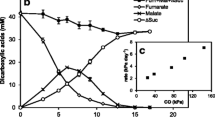
Abstract
Sulfurospirillum deleyianum grew in batch culture under anoxic conditions with sulfide (up to 5 mM) as electron donor, nitrate as electron acceptor, and acetate as carbon source. Nitrate was reduced to ammonia via nitrite, a quantitatively liberated intermediate. Four moles of sulfide were oxidized to elemental sulfur per mole nitrate converted to ammonia. The molar growth yield per mole sulfide consumed, Ym, was 1.5±0.2 g mol−1 for the reduction of nitrate to ammonia. By this type of metabolism,S. deleyianum connected the biogeochemical cycles of sulfur and nitrogen. The sulfur reductase activity inS. deleyianum was inducible, as the activity depended on the presence of sulfide or elemental sulfur during cultivation with nitrate or fumarate as electron acceptor. Hydrogenase activity was always high, indicating that the enzyme is constitutively expressed. The ammonia-forming nitrite reductase was an inducible enzyme, expressed when cells were cultivated with nitrate, nitrite, or elemental sulfur, but repressed after cultivation with fumarate.
Similar content being viewed by others
Abbreviations
- <S 0}:
-
Biogenic elemental sulfur
- S 8R :
-
Crystalline elemental sulfur
References
Abou-Jaoudé A, Chippaux M, Pascal MC (1979) Formate-nitrite reduction inEscherichia coli K12. 1. Physiological study of the system. Eur J Biochem 95:309–314
Aminuddin M, Nicholas DJD (1973) Sulphide oxidation linked to the reduction of nitrate and nitrite inThiobacillus denitrificans, Biochim Biophys Acta 325:81–93
Bokranz MJ, Katz J, Schröder I, Roberton AM, Kröger A (1983) Energy metabolism and biosynthesis ofVibrio succinogenes growing with nitrate or nitrite as terminal electron acceptor. Arch Microbiol 135:36–41
Boltz DF, Taras MJ (1978) Nitrogen. In: Boltz DF, Howell JA (eds) Colorimetric determination of nonmetals. Chemical analysis, vol 8. Wiley, New York, pp 197–251
Cline JD (1969) Spectrophotometric determination of hydrogen sulfide in natural waters. Limnol Oceanogr 14:454–458
Collins MD, Widdel F (1986) Respiratory quinones of sulphate-reducing and sulphur-reducing bacteria: a systematic investigation. Sustem Appl Microbiol 8:8–18
Dalsgaard T, Bak F (1994) Nitrate reduction in a sulfate-reducing bacterium,Desulfovibrio desulfuricans, isolated from rice paddy soil: sulfide inhibition, kinetics, and regulation. Appl Environ Microbiol 60:291–297
Dannenberg S, Kroder M, Dilling W, Cypionka H (1992) Oxidation of H2, organic compounds and inorganic sulfur compounds coupled to reduction of O2 or nitrate by sulfate-reducing bacteria. Arch Microbiol 158:93–99
De Vries W, Niekus HGD, Boellaard M, Stouthamer AH (1980) Growth yields and energy generation byCampylobacter sputorum subspeciesbubulus during growth in continuous culture with different hydrogen acceptors. Arch Microbiol 124:221–227
Fauque G, Herve D, LeGall J (1979) Structure-function relationships in hemoproteins: the role of cytochromec 3 in the reduction of colloidal sulfur by sulfate-reducing bacteria. Arch Microbiol 121:261–264
Gebhardt NA, Thauer RK, Linder D, Kaulfers PM, Pfennig N (1985) Mechanism of acetate oxidation to CO2 with elemental sulfur inDesulfuromonas acetoxidans. Arch Microbiol 141:392–398
Janek A (1933) Ein neues Verfahren zur Herstellung von Schwefelsolen. Kolloid Z 64:31–32
King D, Nedwell DB (1985) The influence of nitrate concentration upon the end product of nitrate dissimilation by bacteria in anaerobic salt marsh sediment. FEMS Microbiol Ecol 31:23–28
King D, Nedwell DB (1987) The adaptation of nitrate-reducing bacterial communities in estuarine sediments in response to overlying nitrate load. FEMS Microbiol Ecol 45:15–20
Kreis-Kleinschmidt V, Fahrenholz F, Kojro E, Kröger A (1995) Periplasmic sulphide dehydrogenase (Sud) fromWolinella succinogenes: isolation, nucleotide sequence of thesud gene and its expression inEscherichia coli. Eur J Biochem 227:137–142
Lang E, Lang H (1972) Spezifische Farbreaktion zum direkten Nachweis der Ameisensäure. Z Anal Chem 260:8–10
Laudenbach DE, Ehrhardt D, Green L, Grossman A (1991) Isolation of a sulfur-regulated gene encoding a periplasmically localized protein with sequence similarity to rhodanese. J Bacteriol 173:2751–2760
Ma K, Adams MWW (1994) Sulfide dehydrogenase from the hyperthermophilic archaeonPyrococcus furiosus: a new multifunctional enzyme involved in the reduction of elemental sulfur. J Bacteriol 176:6509–6517
Macy JM, Schröder I, Thauer RK, Kröger A (1986) Growth ofWolinella succinogenes on H2S plus fumarate and on formate plus sulfur as energy sources. Arch Microbiol 144:147–150
Murray RE, Parsons LL, Smith MS (1989) Kinetics of nitrate utilization by mixed populations of denitrifying bacteria. Appl Environ Microbiol 55:717–721
Schumacher W, Kroneck PMH (1992) Anaerobic energy metabolism of the sulfur-reducing bacterium “Spirillum” 5175 during dissimilatory nitrate reduction to ammonia. Arch Microbiol 157:464–470
Schumacher W, Kroneck PMH, Pfennig N (1992) Comparative systematic study on “Spirillum” 5175,Campylobacter andWolinella species. Description of “Spirillum” 5175 asSulfurospirillum deleyianum gen. nov., spec. nov. Arch Microbiol 158:287–293
Schumacher W, Hole U, Kroneck PMH (1994) Ammonia-forming cytochromec nitrite reductase fromSulfurospirillum deleyianum is a tetraheme protein: new aspects of the molecular composition and spectroscopic properties. Biochem Biophys Res Commun 205:911–916
Seitz HJ, Cypionka H (1986) Chemolithotrophic growth ofDesulfovibrio desulfuricans with hydrogen coupled to ammonification of nitrate or nitrite. Arch Microbiol 146:63–67
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano MD, Fujimoto EK, Goeke NM, Olson BJ, Klenk DC (1985) Measurement of protein using bicinchoninic acid. Anal Biochem 150:76–85
Umbreit WW, Burris RH, Stauffer JF (1972) Manometric and biochemical techniques. Burgess, Minneapolis
Wolfe RS, Pfennig N (1977) Reduction of sulfur by spirillum 5175 and syntrophism withChlorobium. Appl Environ Microbiol 33:427–433
Zöphel A, Kennedy MC, Beinert H, Kroneck PMH (1988) Investigations on microbial sulfur respiration. 1. Activation and reduction of elemental sulfur in several strains of eubacteria. Arch Microbiol 150:72–77
Zöphel A, Kennedy MC, Beinert H, Kroneck PMH (1991) Investigations on microbial sulfur respiration. 2. Isolation, purification, and characterization of cellular components from Spirillum 5175. Eur J Biochem 195:849–856
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Prof. Norbert Pfennig on the occasion of his 70th birthday
Rights and permissions
About this article
Cite this article
Eisenmann, E., Beuerle, J., Sulger, K. et al. Lithotrophic growth ofSulfurospirillum deleyianum with sulfide as electron donor coupled to respiratory reduction of nitrate to ammonia. Arch. Microbiol. 164, 180–185 (1995). https://doi.org/10.1007/BF02529969
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02529969