Abstract
Background
While mesh reinforcement is recognized as the optimal strategy for many hernia repairs, there remains debate on the optimal position for deployment and characteristics that lead to improved biocompatibility. Coatings are an avenue by which integration may be improved. Our aim was to evaluate tissue integration between uncoated, fibroblast- and mesenchymal stem cell-coated meshes placed as subcutaneous onlay (ON) or intraperitoneal underlay (UN).
Methods
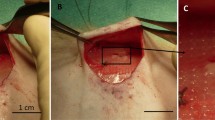
Three commonly used biologic and synthetic hernia meshes were tested including Parietex, TIGR and Strattice. Each mesh was coated with rat kidney fibroblasts (NRKs) or rat mesenchymal stem cells (MSCs) along with an uncoated group. In the ON group, mesh was fixated on top of the external oblique fascia. In the UN group, mesh was placed against the intact peritoneum. Animals were survived for 30 days and killed for biomechanical and histologic analysis. A “T”-peel test was performed on a mesh–tissue explant from each sample to analyze the strength of integration at the mesh–tissue interface. Tissue integration was evaluated histologically using an established scoring system.
Results
All uncoated meshes demonstrated significantly higher tissue ingrowth in the UN compared to ON position. Cell-coating of synthetic meshes decreased tissue ingrowth as UN, but increased it as ON, with a net effect of minimizing biomechanical difference between the two positions. In the biologic group however, NRK-coating decreased tissue ingrowth regardless of position, while MSC-coating increased it in both ON and UN positions.
Conclusions
Both cell-coating and positioning affect mesh–tissue integration. Integration is superior in the underlay position compared to onlay when uncoated. Cell-coating of selected synthetic meshes can improve integration, particularly in the onlay position. Furthermore, MSCs appear to be a viable choice for biologic mesh coating, especially when implanted as an onlay. Overall, cell-coating of surgical meshes appears to a have a potential to improve mesh–tissue integration.







Similar content being viewed by others
References
Orenstein SB, Dumeer JL, Monteagudo J, Poi MJ, Novitsky YW (2011) Outcomes of laparoscopic ventral hernia repair with routine defect closure using “shoelacing” technique. Surg Endosc 25(5):1452–1457
Rosen MJ, Jin J, McGee MF, Williams C, Marks J, Ponsky JL (2007) Laparoscopic component separation in the single-stage treatment of infected abdominal wall prosthetic removal. Hernia 11(5):435–440
Breuing K, Butler CE, Ferzoco S, Franz M, Hultman CS, Kilbridge JF et al (2010) Incisional ventral hernias: review of the literature and recommendations regarding the grading and technique of repair. Surgery 148(3):544–558
Timmermans L, de Goede B, van Dijk SM, Kleinrensink G-J, Jeekel J, Lange JF (2014) Meta-analysis of sublay versus onlay mesh repair in incisional hernia surgery. Am J Surg 207(6):980–988
Benhidjeb T, Benecke C, Strik MW (2008) [Incisional hernia repair: sublay or intraperitoneal onlay mesh (IPOM)?]. Zentralbl Chir 133(5):458–463
Demetrashvili ZM, kerkadze VN, Pipia IN, Topchishvili GG (2009) [Comparative evaluate of methods of placement of polypropylene meshes in alloplastic of incisional hernias]. Georgian Med News 175:7–9
Petter-Puchner AH, Fortelny RH, Silic K, Brand J, Gruber-Blum S, Redl H (2011) Biologic hernia implants in experimental intraperitoneal onlay mesh plasty repair: the impact of proprietary collagen processing methods and fibrin sealant application on tissue integration. Surg Endosc 25(10):3245–3252
Anderson JM, Rodriguez A, Chang DT (2008) Foreign body reaction to biomaterials. Semin Immunol 20(2):86–100
Junge K, Binnebösel M, von Trotha KT, Rosch R, Klinge U, Neumann UP et al (2012) Mesh biocompatibility: effects of cellular inflammation and tissue remodelling. Langenbecks Arch Surg 397(2):255–270
Brandt CJ, Kammer D, Fiebeler A, Klinge U (2011) Beneficial effects of hydrocortisone or spironolactone coating on foreign body response to mesh biomaterial in a mouse model. J Biomed Mater Res A 99(3):335–343
Dolce CJ, Stefanidis D, Keller JE, Walters KC, Newcomb WL, Heath JJ et al (2010) Pushing the envelope in biomaterial research: initial results of prosthetic coating with stem cells in a rat model. Surg Endosc 24(11):2687–2693
Holt DJ, Chamberlain LM, Grainger DW (2010) Cell-cell signaling in co-cultures of macrophages and fibroblasts. Biomaterials 31(36):9382–9394
Otto WR, Wright NA (2011) Mesenchymal stem cells: from experiment to clinic. Fibrogenesis Tissue Repair 4:20
Gao Y, Liu L-J, Blatnik JA, Krpata DM, Anderson JM, Criss CN et al (2014) Methodology of fibroblast and mesenchymal stem cell coating of surgical meshes: a pilot analysis. J Biomed Mater Res B Appl Biomater 102(4):797–805
Zhou HY, Zhang J, Yan RL, Wang Q, Fan LY, Zhang Q et al (2011) Improving the antibacterial property of porcine small intestinal submucosa by nano-silver supplementation: a promising biological material to address the need for contaminated defect repair. Ann Surg 253(5):1033–1041
Binnebösel M, von Trotha KT, Ricken C, Klink CD, Junge K, Conze J et al (2012) Gentamicin supplemented polyvinylidenfluoride mesh materials enhance tissue integration due to a transcriptionally reduced MMP-2 protein expression. BMC Surg 12:1
Israelsson LA (2008) Parastomal hernias. Surg Clin North Am 88(1):113–125
Albino FP, Patel KM, Nahabedian MY, Sosin M, Attinger CE, Bhanot P (2013) Does mesh location matter in abdominal wall reconstruction? A systematic review of the literature and a summary of recommendations. Plast Reconstr Surg 132(5):1295–1304
Hawn MT, Snyder CW, Graham LA, Gray SH, Finan KR, Vick CC (2010) Long-term follow-up of technical outcomes for incisional hernia repair. J Am Coll Surg 210(5): 648–55, 655–7
Venclauskas L, Maleckas A, Kiudelis M (2010) One-year follow-up after incisional hernia treatment: results of a prospective randomized study. Hernia 14(6):575–582
Novitsky YW, Rosen MJ (2012) The biology of biologics: basic science and clinical concepts. Plast Reconstr Surg 130(5 Suppl 2):9S–17S
Petter-Puchner AH, Fortelny RH, Mika K, Hennerbichler S, Redl H, Gabriel C (2011) Human vital amniotic membrane reduces adhesions in experimental intraperitoneal onlay mesh repair. Surg Endosc 25(7):2125–2131
Herrero C, Pérez-Simón JA (2010) Immunomodulatory effect of mesenchymal stem cells. Braz J Med Biol Res 43(5):425–430
Asarias JR, Nguyen PT, Mings JR, Gehrich AP, Pierce LM (2011) Influence of mesh materials on the expression of mediators involved in wound healing. J Invest Surg 24(2):87–98
Nguyen PT, Asarias JR, Pierce LM (2012) Influence of a new monofilament polyester mesh on inflammation and matrix remodeling. J Invest Surg 25(5):330–339
Gonzalez R, Ramshaw BJ (2003) Comparison of tissue integration between polyester and polypropylene prostheses in the preperitoneal space. Am Surg 69(6):471–476 (Discussion 476–7)
Vereczkei A, Varga G, Tornoczky T, Papp A, Horvath ÖP (2012) A new experimental method for hiatal reinforcement using connective tissue patch transfer. Dis Esophagus 25(5):465–469
Hjort H, Mathisen T, Alves A, Clermont G, Boutrand JP (2012) Three-year results from a preclinical implantation study of a long-term resorbable surgical mesh with time-dependent mechanical characteristics. Hernia 16(2):191–197
Peeters E, van Barneveld KWY, Schreinemacher MH, De Hertogh G, Ozog Y, Bouvy N et al (2013) One-year outcome of biological and synthetic bioabsorbable meshes for augmentation of large abdominal wall defects in a rabbit model. J Surg Res 180(2):274–283
Orenstein SB, Qiao Y, Klueh U, Kreutzer DL, Novitsky YW (2010) Activation of human mononuclear cells by porcine biologic meshes in vitro. Hernia 14(4):401–407
Orenstein SB, Qiao Y, Kaur M, Klueh U, Kreutzer DL, Novitsky YW (2010) Human monocyte activation by biologic and biodegradable meshes in vitro. Surg Endosc 24(4):805–811
Deeken CR, Matthews BD (2012) Comparison of contracture, adhesion, tissue ingrowth, and histologic response characteristics of permanent and absorbable barrier meshes in a porcine model of laparoscopic ventral hernia repair. Hernia 16(1):69–76
McGinty JJ, Hogle NJ, McCarthy H, Fowler DL (2005) A comparative study of adhesion formation and abdominal wall ingrowth after laparoscopic ventral hernia repair in a porcine model using multiple types of mesh. Surg Endosc 19(6):786–790
Jacob BP, Hogle NJ, Durak E, Kim T, Fowler DL (2007) Tissue ingrowth and bowel adhesion formation in an animal comparative study: polypropylene versus Proceed versus Parietex Composite. Surg Endosc 21(4):629–633
Jenkins ED, Melman L, Deeken CR, Greco SC, Frisella MM, Matthews BD (2010) Evaluation of fenestrated and non-fenestrated biologic grafts in a porcine model of mature ventral incisional hernia repair. Hernia 14(6):599–610
Acknowledgments
This study was supported by the internal grant from Department of Surgery University Hospitals Case Medical Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Arnab Majumder, Yue Gao, Emanuel E. Sadava, and James M. Anderson declare no conflict of interest. Yuri W. Novitsky is a consultant for C.R. Bard, Inc., LifeCell Corp. and Cooper Surgical, Inc., and has received research support from C.R. Bard, Inc.
Additional information
This study was supported by the internal grant from Department of Surgery University Hospitals Case Medical Center.
Rights and permissions
About this article
Cite this article
Majumder, A., Gao, Y., Sadava, E.E. et al. Cell-coating affects tissue integration of synthetic and biologic meshes: comparative analysis of the onlay and underlay mesh positioning in rats. Surg Endosc 30, 4445–4453 (2016). https://doi.org/10.1007/s00464-016-4764-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-016-4764-6