Abstract
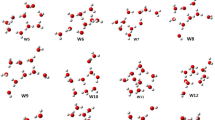
The structure, stability and infrared spectral signatures of triflic acid (TA) with water clusters (Wn) and protonated water clusters (TAH+Wn, n = 1 − 6) were computed using DFT and MP2 methods. Our calculations show that a minimum of three water molecules are necessary to stabilize the dissociated zwitterionic form of TA. It can be seen from the results that there is no significant movement of protons in smaller (n = 1 and 2) and linear (n = 1 – 6) types of water clusters. Further, the geometries of TAWn clusters first form a neutral pair (NP) to contact ion pair (CIP), then form a solvent separated ion pair (SSIP) in a water hexamer. These findings reveal that proton transfer may take place through NP to CIP and then CIP to SSIP. The calculated binding energies (BEs) of ion pair clusters is always higher than that of NP clusters (i.e., more stable than the NP). Existing excess proton linear chain clusters transfer a proton to adjacent water molecules via a Grotthuss mechanism, whereas the same isomers in the branched motifs do not conduct protons. Examination of geometrical parameters and infrared frequencies reveals hydronium ion (H3O+ also called Eigen cation) formation in both TAWn and protonated TAWn clusters. The stability of Eigen water clusters is three times higher than that of other non-Eigen water clusters. Our study shows clearly that formation of ion pairs in TAWn and TAH+Wn clusters greatly favors proton transfer to neighboring water molecules and also enhances the stability of these complexes.





Similar content being viewed by others
References
Schmitt UW, Voth GA (1998) J Phys Chem B 102:5547–5551
Cuma M, Schmitt UW, Voth GA (2001) J Phys Chem A 105:2814–2823
Day TJF, Voth GA (2005) Int J Mass Spectrom 241:197–204
Prakash M, Subramanian V (2011) Phys Chem Chem Phys 13:21479–21486
Mauritz KA, Moore RB (2004) Chem Rev 104:4535–4586
Kreuer KD (2001) J Membr Sci 185:29–39
Kreuer KD, Paddison SJ, Spohr E, Schuster M (2004) Chem Rev 104:4637–4678
Jang SS, Molinero V, Cagin T, Goddard WA III (2004) J Phys Chem B 108:3149–3157
Urata S, Irisawa J, Takada A, Shinoda W, Tsuzuki S, Mikami M (2005) J Phys Chem B 10:4269–4278
Tsuda M, Arboleda NB Jr, Kasai H (2006) Chem Phys 324:393–397
Li X, Liao S (2009) J Mol Struct (Theochem) 897:66–68
Eikerling M, Paddison SJ, Pratt LR, Zawodzinski TA Jr (2003) Chem Phys Lett 368:108–114
Elliott JA, Paddison SJ (2005) J Phys Chem A 093:7583–7593
Elliott JA, Paddison SJ (2007) Phys Chem Chem Phys 9:2602–2618
Blake NP, Mills G, Metiu H (2007) J Phys Chem B 111:2490–2494
Hristov IH, Paddison SJ, Paul R (2008) J Phys Chem B 112:2937–2949
Hayes RL, Paddison SJ (2009) J Phys Chem B 113:16574–16589
Hayes RL, Paddison SJ, Tuckerman ME (2001) J Phys Chem A 115:6112–6124
Karo J, Aabloo A, Thomas JO, Brandell D (2010) J Phys Chem B 114:6056–6064
Wu DS, Paddison SJ, Elliott JA (2009) Macromolecules 43:3358–3367
Phonyiem M, Chaiwongwattana S, Lao-ngam C, Sagarik K (2011) Phys Chem Chem Phys 13:10923–10939
Sagarik K, Phonyiem M, Lao-ngam C, Chaiwongwattana S (2008) Phys Chem Chem Phys 10:2098–2112
Wescott JT, Qi Y, Subramanian L, Capehart TW (2006) J Chem Phys 124:134702
Petersen MK, Voth GA (2006) J Phys Chem B 110:8594–18600
Chen H, Yan T, Voth GA (2009) J Phys Chem A 113:4507–4517
Knox CK, Voth GA (2010) J Phys Chem B 114:3205–3218
Wang S, Bianco R, Hynes JT (2009) J Phys Chem A 113:1295–1307
Wang S, Bianco R, Hynes JT (2010) Phys Chem Chem Phys 12:8241–8249
Lee C, Sosa C, Planas M, Novoa JA (1996) J Chem Phys 104:7081–7085
Fennell CJ, Bizjak A, Vlachy V, Dill KA (2009) J Phys Chem B 113:6782–6791
Fulton JL, Balasubramanian M (2010) J Am Chem Soc 132:12597–12604
Walewski L, Forbert H, Marx D (2011) J Phys Chem Lett 2:3069–3074
Pluhařová E, Marsalek O, Schmidt B, Jungwirth P (2013) J Phys Chem Lett 4:4177–4181
van der Post ST, Hunger J, Bonn M, Bakker HJ (2014) J Phys Chem B 118:4397–4403
Chen H, Ruckenstein E (2015) J Phys Chem B 119:12671–12676
Møller C, Plesset MS (1934) Phys Rev 46:618
Curtiss LA, Redfern PC, Raghavachari K, Rassolov V, Pople JA (1999) J Chem Phys 110:4703
Becke AD (1993) J Chem Phys 98:5648
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785
Zhao Y, Truhlar DG (2005) Phys Chem Chem Phys 7:2701–2705
Jiang JC, Wang YS, Chang HC, Lin SH, Lee YT, Niedner-Schatteburg G, Chang HC (2000) J Am Chem Soc 122:1398–1410
Jiang JC, Chaudhuri C, Lee YT, Chang HC (2002) J Phys Chem A 106:0937–10944
Lin CK, Wu CC, Wang YS, Lee YT, Chang HC, Kuo JL, Klein ML (2005) Phys Chem Chem Phys 7:938–944
Wu CC, Lin CK, Chang HC, Jiang JC, Kuo JL, Klein ML (2005) J Chem Phys 122:074315
Suhara K, Fujii A, Mizuse K, Mikami N, Kuo JL (2007) J Chem Phys 126:194306
Kuo JL, Xie ZZ, Bing D, Fujii A, Hamashima T, Suhara KI, Mikami N (2008) J Phys Chem A 112:10125–10133
Prakash M, Subramanian V, Gadre SR (2009) J Phys Chem A 113:12260–12275
Prakash M, Gopalsamy K, Subramanian V (2009) J Phys Chem A 113:13845–13852
Prakash M, Mathivon K, Benoit DM, Chambaud G, Hochlaf M (2014) Phys Chem Chem Phys 16:12503–12509
Prakash M, Chambaud G, Al-Mogren MM, Hochlaf M (2014) J Mol Mod 20:2534
Boussouf K, Boulmene R, Prakash M, Komiha N, Taleb M, Al-Mogren MM, Hochlaf M (2015) Phys Chem Chem Phys 17:14417–14426
Boussouf K, Khairat T, Prakash M, Komiha N, Chambaud G, Hochlaf M (2015) J Phys Chem A 119:11928–11940
Boulmene R, Boussouf K, Prakash M, Komiha N, Al-Mogren MM, Hochlaf M (2016) Chem Phys Chem 17:1–13
Boys SF, Bernardi F (1970) Mol Phys 19:553–566
Frisch MJ et al (2009) Gaussian 09 Revision A 0.2. Gaussian Inc, Wallingford, CT
Mandal A, Prakash M, Kumar RM, Parthasarathi R, Subramanian V (2010) J Phys Chem A 114:2250–2258
Headrick JM, Diken EG, Walters RS, Hammer NI, Christie RA, Cui J, Myshakin EM, Duncan MA, Johnson MA, Jordan KD (2005) Science 308:1765–1769
Miyazaki M, Fujii A, Ebata T, Mikami N (2004) Science 304:1134–1137
Shin JW, Hammer NI, Diken EG, Johnson MA, Walters RS, Jaeger TD, Duncan MA, Christie RA, Jordan KD (2004) Science 304:1137–1140
Bgemann MH, Gudeman CS, Pfaff J, Saykally R (1983) J Phys Rev Lett 51:554
Schwartz HA (1977) J Chem Phys 67:5525
Asmis KN, Pivonka NL, Santambrogio G, Brümmer M, Kaposta C, Neumark DM, Wöste L (2003) Science 299:1375–1377
Fridgen TD, McMahon TB, MacAleese L, Lemaire J, Maitre P (2004) J Phys Chem A 108:9008–9010
Park M, Shin I, Singh NJ, Kim KS (2007) J Phys Chem A 111:10692–10702
Prakash M, Gopalsamy K, Subramanian V (2011) J Chem Phys 135:214308
Frank RAW, Titman CM, Pratap JV, Luisi BF, Perham RN (2004) Science 306:872–876
Falk M (1980) Can J Chem 58:1495–1501
Quezado S, Kwak JCT, Falk M (1984) Can J Chem 62:958–966
Moilanen DE, Piletic IR, Fayer MD (2006) J Phys Chem A 110:9084–9088
Moilanen DE, Spry DB, Fayer MD (2008) Langmuir 24:3690–3698
Liu S, Aquino AJA, Korzeniewski C (2013) Langmuir 29:13890–13897
Kabrane J, Aquino AJA (2015) J Phys Chem A 119:1754–1764
Acknowledgements
This work is supported by DST India-European Union sponsored project (HYPOMAP 8/233482/2008) and the Council of Scientific and Industrial Research (CSIR), India. We also thank the CSIR-CLRI for high performance computing time.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 30 kb)
Rights and permissions
About this article
Cite this article
Prakash, M., Subramanian, V. Ab initio and density functional theory (DFT) studies on triflic acid with water and protonated water clusters. J Mol Model 22, 293 (2016). https://doi.org/10.1007/s00894-016-3158-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-016-3158-y