Abstract
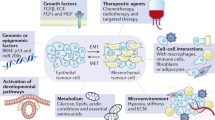
As yet, there is no cure for metastatic breast cancer. Historically, considerable research effort has been concentrated on understanding the processes of metastasis, how a primary tumour locally invades and systemically disseminates using the phenotypic switching mechanism of epithelial to mesenchymal transition (EMT); however, much less is understood about how metastases are then formed. Breast cancer metastases often look (and may even function) as ‘normal’ breast tissue, a bizarre observation against the backdrop of the organ structure of the lung, liver, bone or brain. Mesenchymal to epithelial transition (MET), the opposite of EMT, has been proposed as a mechanism for establishment of the metastatic neoplasm, leading to questions such as: Can MET be clearly demonstrated in vivo? What factors cause this phenotypic switch within the cancer cell? Are these signals/factors derived from the metastatic site (soil) or expressed by the cancer cells themselves (seed)? How do the cancer cells then grow into a detectable secondary tumour and further disseminate? And finally—Can we design and develop therapies that may combat this dissemination switch? This review aims to address these important questions by evaluating long-standing paradigms and novel emerging concepts in the field of epithelial mesencyhmal plasticity.




Similar content being viewed by others
References
Jemal, A., Bray, F., Center, M. M., Ferlay, J., Ward, E., & Forman, D. (2011). Global cancer statistics. CA: A Cancer Journal for Clinicians, 61, 69–90.
Desantis, C., Siegel, R., Bandi, P., & Jemal, A. (2011). Breast cancer statistics, 2011. CA: A Cancer Journal for Clinicians, 61, 408–418.
Jones, S. E. (2008). Metastatic breast cancer: the treatment challenge. Clinical Breast Cancer, 8, 224–233.
Lopez-Tarruella, S., & Martin, M. (2009). Recent advances in systemic therapy: advances in adjuvant systemic chemotherapy of early breast cancer. Breast Cancer Research, 11, 204.
Fisher, B., Jeong, J. H., Bryant, J., et al. (2004). Treatment of lymph-node-negative, oestrogen-receptor-positive breast cancer: long-term findings from National Surgical Adjuvant Breast and Bowel Project randomised clinical trials. Lancet, 364, 858–868.
Early Breast Cancer Trialists' Collaborative Group. (1998). Polychemotherapy for early breast cancer: an overview of the randomised trials. Lancet, 352, 930–942.
Hanahan, D., & Weinberg, R. A. (2000). The hallmarks of cancer. Cell, 100, 57–70.
Woodhouse, E. C., Chuaqui, R. F., & Liotta, L. A. (1997). General mechanisms of metastasis. Cancer, 80, 1529–1537.
Chambers, A. F., Groom, A. C., & MacDonald, I. C. (2002). Dissemination and growth of cancer cells in metastatic sites. Nature Reviews. Cancer, 2, 563–572.
Weidner, N., Folkman, J., Pozza, F., et al. (1992). Tumor angiogenesis: a new significant and independent prognostic indicator in early-stage breast carcinoma. Journal of the National Cancer Institute, 84, 1875–1887.
Folkman, J., & Shing, Y. (1992). Angiogenesis. Journal of Biological Chemistry, 267, 10931–10934.
Folkman, J. (1992). The role of angiogenesis in tumor growth. Seminars in Cancer Biology, 3, 65–71.
Kiaris, H., Chatzistamou, I., Kalofoutis, C., Koutselini, H., Piperi, C., & Kalofoutis, A. (2004). Tumour-stroma interactions in carcinogenesis: basic aspects and perspectives. Molecular and Cellular Biochemistry, 261, 117–122.
Pupa, S. M., Menard, S., Forti, S., & Tagliabue, E. (2002). New insights into the role of extracellular matrix during tumor onset and progression. Journal of Cellular Physiology, 192, 259–267.
Wells, A., Chao, Y. L., Grahovac, J., Wu, Q., & Lauffenburger, D. A. (2011). Epithelial and mesenchymal phenotypic switchings modulate cell motility in metastasis. Frontiers in Bioscience, 16, 815–837.
Kienast, Y., von Baumgarten, L., Fuhrmann, M., et al. (2010). Real-time imaging reveals the single steps of brain metastasis formation. Nature Medicine, 16, 116–122.
Luzzi, K. J., MacDonald, I. C., Schmidt, E. E., et al. (1998). Multistep nature of metastatic inefficiency: dormancy of solitary cells after successful extravasation and limited survival of early micrometastases. American Journal of Pathology, 153, 865–873.
Howlett, A. R., & Bissell, M. J. (1993). The influence of tissue microenvironment (stroma and extracellular matrix) on the development and function of mammary epithelium. Epithelial Cell Biology, 2, 79–89.
Jechlinger, M., Grunert, S., & Beug, H. (2002). Mechanisms in epithelial plasticity and metastasis: insights from 3D cultures and expression profiling. Journal of Mammary Gland Biology and Neoplasia, 7, 415–432.
de Herreros, A. G., Peiro, S., Nassour, M., & Savagner, P. (2010). Snail family regulation and epithelial mesenchymal transitions in breast cancer progression. Journal of Mammary Gland Biology and Neoplasia, 15, 135–147.
Creighton, C. J., Chang, J. C., & Rosen, J. M. (2010). Epithelial-mesenchymal transition (EMT) in tumor-initiating cells and its clinical implications in breast cancer. Journal of Mammary Gland Biology and Neoplasia, 15, 253–260.
Wang, Y., & Zhou, B. P. (2011). Epithelial-mesenchymal transition in breast cancer progression and metastasis. Chinese Journal of Cancer, 30, 603–611.
Chao, Y. L., Shepard, C. R., & Wells, A. (2010). Breast carcinoma cells re-express E-cadherin during mesenchymal to epithelial reverting transition. Molecular Cancer, 9, 179.
Chaffer, C. L., Brennan, J. P., Slavin, J. L., Blick, T., Thompson, E. W., & Williams, E. D. (2006). Mesenchymal-to-epithelial transition facilitates bladder cancer metastasis: role of fibroblast growth factor receptor-2. Cancer Research, 66, 11271–11278.
Chaffer, C. L., Thompson, E. W., & Williams, E. D. (2007). Mesenchymal to epithelial transition in development and disease. Cells, Tissues, Organs, 185, 7–19.
Hugo, H., Ackland, M. L., Blick, T., et al. (2007). Epithelial–mesenchymal and mesenchymal–epithelial transitions in carcinoma progression. Journal of Cellular Physiology, 213, 374–383.
Bernards, R., & Weinberg, R. A. (2002). A progression puzzle. Nature, 418, 823.
Weinberg, R. A. (2008). Leaving home early: reexamination of the canonical models of tumor progression. Cancer Cell, 14, 283–284.
Al-Hajj, M., Wicha, M. S., Benito-Hernandez, A., Morrison, S. J., & Clarke, M. F. (2003). Prospective identification of tumorigenic breast cancer cells. Proceedings of the National Academy of Sciences of the United States of America, 100, 3983–3988.
Blick, T., Hugo, H., Widodo, E., et al. (2010). Epithelial mesenchymal transition traits in human breast cancer cell lines parallel the CD44(hi/)CD24 (lo/-) stem cell phenotype in human breast cancer. Journal of Mammary Gland Biology and Neoplasia, 15, 235–252.
Mani, S. A., Guo, W., Liao, M. J., et al. (2008). The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell, 133, 704–715.
Kowalski, P. J., Rubin, M. A., & Kleer, C. G. (2003). E-cadherin expression in primary carcinomas of the breast and its distant metastases. Breast Cancer Research, 5, R217–R222.
Stessels, F., Van den Eynden, G., Van der Auwera, I., et al. (2004). Breast adenocarcinoma liver metastases, in contrast to colorectal cancer liver metastases, display a non-angiogenic growth pattern that preserves the stroma and lacks hypoxia. British Journal of Cancer, 90, 1429–1436.
Chao, Y., Wu, Q., Acquafondata, M., Dhir, R., & Wells, A. (2012). Partial mesenchymal to epithelial reverting transition in breast and prostate cancer metastases. Cancer Microenvironment, 5, 19–28.
Korpal, M., Ell, B. J., Buffa, F. M., et al. (2011). Direct targeting of Sec23a by miR-200s influences cancer cell secretome and promotes metastatic colonization. Nature Medicine, 17, 1101–1108.
Hurteau, G. J., Carlson, J. A., Spivack, S. D., & Brock, G. J. (2007). Overexpression of the microRNA hsa-miR-200c leads to reduced expression of transcription factor 8 and increased expression of E-cadherin. Cancer Research, 67, 7972–7976.
Bendoraite, A., Knouf, E. C., Garg, K. S., et al. (2010). Regulation of miR-200 family microRNAs and ZEB transcription factors in ovarian cancer: evidence supporting a mesothelial-to-epithelial transition. Gynecologic Oncology, 116, 117–125.
Brabletz, S., & Brabletz, T. (2010). The ZEB/miR-200 feedback loop—a motor of cellular plasticity in development and cancer? EMBO Reports, 11, 670–677.
Gregory, P. A., Bracken, C. P., Smith, E., et al. (2011). An autocrine TGF-beta/ZEB/miR-200 signaling network regulates establishment and maintenance of epithelial-mesenchymal transition. Molecular Biology of the Cell, 22, 1686–1698.
Burk, U., Schubert, J., Wellner, U., et al. (2008). A reciprocal repression between ZEB1 and members of the miR-200 family promotes EMT and invasion in cancer cells. EMBO Reports, 9, 582–589.
Bullock, M. D., Sayan, A. E., Packham, G. K., & Mirnezami, A. H. (2012). MicroRNAs: critical regulators of epithelial to mesenchymal (EMT) and mesenchymal to epithelial transition (MET) in cancer progression. Biology of the Cell, 104, 3–12.
Celia-Terrassa, T., Meca-Cortes, O., Mateo, F., et al. (2012). Epithelial-mesenchymal transition can suppress major attributes of human epithelial tumor-initiating cells. The Journal of Clinical Investigation, 122, 1849–1868.
Sporn, M. B. (1996). The war on cancer. Lancet, 347, 1377–1381.
Mettlin, C. (1999). Global breast cancer mortality statistics. CA: A Cancer Journal for Clinicians, 49, 138–144.
No authors listed (2000). Breast cancer statistics. Journal of the National Cancer Institute, 92, 445.
Kamo, K., & Sobue, T. (2004). Cancer statistics digest. Mortality trend of prostate, breast, uterus, ovary, bladder and “kidney and other urinary tract” cancer in Japan by birth cohort. Japanese Journal of Clinical Oncology, 34, 561–563.
Birchmeier, W., & Behrens, J. (1994). Cadherin expression in carcinomas: role in the formation of cell junctions and the prevention of invasiveness. Biochimica et Biophysica Acta, 1198, 11–26.
Berx, G., Staes, K., van Hengel, J., et al. (1995). Cloning and characterization of the human invasion suppressor gene E-cadherin (CDH1). Genomics, 26, 281–289.
Pecina-Slaus, N. (2003). Tumor suppressor gene E-cadherin and its role in normal and malignant cells. Cancer Cell International, 3, 17.
Perl, A. K., Wilgenbus, P., Dahl, U., Semb, H., & Christofori, G. (1998). A causal role for E-cadherin in the transition from adenoma to carcinoma. Nature, 392, 190–193.
Wells, A., Yates, C., & Shepard, C. R. (2008). E-cadherin as an indicator of mesenchymal to epithelial reverting transitions during the metastatic seeding of disseminated carcinomas. Clinical & Experimental Metastasis, 25, 621–628.
Saha, B., Chaiwun, B., Imam, S. S., et al. (2007). Overexpression of E-cadherin protein in metastatic breast cancer cells in bone. Anticancer Research, 27, 3903–3908.
Bastid, J. (2012). EMT in carcinoma progression and dissemination: facts, unanswered questions, and clinical considerations. Cancer and Metastasis Reviews, 31, 277–283.
Wendt, M. K., Taylor, M. A., Schiemann, B. J., & Schiemann, W. P. (2011). Down-regulation of epithelial cadherin is required to initiate metastatic outgrowth of breast cancer. Molecular Biology of the Cell, 22, 2423–2435.
Cailleau, R., Olive, M., & Cruciger, Q. V. (1978). Long-term human breast carcinoma cell lines of metastatic origin: preliminary characterization. In Vitro, 14, 911–915.
Brinkley, B. R., Beall, P. T., Wible, L. J., Mace, M. L., Turner, D. S., & Cailleau, R. M. (1980). Variations in cell form and cytoskeleton in human breast carcinoma cells in vitro. Cancer Research, 40, 3118–3129.
Thompson, E. W., Paik, S., Brunner, N., et al. (1992). Association of increased basement membrane invasiveness with absence of estrogen receptor and expression of vimentin in human breast cancer cell lines. Journal of Cellular Physiology, 150, 534–544.
Sheikh, M. S., Shao, Z. M., Hussain, A., & Fontana, J. A. (1993). The p53-binding protein MDM2 gene is differentially expressed in human breast carcinoma. Cancer Research, 53, 3226–3228.
Maemura, M., Akiyama, S. K., Woods, V. L., Jr., & Dickson, R. B. (1995). Expression and ligand binding of alpha 2 beta 1 integrin on breast carcinoma cells. Clinical & Experimental Metastasis, 13, 223–235.
Hiraguri, S., Godfrey, T., Nakamura, H., et al. (1998). Mechanisms of inactivation of E-cadherin in breast cancer cell lines. Cancer Research, 58, 1972–1977.
Pishvaian, M. J., Feltes, C. M., Thompson, P., Bussemakers, M. J., Schalken, J. A., & Byers, S. W. (1999). Cadherin-11 is expressed in invasive breast cancer cell lines. Cancer Research, 59, 947–952.
Jo, M., Lester, R. D., Montel, V., Eastman, B., Takimoto, S., & Gonias, S. L. (2009). Reversibility of epithelial-mesenchymal transition (EMT) induced in breast cancer cells by activation of urokinase receptor-dependent cell signaling. Journal of Biological Chemistry, 284, 22825–22833.
Lester, R. D., Jo, M., Montel, V., Takimoto, S., & Gonias, S. L. (2007). uPAR induces epithelial-mesenchymal transition in hypoxic breast cancer cells. The Journal of Cell Biology, 178, 425–436.
Lo, H. W., Hsu, S. C., Xia, W., et al. (2007). Epidermal growth factor receptor cooperates with signal transducer and activator of transcription 3 to induce epithelial-mesenchymal transition in cancer cells via up-regulation of TWIST gene expression. Cancer Research, 67, 9066–9076.
Bonnomet, A., Syne, L., Brysse, A., et al. (2011). A dynamic in vivo model of epithelial-to-mesenchymal transitions in circulating tumor cells and metastases of breast cancer. Oncogene. (In press)
Lee, J. M., Dedhar, S., Kalluri, R., & Thompson, E. W. (2006). The epithelial-mesenchymal transition: new insights in signaling, development, and disease. The Journal of Cell Biology, 172, 973–981.
Klymkowsky, M. W., & Savagner, P. (2009). Epithelial-mesenchymal transition: a cancer researcher's conceptual friend and foe. American Journal of Pathology, 174, 1588–1593.
Martinez, V., & Azzopardi, J. G. (1979). Invasive lobular carcinoma of the breast: incidence and variants. Histopathology, 3, 467–488.
DiCostanzo, D., Rosen, P. P., Gareen, I., Franklin, S., & Lesser, M. (1990). Prognosis in infiltrating lobular carcinoma. An analysis of “classical” and variant tumors. The American Journal of Surgical Pathology, 14, 12–23.
Da Silva, L., Parry, S., Reid, L., et al. (2008). Aberrant expression of E-cadherin in lobular carcinomas of the breast. The American Journal of Surgical Pathology, 32, 773–783.
Oltean, S., Sorg, B. S., Albrecht, T., et al. (2006). Alternative inclusion of fibroblast growth factor receptor 2 exon IIIc in Dunning prostate tumors reveals unexpected epithelial mesenchymal plasticity. Proceedings of the National Academy of Sciences of the United States of America, 103, 14116–14121.
Oltean, S., Febbo, P. G., & Garcia-Blanco, M. A. (2008). Dunning rat prostate adenocarcinomas and alternative splicing reporters: powerful tools to study epithelial plasticity in prostate tumors in vivo. Clinical & Experimental Metastasis, 25, 611–619.
Tsuji, T., Ibaragi, S., Shima, K., et al. (2008). Epithelial-mesenchymal transition induced by growth suppressor p12CDK2-AP1 promotes tumor cell local invasion but suppresses distant colony growth. Cancer Research, 68, 10377–10386.
Martorana, A. M., Zheng, G., Crowe, T. C., O’Grady, R. L., & Lyons, J. G. (1998). Epithelial cells up-regulate matrix metalloproteinases in cells within the same mammary carcinoma that have undergone an epithelial-mesenchymal transition. Cancer Research, 58, 4970–4979.
Kleer, C. G., van Golen, K. L., Braun, T., & Merajver, S. D. (2001). Persistent E-cadherin expression in inflammatory breast cancer. Modern Pathology, 14, 458–464.
Lang, S. H., Sharrard, R. M., Stark, M., Villette, J. M., & Maitland, N. J. (2001). Prostate epithelial cell lines form spheroids with evidence of glandular differentiation in three-dimensional Matrigel cultures. British Journal of Cancer, 85, 590–599.
Kurahara, H., Takao, S., Maemura, K., et al. (2012). Epithelial-mesenchymal transition and mesenchymal-epithelial transition via regulation of ZEB-1 and ZEB-2 expression in pancreatic cancer. Journal of Surgical Oncology, 105, 655–61.
Chao, Y., Wu, Q., Shepard, C., & Wells, A. (2012). Hepatocyte induced re-expression of E-cadherin in breast and prostate cancer cells increases chemoresistance. Clinical & Experimental Metastasis, 29, 39–50.
Yates, C. C., Shepard, C. R., Stolz, D. B., & Wells, A. (2007). Co-culturing human prostate carcinoma cells with hepatocytes leads to increased expression of E-cadherin. British Journal of Cancer, 96, 1246–1252.
Lopes, N., Carvalho, J., Duraes, C., et al. (2012). 1Alpha,25-dihydroxyvitamin D3 induces de novo E-cadherin expression in triple-negative breast cancer cells by CDH1-promoter demethylation. Anticancer Research, 32, 249–257.
Yilmaz, M., & Christofori, G. (2009). EMT, the cytoskeleton, and cancer cell invasion. Cancer and Metastasis Reviews, 28, 15–33.
Valastyan, S., & Weinberg, R. A. (2011). Tumor metastasis: molecular insights and evolving paradigms. Cell, 147, 275–292.
Yang, J., & Weinberg, R. A. (2008). Epithelial-mesenchymal transition: at the crossroads of development and tumor metastasis. Developmental Cell, 14, 818–829.
Jung, A., Schrauder, M., Oswald, U., et al. (2001). The invasion front of human colorectal adenocarcinomas shows co-localization of nuclear beta-catenin, cyclin D1, and p16INK4A and is a region of low proliferation. American Journal of Pathology, 159, 1613–1617.
Vega, S., Morales, A. V., Ocana, O. H., Valdes, F., Fabregat, I., & Nieto, M. A. (2004). Snail blocks the cell cycle and confers resistance to cell death. Genes & Development, 18, 1131–1143.
Mejlvang, J., Kriajevska, M., Vandewalle, C., et al. (2007). Direct repression of cyclin D1 by SIP1 attenuates cell cycle progression in cells undergoing an epithelial mesenchymal transition. Molecular Biology of the Cell, 18, 4615–4624.
Rubio, C. A. (2006). Cell proliferation at the leading invasive front of colonic carcinomas. Preliminary observations. Anticancer Research, 26, 2275–2278.
Rubio, C. A. (2007). Further studies on the arrest of cell proliferation in tumor cells at the invading front of colonic adenocarcinoma. Journal of Gastroenterology and Hepatology, 22, 1877–1881.
Spaderna, S., Schmalhofer, O., Hlubek, F., et al. (2006). A transient, EMT-linked loss of basement membranes indicates metastasis and poor survival in colorectal cancer. Gastroenterology, 131, 830–840.
Gao, D., Joshi, N., Choi, H., et al. (2012). Myeloid progenitor cells in the premetastatic lung promote metastases by inducing mesenchymal to epithelial transition. Cancer Research, 72, 1384–94.
Thiery, J. P., Acloque, H., Huang, R. Y., & Nieto, M. A. (2009). Epithelial-mesenchymal transitions in development and disease. Cell, 139, 871–890.
Thomson, S., Buck, E., Petti, F., et al. (2005). Epithelial to mesenchymal transition is a determinant of sensitivity of non-small-cell lung carcinoma cell lines and xenografts to epidermal growth factor receptor inhibition. Cancer Research, 65, 9455–9462.
Thomson, S., Petti, F., Sujka-Kwok, I., Epstein, D., & Haley, J. D. (2008). Kinase switching in mesenchymal-like non-small cell lung cancer lines contributes to EGFR inhibitor resistance through pathway redundancy. Clinical & Experimental Metastasis, 25, 843–854.
Yauch, R. L., Januario, T., Eberhard, D. A., et al. (2005). Epithelial versus mesenchymal phenotype determines in vitro sensitivity and predicts clinical activity of erlotinib in lung cancer patients. Clinical Cancer Research, 11, 8686–8698.
Creighton, C. J., Reid, J. G., & Gunaratne, P. H. (2009). Expression profiling of microRNAs by deep sequencing. Briefings in Bioinformatics, 10, 490–497.
Cooke, V. G., LeBleu, V. S., Keskin, D., et al. (2012). Pericyte depletion results in hypoxia-associated epithelial-to-mesenchymal transition and metastasis mediated by MET signaling pathway. Cancer Cell, 21, 66–81.
Valdes, F., Alvarez, A. M., Locascio, A., et al. (2002). The epithelial mesenchymal transition confers resistance to the apoptotic effects of transforming growth factor beta in fetal rat hepatocytes. Molecular Cancer Research, 1, 68–78.
Ansieau, S., Bastid, J., Doreau, A., et al. (2008). Induction of EMT by twist proteins as a collateral effect of tumor-promoting inactivation of premature senescence. Cancer Cell, 14, 79–89.
Gal, A., Sjoblom, T., Fedorova, L., Imreh, S., Beug, H., & Moustakas, A. (2008). Sustained TGF beta exposure suppresses Smad and non-Smad signalling in mammary epithelial cells, leading to EMT and inhibition of growth arrest and apoptosis. Oncogene, 27, 1218–1230.
Sayan, A. E., Griffiths, T. R., Pal, R., et al. (2009). SIP1 protein protects cells from DNA damage-induced apoptosis and has independent prognostic value in bladder cancer. Proceedings of the National Academy of Sciences of the United States of America, 106, 14884–14889.
Straub, B. K., Rickelt, S., Zimbelmann, R., et al. (2011). E-N-cadherin heterodimers define novel adherens junctions connecting endoderm-derived cells. The Journal of Cell Biology, 195, 873–887.
Thiery, J. P. (2002). Epithelial to mesenchymal transitions in tumour progression. Nature Cancer, 2, 442–454.
Acknowledgments
The authors wish to thank members of the Thompson and Wells laboratories for ideas and discussions which shaped the work and conception of this review and gratefully acknowledge the following sources of financial support: the National Breast Cancer Foundation, particularly the National Collaborative Research Program (EMPathy Breast Cancer Network) (Australia), Cancer Council Victoria, The Australian Government's Endeavour Awards Scholarship Program, the Victorian Government's Operational Infrastructure Support Program, the DoD CDMRP on Breast Cancer and the VA Merit Award Program, USA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gunasinghe, N.P.A.D., Wells, A., Thompson, E.W. et al. Mesenchymal–epithelial transition (MET) as a mechanism for metastatic colonisation in breast cancer. Cancer Metastasis Rev 31, 469–478 (2012). https://doi.org/10.1007/s10555-012-9377-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10555-012-9377-5