Abstract
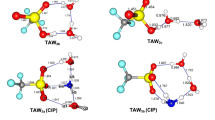
The microsolvation of taurine (TA) with one, two or three water molecules was investigated by a density functional theory (DFT) approach. Quantum theory of atoms in molecules (QTAIM) analyses were employed to elucidate the hydrogen bond (H-bond) interaction characteristics in TA-(H2O)n (n = 1–3) complexes. The results showed that the intramolecular H-bond formed between the hydroxyl and the N atom of TA are retained in most TA-(H2O)n (n = 1–3) complexes, and are strengthened via cooperative effects among multiple H-bonds from n = 1–3. A trend of proton transformation exists from the hydroxyl to the N atom, which finally results in the cleavage of the origin intramolecular H-bond and the formation of a new intramolecular H-bond between the amino and the O atom of TA. Therefore, the most stable TA-(H2O)3 complex becomes a zwitterionic complex rather than a neutral type. A many-body interaction analysis showed that the major contributors to the binding energies for complexes are the two-body energies, while three-body energies and relaxation energies make significant contributions to the binding energies for some complexes, whereas the four-body energies are too small to be significant.

The microsolvation of taurine (TA) with 1–3 water molecules was investigated by a density functional theory (DFT) approach. Quantum theory of atoms in molecules (QTAIM) and many-body interaction analyses were employed to elucidate hydrogen bonding interaction characteristics in TA-(H2O)n (n = 1–3) complexes




Similar content being viewed by others
References
Sturman JA (1993) Taurine in development. Physiol Rev 73:119–147
Huxtable RJ (1992) Physiological actions of taurine. Physiol Rev 72:101–163
del Olmo N, Bustamante J, del Rio RM, Solis JM (2000) Taurine activates GABA(A) but not GABA(B) receptors in rat hippocampal CA1 area. Brain Res 864:298–307
Jia F, Yue M, Chandra D, Keramidas A, Goldstein PA, Homanics GE, Harrison NL (2008) Taurine is a potent activator of extrasynaptic GABA(A) receptors in the thalamus. J Neurosci 28:106–115
Beukes JA, Mo F, van Beek W (2007) X-Ray induced radiation damage in taurine: a combined X-ray diffraction and Raman study. Phys Chem Chem Phys 9:4709–4720
Dewar MJS, Zoebisch EG, Healy EF, Stewart JJP (1985) Development and use of quantum mechanical molecular models. 76. AM1: a new general purpose quantum mechanical molecular model. J Am Chem Soc 107:3902–3909
Cortijo V, Sanz ME, Lopez JC, Alonso JL (2009) Conformational study of taurine in the gas phase. J Phys Chem A 113:14681–14683
Hibbs DE, Austin-Woods CJ, Platts JA, Overgaard J, Turner P (2003) Experimental and theoretical charge density study of the neurotransmitter taurine. Chem Eur J 9:1075–1084
Waller MP, Howard ST, Platts JA, Piltz RO, Willock DJ, Hibbs DE (2006) Novel properties from experimental charge densities: an application to the zwitterionic neurotransmitter taurine. Chem Eur J 12:7603–7614
Huang ZG, Dai YM, Yu L (2010) Density functional theory and topological analysis on the hydrogen bonding interactions in N-protonated adrenaline-DMSO complexes. Struct Chem 21:863–872
Huang ZG, Yu L, Dai YM (2010) Combined DFT with NBO and QTAIM studies on the hydrogen bonds in (CH3OH) n (n = 2–8) clusters. Struct Chem 21:565–572
Rappe AK, Bernstein ER (2000) Ab initio calculation of nonbonded interactions: are we there yet? J Phys Chem A 104:6117–6128
Grimme S (2004) Accurate description of van der Waals complexes by density functional theory including empirical corrections. J Comput Chem 25:1463–1473
Schwabe T, Grimme S (2007) Double-hybrid density functionals with long-range dispersion corrections: higher accuracy and extended applicability. Phys Chem Chem Phys 9:3397–3406
Zhao Y, Truhlar DG (2006) Comparative DFT study of van der Waals complexes: rare-gas dimers, alkaline-earth dimers, zinc dimer, and zinc-rare-gas dimers. J Phys Chem A 110:5121–5129
Zhao Y, Truhlar DG (2008) The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor Chem Acc 120:215–241
Chai JD, Head-Gordon M (2008) Long-range corrected hybrid density functionals with damped atom-atom dispersion corrections. Phys Chem Chem Phys 10:6615–6620
Johnson ER, Mackie ID, DiLabio GA (2009) Dispersion interactions in density-functional theory. J Phys Org Chem 22:1127–1135
Riley KE, Pitonak M, Jurecka P, Hobza P (2010) Stabilization and structure calculations for noncovalent interactions in extended molecular systems based on wave function and density functional theories. Chem Rev 110:5023–5063
Mandal A, Prakash M, Kumar RM, Parthasarathi R, Subramanian V (2010) Ab initio and DFT studies on methanol-water clusters. J Phys Chem A 114:2250–2258
Rai AK, Fei WX, Lu ZW, Lin ZJ (2009) Effects of microsolvation and aqueous solvation on the tautomers of histidine: a computational study on energy, structure and IR spectrum. Theor Chem Acc 124:37–47
Rao L, Ke HW, Fu G, Xu X, Yan YJ (2009) Performance of several density functional theory methods on describing hydrogen-bond interactions. J Chem Theor Comput 5:86–96
Riley KE, Pitonak M, Cerny J, Hobza P (2010) On the structure and geometry of biomolecular binding motifs (hydrogen-Bonding, stacking, X-H···π): WFT and DFT calculations. J Chem Theor Comput 6:66–80
Huang ZG, Yu L, Dai YM, Wang HK (2010) Hydrogen bonding interactions in cysteine-urea complexes: theoretical studies of structures, properties and topologies. J Mol Struct THEOCHEM 960:98–105
Huang ZG, Dai YM, Wang HK, Yu L (2011) Microsolvation of aminoethanol: A DFT combined with QTAIM study. J Mol Model. doi:10.1007/s00894-011-0973-z
Huang ZG, Dai YM, Yu L, Wang HK (2011) Hydrogen bonding interactions in noradrenaline- DMSO complexes: DFT and QTAIM studies of structure, properties and topology. J Mol Model. doi:10.1007/s00894-011-0956-0
Plumley JA, Dannenberg JJ (2011) A comparison of the behavior of functional/basis set combinations for hydrogen-bonding in the water dimer with emphasis on basis set superposition error. J Comput Chem 32. doi:10.1002/jcc.21729
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian09. Gaussian Inc, Wallingford
McLean AD, Chandler GS (1980) Contracted Gaussian basis sets for molecular calculations. I. Second row atoms, Z=11–18. J Chem Phys 72:5639–5648
Krishnan R, Binkley JS, Seeger R, Pople JA (1980) Self-consistent molecular orbital methods. XX. A basis set for correlated wave functions. J Chem Phys 72:650–654
Boys SF, Bernardi F (1970) The calculation of small molecular interactions by the differences of separate total energies. Some procedures with reduced errors. Mol Phys 19:553–566
Biegler-König F (2000) AIM2000. 1.0 edn. University of Applied Sciences, Bielefeld, Germany
Huang ZG, Yu L, Dai YM (2010) Density functional theory and topological analysis on the hydrogen bonds in cysteine-propanoic acid complexes. Struct Chem 21:855–862
Nozad AG, Meftah S, Ghasemi MH, Kiyani RA, Aghazadeh M (2009) Investigation of intermolecular hydrogen bond interactions in crystalline l-cysteine by DFT calculations of the oxygen-17, nitrogen-14, and hydrogen-2 EFG tensors and AIM analysis. Biophys Chem 141:49–58
Zhou HW, Lai WP, Zhang ZQ, Li WK, Cheung HY (2009) Computational study on the molecular inclusion of andrographolide by cyclodextrin. J Comput Aided Mol Des 23:153–162
Koch U, Popelier PLA (1995) Characterization of C-H-O hydrogen bonds on the basis of the charge density. J Phys Chem 99:9747–9754
Maheshwary S, Patel N, Sathyamurthy N, Kulkarni AD, Gadre SR (2001) Structure and stability of water clusters (H2O)n, n = 8–20: an ab initio investigation. J Phys Chem A 105:10525–10537
Xantheas SS (2000) Cooperativity and hydrogen bonding network in water clusters. Chem Phys 258:225–231
Chaudhari A, Lee SL (2004) A computational study of microsolvation effect on ethylene glycol by density functional method. J Chem Phys 120:7464–7469
Chaudhari A, Sahu PK, Lee SL (2004) Many-body interaction in glycine-(water) 3 complex using density functional theory method. J Chem Phys 120:170–174
Kulkarni AD, Ganesh V, Gadre SR (2004) Many-body interaction analysis: algorithm development and application to large molecular clusters. J Chem Phys 121:5043–5050
Hannachi Y, Silvi B, Bouteiller Y (1992) Ab initio study of the structure, cooperativity, and vibrational properties of the H2O:(HF)2 hydrogen bonded complex. J Chem Phys 97:1911–1918
Mo O, Yanez M, Elguero J (1992) Cooperative (nonpairwise) effects in water trimers: an ab initio molecular orbital study. J Chem Phys 97:6628–6638
Frank HS, Wen WY (1957) Ion-solvent interaction. Structural aspects of ion-solvent interaction in aqueous solutions: a suggested picture of water structure. Discuss Faraday Soc 24:133–140
Acknowledgments
This work is supported by Tianjin Science and Technology Development Fund Projects in Colleges and Universities (No. 20080504).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dai, Y., Wang, Y., Huang, Z. et al. Microsolvation effect and hydrogen-bonding pattern of taurine-water TA-(H2O)n (n = 1–3) complexes. J Mol Model 18, 265–274 (2012). https://doi.org/10.1007/s00894-011-1070-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-011-1070-z