypertension. J Cell Mol Med 14:3–17
Carnevale D, Mascio G, Ajmone-Cat MA, D’Andrea I, Cifelli G, Madonna M, Cocozza G, Frati A, Carullo P, Carnevale L, Alleva E, Branchi I, Lembo G, Minghetti L (2012) Role of neuroinflammation in hypertension-induced brain amyloid pathology. Neurobiol Aging 33:205.e19–205.e29
Janssens JP, Pautex S, Hilleret H, Michel JP (2000) Sleep disordered breathing in the elderly. Aging (Milano) 124:17–29
Bombois S, Derambure P, Pasquier F, Monaca C (2010) Sleep disorders in aging and dementia. J Nutr Health Aging 14:212–217
Drager LF, Bortolotto LA, Lorenzi MC, Figueiredo AC, Krieger EM, Lorenzi-Filho G (2005) Early signs of atherosclerosis in obstructive sleep apnea. Am J Respir Crit Care Med 172:613–618
Pallayova M, Steele KE, Magnuson TH, Schweitzer MA, Hill NR, Bevans-Fonti S, Schwartz AR (2010) Sleep apnea predicts distinct alterations in glucose homeostasis and biomarkers in obese adults with normal and impaired glucose metabolism. Cardiovasc Diabetol 9:83
Dempsey JA, Veasey SC, Morgan BJ, O’Donnell CP (2010) Pathophysiology of sleep apnea. Physiol Rev 90:47–112
Daulatzai MA (2012) Dysfunctional nucleus tractus solitarius: its crucial role in promoting neuropathogentic cascade of Alzheimer’s dementia-a novel hypothesis. Neurochem Res 37:846–868
Daulatzai MA (2012) Memory and cognitive dysfunctions in Alzheimer’s disease are inextricably intertwined with neuroinflammation due to aging, obesity, obstructive sleep apnea, and other upstream risk factors. In: Costa A, Eugenio Villalba E (eds) Memory impairment: causes, management and risk factors, horizons in neuroscience research, vol 8. Nova Science, Hauppauge
Daulatzai MA (2012) Pathogenesis of cognitive dysfunction in patients with obstructive sleep apnea: a hypothesis with emphasis on the nucleus tractus solitarius. Sleep Disorders 2012, Article ID 251096, 18 pp. doi:10.1155/2012/251096
Row BW (2007) Intermittent hypoxia and cognitive function: implications from chronic animal models. Adv Exp Med Biol 618:51–67
Skoog I, Lithell H, Hansson L, Elmfeldt D, Hofman A, Olofsson B, Trenkwalder P, Zanchetti A, SCOPE Study Group (2005) Effect of baseline cognitive function and antihypertensive treatment on cognitive and cardiovascular outcomes: study on cognition and prognosis in the elderly (SCOPE). Am J Hypertens 18:1052–1059
Foster GE, Brugniaux JV, Pialoux V, Duggan CT, Hanly PJ, Ahmed SB, Poulin MJ (2009) Cardiovascular and cerebrovascular responses to acute hypoxia following exposure to intermittent hypoxia in healthy humans. J Physiol 587:3287–3299
Chen TI, Lai CJ, Hsieh CJ, Tsai KL, Yang KT (2011) Differences in left ventricular cardiomyocyte loss induced by chronic intermittent hypoxia between spontaneously hypertensive and Wistar-Kyoto rats. Sleep Breath 15:845–854
Levy P, Tamisier R, Arnaud C, Monneret D, Baguet JP, Stanke-Labesque F, Dematteis M, Godin-Ribuot D, Ribuot C, Pepin JL (2012) Sleep deprivation, sleep apnea and cardiovascular diseases. Front Biosci 4:2007–2021
Peppard PE, Young T, Palta M, Skatrud J (2000) Prospective study of the association between sleep-disordered breathing and hypertension. N Engl J Med 342:1378–1384
Shahar E, Whitney CW, Redline S, Lee ET, Newman AB, Javier NF, O’Connor GT, Boland LL, Schwartz JE, Samet JM (2001) Sleep-disordered breathing and cardiovascular disease: cross-sectional results of the Sleep Heart Health Study. Am J Respir Crit Care Med 163:19–25
McArdle N, Hillman D, Beilin L, Watts G (2007) Metabolic risk factors for vascular disease in obstructive sleep apnea: a matched controlled study. Am J Respir Crit Care Med 175:190–195
Shiina K, Tomiyama H, Takata Y, Yoshida M, Kato K, Saruhara H, Hashimura Y, Matsumoto C, Asano K, Usui Y, Yamashina A (2010) Effects of CPAP therapy on the sympathovagal balance and arterial stiffness in obstructive sleep apnea. Respir Med 104:911–916
Ziegler MG, Mills PJ, Loredo JS, Ancoli-Israel S, Dimsdale JE (2001) Effect of continuus positive airway pressure and placebo treatment on sympathetic nervous activity in patients with obstructive sleep apnea. Chest 120:887–893
Kohler M, Pepperell JC, Casadei B, Craig S, Crosthwaite N, Stradling JR, Davies RJ (2008) CPAP and measures of cardiovascular risk in males with OSAS. Eur Respir J 32:1488–1496
Jaimchariyatam N, Rodriguez CL, Budur K (2010) Does CPAP treatment in mild obstructive sleep apnea affect blood pressure? Sleep Med 11:837–842
Marrone O, Salvaggio A, Bue AL, Bonanno A, Riccobono L, Insalaco G, Bonsignore MR (2011) Blood pressure changes after automatic and fixed CPAP in obstructive sleep apnea: relationship with nocturnal sympathetic activity. Clin Exp Hypertens 33:373–380
Phillips CL, Yang Q, Williams A, Roth M, Yee BJ, Hedner JA, Berend N, Grunstein RR (2007) The effect of short-term withdrawal from continuous positive airway pressure therapy on sympathetic activity and markers of vascular inflammation in subjects with obstructive sleep apnoea. J Sleep Res 16:217–225
Foster GE, Hanly PJ, Ahmed SB, Beaudin AE, Pialoux V, Poulin MJ (2010) Intermittent hypoxia increases arterial blood pressure in humans through a Renin-Angiotensin system-dependent mechanism. Hypertension 56:369–377
Phillips BG, Narkiewicz K, Pesek CA, Haynes WG, Dyken ME, Somers VK (1999) Effects of obstructive sleep apnea on endothelin-1 and blood pressure. J Hypertens 17:61–66
Rey S, Del Rio R, Iturriaga R (2006) Contribution of endothelin-1 to the enhanced carotid body chemosensory responses induced by chronic intermittent hypoxia. Brain Res 108:6152–6159
Drager LF, Bortolotto LA, Figueiredo AC, Krieger EM, Lorenzi-Filho G (2007) Effects of continuous positive airway pressure on early signs of atherosclerosis in obstructive sleep apnea. Am J Respir Crit Care Med 176:706–712
Grote L, Kraiczi H, Hedner J (2000) Reduced α- and β(2)-adrenergic vascular response in patients with obstructive sleep apnea. Am J Respir Crit Care Med 162:1480–1487
Morgan BJ (2007) Vascular consequences of intermittent hypoxia. Adv Exp Med Biol 618:69–84
Ziegler MG, Milic M, Elayan H (2011) Cardiovascular regulation in obstructive sleep apnea. Drug Discov Today Dis Models 8:155–160
Talman WT, Perrone MH, Reis DJ (1981) Acute hypertension after the local injection of kainic acid into the nucleus tractus solitarii of rats. Circ Res 48:292–298
Machado BH (2001) Neurotransmission of the cardiovascular reflexes in the nucleus tractus solitarii of awake rats. Ann N Y Acad Sci 940:179–196
Boscan P, Allen AM, Paton JF (2001) Baroreflex inhibition of cardiac sympathetic outflow is attenuated by angiotensin II in the nucleus of the solitary tract. Neuroscience 103:153–160
Biaggioni I, Whetsell WO, Jobe J, Nadeau JH (1994) Baroreflex failure in a patient with central nervous system lesions involving the nucleus tractus solitarii. Hypertension 23:491–495
Cooper VL, Pearson SB, Bowker CM, Elliott MW, Hainsworth R (2005) Interaction of chemoreceptor and baroreceptor reflexes by hypoxia and hypercapnia—a mechanism for promoting hypertension in obstructive sleep apnoea. J Physiol 568:677–687
Urbano F, Roux F, Schindler J, Mohsenin V (2008) Impaired cerebral autoregulation in obstructive sleep apnea. J Appl Physiol 105:1852–1857
Culebras A (2007) Cerebrovascular disease and the pathophysiology of obstructive sleep apnea. Curr Neurol Neurosci Rep 7:173–179
Nishibayashi M, Miyamoto M, Miyamoto T, Suzuki K, Hirata K (2008) Correlation between severity of obstructive sleep apnea and prevalence of silent cerebrovascular lesions. J Clin Sleep Med 4:242–247
Brickman AM, Reitz C, Luchsinger JA, Manly JJ, Schupf N, Muraskin J, DeCarli C, Brown TR, Mayeux R (2010) Long-term blood pressure fluctuation and cerebrovascular disease in an elderly cohort. Arch Neurol 67:564–569
Somers VK, Dyken ME, Clary MP, Abboud FM (1995) Sympathetic neural mechanisms in obstructive sleep apnea. J Clin Invest 96:1897–1904
Narkiewicz K, van de Borne PJ, Montano N, Dyken ME, Phillips BG, Somers VK (1998) Contribution of tonic chemoreflex activation to sympathetic activity and blood pressure in patients with obstructive sleep apnea. Circulation 97:943–945
Dyken ME, Yamada T, Glenn CL, Berger HA (2004) Obstructive sleep apnea associated with cerebral hypoxemia and death. Neurology 62:491–493
Khoo SM, Mukherjee JJ, Phua J, Shi DX (2009) Obstructive sleep apnea presenting as recurrent cardiopulmonary arrest. Sleep Breath 13:89–92
Hayakawa T, Terashima M, Kayukawa Y, Ohta T, Okada T (1996) Changes in cerebral oxygenation and hemodynamics during obstructive sleep apneas. Chest 109:916–921
Rosenberger P, Schwab JM, Mirakaj V, Masekowsky E, Mager A, Morote-Garcia JC, Unertl K, Eltzschig HK (2009) Hypoxia-inducible factor-dependent induction of netrin-1 dampens inflammation caused by hypoxia. Nat Immunol 10:195–202
Eltzschig HK, Abdulla P, Hoffman E, Hamilton KE, Daniels D, Schönfeld C, Löffler M, Reyes G, Duszenko M, Karhausen J, Robinson A, Westerman KA, Coe IR, Colgan SP (2005) HIF-1-dependent repression of equilibrative nucleoside transporter (ENT) in hypoxia. J Exp Med 202:1493–1505
Eltzschig HK, Carmeliet P (2011) Hypoxia and Inflammation. New Engl J Med 364:656–665
Rae C, Bartlett DJ, Yang Q, Walton D, Denotti A, Sachinwalla T, Grunstein R (2009) Dynamic changes in brain bioenergetics during obstructive sleep apnea. J Cereb Blood Flow Metab 29:1421–1428
Kawano Y, Tamura A, Kadota J (2012) Association between the severity of obstructive sleep apnea and the ratio of low-density lipoprotein cholesterol to high-density lipoprotein cholesterol. Metabolism 61:186–192
Jun J, Reinke C, Bedja D, Berkowitz D, Bevans-Fonti S, Li J, Barouch LA, Gabrielson K, Polotsky VY (2010) Effect of intermittent hypoxia on atherosclerosis in apolipoprotein E-deficient mice. Atherosclerosis 209:381–386
Savransky V, Jun J, Li J, Nanayakkara A, Fonti S, Moser AB, Steele KE, Schweitzer MA, Patil SP, Bhanot S, Schwartz AR, Polotsky VY (2008) Dyslipidemia and atherosclerosis induced by chronic intermittent hypoxia are attenuated by deficiency of stearoyl coenzyme A desaturase. Circ Res 103:1173–1180
Drager LF, Polotsky VY, Lorenzi-Filho G (2011) Obstructive sleep apnea: an emerging risk factor for atherosclerosis. Chest 140:534–542
Vgontzas AN, Papanicolaou DA, Bixler EO, Kales A, Tyson K, Chrousos GP (1997) Elevation of plasma cytokines in disorders of excessive daytime sleepiness: role of sleep disturbance and obesity. J Clin Endocrinol Metab 82:1313–1316
Dyugovskaya L, Lavie P, Lavie L (2002) Increased adhesion molecules expression and production of reactive oxygen species in leukocytes of sleep apnea patients. Am J Respir Crit Care Med 165:934–939
Shamsuzzaman AS, Winnicki M, Lanfranchi P, Wolk R, Kara T, Accurso V, Somers VK (2002) Elevated C-reactive protein in patients with obstructive sleep apnea. Circulation 105:2462–2464
Ohga E, Nagase T, Tomita T, Teramoto S, Matsuse T, Katayama H, Ouchi Y (1999) Increased levels of circulating ICAM-1, VCAM-1, and L-selectin in obstructive sleep apnea syndrome. J Appl Physiol 87:10–14
Ohga E, Tomita T, Wada H, Yamamoto H, Nagase T, Ouchi Y (2003) Effects of obstructive sleep apnea on circulating ICAM-1, IL-8, and MCP-1. J Appl Physiol 94:179–184
El-Solh AA, Mador MJ, Sikka P, Dhillon RS, Amsterdam D, Grant BJ (2002) Adhesion molecules in patients with coronary artery disease and moderate-to-severe obstructive sleep apnea. Chest 121:1541–1547
Jelic S, Le Jemtel TH (2008) Inflammation, oxidative stress, and the vascular endothelium in obstructive sleep apnea. Trends Cardiovasc Med 18:253–260
Jelic S, Lederer DJ, Adams T, Padeletti M, Colombo PC, Factor PH, Le Jemtel TH (2010) Vascular inflammation in obesity and sleep apnea. Circulation 121:1014–1021
Lurie A (2011) Endothelial dysfunction in adults with obstructive sleep apnea. Adv Cardiol 46:139–170
Kohler M, Stradling JR (2010) Mechanisms of vascular damage in obstructive sleep apnea. Nat Rev Cardiol 7:677–685
Firat Guven S, Turkkani MH, Ciftci B, Ulukavak Ciftci T, Erdogan Y (2012) The relationship between high-sensitivity C-reactive protein levels and the severity of obstructive sleep apnea. Sleep Breath 16:217–221
Mausbach BT, Ancoli-Israel S, von Känel R, Patterson TL, Aschbacher K, Mills PJ, Ziegler MG, Dimsdale JE, Calleran S, Grant I (2006) Sleep disturbance, norepinephrine, and D-dimer are all related in elderly caregivers of people with Alzheimer disease. Sleep 29:1347–1352
von Känel R, Dimsdale JE, Ancoli-Israel S, Mills PJ, Patterson TL, McKibbin CL, Archuleta C, Grant I (2006) Poor sleep is associated with higher plasma proinflammatory cytokine interleukin-6 and procoagulant marker fibrin D-dimer in older caregivers of people with Alzheimer’s disease. J Am Geriatr Soc 54:431–437
von Känel R, Loredo JS, Ancoli-Israel S, Mills PJ, Natarajan L, Dimsdale JE (2007) Association between polysomnographic measures of disrupted sleep and prothrombotic factors. Chest 131:733–739
Venugopal SK, Devaraj S, Yuhanna I, Shaul P, Jialal I (2002) Demonstration that C-reactive protein decreases eNOS expression and bioactivity in human aortic endothelial cells. Circulation 106:1439–1441
Woollard KJ, Phillips DC, Griffiths HR (2002) Direct modulatory effect of C-reactive protein on primary human monocyte adhesion to human endothelial cells. Clin Exp Immunol 130:256–262
Giunta B, Fernandez F, Nikolic WV, Obregon D, Rrapo E, Town T, Tan J (2008) Inflammaging as a prodrome to Alzheimer’s disease. J Neuroinflammation 5:51
Büchner NJ, Quack I, Woznowski M, Stähle C, Wenzel U, Rump LC (2011) Microvascular endothelial dysfunction in obstructive sleep apnea is caused by oxidative stress and improved by continuous positive airway pressure therapy. Respiration 82:409–417
Ramar K, Caples SM (2011) Vascular changes, cardiovascular disease and obstructive sleep apnea. Future Cardiol 7:241–249
Dilger RN, Johnson RW (2008) Aging, microglial cell priming, and the discordant central inflammatory response to signals from the peripheral immune system. J Leukoc Biol 84:932–939
Henry CJ, Huang Y, Wynne AM, Godbout JP (2009) Peripheral lipopolysaccharide (LPS) challenge promotes microglial hyperactivity in aged mice that is associated with exaggerated induction of both pro-inflammatory IL-1beta and anti-inflammatory IL-10 cytokines. Brain Behav Immun 23:309–317
Holmes C, Cunningham C, Zotova E, Woolford J, Dean C, Kerr S, Culliford D, Perry VH (2009) Systemic inflammation and disease progression in Alzheimer disease. Neurology 73:768–774
Oga T, Chin K, Tabuchi A, Kawato M, Morimoto T, Takahashi K, Handa T, Takahashi K, Taniguchi R, Kondo H, Mishima M, Kita T, Horiuchi H (2009) Effects of obstructive sleep apnea with intermittent hypoxia on platelet aggregability. J Atheroscler Thromb 16:862–869
Rahangdale S, Yeh SY, Novack V, Stevenson K, Barnard MR, Furman MI, Frelinger AL, Michelson AD, Malhotra A (2011) The influence of intermittent hypoxemia on platelet activation in obese patients with obstructive sleep apnea. J Clin Sleep Med 7:172–178
Nurden AT (2011) Platelets, inflammation and tissue regeneration. Thromb Haemost 105(Suppl 1):S13–S33
Casoli T, Di Stefano G, Balietti M, Solazzi M, Giorgetti B, Fattoretti P (2010) Peripheral inflammatory biomarkers of Alzheimer’s disease: the role of platelets. Biogerontology 11:627–633
Casoli T, Di Stefano G, Giorgetti B, Grossi Y, Balietti M, Fattoretti P, Bertoni-Freddari C (2007) Release of beta-amyloid from high-density platelets: implications for Alzheimer’s disease pathology. Ann N Y Acad Sci 1096:170–178
Ip MS, Tse HF, Lam B, Tsang KW, Lam WK (2004) Endothelial function in obstructive sleep apnea and response to treatment. Am J Respir Crit Care Med 169:348–353
Patt BT, Jarjoura D, Haddad DN, Sen CK, Roy S, Flavahan NA, Khayat RN (2010) Endothelial dysfunction in the microcirculation of patients with obstructive sleep apnea. Am J Respir Crit Care Med 182:1540–1545
Lavie L, Vishnevsky A, Lavie P (2004) Evidence for lipid peroxidation in obstructive sleep apnea. Sleep 27:123–128
Khayat R, Patt B, Hayes D Jr (2009) Obstructive sleep apnea: the new cardiovascular disease. Part I: obstructive sleep apnea and the pathogenesis of vascular disease. Heart Fail Rev 14:143–153
Förstermann U (2010) Nitric oxide and oxidative stress in vascular disease. Pflugers Arch 459:923–939
Li J, Savransky V, Nanayakkara A, Smith PL, O’Donnell CP, Polotsky VY (2007) Hyperlipidemia and lipid peroxidation are dependent on the severity of chronic intermittent hypoxia. J Appl Physiol 102:557–563
Kamba M, Inoue Y, Higami S, Suto Y, Ogawa T, Chen W (2001) Cerebral metabolic impairment in patients with obstructive sleep apnoea: an independent association of obstructive sleep apnoea with white matter change. J Neurol Neurosurg Psychiatry 71:334–339
Raji CA, Lopez OL, Kuller LH, Carmichael OT, Longstreth WT Jr, Gach HM, Boardman J, Bernick CB, Thompson PM, Becker JT (2012) White matter lesions and brain gray matter volume in cognitively normal elders. Neurobiol Aging 33:834.e7–834.e16
Macey PM, Kumar R, Woo MA, Valladares EM, Yan-Go FL, Harper RM (2008) Brain structural changes in obstructive sleep apnea. Sleep 31:967–977
Saunamäki T, Himanen SL, Polo O, Jehkonen M (2009) Executive dysfunction in patients with obstructive sleep apnea syndrome. Eur Neurol 62:237–242
Antonelli Incalzi R, Marra C, Salvigni BL, Petrone A, Gemma A, Selvaggio D, Mormile F (2004) Does cognitive dysfunction conform to a distinctive pattern in obstructive sleep apnea syndrome? J Sleep Res 13:79–86
Torelli F, Moscufo N, Garreffa G, Placidi F, Romigi A, Zannino S, Bozzali M, Fasano F, Giulietti G, Djonlagic I, Malhotra A, Marciani MG, Guttmann CR (2011) Cognitive profile and brain morphological changes in obstructive sleep apnea. Neuroimage 54:787–793
Ancoli-Israel S, Palmer BW, Cooke JR, Corey-Bloom J, Fiorentino L, Natarajan L, Liu L, Ayalon L, He F, Loredo JS (2008) Cognitive effects of treating obstructive sleep apnea in Alzheimer’s disease: a randomized controlled study. J Am Geriatr Soc 56:2076–2081
Weaver TE, Chasens ER (2007) Continuous positive airway pressure treatment for sleep apnea in older adults. Sleep Med Rev 11:99–111
Canessa N, Castronovo V, Cappa SF, Aloia MS, Marelli S, Falini A, Alemanno F, Ferini-Strambi L (2011) Obstructive sleep apnea: brain structural changes and neurocognitive function before and after treatment. Am J Respir Crit Care Med 183:1419–1426
Lau EY, Eskes GA, Morrison DL, Rajda M, Spurr KF (2010) Executive function in patients with obstructive sleep apnea treated with continuous positive airway pressure. J Int Neuropsychol Soc 16:1077–1088
Andresen MC, Kunze DL (1994) Nucleus tractus solitarius—gateway to neural circulatory control. Annu Rev Physiol 56:93–116
Maier SF, Goehler LE, Fleshner M, Watkins LR (1998) The role of the vagus nerve in cytokine-to-brain communication. Ann NY Acad Sci 840:289–300
Groves DA, Brown VJ (2005) Vagal nerve stimulation: a review of its applications and potential mechanisms that mediate its clinical effects. Neurosci Biobehav Rev 29:493–500
Celle S, Peyron R, Faillenot I, Pichot V, Alabdullah M, Gaspoz JM, Laurent B, Barthélémy JC, Roche F (2009) Undiagnosed sleep-related breathing disorders are associated with focal brainstem atrophy in the elderly. Hum Brain Mapp 30:2090–2097
Daulatzai MA (2010) Conversion of elderly to Alzheimer’s dementia: role of confluence of hypothermia and senescent stigmata—the plausible pathway. J Alzheimers Dis 21:1039–1063
Pascual O, Morin-Surun MP, Barna B, Denavit-Saubié M, Pequignot JM, Champagnat J (2002) Progesterone reverses the neuronal responses to hypoxia in rat nucleus tractus solitarius in vitro. J Physiol 544:511–520
Kline DD, Ramirez-Navarro A, Kunze DL (2007) Adaptive depression in synaptic transmission in the nucleus of the solitary tract after in vivo chronic intermittent hypoxia: evidence for homeostatic plasticity. J Neurosci 27:4663–4673
Zhang JH, Fung SJ, Xi M, Sampogna S, Chase MH (2009) Recurrent apnea induces neuronal apoptosis in the guinea pig forebrain. Exp Neurol 216:290–294
Kaur C, Viswanathan S, Ling EA (2011) Hypoxia-induced cellular and vascular changes in the nucleus tractus solitarius and ventrolateral medulla. J Neuropathol Exp Neurol 70:201–217
Xu W, Chi L, Row BW, Xu R, Ke Y, Xu B, Luo C, Kheirandish L, Gozal D, Liu R (2004) Increased oxidative stress is associated with chronic intermittent hypoxia-mediated brain cortical neuronal cell apoptosis in a mouse model of sleep apnea. Neuroscience 126:313–323
Machaalani R, Waters KA (2003) Increased neuronal cell death after intermittent hypercapnic hypoxia in the developing piglet brainstem. Brain Res 985:127–134
Buller KM, Wixey JA, Pathipati P, Carty M, Colditz PB, Williams CE, Scheepens A (2008) Selective losses of brainstem catecholamine neurons after hypoxia-ischemia in the immature rat pup. Pediatr Res 63:364–369
Zhang JH, Fung SJ, Xi M, Sampogna S, Chase MH (2010) Apnea produces neuronal degeneration in the pons and medulla of guinea pigs. Neurobiol Dis 40:251–264
Stecco C, Porzionato A, Macchi V, Sarasin G, Calcagno A, Parenti A, De Caro R (2005) Detection of apoptosis in human brainstem by TUNEL assay. Ital J Anat Embryol 110:255–260
De Caro R, Parenti A, Montisci M, Guidolin D, Macchi V (2000) Solitary tract nuclei in acute heart failure. Stroke 31:1187–1193
Lièvre V, Becuwe P, Bianchi A, Bossenmeyer-Pourié C, Koziel V, Franck P, Nicolas MB, Dauça M, Vert P, Daval JL (2001) Intracellular generation of free radicals and modifications of detoxifying enzymes in cultured neurons from the developing rat forebrain in response to transient hypoxia. Neuroscience 105:287–297
Tan ZS, Seshadri S (2010) Inflammation in the Alzheimer’s disease cascade: culprit or innocent bystander? Alzheimers Res Ther 2:6
Agostinho P, Cunha RA, Oliveira C (2010) Neuroinflammation, oxidative stress and the pathogenesis of Alzheimer’s disease. Curr Pharm Des 16:2766–2778
Rogers J, Mastroeni D, Leonard B, Joyce J, Grover A (2007) Neuroinflammation in Alzheimer’s disease and Parkinson’s disease: are microglia pathogenic in either disorder? Int Rev Neurobiol 82:235–246
Sirivelu MP, MohanKumar PS, MohanKumar SM (2012) Differential effects of systemic interleukin-1β on gene expression in brainstem noradrenergic nuclei. Life Sci 90:77–81
Moalem G, Tracey DJ (2006) Immune and inflammatory mechanisms in neuropathic pain. Brain Res Rev 51:240–264
Del Rio R, Moya EA, Parga MJ, Madrid C, Iturriaga R (2011) Carotid body inflammation and cardiorespiratory alterations in intermittent hypoxia. Eur Respir J Dec 19. [Epub ahead of print]
Stanimirovic D, Shapiro A, Wong J, Hutchison J, Durkin JP (1997) The induction of ICAM-1 in human cerebromicrovascular endothelial cells (HCEC) by ischemia-like conditions promotes enhanced neutrophil/HCEC adhesion. J Neuroimmunol 76:193–205
Kim KS, Rajagopal V, Gonsalves C, Johnson C, Kalra VK (2006) A novel role of hypoxia-inducible factor in cobalt chloride- and hypoxia-mediated expression of IL-8 chemokine in human endothelial cells. J Immunol 177:7211–7224
Zhang W, Smith C, Howlett C, Stanimirovic D (2000) Inflammatory activation of human brain endothelial cells by hypoxic astrocytes in vitro is mediated by IL-1β. J Cereb Blood Flow Metab 20:967–978
Zhang W, Mojsilovic-Petrovic J, Callaghan D, Jones A, Cui H, Howlett C, Stanimirovic D (2006) Evidence that hypoxia-inducible factor-1 (HIF-1) mediates transcriptional activation of Interleukin-1β (IL-1β) in astrocyte cultures. J Neuroimmunol 174:63–73
Mojsilovic-Petrovic J, Callaghan D, Cui H, Dean C, Stanimirovic DB, Zhang W (2007) Hypoxia-inducible factor-1 (HIF-1) is involved in the regulation of hypoxia-stimulated expression of monocyte chemoattractant protein-1 (MCP-1/CCL2) and MCP-5 (Ccl12) in astrocytes. J Neuroinflammation 4:12
Csiszar A, Wang M, Lakatta EG, Ungvari Z (2008) Inflammation and endothelial dysfunction during aging: role of NF-kappaB. J Appl Physiol 105:1333–1341
Fogal B, Li J, Lobner D, McCullough LD, Hewett SJ (2007) System xI- activity and astrocytes are necessary for interleukin-1 beta-mediated hypoxic neuronal injury. J Neurosci 27:10094–10105
Ng KM, Lau CF, Fung ML (2010) Melatonin reduces hippocampal beta-amyloid generation in rats exposed to chronic intermittent hypoxia. Brain Res 1354:163–171
Waki H, Gouraud SS, Maeda M, Paton JF (2010) Evidence of specific inflammatory condition in nucleus tractus solitarii of spontaneously hypertensive rats. Exp Physiol 95:595–600
Waki H, Bhuiyan ME, Gouraud SS, Takagishi M, Hatada A, Kohsaka A, Paton JF, Maeda M (2011) Acute reductions in blood flow restricted to the dorsomedial medulla induce a pressor response in rats. J Hypertens 29:1536–1545
Colton CA, Mott RT, Sharpe H, Xu Q, Van Nostrand WE, Vitek MP (2006) Expression profiles for macrophage alternative activation genes in AD and in mouse models of AD. J Neuroinflammation 27:27
Rathcke CN, Vestergaard H (2006) YKL-40, a new inflammatory marker with relation to insulin resistance and with a role in endothelial dysfunction and atherosclerosis. Inflamm Res 55:221–227
Paton JF, Waki H (2009) Is neurogenic hypertension related to vascular inflammation of the brainstem? Neurosci Biobehav Rev 33:89–94
Nieto FJ, Young TB, Lind BK, Shahar E, Samet JM, Redline S, D’Agostino RB, Newman AB, Lebowitz MD, Pickering TG (2000) Association of sleep-disordered breathing, sleep apnea, and hypertension in a large community-based study. Sleep heart health study. JAMA 283:1829–1836
Can I, Aytemir K, Demir AU, Deniz A, Ciftci O, Tokgozoglu L, Oto A, Sahin A (2009) P-wave duration and dispersion in patients with obstructive sleep apnea. Int J Cardiol 133:e85–e89
Sari I, Davutoglu V, Ozbala B, Ozer O, Baltaci Y, Yavuz S, Aksoy M (2008) Acute sleep deprivation is associated with increased electrocardiographic p-wave dispersion in healthy young men and women. Pacing Clin Electrophysiol 31:438–442
Hung J, Whitford EG, Parsons RW, Hillman DR (1990) Association of sleep apnoea with myocardial infarction in men. Lancet 336:261–264
Peled N, Abinader EG, Pillar G, Sharif D, Lavie P (1999) Nocturnal ischemic events in patients with obstructive sleep apnea syndrome and ischemic heart disease: effects of continuous positive air pressure treatment. J Am Coll Cardiol 34:1744–1749
Peker Y, Hedner J, Kraiczi H, Löth S (2000) Respiratory disturbance index: an independent predictor of mortality in coronary artery disease. Am J Respir Crit Care Med 162:81–86
Vgontzas AN, Zoumakis M, Papanicolaou DA, Bixler EO, Prolo P, Lin HM, Vela-Bueno A, Kales A, Chrousos GP (2002) Chronic insomnia is associated with a shift of interleukin-6 and tumor necrosis factor secretion from nighttime to daytime. Metabolism 51:887–892
Hanly P, Sasson Z, Zuberi N, Lunn K (1993) ST-segment depression during sleep in obstructive sleep apnea. Am J Cardiol 71:1341–1345
Franklin KA, Nilsson JB, Sahlin C, Naslund U (1995) Sleep apnoea and nocturnal angina. Lancet 345:1085–1087
Philip P, Guilleminault C (1993) ST segment abnormality, angina during sleep and obstructive sleep apnea. Sleep 16:558–559
Lau DH, Mackenzie L, Kelly DJ, Psaltis PJ, Worthington M, Rajendram A, Kelly DR, Nelson AJ, Zhang Y, Kuklik P, Brooks AG, Worthley SG, Faull RJ, Rao M, Edwards J, Saint DA, Sanders P (2010) Short-term hypertension is associated with the development of atrial fibrillation substrate: a study in an ovine hypertensive model. Heart Rhythm 7:396–404
Lisman KA, Stetson SJ, Koerner MM, Farmer JA, Torre-Amione G (2002) The role of inflammation in the pathogenesis of heart failure. Curr Cardiol Rep 4:200–205
Schulz R, Schmidt D, Blum A, Lopes-Ribeiro X, Lücke C, Mayer K, Olschewski H, Seeger W, Grimminger F (2000) Decreased plasma levels of nitric oxide derivatives in obstructive sleep apnoea: response to CPAP therapy. Thorax 55:1046–1051
Kato M, Roberts-Thomson P, Phillips BG, Haynes WG, Winnicki M, Accurso V, Somers VK (2000) Impairment of endothelium-dependent vasodilation of resistance vessels in patients with obstructive sleep apnea. Circulation 102:2607–2610
Poredos P, Orehek M, Tratnik E (1999) Smoking is associated with dose-related increase of intima-media thickness and endothelial dysfunction. Angiology 50:201–208
Lee IK, Kim HS, Bae JH (2002) Endothelial dysfunction: its relationship with acute hyperglycaemia and hyperlipidemia. Int J Clin Pract Suppl 129:59–64
Rizzoni D, Porteri E, Guelfi D, Muiesan ML, Piccoli A, Valentini U, Cimino A, Girelli A, Salvetti M, De Ciuceis C, Tiberio GA, Giulini SM, Sleiman I, Monteduro C, Rosei EA (2001) Endothelial dysfunction in small resistance arteries of patients with non-insulin-dependent diabetes mellitus. J Hypertens 19:913–919
Yim-Yeh S, Rahangdale S, Nguyen AT, Jordan AS, Novack V, Veves A, Malhotra A (2010) Obstructive sleep apnea and aging effects on macrovascular and microcirculatory function. Sleep 33:1177–1183
Yim-Yeh S, Rahangdale S, Nguyen AT, Stevenson KE, Novack V, Veves A, Malhotra A (2011) Vascular dysfunction in obstructive sleep apnea and type 2 diabetes mellitus. Obesity (Silver Spring) 19:17–22
Buchner NJ, Quack I, Stegbauer J, Woznowski M, Kaufmann A, Rump LC (2012) Treatment of obstructive sleep apnea reduces arterial stiffness. Sleep Breath 16:123–133
Henning EC, Warach S, Spatz M (2010) Hypertension-induced vascular remodeling contributes to reduced cerebral perfusion and the development of spontaneous stroke in aged SHRSP rats. J Cereb Blood Flow Metab 30:827–836
Johansson BB (1984) Cerebral vascular bed in hypertension and consequences for the brain. Hypertension 6:III81–III86
Bålfors EM, Franklin KA (1994) Impairment of cerebral perfusion during obstructive sleep apneas. Am J Respir Crit Care Med 150:1587–1591
Farkas E, De Vos RA, Jansen Steur EN, Luiten PG (2000) Are Alzheimer’s disease, hypertension, and cerebrocapillary damage related? Neurobiol Aging 21:235–243
Israeli-Korn SD, Masarwa M, Schechtman E, Abuful A, Strugatsky R, Avni S, Farrer LA, Friedland RP, Inzelberg R (2010) Hypertension increases the probability of Alzheimer’s disease and of mild cognitive impairment in an Arab community in northern Israel. Neuroepidemiology 34:99–105
Yang YH, Roe CM, Morris JC (2011) Relationship between late-life hypertension, blood pressure, and Alzheimer’s disease. Am J Alzheimers Dis Other Demen 26:457–462
Corfield DR, Meadows GE (2006) Control of cerebral blood flow during sleep and the effects of hypoxia. Adv Exp Med Biol 588:65–73
Olopade CO, Mensah E, Gupta R, Huo D, Picchietti DL, Gratton E, Michalos A (2007) Noninvasive determination of brain tissue oxygenation during sleep in obstructive sleep apnea: a near-infrared spectroscopic approach. Sleep 30:1747–1755
Namtvedt SK, Randby A, Einvik G, Hrubos-Strøm H, Somers VK, Røsjø H, Omland T (2011) Cardiac arrhythmias in obstructive sleep apnea (from the Akershus Sleep Apnea Project). Am J Cardiol 108:1141–1146
Won CH, Chun HJ, Chandra SM, Sarinas PS, Chitkara RK, Heidenreich PA (2012) Severe obstructive sleep apnea increases mortality in patients with ischemic heart disease and myocardial injury. Sleep Breath Feb 1. [Epub ahead of print]
Foster GE, Poulin MC, Hanly PJ (2007) Intermittent hypoxia and vascular function: implications for obstructive sleep apnoea. Exp Physiol 92:51–65
Furtner M, Staudacher M, Frauscher B, Brandauer E, Esnaola y Rojas MM, Gschliesser V, Poewe W, Schmidauer C, Ritsch-Marte M, Högl B (2009) Cerebral vasoreactivity decreases overnight in severe obstructive sleep apnea syndrome: a study of cerebral hemodynamics. Sleep Med 10:875–881
Hsieh SW, Lai CL, Liu CK, Hsieh CF, Hsu CY (2012) Obstructive sleep apnea linked to wake-up strokes. J Neurol Jan 4. [Epub ahead of print]
Cheng Z, Guo SZ, Lipton AJ, Gozal D (2002) Domoic acid lesions in nucleus of the solitary tract: time-dependent recovery of hypoxic ventilatory response and peripheral afferent axonal plasticity. J Neurosci 22:3215–3226
Snowdon DA, Greiner LH, Mortimer JA, Riley KP, Greiner PA, Markesbery WR (1997) Brain infarction and the clinical expression of Alzheimer’s disease: the nun study. JAMA 277:813–817
Jellinger KA, Attems J (2007) Neuropathological evaluation of mixed dementia. J Neurol Sci 257:80–87
Jefferson AL, Himali JJ, Au R, Seshadri S, Decarli C, O’Donnell CJ, Wolf PA, Manning WJ, Beiser AS, Benjamin EJ (2011) Relation of left ventricular ejection fraction to cognitive aging from the Framingham Heart Study. Am J Cardiol 108:1346–1351
Kume K, Hanyu H, Sato T, Hirao K, Shimizu S, Kanetaka H, Sakurai H, Iwamoto T (2011) Vascular risk factors are associated with faster decline of Alzheimer disease: a longitudinal SPECT study. J Neurol 258:1295–1303
van Beek AH, Lagro J, Olde-Rikkert MG, Zhang R, Claassen JA (2012) Oscillations in cerebral blood flow and cortical oxygenation in Alzheimer’s disease. Neurobiol Aging 33:428.e21–428.e31
Okamoto Y, Yamamoto T, Kalaria RN, Senzaki H, Maki T, Hase Y, Kitamura A, Washida K, Yamada M, Ito H, Tomimoto H, Takahashi R, Ihara M (2012) Cerebral hypoperfusion accelerates cerebral amyloid angiopathy and promotes cortical microinfarcts. Acta Neuropathol 123:381–394
Soontornniyomkij V, Lynch MD, Mermash S, Pomakian J, Badkoobehi H, Clare R, Vinters HV (2010) Cerebral microinfarcts associated with severe cerebral beta-amyloid angiopathy. Brain Pathol 20:459–467
de la Torre JC (2000) Critically attained threshold of cerebral hypoperfusion: can it cause Alzheimer’s disease? Ann N Y Acad Sci 903:424–436
Sun X, He G, Qing H, Zhou W, Dobie F, Cai F, Staufenbiel M, Huang LE, Song W (2006) Hypoxia facilitates Alzheimer’s disease pathogenesis by up-regulating BACE1 gene expression. Proc Natl Acad Sci USA 103:18727–18732
Murray IV, Proza JF, Sohrabji F, Lawler JM (2011) Vascular and metabolic dysfunction in Alzheimer’s disease: a review. Exp Biol Med (Maywood) 236:772–782
Kalaria RN, Bhatti SU, Lust WD, Perry G (1993) The amyloid precursor protein in ischemic brain injury and chronic hypoperfusion. Ann N Y Acad Sci 695:190–193
Sadowski M, Pankiewicz J, Scholtzova H, Li YS, Quartermain D, Duff K, Wisniewski T (2004) Links between the pathology of Alzheimer’s disease and vascular dementia. Neurochem Res 29:1257–1266
Wang X, Xing A, Xu C, Cai Q, Liu H, Li L (2010) Cerebrovascular hypoperfusion induces spatial memory impairment, synaptic changes, and amyloid-β oligomerization in rats. J Alzheimers Dis 21:813–822
Sun MK, Alkon DL (2004) Cerebral hypoperfusion and amyloid-induced synergistic impairment of hippocampal CA1 synaptic efficacy and spatial memory in young adult rats. J Alzheimers Dis 6:355–366
Liu H, Xing A, Wang X, Liu G, Li L (2012) Regulation of β-amyloid level in the brain of rats with cerebrovascular hypoperfusion. Neurobiol Aging 33:826.e31-826.e42
Ip MS, Lam B, Ng MM, Lam WK, Tsang KW, Lam KS (2002) Obstructive sleep apnea is independently associated with insulin resistance. Am J Respir Crit Care Med 165:670–676
Spiegel K, Knutson K, Leproult R, Tasali E, Van Cauter E (2005) Sleep loss: a novel risk factor for insulin resistance and Type 2 diabetes. J Appl Physiol 99:2008–2019
Spiegel K, Tasali E, Leproult R, Van Cauter E (2009) Effects of poor and short sleep on glucose metabolism and obesity risk. Nat Rev Endocrinol 5:253–261
Ronksley PE, Hemmelgarn BR, Heitman SJ, Hanly PJ, Faris PD, Quan H, Tsai WH (2009) Obstructive sleep apnoea is associated with diabetes in sleepy subjects. Thorax 64:834–839
Morselli L, Leproult R, Balbo M, Spiegel K (2010) Role of sleep duration in the regulation of glucose metabolism and appetite. Best Pract Res Clin Endocrinol Metab 24:687–702
Nena E, Steiropoulos P, Papanas N, Tsara V, Fitili C, Froudarakis ME, Maltezos E, Bouros D (2012) Sleepiness as a marker of glucose deregulation in obstructive sleep apnea. Sleep Breath 16:181–186
Punjabi NM, Shahar E, Redline S, Gottlieb DJ, Givelber R, Resnick HE (2004) Sleep heart health study investigators. Sleep-disordered breathing, glucose intolerance, and insulin resistance: the sleep heart health study. Am J Epidemiol 160:521–530
Elmasry A, Lindberg E, Berne C, Janson C, Gislason T, Awad Tageldin M, Boman G (2001) Sleep-disordered breathing and glucose metabolism in hypertensive men: a population-based study. J Intern Med 249:153–161
Tamura A, Kawano Y, Watanabe T, Kadota J (2008) Relationship between the severity of obstructive sleep apnea and impaired glucose metabolism in patients with obstructive sleep apnea. Respir Med 102:1412–1416
Pamidi S, Aronsohn RS, Tasali E (2010) Obstructive sleep apnea: role in the risk and severity of diabetes. Best Pract Res Clin Endocrinol Metab 24:703–715
Pandey A, Demede M, Zizi F, Al Haija’a OA, Nwamaghinna F, Jean-Louis G, McFarlane SI (2011) Sleep apnea and diabetes: insights into the emerging epidemic. Curr Diab Rep 11:35–40
Iiyori N, Alonso LC, Li J, Sanders MH, Garcia-Ocana A, O’Doherty RM, Polotsky VY, O’Donnell CP (2007) Intermittent hypoxia causes insulin resistance in lean mice independent of autonomic activity. Am J Respir Crit Care Med 175:851–857
Bertuglia S, Reiter RJ (2009) Melatonin reduces microvascular damage and insulin resistance in hamsters due to chronic intermittent hypoxia. J Pineal Res 46:307–313
Brooks B, Cistulli PA, Borkman M, Ross G, McGhee S, Grunstein RR, Sullivan CE, Yue DK (1994) Obstructive sleep apnea in obese noninsulin-dependent diabetic patients: effect of continuous positive airway pressure treatment on insulin responsiveness. Clin Endocrinol Metab 79:1681–1685
Dawson A, Abel SL, Loving RT, Dailey G, Shadan FF, Cronin JW, Kripke DF, Kline LE (2008) CPAP therapy of obstructive sleep apnea in type 2 diabetics improves glycemic control during sleep. J Clin Sleep Med 4:538–542
Lam JC, Tan KC, Lai AY, Lam DC, Ip MS (2012) Increased serum levels of advanced glycation end-products is associated with severity of sleep disordered breathing but not insulin sensitivity in non-diabetic men with obstructive sleep apnoea. Sleep Med 13:15–20
Peltier AC, Bagai K, Artibee K, Diedrich A, Garland E, Elasy T, Shi Y, Wang L, Feldman EL, Robertson D, Malow BA (2012) Effect of mild hyperglycemia on autonomic function in obstructive sleep apnea. Clin Auton Res 22:1–8
Drager LF, Jun JC, Polotsky VY (2010) Metabolic consequences of intermittent hypoxia: relevance to obstructive sleep apnea. Best Pract Res Clin Endocrinol Metab 24:843–851
Deboer MD, Mendoza JP, Liu L, Ford G, Yu PL, Gaston BM (2011) Increased systemic inflammation overnight correlates with insulin resistance among children evaluated for obstructive sleep apnea. Sleep Breath Mar 1 [Epub ahead of print]
Knutson KL, Van Cauter E (2008) Associations between sleep loss and increased risk of obesity and diabetes. Ann N Y Acad Sci 1129:287–304
Harsch IA, Schahin SP, Brückner K, Radespiel-Tröger M, Fuchs FS, Hahn EG, Konturek PC, Lohmann T, Ficker JH (2004) The effect of continuous positive airway pressure treatment on insulin sensitivity in patients with obstructive sleep apnoea syndrome and type 2 diabetes. Respiration 71:252–259
Hassaballa HA, Tulaimat A, Herdegen JJ, Mokhlesi B (2005) The effect of continuous positive airway pressure on glucose control in diabetic patients with severe obstructive sleep apnea. Sleep Breath 9:176–180
Leenders KL, Perani D, Lammertsma AA, Heather JD, Buckingham P, Healy MJ, Gibbs JM, Wise RJ, Hatazawa J, Herold S (1990) Cerebral blood flow, blood volume and oxygen utilization. Normal values and effect of age. Brain 113:27–47
Bangen KJ, Restom K, Liu TT, Jak AJ, Wierenga CE, Salmon DP, Bondi MW (2009) Differential age effects on cerebral blood flow and BOLD response to encoding: associations with cognition and stroke risk. Neurobiol Aging 30:1276–1287
Takada H, Nagata K, Hirata Y, Satoh Y, Watahiki Y, Sugawara J (1992) Age-related decline of cerebral oxygen metabolism in normal population detected with positron emission tomography. Neurol Res 14:128–131
Hoyer S (1982) The young-adult and normally aged brain. Its blood flow and oxidative metabolism. A review—part I. Arch Gerontol Geriatr 1:101–116
Powers WJ, Videen TO, Markham J, Walter V, Perlmutter JS (2011) Metabolic control of resting hemispheric cerebral blood flow is oxidative, not glycolytic. J Cereb Blood Flow Metab 31:1223–1228
Claus JJ, Breteler MM, Hasan D, Krenning EP, Bots ML, Grobbee DE, Van Swieten JC, Van Harskamp F, Hofman A (1998) Regional cerebral blood flow and cerebrostroke risk factors in the elderly population. Neurobiol Aging 19:57–64
D’Esposito M, Deouell LY, Gazzaley A (2003) Alterations in the BOLD fMRI signal with ageing and disease: a challenge for neuroimaging. Nat Rev Neurosci 4:863–872
Yamaguchi T, Kanno I, Uemura K, Shishido F, Inugami A, Ogawa T, Murakami M, Suzuki K (1986) Reduction in regional cerebral metabolic rate of oxygen during human aging. Stroke 17:1220–1228
Hagstadius S, Risberg J (1989) Regional cerebral blood flow characteristics and variations with age in resting normal subjects. Brain Cogn 10:28–43
Parkes LM, Rashid W, Chard DT, Tofts PS (2004) Normal cerebral perfusion measurements using arterial spin labeling: reproducibility, stability, and age and gender effects. Magn Reson Med 51:736–743
Petit-Taboué MC, Landeau B, Desson JF, Desgranges B, Baron JC (1998) Effects of healthy aging on the regional cerebral metabolic rate of glucose assessed with statistical parametric mapping. Neuroimage 7:176–184
Kadir A, Almkvist O, Forsberg A, Wall A, Engler H, Långström B, Nordberg A (2012) Dynamic changes in PET amyloid and FDG imaging at different stages of Alzheimer’s disease. Neurobiol Aging 33:198.e1–198.e14
Klunk WE, Engler H, Nordberg A, Wang Y, Blomqvist G, Holt DP, Bergström M, Savitcheva I, Huang GF, Estrada S, Ausén B, Debnath ML, Barletta J, Price JC, Sandell J, Lopresti BJ, Wall A (2004) Imaging brain amyloid in Alzheimer’s disease with Pittsburgh Compound-B. Ann Neurol 55:306–319
Langbaum JB, Chen K, Launer LJ, Fleisher AS, Lee W, Liu X, Protas HD, Reeder SA, Bandy D, Yu M, Caselli RJ, Reiman EM (2012) Blood pressure is associated with higher brain amyloid burden and lower glucose metabolism in healthy late middle-age persons. Neurobiol Aging 33:827.e11–827.e19
Zlokovic BV (2011) Neurovascular pathways to neurodegeneration in Alzheimer’s disease and other disorders. Nat Rev Neurosci 12:723–738
Muller M, van der Graaf Y, Visseren FL, Mali WP, Geerlings MI (2012) Hypertension and longitudinal changes in cerebral blood flow: the SMART-MR study. Ann Neurol Feb 3. doi:10.1002/ana.23554. [Epub ahead of print]
Hoyer S (1982) The abnormally aged brain. Its blood flow and oxidative metabolism. A review—part II. Arch Gerontol Geriatr 1:195–207
Thal DR, Griffin WS, de Vos RA, Ghebremedhin E (2008) Cerebral amyloid angiopathy and its relationship to Alzheimer’s disease. Acta Neuropathol 115:599–609
Hunt A, Schönknecht P, Henze M, Seidl U, Haberkorn U, Schröder J (2007) Reduced cerebral glucose metabolism in patients at risk for Alzheimer’s disease. Psychiatry Res 155:147–154
Furst AJ, Lal RA (2011) Amyloid-β and glucose metabolism in Alzheimer’s disease. J Alzheimers Dis 26(Suppl 3):105–116
Ishii H, Ishikawa H, Meguro K, Tashiro M, Yamaguchi S (2009) Decreased cortical glucose metabolism in converters from CDR 0.5 to Alzheimer’s disease in a community: the Osaki-Tajiri Project. Int Psychogeriatr 21:148–156
Herholz K (2010) Cerebral glucose metabolism in preclinical and prodromal Alzheimer’s disease. Expert Rev Neurother 10:1667–1673
Mosconi L, Sorbi S, de Leon MJ, Li Y, Nacmias B, Myoung PS, Tsui W, Tsui W, Ginestroni A, Bessi V, Fayyazz M, Caffarra P, Pupi A (2006) Hypometabolism exceeds atrophy in presymptomatic early-onset familial Alzheimer’s disease. J Nucl Med 47:1778–1786
Mosconi L, De Santi S, Li J, Tsui WH, Li Y, Boppana M, Laska E, Rusinek H, de Leon MJ (2008) Hippocampal hypometabolism predicts cognitive decline from normal aging. Neurobiol Aging 29:676–692
Drzezga A, Lautenschlager N, Siebner H, Riemenschneider M, Willoch F, Minoshima S, Schwaiger M, Kurz A (2003) Cerebral metabolic changes accompanying conversion of mild cognitive impairment into Alzheimer’s disease: a PET follow-up study. Eur J Nucl Med Mol Imaging 30:1104–1113
Mosconi L (2005) Brain glucose metabolism in the early and specific diagnosis of Alzheimer’s disease. FDG-PET studies in MCI and AD. Eur J Nucl Med Mol Imaging 32:486–510
Nestor PJ, Fryer TD, Smielewski P, Hodges JR (2003) Limbic hypometabolism in Alzheimer’s disease and mild cognitive impairment. Ann Neurol 54:343–351
Poisnel G, Hérard AS, El Tannir El Tayara N, Bourrin E, Volk A, Kober F, Delatour B, Delzescaux T, Debeir T, Rooney T, Benavides J, Hantraye P, Dhenain M (2011) Increased regional cerebral glucose uptake in an APP/PS1 model of Alzheimer’s disease. Neurobiol Aging Nov 11. [Epub ahead of print]
de la Monte SM (2012) Contributions of brain insulin resistance and deficiency in amyloid-related neurodegeneration in Alzheimer’s disease. Drugs 72:49–66
Craft S (2007) Insulin resistance and Alzheimer’s disease pathogenesis: potential mechanisms and implications for treatment. Curr Alzheimer Res 4:147–152
Neumann KF, Rojo L, Navarrete LP, Farías G, Reyes P, Maccioni RB (2008) Insulin resistance and Alzheimer’s disease: molecular links and clinical implications. Curr Alzheimer Res 5:438–447
Sroriz-Filho J, Sá-Roriz TM, Rosset I, Camozzato AL, Santos AC, Chaves ML, Moriguti JC, Roriz-Cruz M (2009) (Pre)diabetes, brain aging, and cognition. Biochim Biophys Acta 1792:432–443
Sato N, Takeda S, Uchio-Yamada K, Ueda H, Fujisawa T, Rakugi H, Morishita R (2011) Role of insulin signaling in the interaction between Alzheimer disease and diabetes mellitus: a missing link to therapeutic potential. Curr Aging Sci 4:118–127
Ding Y, Kantarci A, Hasturk H, Trackman PC, Malabanan A, Van Dyke TE (2007) Activation of RAGE induces elevated O2- generation by mononuclear phagocytes in diabetes. J Leukoc Biol 81:520–527
Hallam KM, Li Q, Ananthakrishnan R, Kalea A, Zou YS, Vedantham S, Schmidt AM, Yan SF, Ramasamy R (2010) Aldose reductase and AGE-RAGE pathways: central roles in the pathogenesis of vascular dysfunction in aging rats. Aging Cell 9:776–784
Srikanth V, Maczurek A, Phan T, Steele M, Westcott B, Juskiw D, Münch G (2011) Advanced glycation endproducts and their receptor RAGE in Alzheimer’s disease. Neurobiol Aging 32:763–777
Freude S, Schilbach K, Schubert M (2009) The role of IGF-1 receptor and insulin receptor signaling for the pathogenesis of Alzheimer’s disease: from model organisms to human disease. Curr Alzheimer Res 6:213–223
Zemva J, Schubert M (2011) Central insulin and insulin-like growth factor-1 signaling: implications for diabetes associated dementia. Curr Diabetes Rev 7:356–366
Lester-Coll N, Rivera EJ, Soscia SJ, Doiron K, Wands JR, de la Monte SM (2006) Intracerebral streptozotocin model of type 3 diabetes: relevance to sporadic Alzheimer’s disease. J Alzheimers Dis 9:13–33
Seigel GM, Lupien SB, Campbell LM, Ishii DN (2006) Systemic IGF-I treatment inhibits cell death in diabetic rat retina. J Diabetes Complicat 20:196–204
Mattson MP (1997) Cellular actions of beta-amyloid precursor protein and its soluble and fibrillogenic derivatives. Physiol Rev 77:1081–1132
Desmond DW, Moroney JT, Sano M, Stern Y (2002) Incidence of dementia after ischemic stroke: results of a longitudinal study. Stroke 33:2254–2260
Grammas P, Tripathy D, Sanchez A, Yin X, Luo J (2011) Brain microvasculature and hypoxia-related proteins in Alzheimer’s disease. Int J Clin Exp Pathol 4:616–627
Lee S-R, Lo EH (2004) Induction of caspase mediated cell death by matrix metalloproteinases in cerebral endothelial cells after hypoxia-reoxygenation. J Cereb Blood Flow Metab 24:720–727
Egashira N, Iwasaki K, Ishibashi M, Hatip-Al-Khatib I, Wolozin B, Mishima K, Irie K, Fujiwara M (2002) Hypoxia enhances beta-amyloid-induced apoptosis in rat cultured hippocampal neurons. Jpn J Pharmacol 90:321–327
Friedman JE, Chow EJ, Haddad GG (1998) State of actin filaments is changed by anoxia in cultured rat neocortical neurons. Neuroscience 82:421–427
Breteler MM (2000) Vascular risk factors for Alzheimer’s disease: an epidemiologic perspective. Neurobiol Aging 21:153–160
de la Monte SM, Neely TR, Cannon J, Wands JR (2000) Oxidative stress and hypoxia-like injury cause Alzheimer-type molecular abnormalities in central nervous system neurons. Cell Mol Life Sci 57:1471–1481
Emerit J, Edeas M, Bricaire F (2004) Neurodegenerative diseases and oxidative stress. Biomed Pharmacother 58:39–46
Paglieri C, Bisbocci D, Caserta M, Rabbia F, Bertello C, Canadè A, Veglio F (2008) Hypertension and cognitive function. Clin Exp Hypertens 30:701–710
Kivipelto M, Helkala EL, Laakso MP, Hänninen T, Hallikainen M, Alhainen K, Soininen H, Tuomilehto J, Nissinen A (2001) Midlife vascular risk factors and Alzheimer’s disease in later life: longitudinal, population based study. BMJ 322:1447–1451
Kehoe PG, Miners S, Love S (2009) Angiotensins in Alzheimer’s disease—friend or foe? Trends Neurosci 32:619–628
Takeda S, Sato N, Ogihara T, Morishita R (2008) The renin-angiotensin system, hypertension and cognitive dysfunction in Alzheimer’s disease: new therapeutic potential. Front Biosci 13:2253–2265
Elias MF, D’Agostino RB, Elias PK, Wolf PA (1995) Neuropsychological test performance, cognitive functioning, blood pressure, and age: the Framingham Heart Study. Exp Aging Res 21:369–391
Farmer ME, Kittner SJ, Abbott RD, Wolz MM, Wolf PA, White LR (1990) Longitudinally measured blood pressure, antihypertensive medication use, and cognitive performance: the Framingham Study. J Clin Epidemiol 43:475–480
Elias MF, Wolf PA, D’Agostino RB, Cobb J, White LR (1993) Untreated blood pressure level is inversely related to cognitive functioning: the Framingham Study. Am J Epidemiol 138:353–364
Poon IO (2008) Effects of antihypertensive drug treatment on the risk of dementia and cognitive impairment. Pharmacotherapy 28:366–375
Dickstein DL, Walsh J, Brautigam H, Stockton SD Jr, Gandy S, Hof PR (2010) Role of vascular risk factors and vascular dysfunction in Alzheimer’s disease. Mt Sinai J Med 77:82–102
Svatikova A, Wolk R, Shamsuzzaman AS, Kara T, Olson EJ, Somers VK (2003) Serum amyloid a in obstructive sleep apnea. Circulation 108:1451–1454
Poitou C, Coupaye M, Laaban JP, Coussieu C, Bedel JF, Bouillot JL, Basdevant A, Clément K, Oppert JM (2006) Serum amyloid A and obstructive sleep apnea syndrome before and after surgically-induced weight loss in morbidly obese subjects. Obes Surg 16:1475–1481
Wright JW, Harding JW (2010) The brain RAS and Alzheimer’s disease. Exp Neurol 223:326–333
Gentile MT, Poulet R, Di Pardo A, Cifelli G, Maffei A, Vecchione C, Passarelli F, Landolfi A, Carullo P, Lembo G (2009) β-amyloid deposition in brain is enhanced in mouse models of arterial hypertension. Neurobiol Aging 30:222–228
Bloomfield SM, McKinney J, Smith L, Brisman J (2007) Reliability of S100B in predicting severity of central nervous system injury. Neurocrit Care 6:121–138
Braga CW, Martinez D, Wofchuk S, Portela LV, Souza DO (2006) S100B and NSE serum levels in obstructive sleep apnea syndrome. Sleep Med 7:431–435
Sonka K, Kelemen J, Kemlink D, Volná J, Pretl M, Zima T, Benakova H, Rambousek P, Foltán R, Donev F (2007) Evening and morning plasma levels of protein S100B in patients with obstructive sleep apnea. Neuro Endocrinol Lett 28:575–579
Breteler MM (2000) Vascular involvement in cognitive decline and dementia. Epidemiologic evidence from the Rotterdam Study and the Rotterdam Scan Study. Ann NY Acad Sci 903:457–465
Devisser A, Yang C, Herring A, Martinez JA, Rosales-Hernandez A, Poliakov I, Ayer A, Garven A, Zaver S, Rincon N, Xu K, Tuor UI, Schmidt AM, Toth C (2011) Differential impact of diabetes and hypertension in the brain: adverse effects in grey matter. Neurobiol Dis 44:161–173
Qiu C, Winblad B, Fratiglioni L (2005) The age-dependent relation of blood pressure to cognitive function and dementia. Lancet Neurol 4:487–499
Razay G, Williams J, King E, Smith AD, Wilcock G (2009) Blood pressure, dementia and Alzheimer’s disease: the OPTIMA longitudinal study. Dement Geriatr Cogn Disord 28:70–74
Joas E, Bäckman K, Gustafson D, Ostling S, Waern M, Guo X, Skoog I (2012) Blood pressure trajectories from midlife to late life in relation to dementia in women followed for 37 years. Hypertension 59:796–801
Wirths O, Breyhan H, Marcello A, Cotel MC, Brück W, Bayer TA (2010) Inflammatory changes are tightly associated with neurodegeneration in the brain and spinal cord of the APP/PS1KI mouse model of Alzheimer’s disease. Neurobiol Aging 31:747–757
Desai AK (2011) Revitalizing the aged brain. Med Clin North Am 95:463–475
Sachdev PS, Brodaty H, Reppermund S, Kochan NA, Trollor JN, Draper B, Slavin MJ (2010) The Sydney memory and ageing study (MAS): methodology and baseline medical and neuropsychiatric characteristics of an elderly epidemiological non-demented cohort of Australians aged 70–90 years. Int Psychogeriatr 22:1248–1264
Hohman TJ, Beason-Held LL, Resnick SM (2011) Cognitive complaints, depressive symptoms, and cognitive impairment: are they related? J Am Geriatr Soc 59:1908–1912
Saunamäki T, Jehkonen M (2007) Depression and anxiety in obstructive sleep apnea syndrome: a review. Acta Neurol Scand 116:277–288
Choi NG, Dinitto DM (2011) Heavy/binge drinking and depressive symptoms in older adults: gender differences. Int J Geriatr Psychiatry 26:860–868
Godbout JP, Moreau M, Lestage J, Chen J, Sparkman NL, O’ Connor J, Castanon N, Kelley KW, Dantzer R, Johnson RW (2008) Aging exacerbates depressive-like behavior in mice in response to activation of the peripheral innate immune system. Neuropsychopharmacology 33:2341–2351
Maes M, Kubera M, Obuchowiczwa E, Goehler L, Brzeszcz J (2011) Depression’s multiple comorbidities explained by (neuro)inflammatory and oxidative & nitrosative stress pathways. Neuro Endocrinol Lett 32:7–24
Akiyama H, Meyer JS, Mortel KF, Terayama Y, Thornby JI, Konno S (1997) Normal human aging: factors contributing to cerebral atrophy. J Neurol Sci 152:39–49
Shimada A (1999) Age-dependent cerebral atrophy and cognitive dysfunction in SAMP10 mice. Neurobiol Aging 20:125–136
Clyburn EB, DiPette DJ (1995) Hypertension induced by drugs and other substances. Semin Nephrol 15:72–86
Migliore L, Coppedè F (2009) Environmental-induced oxidative stress in neurodegenerative disorders and aging. Mutat Res 674:73–84
Ogasawara K, Nakamura Y, Aleksic B, Yoshida K, Ando K, Iwata N, Kayukawa Y, Ozaki N (2011) Depression associated with alcohol intake and younger age in Japanese office workers: a case- control and a cohort study. J Affect Disord 128:33–40
Tobe SW, Soberman H, Kiss A, Perkins N, Baker B (2006) The effect of alcohol and gender on ambulatory blood pressure: results from the Baseline Double Exposure study. Am J Hypertens 19:136–139
McFarlane SI, von Gizycki H, Salifu M, Deshmukh M, Manieram M, Gebreegziabher Y, Gliwa AA, Bordia S, Shah S, Sowers JR (2007) Alcohol consumption and blood pressure in the adult US population: assessment of gender-related effects. J Hypertens 25:965–970
Estruch R, Nicolás JM, Salamero M, Aragón C, Sacanella E, Fernández-Solà J, Urbano-Márquez A (1997) Atrophy of the corpus callosum in chronic alcoholism. J Neurol Sci 146:145–151
Teipel SJ, Bayer W, Alexander GE, Bokde AL, Zebuhr Y, Teichberg D, Müller-Spahn F, Schapiro MB, Möller HJ, Rapoport SI, Hampel H (2003) Regional pattern of hippocampus and corpus callosum atrophy in Alzheimer’s disease in relation to dementia severity: evidence for early neocortical degeneration. Neurobiol Aging 24:85–94
Teipel SJ, Bayer W, Alexander GE, Zebuhr Y, Teichberg D, Kulic L, Schapiro MB, Möller HJ, Rapoport SI, Hampel H (2002) Progression of corpus callosum atrophy in Alzheimer disease. Arch Neurol 59:243–248
Nicolás JM, Catafau AM, Estruch R, Lomeña FJ, Salamero M, Herranz R, Monforte R, Cardenal C, Urbano-Marquez A (1993) Regional cerebral blood flow-SPECT in chronic alcoholism: relation to neuropsychological testing. J Nucl Med 34:1452–1459
Williams-Hemby L, Porrino LJ (1994) Low and moderate doses of ethanol produce distinct patterns of cerebral metabolic changes in rats. Alcohol Clin Exp Res 18:982–988
Christie IC, Price J, Edwards L, Muldoon M, Meltzer CC, Jennings JR (2008) Alcohol consumption and cerebral blood flow among older adults. Alcohol 42:269–275
Pach D, Hubalewska-Dydejczyk A, Szurkowska M, Kamenczak A, Targosz D, Gawlikowski T, Huszno B, Głowa B (2007) Evaluation of regional cerebral blood flow using 99mTc-ECD SPECT in ethanol dependent patients: pilot study. Przegl Lek 64:204–207
Talukder MA, Johnson WM, Varadharaj S, Lian J, Kearns PN, El-Mahdy MA, Liu X, Zweier JL (2011) Chronic cigarette smoking causes hypertension, increased oxidative stress, impaired NO bioavailability, endothelial dysfunction, and cardiac remodeling in mice. Am J Physiol Heart Circ Physiol 300:H388–H396
Halterman MW, Miller CC, Federoff HJ (1999) Hypoxia-inducible factor-1alpha mediates hypoxia-induced delayed neuronal death that involves p53. J Neurosci 19:6818–6824
Pulsinelli WA, Brierley JB, Plum F (1982) Temporal profile of neuronal damage in a model of transient forebrain ischemia. Ann Neurol 11:491–498
Chen J, Nagayama T, Jin K, Stetler RA, Zhu RL, Graham SH, Simon RP (1998) Induction of caspase-3-like protease may mediate delayed neuronal death in the hippocampus after transient cerebral ischemia. J Neurosci 18:4914–4928
Rabinovici GD, Jagust WJ (2009) Amyloid imaging in aging and dementia: testing the amyloid hypothesis in vivo. Behav Neurol 21:117–128
Sadowski M, Pankiewicz J, Scholtzova H, Li YS, Quartermain D, Duff K, Wisniewski T (2004) Links between the pathology of Alzheimer’s disease and vascular dementia. Neurochem Res 29:1257–1266
Austin BP, Nair VA, Meier TB, Xu G, Rowley HA, Carlsson CM, Johnson SC, Prabhakaran V (2011) Effects of hypoperfusion in Alzheimer’s disease. J Alzheimers Dis 26(Suppl 3):123–133
Sun X, He G, Qing H, Zhou W, Dobie F, Cai F, Staufenbiel M, Huang LE, Song W (2006) Hypoxia facilitates Alzheimer’s disease pathogenesis by up-regulating BACE1 gene expression. Proc Natl Acad Sci USA 103:18727–18732
Anglade P, Vyas S, Hirsch EC, Agid Y (1997) Apoptosis in dopaminergic neurons of the human substantia nigra during normal aging. Histol Histopathol 12:603–610
Zhang F, Yin W, Chen J (2004) Apoptosis in cerebral ischemia: executional and regulatory signaling mechanisms. Neurol Res 26:835–845
Gilles FH (1969) Hypotensive brain stem necrosis: selective symmetrical necrosis of tegmental neuronal aggregates following cardiac arrest. Arch Pathol 88:32–41
Revesz T, Geddes JF (1988) Symmetrical columnar necrosis of the basal ganglia and brain stem in an adult following cardiac arrest. Clin Neuropathol 6:294–298
De Caro R, Parenti A, Montisci M, Guidolin D, Macchi V (2003) Symmetrical selective neuronal necrosis in solitary tract nuclei. Int J Legal Med 117:253–254
Parenti A, Macchi V, Snenghi R, Porzionato A, Scaravilli T, Ferrara SD, De Caro R (2005) Selective stroke of the solitary tract nuclei in two cases of central sleep apnoea. Clin Neuropathol 24:239–246
Gustafson D, Lissner L, Bengtsson C, Björkelund C, Skoog I (2004) A 24-year follow-up of body mass index and cerebral atrophy. Neurology 63:1876–1881
Kivipelto M, Ngandu T, Fratiglioni L, Viitanen M, Kåreholt I, Winblad B, Helkala EL, Tuomilehto J, Soininen H, Nissinen A (2005) Obesity and vascular risk factors at midlife and the risk of dementia and Alzheimer disease. Arch Neurol 62:1556–1560
Rosengren A, Skoog I, Gustafson D, Wilhelmsen L (2005) Body mass index, other cardiovascular risk factors, and hospitalization for dementia. Arch Intern Med 165:321–326
Whitmer RA, Gunderson EP, Barrett-Connor E (2005) Obesity in middle age and future risk of dementia: a 27 year longitudinal population based study. BMJ 330:1360
Zanni MV, Stanley TL, Makimura H, Chen CY, Grinspoon SK (2010) Effects of TNF-alpha antagonism on E-selectin in obese subjects with metabolic dysregulation. Clin Endocrinol (Oxf) 73:48–54
Iannone F, Lapadula G (2010) Obesity and inflammation–targets for OA therapy. Curr Drug Targets 11:586–598
Walther K, Birdsill AC, Glisky EL, Ryan L (2010) Structural brain differences and cognitive functioning related to body mass index in older females. Hum Brain Mapp 31:1052–1064
Raji CA, Ho AJ, Parikshak NN, Becker JT, Lopez OL, Kuller LH, Hua X, Leow AD, Toga AW, Thompson PM (2010) Brain structure and obesity. Hum Brain Mapp 31:353–364
Luchsinger JA, Mayeux R (2007) Adiposity and Alzheimer’s disease. Curr Alzheimer Res 4:127–134
Erol A (2008) An integrated and unifying hypothesis for the metabolic basis of sporadic Alzheimer’s disease. J Alzheimers Dis 13:241–253
Ho L, Qin W, Pompl PN, Xiang Z, Wang J, Zhao Z, Peng Y, Cambareri G, Rocher A, Mobbs CV, Hof PR, Pasinetti GM (2004) Diet-induced insulin resistance promotes amyloidosis in a transgenic mouse model of Alzheimer’s disease. FASEB J 18:902–904
Julien C, Tremblay C, Phivilay A, Berthiaume L, Emond V, Julien P, Calon F (2010) High-fat diet aggravates amyloid-beta and tau pathologies in the 3xTg-AD mouse model. Neurobiol Aging 31:1516–1531
Wyss-Coray T (2006) Inflammation in Alzheimer disease: driving force, bystander or beneficial response? Nat Med 12:1005–1015
Tan J, Town T, Crawford F, Mori T, DelleDonne A, Crescentini R, Obregon D, Flavell RA, Mullan MJ (2002) Role of CD40 ligand in amyloidosis in transgenic Alzheimer’s mice. Nat Neurosci 5:1288–1293
Kitazawa M, Oddo S, Yamasaki TR, Green KN, LaFerla FM (2005) Lipopolysaccharide-induced inflammation exacerbates tau pathology by a cyclin-dependent kinase 5-mediated pathway in a transgenic model of Alzheimer’s disease. J Neurosci 25:8843–8853
Luchsinger JA, Reitz C, Patel B, Tang MX, Manly JJ, Mayeux R (2007) Relation of diabetes to mild cognitive impairment. Arch Neurol 64:570–575
Takeda S, Sato N, Rakugi H, Morishita R (2011) Molecular mechanisms linking diabetes mellitus and Alzheimer disease: beta-amyloid peptide, insulin signaling, and neuronal function. Mol BioSyst 7:1822–1827
Strachan MW, Reynolds RM, Marioni RE, Price JF (2011) Cognitive function, dementia and type 2 diabetes mellitus in the elderly. Nat Rev Endocrinol 7:108–114
Hölscher C (2011) Diabetes as a risk factor for Alzheimer’s disease: insulin signalling impairment in the brain as an alternative model of Alzheimer’s disease. Biochem Soc Trans 39:891–897
Murakami K, Yokoyama S, Murata N, Ozawa Y, Irie K, Shirasawa T, Shimizu T (2011) Insulin receptor mutation results in insulin resistance and hyperinsulinemia but does not exacerbate Alzheimer’s-like phenotypes in mice. Biochem Biophys Res Commun 409:34–39
Harsch IA, Hahn EG, Konturek PC (2005) Insulin resistance and other metabolic aspects of the obstructive sleep apnea syndrome. Med Sci Monit 11:RA70–RA75
Makino S, Handa H, Suzukawa K, Fujiwara M, Nakamura M, Muraoka S, Takasago I, Tanaka Y, Hashimoto K, Sugimoto T (2006) Obstructive sleep apnoea syndrome, plasma adiponectin levels, and insulin resistance. Clin Endocrinol (Oxf) 64:12–19
Steiropoulos P, Papanas N, Nena E, Tsara V, Fitili C, Tzouvelekis A, Christaki P, Maltezos E, Bouros D (2009) Markers of glycemic control and insulin resistance in non-diabetic patients with obstructive sleep apnea hypopnea syndrome: does adherence to CPAP treatment improve glycemic control? Sleep Med 10:887–891
Bonsignore MR, Esquinas C, Barceló A, Sanchez-de-la-Torre M, Paternó A, Duran-Cantolla J, Marín JM, Barbé F (2011) Metabolic syndrome, insulin resistance and sleepiness in real-life obstructive sleep apnoea. Eur Respir J Nov 10. [Epub ahead of print]
Deboer MD, Mendoza JP, Liu L, Ford G, Yu PL, Gaston BM (2012) Increased systemic inflammation overnight correlates with insulin resistance among children evaluated for obstructive sleep apnea. Sleep Breath 16:349–354
Yang D, Liu Z, Yang H, Luo Q (2012) Effects of continuous positive airway pressure on glycemic control and insulin resistance in patients with obstructive sleep apnea: a meta-analysis. Sleep Breath Mar 13. [Epub ahead of print]
Moroz N, Tong M, Longato L, Xu H, de la Monte SM (2008) Limited Alzheimer-type neurodegeneration in experimental obesity and type 2 diabetes mellitus. J Alzheimers Dis 15:29–44
Correia SC, Santos RX, Perry G, Zhu X, Moreira PI, Smith MA (2011) Insulin-resistant brain state: the culprit in sporadic Alzheimer’s disease? Ageing Res Rev 10:264–273
Moreira PI, Carvalho C, Zhu X, Smith MA, Perry G (2010) Mitochondrial dysfunction is a trigger of Alzheimer’s disease pathophysiology. Biochim Biophys Acta 1802:2–10
Horan MP, Pichaud N, Ballard JW (2012) Review: Quantifying Mitochondrial Dysfunction in Complex Diseases of Aging. J Gerontol A Biol Sci Med Sci Mar 29. [Epub ahead of print]
Scaglia F (2010) The role of mitochondrial dysfunction in psychiatric disease. Dev Disabil Res Rev 16:136–143
Kagan VE, Tyurina YY, Bayir H, Chu CT, Kapralov AA, Vlasova II, Belikova NA, Tyurin VA, Amoscato A, Epperly M, Greenberger J, Dekosky S, Shvedova AA, Jiang J (2006) The “pro-apoptotic genies” get out of mitochondria: oxidative lipidomics and redox activity of cytochrome c/cardiolipin complexes. Chem Biol Interact 163:15–28
Yin H, Zhu M (2012) Free Radical Oxidation of Cardiolipin: Chemical Mechanisms, Detection, and Implication in Apoptosis, Mitochondrial Dysfunction, and Human Diseases. Free Radic Res Apr 3. [Epub ahead of print]
Chakrabarti S, Munshi S, Banerjee K, Thakurta IG, Sinha M, Bagh MB (2011) Mitochondrial dysfunction during brain aging: role of oxidative stress and modulation by antioxidant supplementation. Aging Dis 2:242–256
Lloret A, Buj J, Badia MC, Sastre J, Morera J, Viña J (2007) Obstructive sleep apnea: arterial oxygen desaturation coincides with increases in systemic oxidative stress markers measured with continuous monitoring. Free Radic Biol Med 42:893–894
Eckert A, Schmitt K, Götz J (2011) Mitochondrial dysfunction—the beginning of the end in Alzheimer’s disease? Separate and synergistic modes of tau and amyloid-β toxicity. Alzheimers Res Ther 3:15
Lezi E, Swerdlow RH (2012) Mitochondria in neurodegeneration. Adv Exp Med Biol 942:269–286
Trushina E, Nemutlu E, Zhang S, Christensen T, Camp J, Mesa J, Siddiqui A, Tamura Y, Sesaki H, Wengenack TM, Dzeja PP, Poduslo JF (2012) Defects in mitochondrial dynamics and metabolomic signatures of evolving energetic stress in mouse models of familial Alzheimer’s disease. PLoS ONE 7:e32737
Karbowski M, Neutzner A (2012) Neurodegeneration as a consequence of failed mitochondrial maintenance. Acta Neuropathol 123:157–171
Aliev G, Palacios HH, Walrafen B, Lipsitt AE, Obrenovich ME, Morales L (2009) Brain mitochondria as a primary target in the development of treatment strategies for Alzheimer disease. Int J Biochem Cell Biol 41:1989–2004
Wang Y, Zhang SX, Gozal D (2010) Reactive oxygen species and the brain in sleep apnea. Respir Physiol Neurobiol 174:307–316
Shi Q, Gibson GE (2007) Oxidative stress and transcriptional regulation in Alzheimer disease. Alzheimer Dis Assoc Disord 21:276–291
Ferrer I, Planas AM (2003) Signaling of cell death and cell survival following focal cerebral ischemia: life and death struggle in the penumbra. J Neuropathol Exp Neurol 62:329–339
Kim GS, Jung JE, Narasimhan P, Sakata H, Yoshioka H, Song YS, Okami N, Chan PH (2011) Release of mitochondrial apoptogenic factors and cell death are mediated by CK2 and NADPH oxidase. J Cereb Blood Flow Metab 32:720–730
Ferreira IL, Resende R, Ferreiro E, Rego AC, Pereira CF (2010) Multiple defects in energy metabolism in Alzheimer’s disease. Curr Drug Targets 11:1193–1206
Fung SJ, Xi MC, Zhang JH, Sampogna S, Yamuy J, Morales FR, Chase MH (2007) Apnea promotes glutamate-induced excitotoxicity in hippocampal neurons. Brain Res 1179:42–50
Xie SX, Libon DJ, Wang X, Massimo L, Moore P, Vesely L, Khan A, Chatterjee A, Coslett HB, Hurtig HI, Liang TW, Grossman M (2010) Longitudinal patterns of semantic and episodic memory in frontotemporal lobar degeneration and Alzheimer’s disease. J Int Neuropsychol Soc 16:278–286
Gozal D, Daniel JM, Dohanich GP (2001) Behavioral and anatomical correlates ofchronic episodic hypoxia during sleep in the rat. J Neurosci 21:2442–2450
Gozal D, Row BW, Kheirandish L, Liu R, Guo SZ, Qiang F, Brittian KR (2003) Increased susceptibility to intermittent hypoxia in aging rats: changes in proteasomal activity, neuronal apoptosis and spatial function. J Neurochem 86:1545–1552
Goldbart A, Row BW, Kheirandish L, Schurr A, Gozal E, Guo SZ, Payne RS, Cheng Z, Brittian K, Gozal D (2003) Intermittent hypoxic exposure during light phase induces changes in cAMP response element binding protein activity in the rat CA1 hippocampal region: water maze performance correlates. Neuroscience 122:583–590
Ward CP, McCoy JG, McKenna JT, Connolly NP, McCarley RW, Strecker RE (2009) Spatial learning and memory deficits following exposure to 24 h of sleep fragmentation or intermittent hypoxia in a rat model of obstructive sleep apnea. Brain Res 1294:128–137
Row BW, Liu R, Xu W, Kheirandish L, Gozal D (2003) Intermittent hypoxia is associated with oxidative stress and spatial learning deficits in the rat. Am J Respir Crit Care Med 167:1548–1553
Li RC, Row BW, Kheirandish L, Brittian KR, Gozal E, Guo SZ, Sachleben LR Jr, Gozal D (2004) Nitric oxide synthase and intermittent hypoxia-induced spatial learning deficits in the rat. Neurobiol Dis 17:44–53
Li RC, Row BW, Gozal E, Kheirandish L, Fan Q, Brittian KR, Guo SZ, Sachleben LR Jr, Gozal D (2003) Cyclooxygenase 2 and intermittent hypoxia-induced spatial deficits in the rat. Am J Respir Crit Care Med 168:469–475
Bourgeat P, Chételat G, Villemagne VL, Fripp J, Raniga P, Pike K, Acosta O, Szoeke C, Ourselin S, Ames D, Ellis KA, Martins RN, Masters CL, Rowe CC, Salvado O (2010) Beta-amyloid burden in the temporal neocortex is related to hippocampal atrophy in elderly subjects without dementia. Neurology 74:121–127
Dai W, Lopez OL, Carmichael OT, Becker JT, Kuller LH, Gach HM (2008) Abnormal regional cerebral blood flow in cognitively normal elderly subjects with hypertension. Stroke 39:349–354
Korf ES, White LR, Scheltens P, Launer LJ (2004) Midlife blood pressure and the risk of hippocampal atrophy: the Honolulu Asia Aging Study. Hypertension 44:29–34
Wiseman RM, Saxby BK, Burton EJ, Barber R, Ford GA, O’Brien JT (2004) Hippocampal atrophy, whole brain volume, and white matter lesions in older hypertensive subjects. Neurology 63:1892–1897
Qiu C, Winblad B, Fratiglioni L (2005) The age-dependent relation of blood pressure to cognitive function and dementia. Lancet Neurol 4:487–499
Razay G, Williams J, King E, Smith AD, Wilcock G (2009) Blood pressure, dementia and Alzheimer’s disease: the OPTIMA longitudinal study. Dement Geriatr Cogn Disord 28:70–74
Espinosa-Oliva AM, de Pablos RM, Villarán RF, Argüelles S, Venero JL, Machado A, Cano J (2011) Stress is critical for LPS-induced activation of microglia and damage in the rat hippocampus. Neurobiol Aging 32:85–102
Tagliari B, Tagliari AP, Schmitz F, da Cunha AA, Dalmaz C, Wyse AT (2011) Chronic variable stress alters inflammatory and cholinergic parameters in hippocampus of rats. Neurochem Res 36:487–493
Castle M, Comoli E, Loewy AD (2005) Autonomic brainstem nuclei are linked to the hippocampus. Neuroscience 134:657–669
Loewy AD (1998) Viruses as transneuronal tracers for defining neural circuits. Neurosci Biobehav Rev 22:679–684
Dong HW, Petrovich GD, Watts AG, Swanson LW (2001) Basic organization of projections from the oval and fusiform nuclei of the bed nuclei of the stria terminalis in adult rat brain. J Comp Neurol 436:430–455
Loy R, Koziell DA, Lindsey JD, Moore RY (1980) Noradrenergic innervation of the adult rat hippocampal formation. J Comp Neurol 189:699–710
Rye DB, Wainer BH, Mesulam MM, Mufson EJ, Saper CB (1984) Cortical projections arising from the basal forebrain: a study of cholinergic and noncholinergic components employing combined retrograde tracing and immunohistochemical localization of choline acetyltransferase. Neuroscience 13:627–643
Vgontzas AN, Bixler EO, Chrousos GP (2005) Sleep apnea is a manifestation of the metabolic syndrome. Sleep Med Rev 9:211–224
Vgontzas AN (2008) Does obesity play a major role in the pathogenesis of sleep apnoea and its associated manifestations via inflammation, visceral adiposity, and insulin resistance? Arch Physiol Biochem 114:211–223
Carneiro G, Togeiro SM, Ribeiro-Filho FF, Truksinas E, Ribeiro AB, Zanella MT, Tufik S (2009) Continuous positive airway pressure therapy improves hypoadiponectinemia in severe obese men with obstructive sleep apnea without changes in insulin resistance. Metab Syndr Relat Disord 7:537–542
Mokhlesi B, Scoccia B, Mazzone T, Sam S (2012) Risk of obstructive sleep apnea in obese and nonobese women with polycystic ovary syndrome and healthy reproductively normal women. Fertil Steril 97:786–791
Nitsche K, Ehrmann DA (2010) Obstructive sleep apnea and metabolic dysfunction in polycystic ovary syndrome. Best Pract Res Clin Endocrinol Metab 24:717–730
Lin O, Zhang X, Chen G, Huang D, Din H, Tang A (2012) Obstructive sleep apnea syndrome is associated with some components of metabolic syndrome in nonobese adults. Sleep Breath 16:571–578
Tasali E, Van Cauter E, Hoffman L, Ehrmann DA (2008) Impact of obstructive sleep apnea on insulin resistance and glucose tolerance in women with polycystic ovary syndrome. J Clin Endocrinol Metab 93:3878–3884
Liu A, Kushida CA, Reaven GM (2012) Risk for obstructive sleep apnea in obese, nondiabetic adults varies with insulin resistance status. Sleep Breath Apr 7. [Epub ahead of print]
Moroz N, Tong M, Longato L, Xu H, de la Monte SM (2008) Limited Alzheimer-type neurodegeneration in experimental obesity and type 2 diabetes mellitus. J Alzheimers Dis 15:29–44
Musicco M, Palmer K, Salamone G, Lupo F, Perri R, Mosti S, Spalletta G, di Iulio F, Pettenati C, Cravello L, Caltagirone C (2009) Predictors of progression of cognitive decline in Alzheimer’s disease: the role of vascular and sociodemographic factors. J Neurol 256:1288–1295
Dong YF, Kataoka K, Toyama K, Sueta D, Koibuchi N, Yamamoto E, Yata K, Tomimoto H, Ogawa H, Kim-Mitsuyama S (2011) Attenuation of brain damage and cognitive impairment by direct ennin inhibition in mice with chronic cerebral hypoperfusion. Hypertension 58:635–642
Inaba S, Iwai M, Furuno M, Tomono Y, Kanno H, Senba I, Okayama H, Mogi M, Higaki J, Horiuchi M (2009) Continuous activation of rennin-angiotensin system impairs cognitive function in ennin/angiotensinogen transgenic mice. Hypertension 53:356–362
Inaba S, Iwai M, Tomono Y, Senba I, Furuno M, Kanno H, Okayama H, Mogi M, Higaki J, Horiuchi M (2009) Exaggeration of focal cerebral ischemia in transgenic mice carrying human Renin and human angiotensinogen genes. Stroke 40:597–603
Díaz-Ruiz C, Wang J, Ksiezak-Reding H, Ho L, Qian X, Humala N, Thomas S, Martínez-Martín P, Pasinetti GM (2009) Role of hypertension in aggravating abeta neuropathology of AD type and tau-mediated motor impairment. Cardiovasc Psychiatry Neurol 2009:107286
Sun X, He G, Qing H, Zhou W, Dobie F, Cai F, Staufenbiel M, Huang LE, Song W (2006) Hypoxia facilitates Alzheimer’s disease pathogenesis by up-regulating BACE1 gene expression. Proc Natl Acad Sci USA 103:18727–18732
Zhang X, Zhou K, Wang R, Cui J, Lipton SA, Liao FF, Xu H, Zhang YW (2007) Hypoxia-inducible factor 1alpha (HIF-1alpha)-mediated hypoxia increases BACE1 expression and beta-amyloid generation. J Biol Chem 282:10873–10880
Rosenberg PB, Drye LT, Martin BK, Frangakis C, Mintzer JE, Weintraub D, Porsteinsson AP, Schneider LS, Rabins PV, Munro CA, Meinert CL, Lyketsos CG (2010) Sertraline for the treatment of depression in Alzheimer disease. Am J Geriatr Psychiatry 18:136–145
O’Collins VE, Macleod MR, Donnan GA, Horky LL, van der Worp BH, Howells DW (2006) 1,026 experimental treatments in acute stroke. Ann Neurol 59:467–477
DeGracia DJ (2010) Towards a dynamical network view of brain ischemia and reperfusion. Part IV: additional considerations. J Exp Stroke Transl Med 3:104–114
Huang S, Wikswo J (2006) Dimensions of systems biology. Rev Physiol Biochem Pharmacol 157:81–104
Tyson JJ, Chen K, Novak B (2001) Network dynamics and cell physiology. Nat Rev Mol Cell Biol 2:908–916
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Daulatzai, M.A. Quintessential Risk Factors: Their Role in Promoting Cognitive Dysfunction and Alzheimer’s Disease. Neurochem Res 37, 2627–2658 (2012). https://doi.org/10.1007/s11064-012-0854-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-012-0854-6