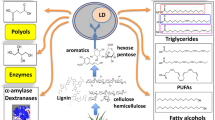
Abstract
Strains of the yeast genus Blastobotrys (subphylum Saccharomycotina) represent a valuable biotechnological resource for basic biochemistry research, single-cell protein, and heterologous protein production processes. Species of this genus are dimorphic, non-pathogenic, thermotolerant, and can assimilate a variety of hydrophilic and hydrophobic substrates. These can constitute a single-cell oil platform in an emerging bio-based economy as oleaginous traits have been discovered recently. However, the regulatory network of lipogenesis in these yeasts is poorly understood. To keep pace with the growing market demands for lipid-derived products, it is critical to understand the lipid biosynthesis in these unconventional yeasts to pinpoint what governs the preferential channelling of carbon flux into lipids instead of the competing pathways. This review summarizes information relevant to the regulation of lipid metabolic pathways and prospects of metabolic engineering in Blastobotrys yeasts for their application in food, feed, and beyond, particularly for fatty acid-based fuels and oleochemicals.
Key points
• The production of biolipids by heterotrophic yeasts is reviewed.
• Summary of information concerning lipid metabolism regulation is highlighted.
• Special focus on the importance of diacylglycerol acyltransferases encoding genes in improving lipid production is made.






Similar content being viewed by others
References
Abe M, Naohiro T, Yoshito I, Chihiro T, Takuji I, Kiyohiko N (2008) Characteristic fungi observed in the fermentation process for Puer tea. Int J Food Microbiol 124(2):199–203. https://doi.org/10.1016/j.ijfoodmicro.2008.03.008
Aggelis G, Sourdis J (1997) Prediction of lipid accumulation-degradation in oleaginous micro-organisms growing on vegetable oils. Antonie Van Leeuwenhoek 72(2):159–165. https://doi.org/10.1023/a:1000364402110
Álvaro-Benito M, Fernández-Lobato M, Baronian K, Kunze G (2013) Assessment of Schwanniomyces occidentalis as a host for protein production using the wide-range Xplor®2 expression platform. Appl Microbiol Biotechnol 97(10):4443–4456
Anschau A, Xavier Michelle CA, Saartje H, Franco T (2014) Effect of feeding strategies on lipid production by Lipomyces starkeyi. Bioresour Technol 157:214–222. https://doi.org/10.1016/j.biortech.2014.01.104
Augstein A, Kathrin B, Marcus G, Sepp KD, Gerold B (2003) Characterization, localization and functional analysis of Gpr1p, a protein affecting sensitivity to acetic acid in the yeast Yarrowia lipolytica. Microbiology (Reading) 149(Pt 3):589–600. https://doi.org/10.1099/mic.0.25917-0
Awad D, Frank B, Norbert M, Thomas B (2019) Multi-factorial-guided media optimization for enhanced biomass and lipid formation by the oleaginous yeast Cutaneotrichosporon oleaginosus. Front Bioeng Biotechnol 7:54. https://doi.org/10.3389/fbioe.2019.00054
Bandhu S, Neha B, Diptarka D, Vivek J, Arushdeep S, Gazal K, Saugata H, Debashish G (2019) Overproduction of single cell oil from xylose rich sugarcane bagasse hydrolysate by an engineered oleaginous yeast Rhodotorula mucilaginosa IIPL32. Fuel 254:115653. https://doi.org/10.1016/j.fuel.2019.115653
Bansal N, Diptarka D, Saugata H, Thallada B, Anjan R, Debashish G (2020) Effect of utilization of crude glycerol as substrate on fatty acid composition of an oleaginous yeast Rhodotorula mucilagenosa IIPL32: Assessment of nutritional indices. Bioresour Technol 309:123330. https://doi.org/10.1016/j.biortech.2020.123330
Beopoulos A, Thierry C, Jean-Marc N (2009) Yarrowia lipolytica: a model and a tool to understand the mechanisms implicated in lipid accumulation. Biochimie 91(6):692–696. https://doi.org/10.1016/j.biochi.2009.02.004
Beopoulos A, Zuzana M, France T, Marie-Thérèse LD, Ivan H, Seraphim P, Thierry C, Jean-Marc N (2008) Control of lipid accumulation in the yeast Yarrowia lipolytica. Appl Environ Microbiol 74(24):7779–7789. https://doi.org/10.1128/aem.01412-08
Biernacki M, Marek M, Thomas R, Reinhard P, Kim B, Rüdiger B, Gotthard K (2017) Enhancement of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) accumulation in Arxula adeninivorans by stabilization of production. Microb Cell Factories 16(1):144. https://doi.org/10.1186/s12934-017-0751-4
Bischoff F, Katarzyna L, Arno C, Keith B, Rüdiger B, Frieder S, Gothhard K (2015) Three new cutinases from the yeast Arxula adeninivorans that are suitable for biotechnological applications. Appl Environ Microbiol 81(16):5497–5510. https://doi.org/10.1128/aem.00894-15
Blomqvist J, Jana P, Khalili TS, Sabine S, Nils M, Jude B, Mats S, Volkmar P (2018) Oleaginous yeast as a component in fish feed. Sci Rep 8(1):15945. https://doi.org/10.1038/s41598-018-34232-x
Böer E, Thomas W, Bianka L, Renate M, Rüdiger B, Gerd G, Gotthard K (2004a) Characterization of the AINV gene and the encoded invertase from the dimorphic yeast Arxula adeninivorans. Antonie Van Leeuwenhoek 86(2):121–134. https://doi.org/10.1023/B:ANTO.0000036135.69810.df
Böer E, Thomas W, Karen D, Gerd G, Gotthard K (2004b) Characterization of the Arxula adeninivorans AHOG1 gene and the encoded mitogen-activated protein kinase. Curr Genet 46(5):269–276. https://doi.org/10.1007/s00294-004-0535-3
Böer E, Thomas W, Silvia S, Rüdiger B, Gerd G, Gotthard K (2005a) Characterization of the AXDH gene and the encoded xylitol dehydrogenase from the dimorphic yeast Arxula adeninivorans. Antonie Van Leeuwenhoek 87(3):233–243. https://doi.org/10.1007/s10482-004-3832-4
Böer E, Peter MH, Rüdiger B, Gerd G, Gotthard K (2005b) An extracellular lipase from the dimorphic yeast Arxula adeninivorans: molecular cloning of the ALIP1 gene and characterization of the purified recombinant enzyme. Yeast 22(7):523–535. https://doi.org/10.1002/yea.1230
Böer E, Steinborn G, Florschütz K, Körner M, Gellissen G, Kunze G (2008) Arxula adeninivorans (Blastobotrys adeninivorans) — A dimorphic yeast of great biotechnological potential. In: Satyanarayana T, Kunze G (eds) Yeast biotechnology: diversity and applications. Springer, Dordrecht. https://doi.org/10.1007/978-1-4020-8292-4_27
Böer E, Schröter A, Rüdiger B, Michael P, Gotthard K (2009a) Characterization and expression analysis of a gene cluster for nitrate assimilation from the yeast Arxula adeninivorans. Yeast 26(2):83–93. https://doi.org/10.1002/yea.1653
Böer E, Gamal EM, Ayman EF, Peggy B, Thomas W, Michael P, Gotthard K (2009b) The MAPk ASTE11 is involved in the maintenance of cell wall integrity and in filamentation in Arxula adeninivorans, but not in adaptation to hypertonic stress. FEMS Yeast Res 9(3):468–477. https://doi.org/10.1111/j.1567-1364.2009.00496.x
Böer E, Rüdiger B, Hans-Peter M, Michael P, Gotthard K (2009c) Atan1p-an extracellular tannase from the dimorphic yeast Arxula adeninivorans: molecular cloning of the ATAN1 gene and characterization of the recombinant enzyme. Yeast 26(6):323–337. https://doi.org/10.1002/yea.1669
Böer E, Michael P, Gotthard K (2009d) Xplor 2—an optimized transformation/expression system for recombinant protein production in the yeast Arxula adeninivorans. Appl Microbiol Biotechnol 84(3):583–594. https://doi.org/10.1007/s00253-009-2167-5
Borelli G, Bernabe FM, Vieira DSL, Falsarella CM, Guimarães PGA, Juliana J (2019) Positive selection evidence in xylose-related genes suggests methylglyoxal reductase as a target for the improvement of yeasts’ fermentation in industry. Genome Biol Evol 11(7):1923–1938. https://doi.org/10.1093/gbe/evz036
Bui Duc M, Irene K, Horstmann C, Schmidt T, Breunig Karin D, Gotthard K (1996) Expression of the Arxula adeninivorans glucoamylase gene in Kluyveromyces lactis. Appl Microbiol Biotechnol 45(1-2):102–106. https://doi.org/10.1007/s002530050655
Cai P, Jiaoqi G, Yongjin Z (2019) CRISPR-mediated genome editing in non-conventional yeasts for biotechnological applications. Microb Cell Factories 18(1):63. https://doi.org/10.1186/s12934-019-1112-2
Casal M, Helena C, Cecilia L (1996) Mechanisms regulating the transport of acetic acid in Saccharomyces cerevisiae. Microbiology (Reading) 142(Pt 6):1385–1390. https://doi.org/10.1099/13500872-142-6-1385
Cases S, Smith Steven J, Yao-Wu Z, Myers Heather M, Lear Steven R, Eric S, Sabine N, Colin C, Welch Carrie B, Lusis Aldons J, Erickson Sandra K, Farese Robert V Jr (1998) Identification of a gene encoding an acyl CoA:diacylglycerol acyltransferase, a key enzyme in triacylglycerol synthesis. Proc Natl Acad Sci U S A 95(22):13018–13023. https://doi.org/10.1073/pnas.95.22.13018
Castañeda MT, Sebastián N, Claudio V, De BH (2019) In silico optimization of lipid production in Rhodosporidium toruloides by gene knockout strategies*. This work was supported by funding projects from Agencia Na-cional de Promocion Científica y Tecnologica (PICT 2014-2394, 2016-2258), CONICET (PIP 2015-0837) and Universidad Nacional de La Plata (I216, PPID-I008) - Argentina. IFAC-PapersOnLine 52(1):94–99. https://doi.org/10.1016/j.ifacol.2019.06.043
Chang W, Ming Z, Sha Z, Ying L, Xiaobin L, Wei L, Gang L, Zhaomin L, Zhiyu X, Zuntian Z, Hongxiang L (2015) Trapping toxins within lipid droplets is a resistance mechanism in fungi. Sci Rep 5:15133. https://doi.org/10.1038/srep15133
Chattopadhyay A, Maiti MK (2020) Efficient xylose utilization leads to highest lipid productivity in Candida tropicalis SY005 among six yeast strains grown in mixed sugar medium. Appl Microbiol Biotechnol 104(7):3133–3144. https://doi.org/10.1007/s00253-020-10443-z
Chen H, Guangfei H, Lei W, Hongchao W, Zhennan G, Liming L, Hao Z, Wei C, Chen Yong Q (2015a) Identification of a critical determinant that enables efficient fatty acid synthesis in oleaginous fungi. Sci Rep 5:11247. https://doi.org/10.1038/srep11247
Chen C-X, Zheng S, Hai-Sheng C, Feng-Li F, Long-Ling O, Zhi-Gang Z (2015b) Identification and characterization of three genes encoding acyl-CoA: diacylglycerol acyltransferase (DGAT) from the microalga Myrmecia incisa Reisigl. Algal Res 12:280–288. https://doi.org/10.1016/j.algal.2015.09.007
Choudhary J, Singh S, Nain L (2016) Thermotolerant fermenting yeasts for simultaneous saccharification fermentation of lignocellulosic biomass. Electron J Biotechnol 21:82–92. https://doi.org/10.1016/j.ejbt.2016.02.007
de la Peña MM, Yisheng K, Keasling Jay D (2010) Feast: Choking on Acetyl-CoA, the Glyoxylate Shunt, and Acetyl-CoA-Driven Metabolism. In: Timmis (ed) Handbook of hydrocarbon and lipid microbiology. Springer Berlin Heidelberg, Berlin, pp 1649–1660
Delic M, Graf Alexandra B, Gunda K, Christina H-T, Stephan H, Diethard M, Brigitte G (2014) Overexpression of the transcription factor Yap1 modifies intracellular redox conditions and enhances recombinant protein secretion. Microb Cell 1(11):376–386. https://doi.org/10.15698/mic2014.11.173
Dulermo T, Zbigniew L, Rémi D, Magdalena R, Ramedane H, Jean-Marc N (2015) Analysis of ATP-citrate lyase and malic enzyme mutants of Yarrowia lipolytica points out the importance of mannitol metabolism in fatty acid synthesis. Biochim Biophys Acta 1851(9):1107–1117. https://doi.org/10.1016/j.bbalip.2015.04.007
El Fiki A, El MG, Carmen B, Thomas W, Rūdiger B, Gerd G, Gotthard K (2007) The Arxula adeninivorans ATAL gene encoding transaldolase-gene characterization and biotechnological exploitation. Appl Microbiol Biotechnol 74(6):1292–1299. https://doi.org/10.1007/s00253-006-0785-8
Fakas S (2017) Lipid biosynthesis in yeasts: a comparison of the lipid biosynthetic pathway between the model nonoleaginous yeast Saccharomyces cerevisiae and the model oleaginous yeast Yarrowia lipolytica. Eng Life Sci 17(3):292–302. https://doi.org/10.1002/elsc.201600040
Friedlander J, Vasiliki T, Annapuma K, Greenhagen Emily H, Consiglio Andrew L, Kyle ME, Crabtree Donald V, Jonathan A, Nugent Rebecca L, Hamilton Maureen A, Joe Shaw A, South Colin R, Gregory S, Brevnova Elena E (2016) Engineering of a high lipid producing Yarrowia lipolytica strain. Biotechnol Biofuels 9:77. https://doi.org/10.1186/s13068-016-0492-3
Galli A, Schiestl RH (1996) Hydroxyurea induces recombination in dividing but not in G1 or G2 cell cycle arrested yeast cells. Mutat Res 354(1):69–75. https://doi.org/10.1016/0027-5107(96)00037-1
Gellissen G, Kunze G, Gaillardin C, Cregg JM, Berardi E, Veenhuis M, van der Klei I (2005) New yeast expression platforms based on methylotrophic Hansenula polymorpha and Pichia pastoris and on dimorphic Arxula adeninivorans and Yarrowia lipolytica - a comparison. FEMS Yeast Res 5(11): 1079–1096. https://doi.org/10.1016/j.femsyr.2005.06.004
Giacomello M, Pellegrini L (2016) The coming of age of the mitochondria-ER contact: a matter of thickness. Cell Death Differ 23(9):1417–1427. https://doi.org/10.1038/cdd.2016.52
Gienow U, Gotthard K, Frieder S, Rüdiger B, Hofemeister J (1990) The Yeast Genus Trichosporon spec. LS3; Molecular characterization of genomic complexity. Zentralblatt für Mikrobiologie 145(1):3–12. https://doi.org/10.1016/S0232-4393(11)80001-6
Giersberg M, Adelheld D, Rüdiger B, Michael P, Gotthard K (2012) Production of a thermostable alcohol dehydrogenase from Rhodococcus ruber in three different yeast species using the Xplor®2 transformation/expression platform. J Ind Microbiol Biotechnol 39(9):1385–1396. https://doi.org/10.1007/s10295-012-1134-9
Guerfali M, Ines A, Nadia M, Wajdi A, Hafedh B, Rosário BM, Ribeiro Maria HL, Ali G (2019) Triacylglycerols accumulation and glycolipids secretion by the oleaginous yeast Rhodotorula babjevae Y-SL7: Structural identification and biotechnological applications. Bioresour Technol 273:326–334. https://doi.org/10.1016/j.biortech.2018.11.036
Haïli N, Julien L, Michel C, Franjo J, Christelle L-M, Thierry C, Pierre B (2016) Expression of soluble forms of yeast diacylglycerol acyltransferase 2 that integrate a broad range of saturated fatty acids in triacylglycerols. PLoS One 11(10):e0165431. https://doi.org/10.1371/journal.pone.0165431
Ito Y, Takao K, Mamoru Y, Satoshi K, Shingo I, Kenji I, Makoto F-S, Takashi M (2016) Enhancement of protein production via the strong DIT1 terminator and two RNA-binding proteins in Saccharomyces cerevisiae. Sci Rep 6:36997. https://doi.org/10.1038/srep36997
Jackson CL (2019) Lipid droplet biogenesis. Curr Opin Cell Biol 59:88–96. https://doi.org/10.1016/j.ceb.2019.03.018
Jankowska DA, Anke T-S, Arno C, Hoferichter P, Klein C, Rüdiger B, Keith B, Gotthard K (2013b) Arxula adeninivorans xanthine oxidoreductase and its application in the production of food with low purine content. J Appl Microbiol 115(3):796–807. https://doi.org/10.1111/jam.12284
Jankowska DA, Anke T-S, Arno C, Rüdiger B, Keith B, Gotthard K (2015) A novel enzymatic approach in the production of food with low purine content using Arxula adeninivorans endogenous and recombinant purine degradative enzymes. Bioengineered 6(1):20–25. https://doi.org/10.4161/21655979.2014.991667
Jankowska DA, Faulwasser K, Trautwein-Schult A, Arno C, Hoferichter P, Klein C, Rüdiger B, Keith B, Gotthard K (2013a) Arxula adeninivorans recombinant adenine deaminase and its application in the production of food with low purine content. J Appl Microbiol 115(5):1134–1146. https://doi.org/10.1111/jam.12317
Joshi AS, Benjamin N, Vineet C, Prasanna S-K, Levine Tim P, Andy G, Prinz William A (2018) Lipid droplet and peroxisome biogenesis occur at the same ER subdomains. Nat Commun 9(1):2940. https://doi.org/10.1038/s41467-018-05277-3
Juanssilfero AB, Prihardi K, Amza Rezky L, Nao M, Hiromi O, Hana M, Chie K, Ahmad T, Yopi OC, Bambang P, Akihiko K (2018) Effect of inoculum size on single-cell oil production from glucose and xylose using oleaginous yeast Lipomyces starkeyi. J Biosci Bioeng 125(6):695–702. https://doi.org/10.1016/j.jbiosc.2017.12.020
Juergens H, Varela Javier A, Gorter de Vries Arthur R, Thomas P, Gast Veronica JM, Gyurchev Nikola Y, Rajkumar Arun S, Robert M, Pronk Jack T, Morrissey John P, Daran Jean-Marc G (2018) Genome editing in Kluyveromyces and Ogataea yeasts using a broad-host-range Cas9/gRNA co-expression plasmid. FEMS Yeast Res 18(3). https://doi.org/10.1093/femsyr/foy012
Kabran P, Tristan R, Claude G, Jean-Marc N, Cécile N (2012) Alternative splicing regulates targeting of malate dehydrogenase in Yarrowia lipolytica. DNA Res 19(3):231–244. https://doi.org/10.1093/dnares/dss007
Kakimoto Y, Shinya T, Rieko K, Yuki M, Toshiya E, Yasushi T (2018) Visualizing multiple inter-organelle contact sites using the organelle-targeted split-GFP system. Sci Rep 8(1):6175. https://doi.org/10.1038/s41598-018-24466-0
Kalscheuer R, Alexander S (2003) A novel bifunctional wax ester synthase/acyl-CoA:diacylglycerol acyltransferase mediates wax ester and triacylglycerol biosynthesis in Acinetobacter calcoaceticus ADP1. J Biol Chem 278(10):8075–8082. https://doi.org/10.1074/jbc.M210533200
Kamineni A, Shaw J (2020) Engineering triacylglycerol production from sugars in oleaginous yeasts. Curr Opin Biotechnol 62:239–247. https://doi.org/10.1016/j.copbio.2019.12.022
Kamzolova SV, Morgunov IG (2016) Biosynthesis of pyruvic acid from glucose by Blastobotrys adeninivorans. Appl Microbiol Biotechnol 100(17):7689–7697. https://doi.org/10.1007/s00253-016-7618-1
Kasprzak J, Marion R, Sylvia D, Karin B, Kim B, Rüdiger B, Frieder S, Michael P, H-Mathias V, Gotthard K (2016a) Synthesis of ethyl (R)-mandelate using recombinant Carboxydothermus hydrogenoformans alcohol dehydrogenase produced by two yeast species. J Mol Catal B Enzym 133:176–186. https://doi.org/10.1016/j.molcatb.2016.08.012
Kasprzak J, Marion R, Jan R, Sebastian W, Kim B, Rüdiger B, Frieder S, Gotthard K (2016b) Characterization of an Arxula adeninivorans alcohol dehydrogenase involved in the metabolism of ethanol and 1-butanol. FEMS Yeast Res 16(3). https://doi.org/10.1093/femsyr/fow018
Katongole CB, Albina B, Volkmar P, Erik LJ (2017) Effect of solid-state fermentation with Arxula adeninivorans or Hypocrea jecorina (anamorph Trichoderma reesei) on hygienic quality and in-vitro digestibility of banana peels by mono-gastric animals. Livest Sci 199:14–21. https://doi.org/10.1016/j.livsci.2017.03.002
Kaur P, Anja L, Bijinder S, Erik B, Jelena P, Gerhard S, Rüdiger B, Gerd G, Tulasi S, Gotthard K (2007) APHO1 from the yeast Arxula adeninivorans encodes an acid phosphatase of broad substrate specificity. Antonie Van Leeuwenhoek 91(1):45–55. https://doi.org/10.1007/s10482-006-9094-6
Knoll A, Stefan B, Bernward H, Philip E, Kirsten S, Betina R, Christoph S, Juri S, Jochen B (2007) High cell density cultivation of recombinant yeasts and bacteria under non-pressurized and pressurized conditions in stirred tank bioreactors. J Biotechnol 132(2):167–179. https://doi.org/10.1016/j.jbiotec.2007.06.010
Knothe G (2009) Improving biodiesel fuel properties by modifying fatty ester composition. Energy Environ Sci 2(7):759–766. https://doi.org/10.1039/B903941D
Kot AM, Stanislaw B, Agnieszka K, Joanna B, Iwona G, Anna B-W, Maliszewska M, Lidia R (2017) Effect of initial pH of medium with potato wastewater and glycerol on protein, lipid and carotenoid biosynthesis by Rhodotorula glutinis. Electron J Biotechnol 27:25–31. https://doi.org/10.1016/j.ejbt.2017.01.007
Kumari A, Keith B, Gotthard K, Rani G (2015) Extracellular expression of YlLip11 with a native signal peptide from Yarrowia lipolytica MSR80 in three different yeast hosts. Protein Expr Purif 110:138–144. https://doi.org/10.1016/j.pep.2015.02.016
Kunze G, Kunze I (1996) Arxula adeninivorans. In: Nonconventional yeasts in biotechnology. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-79856-6_11
Kurtzman CP, Fell JW, Teun B (eds) (2011) The yeasts: a taxonomic study, vol 1–3, 5th edn. Elsevier, Amsterdam
Kunze G, Claude G, Malgorzata C, Pascal D, Tiphaine M, Erik B, Toni G, Cruz Jose A, Emmanuel T, Christian M, André G, Valérie B, Philippe B, Keith B, Sebastian B, Claudine B, Rüdiger B, Serge C, Laurence D, Cécile F, Martin G, Piotr GP, Urs H, Anja H, Dagmara J, Claire J, Paul J, Ingrid L, Véronique L-L, Marc L, Marina M-H, Martin M, Guillaume M, Guy-Franck R, Jan R, Christine S, Anasua S, Guilhem S, Joseph S, Sherman David J, Nils S, Marie-Laure S, Agnès T, Anke T-S, Benoit V, Eric W, Sebastian W, Bernard D, Jean-Luc S, Patrick W, Uwe S, Cécile N (2014) The complete genome of Blastobotrys (Arxula) adeninivorans LS3 - a yeast of biotechnological interest. Biotechnol Biofuels 7:66. https://doi.org/10.1186/1754-6834-7-66
Kurtzman CP (2007) Blastobotrys americana sp. nov., Blastobotrys illinoisensis sp. nov., Blastobotrys malaysiensis sp. nov., Blastobotrys muscicola sp. nov., Blastobotrys peoriensis sp. nov. and Blastobotrys raffinosifermentans sp. nov., novel anamorphic yeast species. Int J Syst Evol Microbiol 57(Pt 5):1154–1162. https://doi.org/10.1099/ijs.0.64847-0
Kurtzman CP, Quintilla MR, Anna K, Bart T, Vincent R, Teun B (2015) Advances in yeast systematics and phylogeny and their use as predictors of biotechnologically important metabolic pathways. FEMS Yeast Res 15(6). https://doi.org/10.1093/femsyr/fov050
Kurtzman CP, Robnett CJ (2007) Multigene phylogenetic analysis of the Trichomonascus, Wickerhamiella and Zygoascus yeast clades, and the proposal of Sugiyamaella gen. nov. and 14 new species combinations. FEMS Yeast Res 7(1):141–151. https://doi.org/10.1111/j.1567-1364.2006.00157.x
Kwon Y-J, An-Zhou M, Qian L, Feng W, Guo-Qiang Z, Chun-Zhao L (2011) Effect of lignocellulosic inhibitory compounds on growth and ethanol fermentation of newly-isolated thermotolerant Issatchenkia orientalis. Bioresour Technol 102(17):8099–8104. https://doi.org/10.1016/j.biortech.2011.06.035
Lapeña D, Gergley K, Hansen Line D, Mydland Liv T, Volkmar P, Horn Svein J, Eijsink Vincent GH (2020) Production and characterization of yeasts grown on media composed of spruce-derived sugars and protein hydrolysates from chicken by-products. Microb Cell Factories 19(1):19. https://doi.org/10.1186/s12934-020-1287-6
Ledesma-Amaro R, Zbigniew L, Magdalena R, Zhongpeng G, Florian F, Coq CA-MC-L, Jean-Marc N (2016) Metabolic engineering of Yarrowia lipolytica to produce chemicals and fuels from xylose. Metab Eng 38:115–124. https://doi.org/10.1016/j.ymben.2016.07.001
Li Z, Hanxiao S, Xuemei M, Xiuying L, Bo X, Peng T (2013) Overexpression of malic enzyme (ME) of Mucor circinelloides improved lipid accumulation in engineered Rhodotorula glutinis. Appl Microbiol Biotechnol 97(11):4927–4936. https://doi.org/10.1007/s00253-012-4571-5
Litwińska K, Felix B, Falko M, Rüdiger B, Twan R, Gotthard K (2019) Characterization of recombinant laccase from Trametes versicolor synthesized by Arxula adeninivorans and its application in the degradation of pharmaceuticals. AMB Express 9(1):102. https://doi.org/10.1186/s13568-019-0832-3
Liu Y, Amy YS, John KCM, Lianghui J (2016) Developing a set of strong intronic promoters for robust metabolic engineering in oleaginous Rhodotorula (Rhodosporidium) yeast species. Microb Cell Factories 15(1):200. https://doi.org/10.1186/s12934-016-0600-x
Liu L, Anny P, Caitlin S, Nijia Z, Alper Hal S (2015) An evolutionary metabolic engineering approach for enhancing lipogenesis in Yarrowia lipolytica. Metab Eng 29:36–45. https://doi.org/10.1016/j.ymben.2015.02.003
Liu L-p, Min-hua Z, Yang H, Ning L, Wen-yong L, Hong W (2017) Efficient microbial oil production on crude glycerol by Lipomyces starkeyi AS 2.1560 and its kinetics. Process Biochem 58:230–238. https://doi.org/10.1016/j.procbio.2017.03.024
Macías LG, Miguel M, Christina T, Eladio B (2019) Comparative Genomics Between Saccharomyces kudriavzevii and S. cerevisiae Applied to Identify Mechanisms Involved in Adaptation. Front Genet 10:187. https://doi.org/10.3389/fgene.2019.00187
Madzak C (2015) Yarrowia lipolytica: recent achievements in heterologous protein expression and pathway engineering. Appl Microbiol Biotechnol 99(11):4559–4577. https://doi.org/10.1007/s00253-015-6624-z
Maina S, Chrysanthi P, Nikolaos K, Spiros P, Drosinos Eleftherios H, Seraphim P, Apostolis K (2017) Microbial oil production from various carbon sources by newly isolated oleaginous yeasts. Eng Life Sci 17(3):333–344. https://doi.org/10.1002/elsc.201500153
Malak A, Keith B, Gotthard K (2016) Blastobotrys (Arxula) adeninivorans: a promising alternative yeast for biotechnology and basic research. Yeast 33(10):535–547. https://doi.org/10.1002/yea.3180
Maza DD, Viñarta Silvana C, Ying S, Manuel GJ, Aybar Manuel J (2020) Growth and lipid production of Rhodotorula glutinis R4, in comparison to other oleaginous yeasts. J Biotechnol 310:21–31. https://doi.org/10.1016/j.jbiotec.2020.01.012
Meesters PAE, Eggink G (1996) Isolation and characterization of a delta-9 fatty acid desaturase gene from the oleaginous yeast Cryptococcus curvatus CBS 570. Yeast 12(8):723–730. https://doi.org/10.1002/(sici)1097-0061(19960630)12:8<723::aid-yea963>3.0.co;2-o
Middelhoven WJ, Hoogkamer-Te Niet MC, Kreger-Van Rij NJ (1984) Trichosporon adeninovorans sp. nov., a yeast species utilizing adenine, xanthine, uric acid, putrescine and primary n-alkylamines as the sole source of carbon, nitrogen and energy. Antonie Van Leeuwenhoek 50(4):369–378. https://doi.org/10.1007/bf00394651
Ng T-K, Ai-Qun Y, Hua L, Kurniasih PJN, Jae CW, Jan LSS, Wook CM (2020) Engineering Yarrowia lipolytica towards food waste bioremediation: Production of fatty acid ethyl esters from vegetable cooking oil. J Biosci Bioeng 129(1):31–40. https://doi.org/10.1016/j.jbiosc.2019.06.009
Olstorpe M, Schnürer J, Passoth V (2009) Screening of yeast strains for phytase activity. FEMS Yeast Res 9(3):478–488. https://doi.org/10.1111/j.1567-1364.2009.00493.x
Olstorpe M, Jana P, Anders K, Volkmar P (2014) Strain- and temperature-dependent changes of fatty acid composition in Wickerhamomyces anomalus and Blastobotrys adeninivorans. Biotechnol Appl Biochem 61(1):45–50. https://doi.org/10.1002/bab.1130
Park Y-K, Jean-Marc N, Rodrigo L-A (2018) The engineering potential of Rhodosporidium toruloides as a workhorse for biotechnological applications. Trends Biotechnol 36(3):304–317. https://doi.org/10.1016/j.tibtech.2017.10.013
Patel A, Matsakas L (2019) A comparative study on de novo and ex novo lipid fermentation by oleaginous yeast using glucose and sonicated waste cooking oil. Ultrason Sonochem 52:364–374. https://doi.org/10.1016/j.ultsonch.2018.12.010
Pham HTM, Martin G, Linda G, Karima H, Jochen T, Steffen U, Gerold H, Peter W, Kirsten S, Keith B, Gotthard K (2015) The determination of pharmaceuticals in wastewater using a recombinant Arxula adeninivorans whole cell biosensor. Sensors Actuators B Chem 211:439–448. https://doi.org/10.1016/j.snb.2015.01.107
Poontawee R, Wichien Y, Savitree L (2017) Efficient oleaginous yeasts for lipid production from lignocellulosic sugars and effects of lignocellulose degradation compounds on growth and lipid production. Process Biochem 53:44–60. https://doi.org/10.1016/j.procbio.2016.11.013
Pretorius IS (2017) Synthetic genome engineering forging new frontiers for wine yeast. Crit Rev Biotechnol 37(1):112–136. https://doi.org/10.1080/07388551.2016.1214945
Qian X, Xinhai Z, Lin C, Xiaoyu Z, Fengxue X, Weiliang D, Wenming Z, Katrin O, Min J (2020) Bioconversion of volatile fatty acids into lipids by the oleaginous yeast Apiotrichum porosum DSM27194. Fuel 119811. https://doi.org/10.1016/j.fuel.2020.119811
Qin L, Lu L, An-Ping Z, Dong W (2017) From low-cost substrates to Single Cell Oils synthesized by oleaginous yeasts. Bioresour Technol 245(Pt B):1507–1519. https://doi.org/10.1016/j.biortech.2017.05.163
Ratledge C (2014) The role of malic enzyme as the provider of NADPH in oleaginous microorganisms: a reappraisal and unsolved problems. Biotechnol Lett 36(8):1557–1568. https://doi.org/10.1007/s10529-014-1532-3
Rauter M, Alexandra P, Jakub K, Karin B, Keith B, Rudiger B, Gothhard K, Vorbrodt Matthias H (2015) Coexpression of Lactobacillus brevis ADH with GDH or G6PDH in Arxula adeninivorans for the synthesis of 1-(R)-phenylethanol. Appl Microbiol Biotechnol 99(11):4723–4733. https://doi.org/10.1007/s00253-014-6297-z
Rauter M, Jakub K, Karin B, Jan R, Sebastian W, Anja H, Martin M, Uwe S, Keith B, Rudiger B, Frieder S, Matthias Vorbrodt H, Gotthard K (2016) Aadh2p: an Arxula adeninivorans alcohol dehydrogenase involved in the first step of the 1-butanol degradation pathway. Microb Cell Factories 15(1):175. https://doi.org/10.1186/s12934-016-0573-9
Reiser J, Ochsner UA, Kälin M, Glumoff V, Fiechter A (1996) Trichosporon. In: Wolf K (ed) Nonconventional yeasts in biotechnology: a handbook. Springer Berlin Heidelberg, Berlin, Heidelberg, pp 581–606
Rösel H, Kunze G (1995) Cloning and characterization of a TEF gene for elongation factor 1α from the yeast Arxula adeninivorans. Curr Genet 28(4):360–366. https://doi.org/10.1007/BF00326434
Rösel H, Kunze G (1998) Integrative transformation of the dimorphic yeast Arxula adeninivorans LS3 based on hygromycin B resistance. Curr Genet 33(2):157–163. https://doi.org/10.1007/s002940050322
Saha S, Balaji E, Sona R, Ram R (2006) Cytosolic triacylglycerol biosynthetic pathway in oilseeds. Molecular cloning and expression of peanut cytosolic diacylglycerol acyltransferase. Plant Physiol 141(4):1533–1543. https://doi.org/10.1104/pp.106.082198
Samsonova IA, Gotthard K, Rüdiger B, Böttcher F (1996) A set of genetic markers for the chromosomes of the imperfect yeast Arxula adeninivorans. Yeast 12(12):1209–1217. https://doi.org/10.1002/(sici)1097-0061(19960930)12:12<1209::aid-yea12>3.0.co;2-w
Sano K, Fukuhara H, Nakamura Y (1999) Phytase of the yeast Arxula adeninivorans. Biotechnol Lett 21:33–38. https://doi.org/10.1023/A:1005438121763
Sanya DRA, Djamila O, Gotthard K, Cécile N, Crutz L-CA-M (2020) The native acyltransferase-coding genes DGA1 and DGA2 affect lipid accumulation in Blastobotrys raffinosifermentans differently when overexpressed. FEMS Yeast Res. https://doi.org/10.1093/femsyr/foaa060
Sara M, Kaur BS, François BJ (2016) Lipid production by Yarrowia lipolytica grown on biodiesel-derived crude glycerol: optimization of growth parameters and their effects on the fermentation efficiency. RSC Adv 6(93):90547–90558. https://doi.org/10.1039/C6RA16382C
Sędzielewska KA, Erik B, Carmen B, Thomas W, Rüdiger B, Michael M, Keith B, Gotthard K (2012) Role of the AFRD1-encoded fumarate reductase in hypoxia and osmotolerance in Arxula adeninivorans. FEMS Yeast Res 12(8):924–937. https://doi.org/10.1111/j.1567-1364.2012.00842.x
Shai N, Eden Y, van Roermund Carlo WT, Nir C, Chen B, Lodewijk I, Laetitia C, Julie M, Ramona S, Lior Z, Mari Muriel C, Reggiori Fulvio M, Hughes Adam L, Mafalda E-H, Cohen Mickael M, Waterham Hans R, Wanders Ronald JA, Maya S, Einat Z (2018) Systematic mapping of contact sites reveals tethers and a function for the peroxisome-mitochondria contact. Nat Commun 9(1):1761. https://doi.org/10.1038/s41467-018-03957-8
Shapaval V, Jule B, Johanna B, Valeria T, Volkmar P, Mats S, Achim K (2019) Biochemical profiling, prediction of total lipid content and fatty acid profile in oleaginous yeasts by FTIR spectroscopy. Biotechnol Biofuels 12:140. https://doi.org/10.1186/s13068-019-1481-0
Shen X-X, Xiaofan Z, Jacek K, Kurtzman Cletus P, Todd HC, Antonis R (2016) Reconstructing the backbone of the Saccharomycotina yeast phylogeny using genome-scale data. G3 (Bethesda) 6(12):3927–3939. https://doi.org/10.1534/g3.116.034744
Silverman AM, Kangjian Q, Peng X, Gregory S (2016) Functional overexpression and characterization of lipogenesis-related genes in the oleaginous yeast Yarrowia lipolytica. Appl Microbiol Biotechnol 100(8):3781–3798. https://doi.org/10.1007/s00253-016-7376-0
Sitepu IR, Garay Luis A, Ryan S, David L, Block David E, Bruce GJ, Boundy-Mills Kyria L (2014) Oleaginous yeasts for biodiesel: current and future trends in biology and production. Biotechnol Adv 32(7):1336–1360. https://doi.org/10.1016/j.biotechadv.2014.08.003
Stehlik T, Marco K, Jörg K, Michael B, Johannes F (2020) Peroxisomal targeting of a protein phosphatase type 2C via mitochondrial transit. Nat Commun 11(1):2355. https://doi.org/10.1038/s41467-020-16146-3
Stehr F, Kretschmar M, Kröger C, Hube B, Schäfer W (2003) Microbial lipases as virulence factors. J Mol Catal B Enzym 22(5):347–355. https://doi.org/10.1016/S1381-1177(03)00049-3
Steinborn G, Erik B, Anja S, Kristina T, Gotthard K, Gerd G (2006) Application of a wide-range yeast vector (CoMed) system to recombinant protein production in dimorphic Arxula adeninivorans, methylotrophic Hansenula polymorpha and other yeasts. Microb Cell Factories 5:33. https://doi.org/10.1186/1475-2859-5-33
Steinborn G, Gerd G, Gotthard K (2007) A novel vector element providing multicopy vector integration in Arxula adeninivorans. FEMS Yeast Res 7(7):1197–1205. https://doi.org/10.1111/j.1567-1364.2007.00280.x
Stöckmann C, Marco S, Barbara D, Armin M, Grit H, Georg M, Doris K, Jochen B, Ah KH, Gerd G (2009) Process development in Hansenula polymorpha and Arxula adeninivorans, a re-assessment. Microb Cell Factories 8:22. https://doi.org/10.1186/1475-2859-8-22
Stoltenburg R, Lösche O, Klappach G, Kunze G (1999) Molecular cloning and expression of the ARFC3 gene, a component of the replication factor C from the salt-tolerant, dimorphic yeast Arxula adeninivorans LS3. Curr Genet 35(1):8–13. https://doi.org/10.1007/s002940050426
Sutanto S, Siti Z, Lan T-NP, Suryadi I, Yi-Hsu J (2018) Lipomyces starkeyi: its current status as a potential oil producer. Fuel Process Technol 177:39–55. https://doi.org/10.1016/j.fuproc.2018.04.012
Takaku H, Sayaka E, Kotoha K, Rikako S, Satoshi A, Haruka K, Tomohiko M, Katsuno Y, Hideo A, Yosuke S, Wataru O, Koji I, Sachiyo A, Harutake Y (2021) Isolation and characterization of Lipomyces starkeyi mutants with greatly increased lipid productivity following UV irradiation. J Biosci Bioeng. https://doi.org/10.1016/j.jbiosc.2021.01.006
Tamano K, Bruno Kenneth S, Karagiosis Sue A, Culley David E, Shuang D, Collett James R, Myco U, Hideaki K, Baker Scott E, Masayuki M (2013) Increased production of fatty acids and triglycerides in Aspergillus oryzae by enhancing expressions of fatty acid synthesis-related genes. Appl Microbiol Biotechnol 97(1):269–281. https://doi.org/10.1007/s00253-012-4193-y
Tanimura A, Takashima M, Sugita T, Endoh R, Kikukawa M, Yamaguchi S, Sakuradani E, Ogawa J, Ohkuma M, Shima J (2014) Cryptococcus terricola is a promising oleaginous yeast for biodiesel production from starch through consolidated bioprocessing. Sci Rep 4:4776. https://doi.org/10.1038/srep04776
Tanimura A, Takashima M, Sugita T, Endoh R, Ohkuma M, Kishino S, Ogawa J, Shima J (2016) Lipid production through simultaneous utilization of glucose, xylose, and L-arabinose by Pseudozyma hubeiensis: a comparative screening study. AMB Express 6(1):58. https://doi.org/10.1186/s13568-016-0236-6
Terentiev Y, Huarto PA, Erik B, Thomas W, Jens K, Uta B, Wolfgang B, Manfred S, Gerd G, Gotthard K (2004) A wide-range integrative yeast expression vector system based on Arxula adeninivorans-derived elements. J Ind Microbiol Biotechnol 31(5):223–228. https://doi.org/10.1007/s10295-004-0142-9
Theron CW, Labuschagné M, Gudiminchi R, Albertyn J, Smit MS (2014) A broad-range yeast expression system reveals Arxula adeninivorans expressing a fungal self-sufficient cytochrome P450 monooxygenase as an excellent whole-cell biocatalyst. FEMS Yeast Res 14(4):556–566. https://doi.org/10.1111/1567-1364.12142
Thomas S, Akiola SDR, Florian F, Huu-Vang N, Gotthard K, Cécile N, Anne-Marie C-LC (2019) Blastobotrys adeninivorans and B. raffinosifermentans, two sibling yeast species which accumulate lipids at elevated temperatures and from diverse sugars. Biotechnol Biofuels 12:154. https://doi.org/10.1186/s13068-019-1492-x
Tiukova IA, Sylvain P, Jens N, Mats S, Kerkhoven Eduard J (2019) Genome-scale model of Rhodotorula toruloides metabolism. Biotechnol Bioeng 116(12):3396–3408. https://doi.org/10.1002/bit.27162
Trautwein-Schult A, Dagmara J, Arno C, Petra H, Christina K, Andrea M, Hans-Peter M, Keith B, Rüdiger B, Gotthard K (2013) Arxula adeninivorans recombinant urate oxidase and its application in the production of food with low uric acid content. J Mol Microbiol Biotechnol 23(6):418–430. https://doi.org/10.1159/000353847
Trautwein-Schult A, Dagmara J, Arno C, Petra H, Christina K, Andrea M, Hans-Peter M, Keith B, Rüdiger B, Gotthard K (2014) Arxula adeninivorans recombinant guanine deaminase and its application in the production of food with low purine content. J Mol Microbiol Biotechnol 24(2):67–81. https://doi.org/10.1159/000357674
Tsai Y-Y, Takao O, Chih-Chan W, Dolgormaa B, Ryo M, Savitree L, Kazuhito F (2019) Delta-9 fatty acid desaturase overexpression enhanced lipid production and oleic acid content in Rhodosporidium toruloides for preferable yeast lipid production. J Biosci Bioeng 127(4):430–440. https://doi.org/10.1016/j.jbiosc.2018.09.005
Tsakraklides V, Annapurna K, Consiglio Andrew L, Kyle ME, Jonathan F, Blitzblau Hannah G, Hamilton Maureen A, Crabtree Donald V, Austin S, Jonathan A, Sullivan John E, Greg LW, South Colin R, Greenhagen Emily H, Joe SA, Brevnova Elena E (2018) High-oleate yeast oil without polyunsaturated fatty acids. Biotechnol Biofuels 11:131. https://doi.org/10.1186/s13068-018-1131-y
Tsakraklides V, Elena B, Gregory S, Joe SA (2015) Improved gene targeting through cell cycle synchronization. PLoS One 10(7):e0133434. https://doi.org/10.1371/journal.pone.0133434
Valm AM, Sarah C, Legant Wesley R, Justin M, Uri H, Eric W, Cohen Andrew R, Davidson Michael W, Eric B, Jennifer L-S (2017) Applying systems-level spectral imaging and analysis to reveal the organelle interactome. Nature 546(7656):162–167. https://doi.org/10.1038/nature22369
Van der Walt JP, Smith MT, Yamada Y (1990) Arxula gen. nov. (Candidaceae), a new anamorphic, arthroconidial yeast genus. Antonie Van Leeuwenhoek 57(1):59–61. https://doi.org/10.1007/bf00400338
Visnapuu T, Aivar M, Kristina P, Katrin V, Karin E, Tiina A (2019) Characterization of a maltase from an early-diverged non-conventional yeast Blastobotrys adeninivorans. Int J Mol Sci 21(1). https://doi.org/10.3390/ijms21010297
Vyas S, Chhabra M (2017) Isolation, identification and characterization of Cystobasidium oligophagum JRC1: A cellulase and lipase producing oleaginous yeast. Bioresour Technol 223:250–258. https://doi.org/10.1016/j.biortech.2016.10.039
Wang Y, Sufang Z, Zhiwei Z, Hongwei S, Xinping L, Xiang J, Xiang J, Kent ZZ (2018a) Systems analysis of phosphate-limitation-induced lipid accumulation by the oleaginous yeast Rhodosporidium toruloides. Biotechnol Biofuels 11:148. https://doi.org/10.1186/s13068-018-1134-8
Wang S, Fatima-Zahra I, Martin H, Alexandra G, Ejsing Christer S, Pedro C (2018b) Seipin and the membrane-shaping protein Pex30 cooperate in organelle budding from the endoplasmic reticulum. Nat Commun 9(1):2939. https://doi.org/10.1038/s41467-018-05278-2
Wang G, Delong L, Zhengang M, Shanshan Z, Wenxing L, Lin L (2018c) Comparative transcriptome analysis reveals multiple functions for Mhy1p in lipid biosynthesis in the oleaginous yeast Yarrowia lipolytica. Biochim Biophys Acta Mol Cell Biol Lipids 1863(1):81–90. https://doi.org/10.1016/j.bbalip.2017.10.003
Wang J, Rodrigo L-A, Yongjun W, Boyang J, Xiao-Jun J (2020) Metabolic engineering for increased lipid accumulation in Yarrowia lipolytica—a review. Bioresour Technol 313:123707. https://doi.org/10.1016/j.biortech.2020.123707
Wang Q, Yan C, Biswarup S, Wenmeng M, Lynn ZR, Xianhua L, Guangyi W (2017) Characterization and robust nature of newly isolated oleaginous marine yeast Rhodosporidium spp. from coastal water of Northern China. AMB Express 7(1):30–30. https://doi.org/10.1186/s13568-017-0329-x
Wartmann T, Rösel H, Kunze I, Bode R, Kunze G (1998) AILV1 gene from the yeast Arxula adeninivorans LS3-a new selective transformation marker. Yeast 14(11):1017–1025. https://doi.org/10.1002/(sici)1097-0061(199808)14:11<1017::Aid-yea314>3.0.Co;2-0
Wartmann T, Erik B, Huarto PA, Heike S, Oliver B, Gerd G, Gotthard K (2002) High-level production and secretion of recombinant proteins by the dimorphic yeast Arxula adeninivorans. FEMS Yeast Res 2(3):363–369. https://doi.org/10.1016/s1567-1356(02)00086-7
Wartmann T, Gellissen G, Kunze G (2001) Regulation of the AEFG1 gene, a mitochondrial elongation factor G from the dimorphic yeast Arxula adeninivorans LS3. Curr Genet 40(3):172–178. https://doi.org/10.1007/s002940100254
Wartmann T, Kunze I, Duc Bui M, Manteuffel R, Kunze G (1995) Comparative biochemical, genetical and immunological studies of glucoamylase producing Arxula adeninivorans yeast strains. Microbiol Res 150(2):113–120. https://doi.org/10.1016/s0944-5013(11)80045-5
Wartmann T, Kunze G (2000) Genetic transformation and biotechnological application of the yeast Arxula adeninivorans. Appl Microbiol Biotechnol 54(5):619–624
Wartmann T, Kunze G (2003) Temperature-dependent dimorphism of the non-conventional yeast Arxula adeninivorans. In: Wolf K, Breunig K, Barth G (eds) Non-conventional yeasts in genetics, biochemistry and biotechnology: practical protocols. Springer Berlin Heidelberg, Berlin, pp 3–6
Wartmann T, Regina S, Erik B, Heike S, Oliver B, Gerd G, Gotthard K (2003) The ALEU2 gene—a new component for an Arxula adeninivorans-based expression platform. FEMS Yeast Res 3(2):223–232. https://doi.org/10.1016/s1567-1356(02)00190-3
Williams J, Anke T-S, Gottard K, Kim B (2017) Environmental and metabolic parameters affecting the uric acid production of Arxula adeninivorans. Appl Microbiol Biotechnol 101(11):4725–4736. https://doi.org/10.1007/s00253-017-8199-3
Wu S, Xin Z, Hongwei S, Qian W, Zhao Zongbao K (2011) Microbial lipid production by Rhodosporidium toruloides under sulfate-limited conditions. Bioresour Technol 102(2):1803–1807. https://doi.org/10.1016/j.biortech.2010.09.033
Xavier MCA, ALV C, Carolina DA, Teixeira FT (2017) Lipid production from hemicellulose hydrolysate and acetic acid by Lipomyces starkeyi and the ability of yeast to metabolize inhibitors. Biochem Eng J 118:11–19. https://doi.org/10.1016/j.bej.2016.11.007
Xenopoulos E, Ioannis G, Afroditi C, Apostolis K, Seraphim P (2020) Lipid production by yeasts growing on commercial xylose in submerged cultures with process water being partially replaced by olive mill wastewaters. Processes 8(7). https://doi.org/10.3390/pr8070819
Xie D, Jackson EN, Zhu Q (2015) Sustainable source of omega-3 eicosapentaenoic acid from metabolically engineered Yarrowia lipolytica: from fundamental research to commercial production. Appl Microbiol Biotechnol 99(4):1599–1610. https://doi.org/10.1007/s00253-014-6318-y
Xue Z, Sharpe Pamela L, Seung-Pyo H, Yadav Narenda S, Dongming X, Short David R, Damude Howard G, Rupert Ross A, Seip John E, Jamie W, Pollak Dana W, Bostick Michael W, Bosak Melissa D, Macool Daniel J, Hollerbach Dieter H, Hongxiang Z, Arcilla Dennis M, Bledsoe Sidney A, Kevin C, McCord Elizabeth F, Tyreus Bjorn D, Jackson Ethel N, Quinn Z (2013) Production of omega-3 eicosapentaenoic acid by metabolic engineering of Yarrowia lipolytica. Nat Biotechnol 31(8):734–740. https://doi.org/10.1038/nbt.2622
Yamada R, Azusa Y, Tomomi K, Hiroyasu O (2017) Evaluation of lipid production from xylose and glucose/xylose mixed sugar in various oleaginous yeasts and improvement of lipid production by UV mutagenesis. Biochem Eng J 128:76–82. https://doi.org/10.1016/j.bej.2017.09.010
Yamazaki H, Suzuka K, Sayaka E, Shiho A, Satoshi A, Yosuke S, Wataru O, Katsurou Y, Hideo A, Hiroaki T (2019) Highly selective isolation and characterization of Lipomyces starkeyi mutants with increased production of triacylglycerol. Appl Microbiol Biotechnol 103(15):6297–6308. https://doi.org/10.1007/s00253-019-09936-3
Yang X, Guojie J, Zhiwei G, Hongwei S, Fengwu B, Kent ZZ (2014) Recycling biodiesel-derived glycerol by the oleaginous yeast Rhodosporidium toruloides Y4 through the two-stage lipid production process. Biochem Eng J 91:86–91. https://doi.org/10.1016/j.bej.2014.07.015
Yuzbasheva EY, Mostova Elizaveta B, Andreeva Natalia I, Yuzbashev Tigran V, Laptev Ivan A, Sobolevskaya Tatiana I, Sineoky Sergey P (2017) Co-expression of glucose-6-phosphate dehydrogenase and acyl-CoA binding protein enhances lipid accumulation in the yeast Yarrowia lipolytica. New Biotechnol 39(Pt A):18–21. https://doi.org/10.1016/j.nbt.2017.05.008
Zhang R, Shi-Jue G, Chun-Yan Z, Yu S, Xiao-Ling L, Mou-Ming Z, Cheng-Hua W (2019) Characterization of a novel alkaline Arxula adeninivorans urate oxidase expressed in Escherichia coli and its application in reducing uric acid content of food. Food Chem 293:254–262. https://doi.org/10.1016/j.foodchem.2019.04.112
Zhu Z, Sufang Z, Hongwei L, Hongwei S, Xinping L, Fan Y, Zhou Yongjin J, Guojie J, Mingliang Y, Hanfa Z, Zhao Zongbao K (2012) A multi-omic map of the lipid-producing yeast Rhodosporidium toruloides. Nat Commun 3:1112. https://doi.org/10.1038/ncomms2112
Acknowledgments
Authors acknowledge Hiroko KITAMOTO, Institute for Agro-Environmental Sciences, National Agriculture and Food Research Organisation, Tsukuba, Ibaraki, Japan, and Zongbao Kent Zhao, Laboratory of Biotechnology, Dalian Institute of Chemical Physics, CAS, 457 Zhongshan Road, Dalian, 116023 People’s Republic of China and Mahesh Khot from FONDECYT (Fondo Nacional de Desarrollo Científico y Tecnológico/National Fund for Scientific and Technological Development), Government of Chile, Chile, Santiago in Postdoctoral Project Grant context (No. 3180134), for proofreading this review document.
Author information
Authors and Affiliations
Contributions
DRAS has prepared manuscript, performed literature analysis, and prepared tables and figures. DO, VP, MKM, AC, and MBK have revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants performed by any of the authors
Consent for publication
Not applicable.
Conflict of interest
All authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(PDF 173 kb)
Rights and permissions
About this article
Cite this article
Sanya, D.R.A., Onésime, D., Passoth, V. et al. Yeasts of the Blastobotrys genus are promising platform for lipid-based fuels and oleochemicals production. Appl Microbiol Biotechnol 105, 4879–4897 (2021). https://doi.org/10.1007/s00253-021-11354-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-021-11354-3