Abstract
The rep1 gene of the maize pathogen Ustilago maydis encodes a pre-pro-protein that is processed in the secretory pathway into 11 peptides. These so-called repellents form amphipathic amyloid fibrils at the surface of aerial hyphae. A SG200 strain in which the rep1 gene is inactivated (∆rep1 strain) is affected in aerial hyphae formation. We here assessed changes in global gene expression as a consequence of the inactivation of the rep1 gene. Microarray analysis revealed that only 31 genes in the ∆rep1 SG200 strain had a fold change in expression of ≥2. Twenty-two of these genes were up-regulated and half of them encode small secreted proteins (SSPs) with unknown functions. Seven of the SSP genes and two other genes that are over-expressed in the ∆rep1 SG200 strain encode proteins that can be classified as secreted cysteine-rich proteins (SCRPs). Interestingly, most of the SCRPs are predicted to form amyloids. The SCRP gene um00792 showed the highest up-regulation in the ∆rep1 strain. Using GFP as a reporter, it was shown that this gene is over-expressed in the layer of hyphae at the medium-air interface. Taken together, it is concluded that inactivation of rep1 hardly affects the expression profile of U. maydis, despite the fact that the mutant strain has a strong reduced ability to form aerial hyphae.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The life cycle of the pathogenic heterobasidiomycete Ustilago maydis is characterized by distinct morphological and nuclear states. Fusion of compatible yeast-like sporidia results in the formation of a pathogenic filamentous dikaryon. Upon formation of an appressorium the host (Zea mays [maize] and Euchlaena mexicana [Mexican teosinte]) is invaded. The fungus proliferates and branches inside the plant tissue, resulting in the formation of diploid teliospores. These spores are dispersed into the environment. After germination, meiosis occurs resulting in the formation of haploid sporidia (Banuett and Herskowitz 1988; Banuett 1992; Christensen 1963).
Only when two partners harbor different alleles of the a- and b-loci, mating and pathogenic development can occur. Cell fusion is controlled by the a1 and a2 mating-type loci (Bölker et al. 1992). These loci encode a pheromone and a receptor for the pheromone of the opposite mating type. The multi-allelic b locus regulates the post-mating processes of filament formation and pathogenic development. The b locus encodes two unrelated homeodomain proteins, bE and bW, that form active hetero-dimers when derived from different alleles (Gillissen et al. 1992; Kämper et al. 1995).
Rep1 is one of the genes that is regulated by the bE/bW heterodimer (Bohlmann 1996; Brachmann et al. 2001; Romeis et al. 2000; Wösten et al. 1996). The rep1 gene encodes a pre-pro-protein, which consists of a signal sequence for secretion and 12 repeated sequences. Processing at KEX2 recognition sites in the secretory pathway results in 10 peptides of 34–53 amino acids and one larger protein of 229 amino acids (Wösten et al. 1996). These cleavage products that are collectively known as repellents are secreted into the cell wall of filaments. Here they form amyloid fibrils (Teertstra et al. 2009), which are involved in attachment to hydrophobic surfaces and in formation of hydrophobic aerial hyphae (Teertstra et al. 2006, 2009; Wösten et al. 1996).
Strains in which the rep1 gene has been inactivated form colonies with only few aerial hyphae (Teertstra et al. 2006; Wösten et al. 1996). We here show that expression of only 31 genes is changed at least two-fold in the ∆rep1 strain when compared to the parental SG200 strain under conditions of aerial growth. Most of these genes are up-regulated and encode for small secreted proteins without a predicted enzymatic function.
Methods
Strains and growth conditions
Ustilago maydis strain SG200 (a1mfa2 bE1/bW2) (Bölker et al. 1995) and its derivatives (see below) were used in this study. SG200 is a haploid FB1 (a1b1) strain (Banuett and Herskowitz 1989), which harbors an active b mating type complex. Consequently, the strain grows yeast-like in liquid medium and forms filaments at the water–air interface without the need for mating. U. maydis strains were routinely grown at 25°C using liquid YEPSL medium (0.4% yeast extract, 0.4% peptone, 2% sucrose) or potato dextrose agar (PDA, Sigma) that had either or not been supplemented with 1% (w/v) charcoal. For isolation of RNA for microarray analysis, cells were grown at 22°C on nitrate minimal medium (NM+; Holliday 1974) containing 2% agar (w/v), 1% charcoal (w/v), 20 mg l−1 histidine, 380 mg l−1 leucine, 20 mg l−1 tryptophan, 50 mg l−1 uracil and 76 mg l−1 Yeast Synthetic Drop-out Medium Supplements (Sigma, Y2001). For GFP expression analysis, 1 μl of cell suspension (2 × 107 cells ml−1) was seeded on each side of a 0.25 mm thin 20 × 20 mm square of solidified (1.5% agarose) NM+ medium that was sandwiched between a glass slide and a cover slip. Cells were grown at 25°C under humid conditions.
Constructs
SG200Δrep1 was generated as described (Müller et al. 2008a). The PCR fragment that was used to inactivate rep1 was amplified with oligonucleotide primers RepMUF-fw and RepMDF-rev (Table 1) using Phusion™ polymerase (Finnzymes). The resulting PCR fragment, consisting of a nourseothricin resistance cassette (Brachmann et al. 2004) flanked by the upstream and downstream sequences of the rep1 gene, was used to transform the SG200 strain.
Vector pUC19-Rep-c was used to complement the SG200Δrep1 strain. A 3674 bp PCR fragment was amplified for its construction. This fragment consisted of a 1680 bp rep1 promoter region and the coding sequence of this gene. For this, Phusion™ polymerase (Finnzymes) was used with genomic FB1 DNA as a template and oligonucleotide primers pRep-fw and cRep-rev (Table 1). The latter primer introduces a NotI-site at the 3′ end. The 3674 bp PCR-fragment was introduced in the SmaI-site of pUC19. In the next step, a NotI fragment encompassing a carboxin-resistance cassette (Brachmann et al. 2004) was introduced into the respective site of the pUC19 derivative, resulting in pUC19-Rep-c.
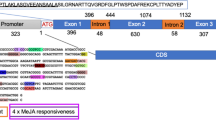
The promoter region of gene um00792 was amplified from genomic DNA of FB1 using PhusionTM polymerase (Finnzymes) with oligonucleotide primers pr792-fw and pr792-rev (Table 1). The resulting 1837 bp fragment was introduced in the SmaI site of pUC19, resulting in vector pUC19-pr792. The 1791 bp ApaI/NcoI promoter fragment of um00792 was digested from pUC19-pr792 and introduced in the corresponding sites of pMF3c (Brachmann et al. 2004). As a result, enhanced GFP (eGFP) was placed under control of the um00792 promoter. The resulting construct pMF3c-pr792 was linearized in the carboxin resistance cassette with AgeI, thus targeting the DNA to the sdh2-locus of U. maydis upon transformation.
Transformation of U. maydis
Ustilago maydis was transformed according to Brachmann et al. (Brachmann et al. 2004). Transformants were selected on PDA plates supplemented with nourseothricin (150 μg ml−1) and carboxin (2 μg ml−1), respectively. Deletion of the rep1 gene was confirmed by Southern analysis. Chromosomal DNA of U. maydis was isolated as described (Hoffman and Winston 1987) and blotted onto Hybond N+ (Amersham). Hybridization was performed at 60°C in 0.5 M phosphate buffer, pH 7.2, containing 7% SDS and 10 mM EDTA. A 936 bp 32P-α-dCTP labeled PCR-fragment was used as a probe, which was derived from the rep1 promoter region of FB1 using the primers RepUF-fw and RepUF-rev (Table 1).
Northern analysis
Ustilago maydis cultures were homogenized with a microdismembrator (B.Braun). RNA was extracted with Trizol (Invitrogen) according to the manufacturer’s protocol. Hybridization was performed using standard protocols (Sambrook et al. 1989). 32P-α-dCTP labeled fragments of genes um00792, um00913 and um03817 were used as a probe.
Microarray analysis
2 × 107 cells of exponentially growing cultures of strains SG200 and SG200Δrep1 were seeded on solid NM+-charcoal medium and grown for 48 h. Cells were harvested, frozen in liquid nitrogen, and homogenized with a dismembrator (Retsch). RNA was isolated with Trizol, after which double stranded cDNA was made according to the Affymetrix protocol (Affymetrix). The cDNA was purified over a cDNA clean up spin column, after which biotin labeled antisense cRNA was obtained using the Enzo BioArray HighYield RNA transcript labeling kit (ENZO Diagnostics). Concentration of the cRNA was determined using the NanoDrop N-1000 (Thermo Scientific) and quality of the cRNA was determined using the 2100 Bioanalyzer (Agilent Technologies). cRNA was fragmented according to the Affymetrix protocol and the resulting 30–200 bp fragments were hybridized to the Affymetrix custom array (MIPIUstilagoA). Array data have been deposited in NCBI’s Gene Expression Omnibus (Edgar et al. 2002) and are accessible through GEO Series accession number GSE21490 (http://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE21490). Data analysis of biological triplicates was performed as described previously (Eichhorn et al. 2006). Genes with a P value of < 0.01, whose mean expression was at least 2-fold changed, were filtered in Excel.
Analysis of protein sequences
Protein sequences of U. maydis were obtained from the MUMDB database (http://mips.gsf.de/genre/proj/ustilago) and analyzed for functional domains with SCOP (Murzin et al. 1995), SMART (Ponting et al. 1999), PFAM (Bateman et al. 2002), SignalP (Bendtsen et al. 2004), TANGO (Fernandez-Escamilla et al. 2004) and Waltz (Maurer-Stroh et al. 2010).
Fluorescence microscopy
Fluorescence microscopy was carried out using a Zeiss Axioscope 2PLUS equipped with a HBO 100 W mercury lamp and a Leica LFC 420C camera (2592 × 1944 pixels). GFP fluorescence was monitored using a FITC filter set.
Results
Absence of repellents has a minor effect on overall gene expression in U. maydis
Gene rep1 was deleted in strain SG200 by introducing a PCR-fragment consisting of a nourseothricin resistance cassette flanked by upstream and downstream sequences of rep1 (see Methods). Five out of 12 transformants showed loss of surface hydrophobicity and reduction of aerial hyphae formation on PDA charcoal. Southern analysis confirmed deletion of rep1 in these strains. Introduction of pUC19-Rep-c encompassing the rep1 gene restored formation of aerial hyphae, showing that the phenotype was solely caused by the gene deletion (Data not shown).
To monitor the effect of the rep1 deletion on overall gene expression during aerial hyphae formation, DNA microarrays (representing ~90% of the genes of U. maydis) were hybridized with RNA from SG200 and a SG200∆rep1 strain. To this end, both strains had been grown for 2 days on a synthetic medium containing charcoal. SG200 formed abundant aerial hyphae, whereas aerial hyphae formation in the mutant strain was severely affected (Fig. 1b). Thirty-one genes showed a fold change ≥2 when expression of the two strains was compared. Twenty-two of these genes were up-regulated, whereas 9 were down-regulated in the SG200∆rep1 strain (Table 2). The microarray data were confirmed with Northern analysis by probing for 3 genes (Fig. 1a). This showed that expression of genes um00792 and um00913 was increased in the SG200∆rep1 strain compared to the parental strain at least 6-fold and 3-fold, respectively, whereas expression of gene um03817 was decreased at least 10-fold. Expression of genes um00792 and um00913 was higher on PDA than on NM+-charcoal, which correlated with the number of aerial hyphae that were formed. In contrast, expression of um03817 was more pronounced on solidified NM+-charcoal. This indicates that expression of at least um03817 is not only influenced by the rep1 deletion, but also by the composition of the medium.
Inactivation of rep1 results in reduced aerial hyphae formation and in changes in gene expression. a Northern analysis of genes um00792, um00913 and um03817 in strains SG200 and SG200Δrep1, 18S rRNA serving as a control. Cultures were grown on PDA-charcoal and NM+-charcoal for 48 h. b Detail of colonies of the SG200 and SG200Δrep1 strain grown on PDA and NM+-charcoal plates. Colonies of strain SG200 form abundant aerial hyphae, whereas in the rep1 deletion strain aerial hyphae formation is severely reduced. Bar represents 0.5 mm
Of the genes that were down-regulated in the SG200∆rep1 strain, 2 encode secreted proteins (Table 2). One of these genes is predicted to have enzymatic activity. This gene, um11112, is related to genes encoding Versicolorin B synthase. The U. maydis gene is proposed to encode a protein with aryl-alcohol oxidase activity (Müller et al. 2008b). The other gene, um04248, encodes a repetitive protein with no similarity to known proteins (Müller et al. 2008a). Eighteen out of the 22 genes with an increased expression in the SG200∆rep1 strain encode secreted proteins (Table 2), as predicted by SignalP and TargetP (see Müller et al. 2008b). Several of these genes have already been deleted in U. maydis, either as a single or part of a multiple gene deletion (Table 2; Kämper et al. 2006; Leuthner et al. 2005; Vraneš 2006). Two up-regulated genes encode secreted proteins with an enzymatic function. Gene um00913 encodes glyoxaloxidase 2 (Leuthner et al. 2005), whereas um04422 is predicted to encode an endo-xylanase (Müller et al. 2008b). Remarkably, the other 16 genes encode secreted proteins with no apparent enzymatic function. Eleven of them belong to the small secreted proteins (SSPs), which have an arbitrary size limit of 300 amino acids. Of these genes, 7 encode secreted cysteine-rich proteins (SCRPs), whereas another 2 up-regulated SCRPs exceed 300 aa (Table 2; Müller et al. 2008b). The TANGO and Waltz algorithms were used to assess whether the 16 secreted proteins that are predicted not to have an enzymatic function have the potential to form amyloid fibrils. The TANGO algorithm predicts regions within a protein that are prone to intermolecular β-sheet formation. These intermolecular β-sheets can result in protein aggregation (Fernandez-Escamilla et al. 2004). It was found that five consecutive residues with a TANGO score above 5% are a good predictor of aggregation (Fernandez-Escamilla et al. 2004). Using standard settings (see http://tango.crg.es/), 14 out of the 16 mature proteins had one or more of these aggregation prone regions and 5 of them had 4 or more. The Waltz algorithm is a position-specific scoring matrix that has been developed to distinguish between amyloid sequences and amorphous β-sheet aggregates (Maurer-Stroh et al. 2010). Using standard settings (http://waltz.switchlab.org/), it was shown that 15 out of the 16 proteins had between 1 and 6 amylogenic regions. Only Mig2-6 that is encoded by um06126 (Zheng et al. 2008) was found not to have such a region. Taken together, the TANGO and Waltz algorithms predict that most of the proteins that are encoded by the up-regulated genes within the rep1 mutant have the propensity to form amyloids.
Gene um00792 is highly induced in filaments of the SG200∆rep1 strain that grow at the water–air interface.
The SCRP gene um00792 shows the highest change in expression when strains SG200 and SG200∆rep1 are compared by micro-array analysis (Table 2). Its expression is 8.75 fold increased in the SG200∆rep1 strain. Expression of this gene was monitored using GFP as a reporter. To this end, strains SG200 and SG200∆rep1 were transformed with construct pMF3c-pr792. This vector contains the eGFP gene, which is placed behind the um00792 promoter. Transformants uG114 and uG115, which are derivatives of SG200 and SG200∆rep1, respectively, showed a fluorescence pattern representative for the majority of the fluorescent transformants. These strains were therefore selected for further study. They were grown along a thin slab of nitrate minimal medium, sandwiched between an object glass and a cover slip. Aerial hyphae and hyphae growing in the substrate showed similar GFP fluorescence in strains uG114 and uG115 (Fig. 2a). However, at the medium-air interface, a 10-fold increase in fluorescence was shown in the SG200∆rep1 derivative uG115 when compared to the wild-type derivative uG114. This layer consists of yeast cells and filaments that have not yet escaped into the air (Fig. 2b, c). Both cell types within this layer showed fluorescence. In contrast, gene expression was not detected in yeast cells in liquid shaken cultures (data not shown).
Localization of expression of um00792 in the SG200 derivative uG114 and the SG200Δrep1 derivative uG115 using eGFP as a reporter. Cells were seeded along a thin layer of solidified medium between a glass slide and a cover slip and grown for 40 h. Cells were monitored by phase contrast and fluorescence microscopy. a Individual hyphae of strain uG114 (A, B) and uG115 (C, D) growing in the substrate (A, C) or in the air (B, D) show similar fluorescence. b Strain uG114 (A, C) and strain uG115 (B, D) growing along the solidified medium visualized by fluorescence (A, B) and light (C, D) microscopy. Total fluorescence of the top layer, consisting of yeasts and developing hyphae, is highly increased in the uG115 strain (B). c Detail of (b, B). Bar represents 100 μm
Discussion
Strains in which the rep1 gene is inactivated form few aerial hyphae (Teertstra et al. 2006; Wösten et al. 1996). We here assessed global changes in gene expression as a consequence of the inactivation of the rep1 gene. It is shown that deletion of this gene has a minor effect on global gene expression under conditions of aerial growth in the wild-type. Interestingly, a majority of the genes that are up-regulated at least two-fold encode small secreted proteins that are predicted to form amyloids.
Gene expression at the stage of aerial hyphae formation was monitored with microarray analysis. Only 31 genes had a fold change in expression of at least two when expression profiles of strains SG200 and SG200Δrep1 were compared. Of these genes, 7 had a fold change of at least 4 and only 1 gene had a fold change of at least 8. This is a striking difference with gene regulation in the basidiomycete Schizophyllum commune and the filamentous bacterium Streptomyces coelicolor. These microorganisms produce repellent-like proteins; i.e. the hydrophobins in S. commune (Wösten et al. 1993, 1994a, b, 1999; Wösten and Willey 2000) and the chaplins in S. coelicolor (Claessen et al. 2003, 2004). Like repellents, hydrophobins and chaplins form functional amyloid fibrils at the hyphal surface (Butko et al. 2001; Claessen et al. 2003; Wösten and de Vocht 2000; de Vocht et al. 2002; Scholtmeijer et al. 2009). Inactivation of the genes encoding these proteins results in a phenotype similar to that of the Δrep1 strain (Claessen et al. 2003; van Wetter et al. 1996, 2000; Wösten et al. 1994b). Over 4000 genes had changed their expression at least two-fold under conditions of aerial growth when expression of a wild-type S. commune strain was compared to that of the hydrophobin knockout strain ΔSC3ΔSC4 (R.A. Ohm, H.A.B. Wösten, unpublished results). In S. coelicolor 244 genes had a ≥2-fold changed expression in the ∆chp strain (de Jong et al. 2009; Claessen et al. 2006). Transcriptional regulators were among the genes with an affected expression in both S. commune and S. coelicolor. In contrast, genes encoding transcriptional activators were not affected in the Δrep1 strain. How can we explain this result? In S. commune and S. coelicolor, aerial hyphae formation is the first step in a differentiation process leading to the formation of fruiting bodies and spore chains, respectively. In contrast, aerial hyphae of U. maydis seem to represent vegetative hyphae that happen to grow into the air.
Remarkably, most of the genes that changed their expression in the Δrep1 strain encode secreted proteins without an enzymatic function. Eleven out of 22 genes that were up-regulated encode small secreted proteins (SSPs) and 9 of them belong to the class of secreted cysteine-rich proteins (SCRPs) (Müller et al. 2008b). These proteins harbor no common protein domains. For these proteins, similarity in three-dimensional structure may be more important than sequence similarity. This is in agreement with the results obtained with the TANGO and Waltz algorithms (Fernandez-Escamilla et al. 2004; Maurer-Stroh et al. 2010). These algorithms predict β-aggregation and amylogenic regions in more than 80% of the SSPs and SCRPs that are up-regulated in the Δrep1 strain. The algorithms also predict amylogenic regions in the repellents of U. maydis and in the S. commune hydrophobin SC3, which are known to form amyloids (Wösten and de Vocht 2000; de Vocht et al. 2002; Scholtmeijer et al. 2009; Teertstra et al. 2009).
We previously showed that repellents form amyloids in the cell wall of hyphae of U. maydis (Teertstra et al. 2009). Interestingly, filaments of the SG200∆rep1 strain also stained with ThT, from which it was concluded that other secreted proteins also have the capability to form amyloid fibrils. The SSPs that are up-regulated in SG200∆rep1 could be candidates for such proteins. These results are in line with previous findings that amyloids can be generally found on microbial surfaces (Claessen et al. 2003; Chapman et al. 2002; Larsen et al. 2007; Otoo et al. 2008; Rauceo et al. 2004; Gebbink et al. 2005). Our results indicate that a variety of proteins can form such structures at a particular cell surface. It may well be that only part of them (e.g. the chaplins, hydrophobins and repellents) form an amphipathic amyloid film, thus enabling aerial growth by lowering the water surface tension. The SSPs of U. maydis may not be able to do so, which would explain why aerial growth in the ∆rep1 strain is strongly affected.
Twelve clusters are found in the genome of U. maydis that contain genes encoding SSPs (Kämper et al. 2006). Deletion of five of them changed virulence (Kämper et al. 2006). Four of these clusters contain SSPs that are up-regulated in the ∆rep1 strain (see Table 2). The symbiotic basidiomycete Laccaria bicolor also harbors a high number of genes encoding SSPs. Ten percent of the secreted proteins belong to this class, many of which are classified as SCRPs. A part of these genes is up-regulated during mycorrhizal interactions, whereas others are down-regulated. This suggests an important role for these proteins in the symbiosis (Martin et al. 2008). SCRPs of fungi and oomycetes have been described to function as apoplastic effectors (Kamoun 2006). Such effectors include toxins, elicitors, virulence and avirulence proteins. The cysteine residues have been proposed to enhance stability of the proteins and yield protection to plant proteases (Joosten et al. 1997; Luderer et al. 2002). Whether (some of) the U. maydis SSP and SCRP genes are also apoplastic effectors remains to be investigated.
The highest up-regulated gene in the ∆rep1 strain is gene um00792. It encodes a SCRP of 144 amino acids with 7 cysteines. The gene is clustered in the genome with genes um00793, um00794 and um00795. It has 28% sequence homology with gene um00793, which is also up-regulated in the SG200∆rep1 strain. Deletion of gene um00792 has no influence on pathogenicity (Vraneš 2006) but its expression depends on biz1. This gene encodes a transcription factor, which functions at the stage of filament and appressorium formation (Flor-Parra et al. 2006). It down-regulates clb1, the mitotic cyclin, (Flor-Parra et al. 2006) and thereby causes G2 cell cycle arrest prior to plant invasion. Of interest, expression of um06126, um01240, um01820 and um01377 is also increased in the absence of repellents and decreased in the absence of Biz1 (This study; Vraneš 2006). The latter indicates a role for these genes in the early stages of pathogenic development.
Expression of gene um00792 was followed using GFP as a reporter. Expression of this gene was detected in strains SG200 and SG200∆rep1 in aerial hyphae and hyphae growing in the substrate. Gene expression was not detected in yeast cells growing in liquid culture, but yeast cells growing in and on the solid substrate, where hyphal growth is initiated, did show fluorescence. Strikingly, over-expression of um00792 was only detected in the ∆rep1 strain in the layer of seeded cells that lay on the solid substrate. This layer contains many hyphae that seem unable to escape from the extracellular matrix. Possibly, the fungus senses absence of the repellents in the extracellular matrix, causing up-regulation of gene um00792. The protein encoded by this gene, however, is not able to complement repellents in formation of aerial hyphae (see above). Future studies should reveal the role of the SSPs and SCRPs that are up-regulated in U. maydis upon inactivation of the rep1 gene. It may well be that the genes are redundant, requiring multiple knock-outs before a phenotype will be observed.
References
Banuett F (1992) Ustilago maydis, the delightful blight. Trends Genet 8:174–180
Banuett F, Herskowitz I (1988) Ustilago maydis, smut of maize. Adv Plant Pathol 6:427–455
Banuett F, Herskowitz I (1989) Different a alleles of Ustilago maydis are necessary for maintenance of filamentous growth but not for meiosis. Proc Natl Acad Sci USA 86:5878–5882
Bateman A, Birney E, Cerruti L, Durbin R, Etwiller L, Eddy SR, Griffiths-Jones S, Howe KL, Marshall M, Sonnhammer ELL (2002) The PFAM protein families database. Nucleic Acids Res 30:276–280
Bendtsen JD, Nielsen H, Von Heijne G, Brunak S (2004) Improved prediction of signal peptides: SignalP 3.0. J Mol Biol 340:783–795
Bohlmann R (1996) Isolierung und Charakterisierung von Filamentspezifisch Exprimierten Genen aus Ustilago maydis. Dissertation, University of Munich
Bölker M, Urban M, Kahmann R (1992) The a mating type locus of U. maydis specifies cell signaling components. Cell 68:441–450
Bölker M, Genin S, Lehmler C, Kahmann R (1995) Regulation of mating and dimorphism in Ustilago maydis. Can J Bot 73:S320–S325
Brachmann A, Weinzierl G, Kämper J, Kahmann R (2001) Identification of genes in the bW/bE regulatory cascade in Ustilago maydis. Mol Microbiol 42:1047–1063
Brachmann A, König J, Julius C, Feldbrügge M (2004) A reverse genetic approach for generating gene replacement mutants in Ustilago maydis. Mol Genet Genomics 272:216–226
Butko P, Buford JP, Goodwin JS, Stroud PA, McCormick CL, Cannon GC (2001) Spectroscopic evidence for amyloid-like interfacial self-assembly of hydrophobin Sc3. Biochem Biophys Res Commun 280:212–215
Chapman MR, Robinson LS, Pinkner JS, Roth R, Heuser J, Hammar M, Normark S, Hultgren SJ (2002) Role of Escherichia coli curli operons in directing amyloid fiber formation. Science 295:851–855
Christensen J. J. (1963) Corn smut caused by Ustilago maydis. The American Phytopathological Society, St Paul, Monograph No.2, 41
Claessen D, Rink R, de Jong W, Siebring J, de Vreugd P, Boersma FGH, Dijkhuizen L, Wösten HAB (2003) A novel class of secreted hydrophobic proteins is involved in aerial hyphae formation in Streptomyces coelicolor by forming amyloid-like fibrils. Genes Dev 17:1714–1726
Claessen D, Stokroos L, Deelstra HJ, Penninga NA, Bormann C, Salas JA, Dijkhuizen L, Wösten HAB (2004) The formation of the rodlet layer of Streptomycetes is the result of the interplay between rodlins and chaplins. Mol Microbiol 53:433–443
Claessen D, de Jong W, Dijkhuizen L, Wösten HAB (2006) Regulation of Streptomyces development: reach for the sky!. Trends Microbiol 14:313–319
de Jong W, Manteca A, Sanchez J, Bucca G, Smith CP, Dijkhuizen L, Claessen D, Wösten HAB (2009) NepA is a structural cell wall protein involved in maintenance of spore dormancy in Streptomyces coelicolor. Mol Microbiol 71:1591–1603
de Vocht ML, Reviakine I, Ulrich WP, Bergsma-Schutter W, Wösten HAB, Vogel H, Brisson A, Wessels JG, Robillard GT (2002) Self-assembly of the hydrophobin SC3 proceeds via two structural intermediates. Protein Sci 11:1199–1205
Edgar R, Domrachev M, Lash AE (2002) Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res 30:207–210
Eichhorn H, Lessing F, Winterberg B, Schirawski J, Kämper J, Müller P, Kahmann R (2006) A ferroxidation/permeation iron uptake system is required for virulence in Ustilago maydis. Plant Cell 18:3332–3345
Fernandez-Escamilla AM, Rousseau F, Schymkowitz J, Serrano L (2004) Prediction of sequence-dependent and mutational effects on the aggregation of peptides and proteins. Nat Biotechnol 22:1302–1306
Flor-Parra I, Vraneš M, Kämper J, Pérez-Martín J (2006) Biz1, a zinc finger protein required for plant invasion by Ustilago maydis, regulates the levels of a mitotic cyclin. Plant Cell 18:2369–2387
Gebbink MF, Claessen D, Bouma B, Dijkhuizen L, Wösten HAB (2005) Amyloids - a functional coat for microorganisms. Nat Rev Microbiol 3:333–341
Gillissen B, Bergemann J, Sandmann C, Schroeer B, Bölker M, Kahmann R (1992) A two-component regulatory system for self/non-self recognition in Ustilago maydis. Cell 68:647–657
Hoffman CS, Winston F (1987) A ten-minute DNA preparation from yeast efficiently releases autonomous plasmids for transformation of Escherichia coli. Gene 57:267–272
Holliday R (1974) Ustilago maydis. In: King RC (ed) Handbook of Genetics. Plenum Press, New York, pp 575–595
Joosten MHAJ, Vogelsang R, Cozijnsen TJ, Verberne MC, de Wit PJGM (1997) The biotrophic fungus Cladosporium fulvum circumvents cf-4-mediated resistance by producing unstable AVR4 elicitors. Plant Cell 9:367–379
Kamoun S (2006) A catalogue of the effector secretome of plant pathogenic oomycetes. Ann Rev Phytopathol 44:41–60
Kämper J, Reichmann M, Romeis T, Bölker M, Kahmann R (1995) Multiallelic recognition: Nonself-dependent dimerization of the bE and bW homeodomain proteins in Ustilago maydis. Cell 81:73–83
Kämper J, Kahmann R, Bölker M, Ma LJ, Brefort T, Saville BJ, Banuett F, Kronstad JW, Gold SE et al (2006) Insights from the genome of the biotrophic fungal plant pathogen Ustilago maydis. Nature 444:97–101
Larsen P, Nielsen JL, Dueholm MS, Wetzel R, Otzen D, Nielsen PH (2007) Amyloid adhesins are abundant in natural biofilms. Environ Microbiol 9:3077–3090
Leuthner B, Aichinger C, Oehmen E, Koopmann E, Müller O, Müller P, Kahmann R, Bölker M, Schreier PH (2005) A H2O2-producing glyoxal oxidase is required for filamentous growth and pathogenicity in Ustilago maydis. Mol Genet Genomics 272:639–650
Luderer R, de Kock MJD, Dees RHL, de Wit PJGM, Joosten MHAJ (2002) Functional analysis of cysteine residues of ECP elicitor proteins of the fungal tomato pathogen Cladosporium fulvum. Mol Plant Pathol 3:91–95
Martin F, Aerts A, Ahrén D, Brun A, Danchin EGJ, Duchaussoy F, Gibon J, Kohler A, Lindquist E et al (2008) The genome of Laccaria bicolor provides insights into mycorrhizal symbiosis. Nature 452:88–92
Maurer-Stroh S, Debulpaep M, Kuemmerer N, dela Lopez Paz M, Cristiano Martins I, Reumers J, Morris KL, Copland A, Serpell L et al (2010) Exploring the sequence determinants of amyloid structure using position-specific scoring matrices. Nat Meth 7:237–242
Müller O, Schreier PH, Uhrig JF (2008a) Identification and characterization of secreted and pathogenesis-related proteins in Ustilago maydis. Mol Genet Genomics 279:27–39
Müller O, Kahmann R, Aguilar G, Trejo-Aguilar B, Wu A, de Vries RP (2008b) The secretome of the maize pathogen Ustilago maydis. Fungal Genet Biol 45:S63–S70
Murzin AG, Brenner SE, Hubbard T, Chothia C (1995) SCOP: A structural classification of proteins database for the investigation of sequences and structures. J Mol Biol 247:536–540
Otoo HN, Kyeng GL, Qiu W, Lipke PN (2008) Candida albicans als adhesins have conserved amyloid-forming sequences. Eukaryot Cell 7:776–782
Ponting CP, Schultz J, Milpetz F, Bork P (1999) SMART: Identification and annotation of domains from signaling and extracellular protein sequences. Nucleic Acids Res 27:229–232
Rauceo JM, Gaur NK, Lee KG, Edwards JE, Klotz SA, Lipke PN (2004) Global cell surface conformational shift mediated by a Candida albicans adhesin. Infect Immun 72:4948–4955
Romeis T, Brachmann A, Kahmann R, Kämper J (2000) Identification of a target gene for the bE-bW homeodomain protein complex in Ustilago maydis. Mol Microbiol 37:54–66
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular Cloning : a Laboratory Manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Scholtmeijer K, de Vocht ML, Rink R, Robillard GT, Wösten HAB (2009) Assembly of the fungal SC3 hydrophobin into functional amyloid fibrils depends on its concentration and is promoted by cell wall polysaccharides. J Biol Chem 284:26309–26314
Teertstra WR, Deelstra HJ, Vranes M, Bohlmann R, Kahmann R, Kämper J, Wösten HAB (2006) Repellents have functionally replaced hydrophobins in mediating attachment to a hydrophobic surface and in formation of hydrophobic aerial hyphae in Ustilago maydis. Microbiology 152:3607–3612
Teertstra WR, van der Velden GJ, de Jong JF, Kruijtzer JA, Liskamp RM, Kroon-Batenburg LM, Müller WH, Gebbink MF, Wösten HAB (2009) The filament-specific Rep1–1 repellent of the phytopathogen Ustilago maydis forms functional surface-active amyloid-like fibrils. J Biol Chem 284:9153–9159
van Wetter MA, Schuren FHJ, Schuurs TA, Wessels JGH (1996) Targeted mutation of the SC3 hydrophobin gene of Schizophyllum commune affects formation of aerial hyphae. FEMS Microbiol Lett 140:265–269
van Wetter MA, Wösten HAB, Wessels JGH (2000) SC3 and SC4 hydrophobins have distinct roles in formation of aerial structures in dikaryons of Schizophyllum commune. Mol Microbiol 36:201–210
Vraneš M (2006) Transkriptom-analyse der frühen infektionsphase von Ustilago maydis: Identifikation neuer pathogenitätsrelevanter gene. Dissertation, University of Marburg
Wösten HAB, de Vocht ML (2000) Hydrophobins, the fungal coat unravelled. Biochim Biophys Acta 1469:79–86
Wösten HAB, Willey JM (2000) Surface-active proteins enable microbial aerial hyphae to grow into the air. Microbiology 146:767–773
Wösten HAB, de Vries OMH, Wessels JGH (1993) Interfacial self-assembly of a fungal hydrophobin into a hydrophobic rodlet layer. Plant Cell 5:1567–1574
Wösten HAB, Asgeirsdottir SA, Krook JHH, Drenth JHH, Wessels JGH (1994a) The fungal hydrophobin Sc3p self-assembles at the surface of aerial hyphae as a protein membrane constituting the hydrophobic rodlet layer. Eur J Cell Biol 63:122–129
Wösten HAB, Schuren FHJ, Wessels JGH (1994b) Interfacial self-assembly of a hydrophobin into an amphipathic protein membrane mediates fungal attachment to hydrophobic surfaces. EMBO J 13:5848–5854
Wösten HAB, Bohlmann R, Eckerskorn C, Lottspeich F, Bölker M, Kahmann R (1996) A novel class of small amphipathic peptides affect aerial hyphal growth and surface hydrophobicity in Ustilago maydis. EMBO J 15:4274–4281
Wösten HAB, van Wetter MA, Lugones LG, van der Mei HC, Busscher HJ, Wessels JGH (1999) How a fungus escapes the water to grow into the air. Curr Biol 9:85–88
Zheng Y, Kief J, Auffarth K, Farfsing JW, Mahlert M, Nieto F, Basse CW (2008) The Ustilago maydis Cys2His2-type zinc finger transcription factor Mzr1 regulates fungal gene expression during the biotrophic growth stage. Mol Microbiol 68:1450–1470
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Teertstra, W.R., Krijgsheld, P. & Wösten, H.A.B. Absence of repellents in Ustilago maydis induces genes encoding small secreted proteins. Antonie van Leeuwenhoek 100, 219–229 (2011). https://doi.org/10.1007/s10482-011-9581-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-011-9581-2