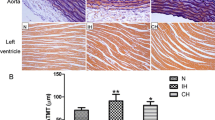
Abstract
Rationale
Chronic intermittent hypoxia (CIH) is thought to induce several cardiovascular effects in patients with obstructive sleep apnoea (OSA). However, the effects of CIH on patients with long-standing hypertension are unknown.
Purpose
This prospective study aimed to investigate the influence of combined OSA and hypertension on cardiomyocyte death.
Methods
Wistar–Kyoto rats (WKY) and spontaneously hypertensive rats (SHR) were exposed to repetitive hypoxia–reoxygenation cycles (30 s of 5% O2; 45 s of 21% O2) or room air for 6 h/day during the light phase (10 a.m.–4 p.m.) for 10, 20, or 30 days, and the levels of necrosis and apoptosis induced in their left ventricular cardiomyocyte were examined.
Results
CIH increased the accumulation of reactive oxygen species, which induced cardiomyocyte necrosis in WKY and SHR (both p < 0.05). Cardiomyocyte oxidative stress levels by CIH were higher in SHR than in WKY (p < 0.05); therefore, cardiomyocyte necrosis was amplified (p < 0.05). Notably, if a superoxide-scavenging agent is injected beforehand, cardiomyocyte necrosis can be effectively inhibited (p < 0.05). When WKY and SHR are exposed to CIH, increases in mitochondria-released cytochrome c and activation of caspase-3 are found in the cytosolic fraction only in WKY.
Conclusions
CIH causes cardiomyocyte loss in SHR mainly through cardiomyocyte necrosis. In WKY however, CIH simultaneously induces apoptosis and necrosis of cardiomyocytes.





Similar content being viewed by others
References
Mohsenin V (2001) Sleep-related breathing disorders and risk of stroke. Stroke 32:1271–1278
Park AM, Suzuki YJ (2007) Effects of intermittent hypoxia on oxidative stress-induced myocardial damage in mice. J Appl Physiol 102:1806–1814
Chen L, Einbinder E, Zhang Q, Hasday J, Balke CW, Scharf SM (2005) Oxidative stress and left ventricular function with chronic intermittent hypoxia in rats. Am J Respir Crit Care Med 172:915–920
Belaidi E, Joyeux-Faure M, Ribuot C, Launois SH, Levy P, Godin-Ribuot D (2009) Major role for hypoxia inducible factor-1 and the endothelin system in promoting myocardial infarction and hypertension in an animal model of obstructive sleep apnea. J Am Coll Cardiol 53:1309–1317
Matsuoka R, Ogawa K, Yaoita H, Naganuma W, Maehara K, Maruyama Y (2002) Characteristics of death of neonatal rat cardiomyocytes following hypoxia or hypoxia-reoxygenation: the association of apoptosis and cell membrane disintegrity. Heart Vessels 16:241–248
Carpagnano GE, Kharitonov SA, Resta O, Foschino-Barbaro MP, Gramiccioni E, Barnes PJ (2003) 8-Isoprostane, a marker of oxidative stress, is increased in exhaled breath condensate of patients with obstructive sleep apnea after night and is reduced by continuous positive airway pressure therapy. Chest 124:1386–1392
Barcelo A, Miralles C, Barbe F, Vila M, Pons S, Agusti AG (2000) Abnormal lipid peroxidation in patients with sleep apnoea. Eur Respir J 16:644–647
Chen L, Zhang J, Gan TX, Chen-Izu Y, Hasday JD, Karmazyn M, Balke CW, Scharf SM (2008) Left ventricular dysfunction and associated cellular injury in rats exposed to chronic intermittent hypoxia. J Appl Physiol 104:218–223
Conrad CH, Brooks WW, Hayes JA, Sen S, Robinson KG, Bing OH (1995) Myocardial fibrosis and stiffness with hypertrophy and heart failure in the spontaneously hypertensive rat. Circulation 91:161–170
Fortuno MA, Ravassa S, Fortuno A, Zalba G, Diez J (2001) Cardiomyocyte apoptotic cell death in arterial hypertension: mechanisms and potential management. Hypertension 38:1406–1412
Lee SD, Kuo WW, Wu CH, Lin YM, Lin JA, Lu MC, Yang AL, Liu JY, Wang SG, Liu CJ, Chen LM, Huang CY (2006) Effects of short- and long-term hypobaric hypoxia on Bcl2 family in rat heart. Int J Cardiol 108:376–384
Lee SD, Kuo WW, Lin JA, Chu YF, Wang CK, Yeh YL, Wang SG, Liu JY, Chang MH, Huang CY (2007) Effects of long-term intermittent hypoxia on mitochondrial and Fas death receptor dependent apoptotic pathways in rat hearts. Int J Cardiol 116:348–356
Buzello M, Boehm C, Orth S, Fischer B, Ehmke H, Ritz E, Mall G, Amann K (2003) Myocyte loss in early left ventricular hypertrophy of experimental renovascular hypertension. Virchows Arch 442:364–371
Gonzalez A, Fortuno MA, Querejeta R, Ravassa S, Lopez B, Lopez N, Diez J (2003) Cardiomyocyte apoptosis in hypertensive cardiomyopathy. Cardiovasc Res 59:549–562
Katz AM (1994) The cardiomyopathy of overload: an unnatural growth response in the hypertrophied heart. Ann Intern Med 121:363–371
Cheng W, Li B, Kajstura J, Li P, Wolin MS, Sonnenblick EH, Hintze TH, Olivetti G, Anversa P (1995) Stretch-induced programmed myocyte cell death. J Clin Invest 96:2247–2259
Drager LF, Bortolotto LA, Figueiredo AC, Silva BC, Krieger EM, Lorenzi-Filho G (2007) Obstructive sleep apnea, hypertension, and their interaction on arterial stiffness and heart remodeling. Chest 131:1379–1386
Fletcher EC, Orolinova N, Bader M (2002) Blood pressure response to chronic episodic hypoxia: the renin-angiotensin system. J Appl Physiol 92:627–633
Lai CJ, Yang CC, Hsu YY, Lin YN, Kuo TB (2006) Enhanced sympathetic outflow and decreased baroreflex sensitivity are associated with intermittent hypoxia-induced systemic hypertension in conscious rats. J Appl Physiol 100:1974–1982
Troncoso Brindeiro CM, da Silva AQ, Allahdadi KJ, Youngblood V, Kanagy NL (2007) Reactive oxygen species contribute to sleep apnea-induced hypertension in rats. Am J Physiol 293:H2971–H2976
Soukhova-O'Hare GK, Ortines RV, Gu Y, Nozdrachev AD, Prabhu SD, Gozal D (2008) Postnatal intermittent hypoxia and developmental programming of hypertension in spontaneously hypertensive rats: the role of reactive oxygen species and L-Ca2+ channels. Hypertension 52:156–162
Siu PM, Bryner RW, Martyn JK, Alway SE (2004) Apoptotic adaptations from exercise training in skeletal and cardiac muscles. FASEB J 18:1150–1152
Touyz RM (2004) Reactive oxygen species, vascular oxidative stress, and redox signaling in hypertension: what is the clinical significance? Hypertension 44:248–252
Yuan YV, Kitts DD, Godin DV (1996) Heart and red blood cell antioxidant status and plasma lipid levels in the spontaneously hypertensive and normotensive Wistar-Kyoto rat. Can J Physiol Pharmacol 74:290–297
Csonka C, Pataki T, Kovacs P, Muller SL, Schroeter ML, Tosaki A, Blasig IE (2000) Effects of oxidative stress on the expression of antioxidative defense enzymes in spontaneously hypertensive rat hearts. Free Radic Biol Med 29:612–619
Narang D, Sood S, Thomas M, Dinda AK, Maulik SK (2005) Dietary palm olein oil augments cardiac antioxidant enzymes and protects against isoproterenol-induced myocardial necrosis in rats. J Pharm Pharmacol 57:1445–1451
Nilakantan V, Zhou X, Hilton G, Shi Y, Baker JE, Khanna AK, Pieper GM (2006) Antagonizing reactive oxygen by treatment with a manganese (III) metalloporphyrin-based superoxide dismutase mimetic in cardiac transplants. J Thorac Cardiovasc Surg 131:898–906
Hickey AJ, Chai CC, Choong SY, de Freitas CS, Skea GL, Phillips AR, Cooper GJ (2009) Impaired ATP turnover and ADP supply depress cardiac mitochondrial respiration and elevate superoxide in nonfailing spontaneously hypertensive rat hearts. Am J Physiol Cell Physiol 297:C766–C774
Nicotera P, Leist M, Ferrando-May E (1998) Intracellular ATP, a switch in the decision between apoptosis and necrosis. Toxicol Lett 102–103:139–142
Gozal E, Sachleben LR Jr, Rane MJ, Vega C, Gozal D (2005) Mild sustained and intermittent hypoxia induce apoptosis in PC-12 cells via different mechanisms. Am J Physiol Cell Physiol 288:C535–C542
Dong Z, Saikumar P, Weinberg JM, Venkatachalam MA (1997) Internucleosomal DNA cleavage triggered by plasma membrane damage during necrotic cell death. Involvement of serine but not cysteine proteases. Am J Pathol 151:1205–1213
Tanaka S, Takehashi M, Iida S, Kitajima T, Kamanaka Y, Stedeford T, Banasik M, Ueda K (2005) Mitochondrial impairment induced by poly(ADP-ribose) polymerase-1 activation in cortical neurons after oxygen and glucose deprivation. J Neurochem 95:179–190
Pieper AA, Walles T, Wei G, Clements EE, Verma A, Snyder SH, Zweier JL (2000) Myocardial postischemic injury is reduced by polyADPripose polymerase-1 gene disruption. Mol Med 6:271–282
Samali A, Nordgren H, Zhivotovsky B, Peterson E, Orrenius S (1999) A comparative study of apoptosis and necrosis in HepG2 cells: oxidant-induced caspase inactivation leads to necrosis. Biochem Biophys Res Commun 255:6–11
Ohkuwa T, Itoh H, Yamamoto T, Minami C, Yamazaki Y (2005) Effect of hypoxia on norepinephrine of various tissues in rats. Wilderness Environ Med 16:22–26
Communal C, Singh K, Pimentel DR, Colucci WS (1998) Norepinephrine stimulates apoptosis in adult rat ventricular myocytes by activation of the beta-adrenergic pathway. Circulation 98:1329–1334
Mann DL, Kent RL, Parsons B, Cooper G 4th (1992) Adrenergic effects on the biology of the adult mammalian cardiocyte. Circulation 85:790–804
Bohm M, Castellano M, Paul M, Erdmann E (1994) Cardiac norepinephrine, beta-adrenoceptors, and Gi alpha-proteins in prehypertensive and hypertensive spontaneously hypertensive rats. J Cardiovasc Pharmacol 23:980–987
Narula J, Kharbanda S, Khaw BA (1997) Apoptosis and the heart. Chest 112:1358–1362
Itoh G, Tamura J, Suzuki M, Suzuki Y, Ikeda H, Koike M, Nomura M, Jie T, Ito K (1995) DNA fragmentation of human infarcted myocardial cells demonstrated by the nick end labeling method and DNA agarose gel electrophoresis. Am J Pathol 146:1325–1331
Clarke M, Bennett M, Littlewood T (2007) Cell death in the cardiovascular system. Heart 93:659–664
Gandhi MS, Kamalov G, Shahbaz AU, Bhattacharya SK, Ahokas RA, Sun Y, Gerling IC, Weber KT (2010) Cellular and molecular pathways to myocardial necrosis and replacement fibrosis. Heart Fail Rev (in press)
Flotats A, Carrio I (2003) Non-invasive in vivo imaging of myocardial apoptosis and necrosis. Eur J Nucl Med Mol Imaging 30:615–630
Davies MJ (2000) The cardiomyopathies: an overview. Heart 83:469–474
Prabhakar NR (2001) Oxygen sensing during intermittent hypoxia: cellular and molecular mechanisms. J Appl Physiol 90:1986–1994
Lerman LO, Chade AR, Sica V, Napoli C (2005) Animal models of hypertension: an overview. J Lab Clin Med 146:160–173
Sarikonda KV, Watson RE, Opara OC, Dipette DJ (2009) Experimental animal models of hypertension. J Am Soc Hypertens 3:158–165
Acknowledgements
This study was supported by the National Science Council, Taiwan, grant NSC96-2320-B-320-017-MY2 and by Tzu Chi University, Taiwan, grant TCIRP 95004-05.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, TI., Lai, CJ., Hsieh, CJ. et al. Differences in left ventricular cardiomyocyte loss induced by chronic intermittent hypoxia between spontaneously hypertensive and Wistar–Kyoto rats. Sleep Breath 15, 845–854 (2011). https://doi.org/10.1007/s11325-010-0448-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-010-0448-y